Substance Release from Polyelectrolyte Microcapsules
Definition
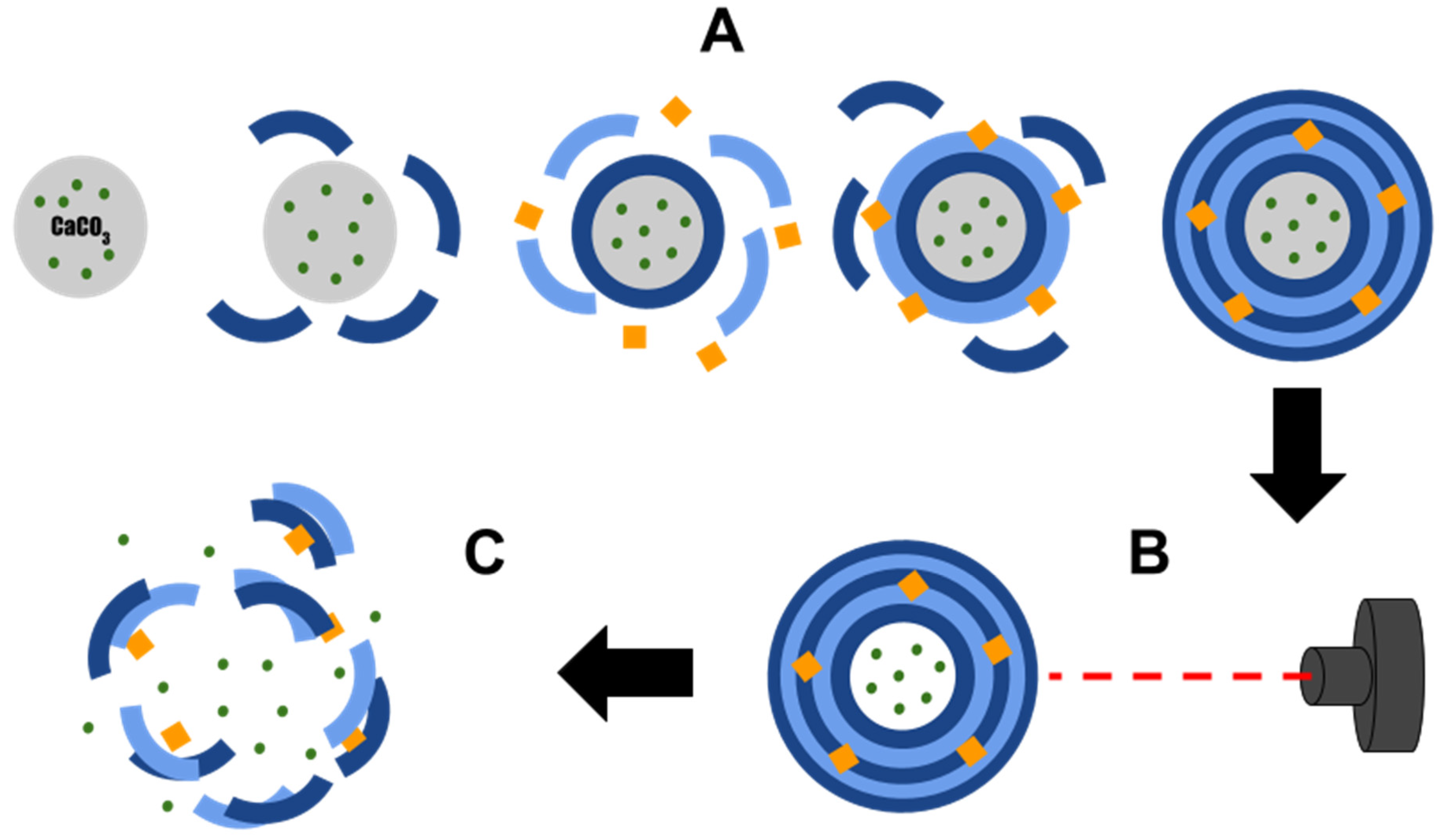
:1. Introduction
2. History
3. The Ways of Encapsulated Substance Release
3.1. Acidity of the Solution (pH)
3.2. Ionic Strength of the Solution
3.3. Glucose Content
3.4. Light Exposure
3.5. Ultrasound
3.6. Magnetic Field
3.7. Redox
3.8. Enzymatic Digestion
3.9. Osmotic Pressure
3.10. Bacterially Driven Decapsulation
4. Practical Relevance and Applications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Entry Link on the Encyclopedia Platform
Abbreviations
| PAD | Poly 3-acrylaminophenylboronic acid: dimethyl aminoethyl acrylate |
| PAH-BOH | Amide bonds between the carboxylic groups of acrilic acid and aminophenylboronic acid hemisulfate |
| PSS | Poly(sodium 4-styrenesulfonate) |
| DDAB | Dioctadecyldimethylammonium bromide |
| MF | Melamine formaldehyde |
| PAH-BP | Benzophenone-modified PAH |
| Bt | Benzotriazole |
| PDADMAC | Poly(diallyldimethylammonium chloride) |
| PMA | Poly(methacrylic acid) |
| NRs | gold nanorods |
| DS | Dextran sulfate |
| PVPON | Poly(vinylpyrrolidone) |
| PMASH | Poly(methacrylic acid) (PMA) conjugated with cysteamine (NH2-(CH2)2)-SH) |
| PLL | Poly(L-lysine) |
| PGA | Poly(L-glutamic acid) |
| PSS-Rh | Rhodamine B-labelled PSS |
| p(HPMA-DMAE | Poly(hydroxypropylmethacrylamide dimethylaminoethyl) |
| CT | Chitosan |
| ALG | Alginate |
| (dex-HEMA-DMAEMA) | Microgel dextran-hydroxyethylmethacrylate dimethylaminoethyl methacrylate |
References
- Decher, G.D.; Hong, J.-D. Buildup of ultrathin multilayer films by a self-assembly process, 1 consecutive adsorption of anionic and cationic bipolar amphiphiles on charged surfaces. In Makromolekulare Chemie. Macromolecular Symposia; Hüthig & Wepf Verlag: Basel, Switzerland, 1991; pp. 321–327. [Google Scholar]
- Decher, G.; Schmitt, J. Fine-Tuning of the film thickness of ultrathin multilayer films composed of consecutively alternating layers of anionic and cationic polyelectrolytes. In Progress in Colloid & Polymer Science; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2007; pp. 160–164. [Google Scholar]
- Donath, E.; Sukhorukov, G.B.; Caruso, F.; Davis, S.A.; Möhwald, H. Novel Hollow Polymer Shells by Colloid-Templated Assembly of Polyelectrolytes. Angew. Chemie Int. 1998, 37, 2201–2205. [Google Scholar] [CrossRef]
- Wang, C.; Ye, S.; Sun, Q.; He, C.; Ye, W.; Liu, X.; Tong, Z. Microcapsules for controlled release fabricated via layer-by-layer self-assembly of polyelectrolytes. J. Exp. Nanosci. 2008, 3, 133–145. [Google Scholar] [CrossRef]
