Effects of High Hydrostatic Pressure on Fungal Spores and Plant Bioactive Compounds
Definition
:1. Introduction
2. Fungal Spore Structures (Special Focus on Ascospores)
3. Prevalence of Fungi and Their Spores on Medicinal Herbs
4. Possible Limitations Caused by Fungi and Their Toxins to the Use of Bioactive Compounds from Food Materials
5. Effects of Nonthermal HPP on Ascospores—Impact on Ultrastructure
6. Potential of HPP to Eliminate Toxin-Forming Fungi in Plants and Herbs
7. Impact of HPP and HPE on Bioactive Compounds
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jahromi, B.; Pirvulescu, I.; Candido, K.D.; Knezevic, N.N. Herbal Medicine for Pain Management: Efficacy and Drug Interactions. Pharmaceutics 2021, 13, 251. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.-P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.-J.; Li, H.-B. Natural Antioxidants in Foods and Medicinal Plants: Extraction, Assessment and Resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Thanushree, M.P.; Vimala, B.S.K.; Moses, J.A.; Anandharamakrishnan, C. Detection Techniques for Insect Infestation in Stored Grains. Agr. Eng. Today 2018, 42, 48–56. [Google Scholar]
- Olicón-Hernández, D.R.; González-López, J.; Aranda, E. Overview on the Biochemical Potential of Filamentous Fungi to Degrade Pharmaceutical Compounds. Front. Microbiol. 2017, 8, 1792. [Google Scholar] [CrossRef] [PubMed]
- Heisel, T.; Montassier, E.; Johnson, A.; Al-Ghalith, G.; Lin, Y.-W.; Wei, L.-N.; Knights, D.; Gale, C.A. High-Fat Diet Changes Fungal Microbiomes and Interkingdom Relationships in the Murine Gut. mSphere 2017, 2, e00351-17. [Google Scholar] [CrossRef]
- Vijayakumar, R.; Sandle, T.; Manohar, C. A Review on Fungal Contamination in Pharmaceutical Products and Phenotypicidentification of Contaminants by Conventional Methods. Eur. J. Parenter. Pharm. Sci. 2012, 17, 4–18. [Google Scholar]
- Halt, M. Moulds and Mycotoxins in Herb Tea and Medicinal Plants. Eur. J. Epidemiol. 1998, 14, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Zhou, X.; Gan, F.; Liu, Z.; Zhou, Y.; Qian, G.; Huang, K. Combination of Selenomethionine and N-Acetylcysteine Alleviates the Joint Toxicities of Aflatoxin B1 and Ochratoxin A by ERK MAPK Signal Pathway in Porcine Alveolar Macrophages. J. Agric. Food Chem. 2018, 66, 5913–5923. [Google Scholar] [CrossRef] [PubMed]
- Thanushree, M.P.; Sailendri, D.; Yoha, K.S.; Moses, J.A.; Anandharamakrishnan, C. Mycotoxin Contamination in Food: An Exposition on Spices. Trends Food Sci. Technol. 2019, 93, 69–80. [Google Scholar] [CrossRef]