- Schüler, C.; Caruso, F. Decomposable Hollow Biopolymer-Based Capsules. Biomacromolecules 2001, 2, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Antipov, A.A.; Sukhorukov, G.B.; Leporatti, S.; Radtchenko, I.L.; Donath, E.; Möhwald, H. Polyelectrolyte multilayer capsule permeability control. Colloids Surf. A Physicochem. Eng. Asp. 2002, 198–200, 535–541. [Google Scholar] [CrossRef]
- Ibarz, G.; Dähne, L.; Donath, E.; Möhwald, H. Controlled Permeability of Polyelectrolyte Capsules via Defined Annealing. Chem. Mater. 2002, 14, 4059–4062. [Google Scholar] [CrossRef]
- Tao, X.; Li, J.; Möhwald, H. Self-Assembly, Optical Behavior, and Permeability of a Novel Capsule Based on an Azo Dye and Polyelectrolytes. Chem. A Eur. J. 2004, 10, 3397–3403. [Google Scholar] [CrossRef]
- Lu, Z.; Prouty, M.D.; Guo, Z.; Golub, V.; Kumar, C.S.S.R.; Lvov, Y.M. Magnetic Switch of Permeability for Polyelectrolyte Microcapsules Embedded with Co@Au Nanoparticles. Langmuir 2005, 21, 2042–2050. [Google Scholar] [CrossRef]
- Gorin, D.A.; Shchukin, D.G.; Mikhailov, A.I.; Köhler, K.; Sergeev, S.A.; Portnov, S.A.; Taranov, I.; Kislov, V.V.; Sukhorukov, G.B. Effect of microwave radiation on polymer microcapsules containing inorganic nanoparticles. Tech. Phys. Lett. 2006, 32, 70–72. [Google Scholar] [CrossRef]
- Shchukin, D.G.; Gorin, D.A.; Möhwald, H. Ultrasonically Induced Opening of Polyelectrolyte Microcontainers. Langmuir 2006, 22, 7400–7404. [Google Scholar] [CrossRef]
- Musin, E.V.; Kim, A.L.; Dubrovskii, A.V.; Kudryashova, E.B.; Tikhonenko, S.A. Decapsulation of Dextran by Destruction of Polyelectrolyte Microcapsule Nanoscale Shell by Bacillus subtilis Bacteria. Nanomaterials 2019, 10, 12. [Google Scholar] [CrossRef]
- Picart, C.; Schneider, A.; Etienne, O.; Mutterer, J.; Schaaf, P.; Egles, C.; Jessel, N.; Voegel, J.-C. Controlled Degradability of Polysaccharide Multilayer Films In Vitro and In Vivo. Adv. Funct. Mater. 2005, 15, 1771–1780. [Google Scholar] [CrossRef]
- Shen, H.-J.; Shi, H.; Ma, K.; Xie, M.; Tang, L.-L.; Shen, S.; Li, B.; Wang, X.-S.; Jin, Y. Polyelectrolyte capsules packaging BSA gels for pH-controlled drug loading and release and their antitumor activity. Acta Biomater. 2013, 9, 6123–6133. [Google Scholar] [CrossRef]
- Antipov, A.A.; Sukhorukov, G.B.; Möhwald, H. Influence of the Ionic Strength on the Polyelectrolyte Multilayers’ Permeability. Langmuir 2003, 19, 2444–2448. [Google Scholar] [CrossRef]
- De Geest, B.G.; Jonas, A.M.; Demeester, J.; De Smedt, S.C. Glucose-Responsive Polyelectrolyte Capsules. Langmuir 2006, 22, 5070–5074. [Google Scholar] [CrossRef] [PubMed]
- Levy, T.; Déjugnat, C.; Sukhorukov, G.B. Polymer Microcapsules with Carbohydrate-Sensitive Properties. Adv. Funct. Mater. 2008, 18, 1586–1594. [Google Scholar] [CrossRef]
- Katagiri, K.; Koumoto, K.; Iseya, S.; Sakai, M.; Matsuda, A.; Caruso, F. Tunable UV-Responsive Organic−Inorganic Hybrid Capsules. Chem. Mater. 2009, 21, 195–197. [Google Scholar] [CrossRef]
- Koo, H.Y.; Lee, H.-J.; Kim, J.K.; Choi, W.S. UV-triggered encapsulation and release from polyelectrolyte microcapsules decorated with photoacid generators. J. Mater. Chem. 2010, 20, 3932–3937. [Google Scholar] [CrossRef]
- Park, M.-K.; Deng, S.; Advincula, R.C. Sustained Release Control via Photo-Cross-Linking of Polyelectrolyte Layer-by-Layer Hollow Capsules. Langmuir 2005, 21, 5272–5277. [Google Scholar] [CrossRef]
- Skirtach, A.G.; Antipov, A.A.; Shchukin, D.G.; Sukhorukov, G.B. Remote Activation of Capsules Containing Ag Nanoparticles and IR Dye by Laser Light. Langmuir 2004, 20, 6988–6992. [Google Scholar] [CrossRef]
- Skorb, E.V.; Sviridov, D.V.; Shchukin, D.G.; Borisenko, V.E.; Gaponenko, S.V.; Gurin, V.S. Light-controllable coatings for corrosion protection. Phys. Chem. Appl. Nanostructures 2009, 515–518. [Google Scholar] [CrossRef]
- Skirtach, A.; Karageorgiev, P.; Bédard, M.F.; Sukhorukov, G.B.; Möhwald, H. Reversibly Permeable Nanomembranes of Polymeric Microcapsules. J. Am. Chem. Soc. 2008, 130, 11572–11573. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Vijay, J.; Ganesh, M.; Sundaramurthy, A. Multilayer capsules encapsulating nimbin and doxorubicin for cancer chemo-photothermal therapy. Int. J. Pharm. 2020, 582, 119350. [Google Scholar] [CrossRef] [PubMed]
- Klitzing, R.; Möhwald, H. A realistic diffusion model for ultrathin polyelectrolyte films. Macromolecules 1996, 29, 6901–6906. [Google Scholar] [CrossRef]
- Skirtach, A.G.; De Geest, B.G.; Mamedov, A.; Antipov, A.A.; Kotov, N.A.; Sukhorukov, G.B. Ultrasound stimulated release and catalysis using polyelectrolyte multilayer capsules. J. Mater. Chem. 2007, 17, 1050–1054. [Google Scholar] [CrossRef]
- Anandhakumar, S.; Mahalakshmi, V.; Raichur, A.M. Silver nanoparticles modified nanocapsules for ultrasonically activated drug delivery. Mater. Sci. Eng. C 2012, 32, 2349–2355. [Google Scholar] [CrossRef]
- Zheng, C.; Ding, Y.; Liu, X.; Wu, Y.; Ge, L. Highly magneto-responsive multilayer microcapsules for controlled release of insulin. Int. J. Pharm. 2014, 475, 17–24. [Google Scholar] [CrossRef]
- Katagiri, K.; Nakamura, M.; Koumoto, K. Magnetoresponsive Smart Capsules Formed with Polyelectrolytes, Lipid Bilayers and Magnetic Nanoparticles. ACS Appl. Mater. Interfaces 2010, 2, 768–773. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, C.; Li, D.; Long, Y.; Song, K.; Tung, C.-H. Magnetic Compression of Polyelectrolyte Microcapsules for Controlled Release. Langmuir 2015, 31, 11195–11199. [Google Scholar] [CrossRef]