- Dijksterhuis, J. Heat-Resistant Ascospores. In Food Mycology: A Multifaceted Approach to Fungi and Food; Dijksterhuis, J., Samson, R.A., Eds.; CRC Press, Taylor and Francis: Boca Raton, FL, USA, 2007; pp. 101–118. ISBN 978-0-8493-9818-6. [Google Scholar]
- Rico-Munoz, E. Heat Resistant Molds in Foods and Beverages: Recent Advances on Assessment and Prevention. Curr. Opin. Food Sci. 2017, 17, 75–83. [Google Scholar] [CrossRef]
- Pinto, C.A.; Moreira, S.A.; Fidalgo, L.G.; Inácio, R.S.; Barba, F.J.; Saraiva, J.A. Effects of High-Pressure Processing on Fungi Spores: Factors Affecting Spore Germination and Inactivation and Impact on Ultrastructure. Compr. Rev. Food Sci. Food Saf. 2020, 19, 553–573. [Google Scholar] [CrossRef] [PubMed]
- Dijksterhuis, J.; Nijsse, J.; Hoekstra, F.A.; Golovina, E.A. High Viscosity and Anisotropy Characterize the Cytoplasm of Fungal Dormant Stress-Resistant Spores. Eukaryot. Cell 2007, 6, 157–170. [Google Scholar] [CrossRef]
- Goh, Y.K.; Vujanovic, V. Ascospore Germination Patterns Revealed Ascomycetous Biotrophic Mycoparasite Specificity to Fusarium Hosts. Botany 2010, 88, 1033–1043. [Google Scholar] [CrossRef]
- Rico-Munoz, E.; Samson, R.A.; Houbraken, J. Mould Spoilage of Foods and Beverages: Using the Right Methodology. Food Microbiol. 2018, 81, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Visagie, C.M.; Yilmaz, N.; Renaud, J.B.; Sumarah, M.W.; Hubka, V.; Frisvad, J.C.; Chen, A.J.; Meijer, M.; Seifert, K.A. A Survey of Xerophilic Aspergillus from Indoor Environment, Including Descriptions of Two New Section Aspergillus Species Producing Eurotium-like Sexual States. MycoKeys 2017, 19, 1–30. [Google Scholar] [CrossRef]
- Mille-Lindblom, C.; Fischer, H.; Tranvik, L.J. Antagonism between Bacteria and Fungi: Substrate Competition and a Possible Tradeoff between Fungal Growth and Tolerance towards Bacteria. Oikos 2006, 113, 233–242. [Google Scholar] [CrossRef]
- Sterflinger, K.; Voitl, C.; Lopandic, K.; Piñar, G.; Tafer, H. Big Sound and Extreme Fungi—Xerophilic, Halotolerant Aspergilli and Penicillia with Low Optimal Temperature as Invaders of Historic Pipe Organs. Life 2018, 8, 22. [Google Scholar] [CrossRef]
- Gock, M.A.; Hocking, A.D.; Pitt, J.I.; Poulos, P.G. Influence of Temperature, Water Activity and PH on Growth of Some Xerophilic Fungi. Int. J. Food Microbiol. 2003, 81, 11–19. [Google Scholar] [CrossRef]