- Carregal-Romero, S.; Guardia, P.; Yu, X.; Hartmann, R.; Pellegrino, T.; Parak, W.J. Magnetically triggered release of molecular cargo from iron oxide nanoparticle loaded microcapsules. Nanoscale 2015, 7, 570–576. [Google Scholar] [CrossRef]
- Zelikin, A.N.; Quinn, J.F.; Caruso, F. Disulfide Cross-Linked Polymer Capsules: En Route to Biodeconstructible Systems. Biomacromolecules 2006, 7, 27–30. [Google Scholar] [CrossRef]
- De Rose, R.; Zelikin, A.N.; Johnston, A.P.R.; Sexton, A.; Chong, S.-F.; Cortez, C.; Mulholland, W.; Caruso, F.; Kent, S.J. Binding, Internalization, and Antigen Presentation of Vaccine-Loaded Nanoengineered Capsules in Blood. Adv. Mater. 2008, 20, 4698–4703. [Google Scholar] [CrossRef]
- De Geest, B.G.; Vandenbroucke, R.E.; Guenther, A.M.; Sukhorukov, G.B.; Hennink, W.E.; Sanders, N.N.; Demeester, J.; De Smedt, S.C. Intracellularly Degradable Polyelectrolyte Microcapsules. Adv. Mater. 2006, 18, 1005–1009. [Google Scholar] [CrossRef]
- De Koker, S.; De Geest, B.G.; Cuvelier, C.; Ferdinande, L.; Deckers, W.; Hennink, W.E.; De Smedt, S.C.; Mertens, N. In vivo Cellular Uptake, Degradation, and Biocompatibility of Polyelectrolyte Microcapsules. Adv. Funct. Mater. 2007, 17, 3754–3763. [Google Scholar] [CrossRef]
- Itoh, Y.; Matsusaki, M.; Kida, T.; Akashi, M. Enzyme-Responsive Release of Encapsulated Proteins from Biodegradable Hollow Capsules. Biomacromolecules 2006, 7, 2715–2718. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ye, S.; Dai, L.; Liu, X.; Tong, Z. Enzymatic desorption of layer-by-layer assembled multilayer films and effects on the release of encapsulated indomethacin microcrystals. Carbohydr. Res. 2007, 342, 2237–2243. [Google Scholar] [CrossRef] [PubMed]
- De Geest, B.G.; Déjugnat, C.; Sukhorukov, G.B.; Braeckmans, K.; De Smedt, S.C.; Demeester, J. Self-Rupturing Microcapsules. Adv. Mater. 2005, 17, 2357–2361. [Google Scholar] [CrossRef]
- De Geest, B.; Dejugnat, C.; Verhoeven, E.; Sukhorukov, G.; Jonas, A.; Plain, J.; Demeester, J.; De Smedt, S. Layer-by-layer coating of degradable microgels for pulsed drug delivery. J. Control. Release 2006, 116, 159–169. [Google Scholar] [CrossRef]
- Rae, J.; Ashokkumar, M.; Eulaerts, O.; von Sonntag, C.; Reisse, J.; Grieser, F. Estimation of ultrasound induced cavitation bubble temperatures in aqueous solutions. Ultrason. Sonochemistry 2005, 12, 325–329. [Google Scholar] [CrossRef]
- Kolesnikova, T.A.; Gorin, D.A.; Fernandes, P.; Kessel, S.; Khomutov, G.B.; Fery, A.; Shchukin, D.G.; Möhwald, H. Nanocomposite Microcontainers with High Ultrasound Sensitivity. Adv. Funct. Mater. 2010, 20, 1189–1195. [Google Scholar] [CrossRef]