- Moss, M. Fungi, Quality and Safety Issues in Fresh Fruits and Vegetables. J. Appl. Microbiol. 2008, 104, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- Stević, T.; Pavlović, S.; Stanković, S.; Šavikin, K. Pathogenic Microorganisms of Medicinal Herbal Drugs. Arch. Biol. Sci. 2012, 64, 49–58. [Google Scholar] [CrossRef]
- Nesic, K.; Ivanovic, S.; Nesic, V. Fusarial Toxins: Secondary Metabolites of Fusarium Fungi. Rev. Environ. Contam. Toxicol. 2014, 228, 101–120. [Google Scholar] [CrossRef] [PubMed]
- Tosun, H.; Arslan, R. Determination of Aflatoxin B1 Levels in Organic Spices and Herbs. Sci. World J. 2013, 2013, e874093. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.; Marín, S.; Sanchis, V.; Ramos, A.J. Screening of Mycotoxin Multicontamination in Medicinal and Aromatic Herbs Sampled in Spain. J. Sci. Food Agric. 2009, 89, 1802–1807. [Google Scholar] [CrossRef]
- Al-juraifani, A.A. Natural Occurrence of Fungi and Aflatoxins of Cinnamon in the Saudi Arabia. Afr. J. Food Sci. 2011, 5, 460–465. [Google Scholar]
- Casas-Junco, P.P.; Ragazzo-Sánchez, J.A.; de Ascencio-Valle, F.J.; Calderón-Santoyo, M. Determination of Potentially Mycotoxigenic Fungi in Coffee (Coffea Arabica L.) from Nayarit. Food Sci. Biotechnol. 2017, 27, 891–898. [Google Scholar] [CrossRef]
- García-Moraleja, A.; Font, G.; Mañes, J.; Ferrer, E. Analysis of Mycotoxins in Coffee and Risk Assessment in Spanish Adolescents and Adults. Food Chem. Toxicol. 2015, 86, 225–233. [Google Scholar] [CrossRef]
- Su, C.; Hu, Y.; Gao, D.; Luo, Y.; Chen, A.J.; Jiao, X.; Gao, W. Occurrence of Toxigenic Fungi and Mycotoxins on Root Herbs from Chinese Markets. J. Food Prot. 2018, 81, 754–761. [Google Scholar] [CrossRef]
- Roy, A.K.; Chourasia, H.K. Mycoflora, Mycotoxin Producibility and Mycotoxins in Traditional Herbal Drugs from India. J. Gen. Appl. Microbiol. 1990, 36, 295–302. [Google Scholar] [CrossRef]
- Huang, X.; Wang, S.; Mao, D.; Miao, S.; Hu, Q.; Ji, S. Optimized QuEChERS Method Combined with UHPLC-MS/MS for the Simultaneous Determination of 15 Mycotoxins in Liquorice. J. AOAC Int. 2018, 101, 633–642. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; García-Campaña, A.M.; Gámiz-Gracia, L. Multiclass Mycotoxin Analysis in Silybum Marianum by Ultra High Performance Liquid Chromatography–Tandem Mass Spectrometry Using a Procedure Based on QuEChERS and Dispersive Liquid–Liquid Microextraction. J. Chromatogr. A 2013, 1282, 11–19. [Google Scholar] [CrossRef]
- Omurtag, G.Z.; Yazicioğilu, D. Determination of Fumonisins B1 and B2 in Herbal Tea and Medicinal Plants in Turkey by High-Performance Liquid Chromatography. J. Food Prot. 2004, 67, 1782–1786. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of Mycotoxin on Immune Response and Consequences for Pig Health. Anim. Nutr. 2016, 2, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B.; Ashiq, S.; Hussain, A.; Bashir, S.; Hussain, M. Evaluation of Mycotoxins, Mycobiota, and Toxigenic Fungi in Selected Medicinal Plants of Khyber Pakhtunkhwa, Pakistan. Fungal Biol. 2014, 118, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Powdery Mildew. Wikipedia. Available online: https://en.wikipedia.org/wiki/Powdery_mildew (accessed on 4 January 2022).
- Draz, I.S.; Esmail, S.M.; Abou-Zeid, M.A.E.-H.; Essa, T.A.E.-M. Powdery Mildew Susceptibility of Spring Wheat Cultivars as a Major Constraint on Grain Yield. Ann. Agric. Sci. 2019, 64, 39–45. [Google Scholar] [CrossRef]
- Jankovics, T.; Komáromi, J.; Fábián, A.; Jäger, K.; Vida, G.; Kiss, L. New Insights into the Life Cycle of the Wheat Powdery Mildew: Direct Observation of Ascosporic Infection in Blumeria graminis f. sp. Tritici. Phytopathology 2015, 105, 797–804. [Google Scholar] [CrossRef]
- Mukasa-Mugerwa, T.T.; Dames, J.F.; Rose, P.D. The Role of a Plant/Fungal Consortium in the Degradation of Bituminous Hard Coal. Biodegradation 2011, 22, 129–141. [Google Scholar] [CrossRef]
- Ashiq, S.; Hussain, M.; Ahmad, B. Natural Occurrence of Mycotoxins in Medicinal Plants: A Review. Fungal Genet. Biol. 2014, 66, 1–10. [Google Scholar] [CrossRef]
- Makhuvele, R.; Naidu, K.; Gbashi, S.; Thipe, V.C.; Adebo, O.A.; Njobeh, P.B. The Use of Plant Extracts and Their Phytochemicals for Control of Toxigenic Fungi and Mycotoxins. Heliyon 2020, 6, e05291. [Google Scholar] [CrossRef]
- Davies, C.R.; Wohlgemuth, F.; Young, T.; Violet, J.; Dickinson, M.; Sanders, J.-W.; Vallieres, C.; Avery, S.V. Evolving Challenges and Strategies for Fungal Control in the Food Supply Chain. Fungal Biol. Rev. 2021, 36, 15–26. [Google Scholar] [CrossRef]
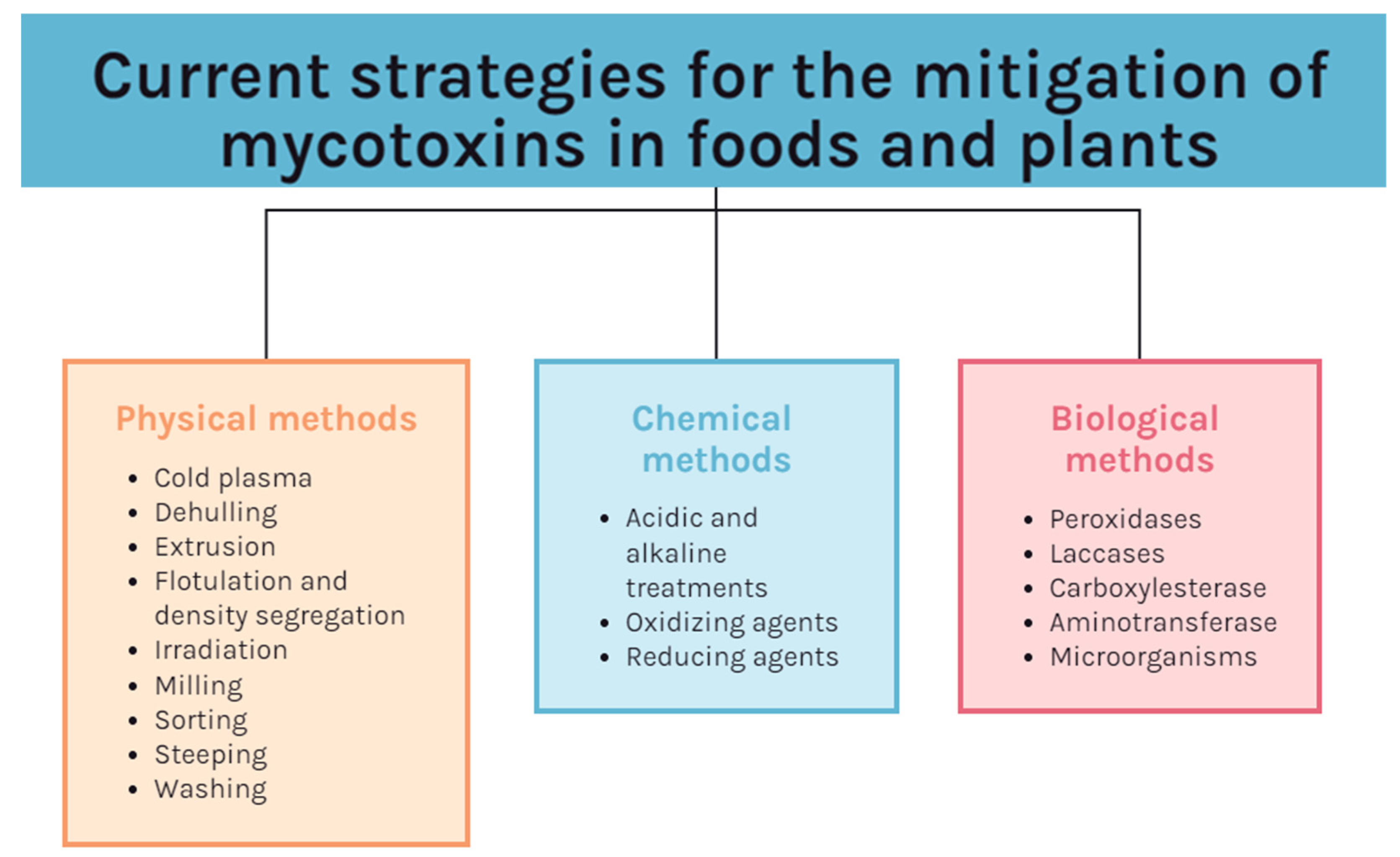
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of Food Processing and Detoxification Treatments on Mycotoxin Contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Vanhoutte, I.; Audenaert, K.; De Gelder, L. Biodegradation of Mycotoxins: Tales from Known and Unexplored Worlds. Front. Microbiol. 2016, 7, 561. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.; da Silva, D.M.; Alvarenga Pereira, R.G.F.; Paiva, L.C.; Prado, G.; Batista, L.R. Effect of Different Roasting Levels and Particle Sizes on Ochratoxin A Concentration in Coffee Beans. Food Control 2013, 34, 651–656. [Google Scholar] [CrossRef]
- Sehrawat, R.; Kaur, B.P.; Nema, P.K.; Tewari, S.; Kumar, L. Microbial Inactivation by High Pressure Processing: Principle, Mechanism and Factors Responsible. Food Sci. Biotechnol. 2021, 30, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Butz, P.; Funtenberger, S.; Haberditzl, T.; Tauscher, B. High Pressure Inactivation of Byssochlamys Nivea Ascospores and Other Heat Resistant Moulds. LWT-Food Sci. Technol. 1996, 29, 404–410. [Google Scholar] [CrossRef]
- Dijksterhuis, J.; Teunissen, P.G.M. Dormant Ascospores of Talaromyces Macrosporus Are Activated to Germinate after Treatment with Ultra High Pressure. J. Appl. Microbiol. 2004, 96, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Reyns, K.M.F.A.; Veraverbeke, E.A.; Michiels, C.W. Activation and Inactivation of Talaromyces Macrosporus Ascospores by High Hydrostatic Pressure. J. Food Prot. 2003, 66, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Aslam, R.; Makroo, H.A. High Pressure Extraction and Its Application in the Extraction of Bio-Active Compounds: A Review. J. Food Process Eng. 2019, 42, e12896. [Google Scholar] [CrossRef]
- Gonelimali, F.D.; Lin, J.; Miao, W.; Xuan, J.; Charles, F.; Chen, M.; Hatab, S.R. Antimicrobial Properties and Mechanism of Action of Some Plant Extracts Against Food Pathogens and Spoilage Microorganisms. Front. Microbiol. 2018, 9, 1639. [Google Scholar] [CrossRef] [PubMed]
- Patras, A.; Brunton, N.P.; Da Pieve, S.; Butler, F. Impact of High Pressure Processing on Total Antioxidant Activity, Phenolic, Ascorbic Acid, Anthocyanin Content and Colour of Strawberry and Blackberry Purées. Innov. Food Sci. Emerg. Technol. 2009, 10, 308–313. [Google Scholar] [CrossRef]
- Corrales, M.; García, A.F.; Butz, P.; Tauscher, B. Extraction of Anthocyanins from Grape Skins Assisted by High Hydrostatic Pressure. J. Food Eng. 2009, 90, 415–421. [Google Scholar] [CrossRef]
- Moreira, S.A.; Pintado, M.E.; Saraiva, J.A. Optimization of High Hydrostatic Pressure Assisted Extraction of Stinging Nettle Leaves Using Response Surface Methodology Experimental Design. Food Meas. 2020, 14, 2773–2780. [Google Scholar] [CrossRef]
- Briones-Labarca, V.; Plaza-Morales, M.; Giovagnoli-Vicuña, C.; Jamett, F. High Hydrostatic Pressure and Ultrasound Extractions of Antioxidant Compounds, Sulforaphane and Fatty Acids from Chilean Papaya (Vasconcellea pubescens) Seeds: Effects of Extraction Conditions and Methods. LWT-Food Sci. Technol. 2015, 60, 525–534. [Google Scholar] [CrossRef]
- Sun, W.; Li, J.; Ramaswamy, H.S.; Yu, Y.; Wang, C.; Zhu, S. Adiabatic Compression Heating of Selected Organic Solvents under High Pressure Processing. High Press. Res. 2018, 38, 325–336. [Google Scholar] [CrossRef]
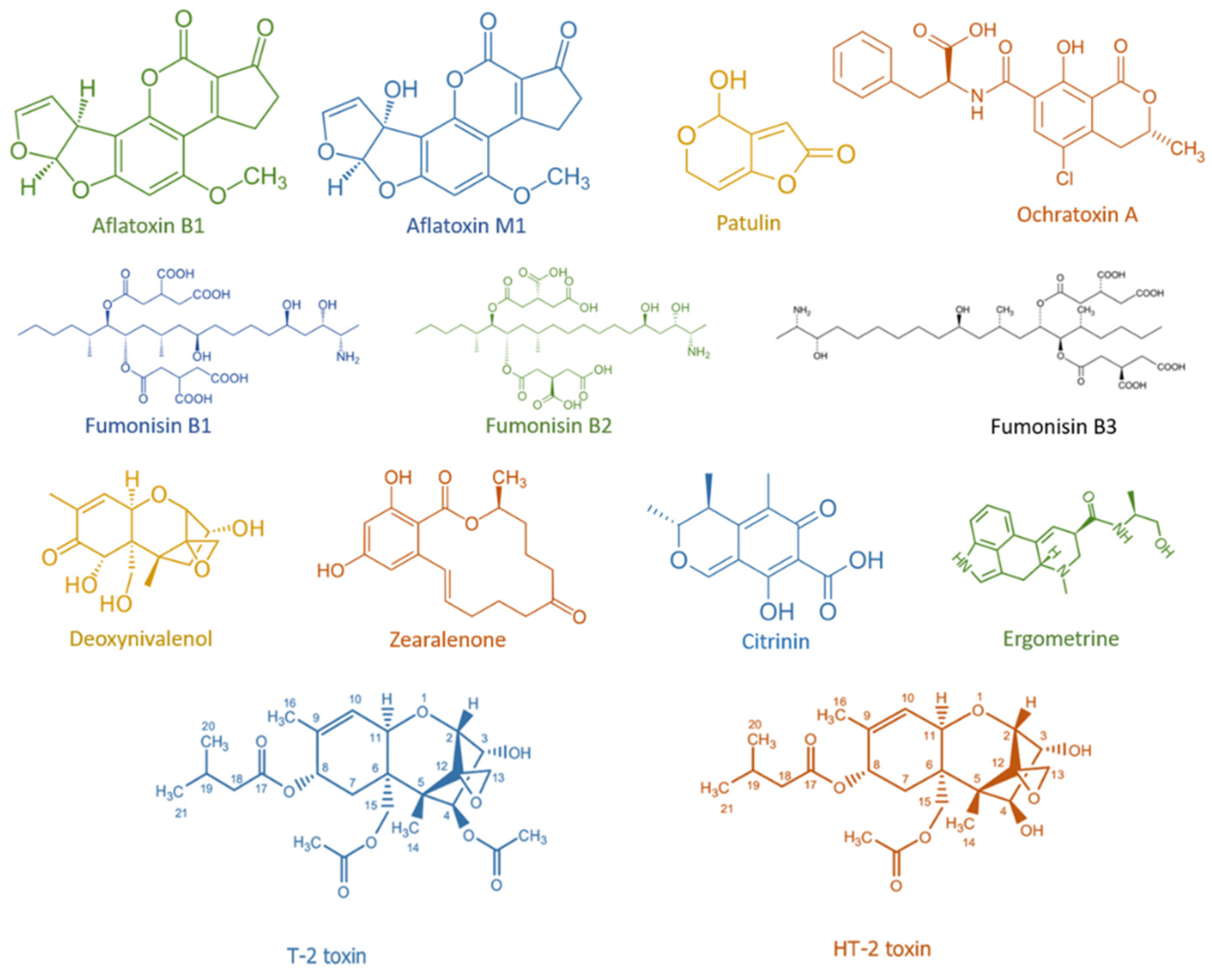


| Plant (Part of the Plant) | Microorganism | Mycotoxin | Reference |
|---|---|---|---|
| Chamomile | - | Aflatoxin | [23] |
| Citrinin | [24] | ||
| Fumonisin | |||
| Cinamomum zeylanicum blume | Aspergillus spp. Penicilium spp. | Aflatoxin B1, B2 ang G2 | [25] |
| Roasted coffee | Aspergillus spp. Penicilium spp. | 15-acetyldeoxynivalenol, Deoxynivalenol, Neosolaniol, Fumonisin B1 and Ochratoxin A | [26,27] |
| Ginseng root | - | Aflatoxin B1 and B2 | [28] |
| Ochratoxin | |||
| Glycyrrhizaglabra (root) | Aspergillus flavus | Aflatoxin B1 and B2 | [29] |
| Fusarium oxysporum | Zearalenone | ||
| Fumonisin B1 | |||
| - | Citrinin | [30] | |
| T-2 and HT-2 | [23] | ||
| Milk thistle | - | Aflatoxin | [31] |
| T-2 | |||
| HT-2 | |||
| Fumonisin | |||
| Mint | - | Aflatoxin | [24] |
| Citrinin | |||
| Fumonisin | [32] | ||
| Tribulus terrestris (seeds) | Aspergillus ochraceus | Ochratoxin A | [29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinto, C.A.; Holovicova, M.; Habanova, M.; Lima, V.; Duarte, R.V.; Barba, F.J.; Saraiva, J.A. Effects of High Hydrostatic Pressure on Fungal Spores and Plant Bioactive Compounds. Encyclopedia 2022, 2, 1453-1463. https://doi.org/10.3390/encyclopedia2030098
Pinto CA, Holovicova M, Habanova M, Lima V, Duarte RV, Barba FJ, Saraiva JA. Effects of High Hydrostatic Pressure on Fungal Spores and Plant Bioactive Compounds. Encyclopedia. 2022; 2(3):1453-1463. https://doi.org/10.3390/encyclopedia2030098
Chicago/Turabian StylePinto, Carlos A., Maria Holovicova, Marta Habanova, Vasco Lima, Ricardo V. Duarte, Francisco J. Barba, and Jorge A. Saraiva. 2022. "Effects of High Hydrostatic Pressure on Fungal Spores and Plant Bioactive Compounds" Encyclopedia 2, no. 3: 1453-1463. https://doi.org/10.3390/encyclopedia2030098
APA StylePinto, C. A., Holovicova, M., Habanova, M., Lima, V., Duarte, R. V., Barba, F. J., & Saraiva, J. A. (2022). Effects of High Hydrostatic Pressure on Fungal Spores and Plant Bioactive Compounds. Encyclopedia, 2(3), 1453-1463. https://doi.org/10.3390/encyclopedia2030098