- De Geest, B.G.; Skirtach, A.G.; Mamedov, A.A.; Antipov, A.A.; Kotov, N.A.; De Smedt, S.C.; Sukhorukov, G.B. Ultrasound-Triggered Release from Multilayered Capsules. Small 2007, 3, 804–808. [Google Scholar] [CrossRef]
- Caruso, F.; Susha, A.S.; Giersig, M.; Möhwald, H. Magnetic Core-Shell Particles: Preparation of Magnetite Multilayers on Polymer Latex Microspheres. Adv. Mater. 1999, 11, 950–953. [Google Scholar] [CrossRef]
- Katagiri, K.; Imai, Y.; Koumoto, K. Variable on-demand release function of magnetoresponsive hybrid capsules. J. Colloid Interface Sci. 2011, 361, 109–114. [Google Scholar] [CrossRef]
- Hu, S.-H.; Tsai, C.-H.; Liao, C.-F.; Liu, D.-M.; Chen, S.-Y. Controlled Rupture of Magnetic Polyelectrolyte Microcapsules for Drug Delivery. Langmuir 2008, 24, 11811–11818. [Google Scholar] [CrossRef]
- Degen, P.; Peschel, S.; Rehage, H. Stimulated aggregation, rotation, and deformation of magnetite-filled microcapsules in external magnetic fields. Colloid Polym. Sci. 2008, 286, 865–871. [Google Scholar] [CrossRef]
- Long, Y.; Liu, C.; Zhao, B.; Song, K.; Yang, G.; Tung, C.-H. Bio-inspired controlled release through compression–relaxation cycles of microcapsules. NPG Asia Mater. 2015, 7, e148. [Google Scholar] [CrossRef]
- Patel, R.; Upadhyay, R.V.; Mehta, R. Microscopic observation of magnetodeformational effects in magnetic nanocomposite micelles. J. Phys. Condens. Matter 2008, 20, 204116. [Google Scholar] [CrossRef] [PubMed]
- Haynie, D.T.; Palath, N.; Liu, Y.; Li, A.B.; Pargaonkar, N. Biomimetic Nanostructured Materials: Inherent Reversible Stabilization of Polypeptide Microcapsules. Langmuir 2005, 21, 1136–1138. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Jeong, Y.; Park, T.G. Shell Cross-Linked Hyaluronic Acid/Polylysine Layer-by-Layer Polyelectrolyte Microcapsules Prepared by Removal of Reducible Hyaluronic Acid Microgel Cores. Biomacromolecules 2007, 8, 3705–3711. [Google Scholar] [CrossRef] [PubMed]
- Szarpak, A.; Cui, D.; Dubreuil, F.; De Geest, B.G.; De Cock, L.J.; Picart, C.; Auzély-Velty, R. Designing Hyaluronic Acid-Based Layer-by-Layer Capsules as a Carrier for Intracellular Drug Delivery. Biomacromolecules 2010, 11, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, K.; Tripathy, J.; Raichur, A.M. Dual enzyme responsive microcapsules simulating an “OR” logic gate for biologically triggered drug delivery applications. Chem. Commun. 2013, 49, 5390–5392. [Google Scholar] [CrossRef]
- Cardoso, M.J.; Caridade, S.G.; Costa, R.R.; Mano, J.F. Enzymatic Degradation of Polysaccharide-Based Layer-by-Layer Structures. Biomacromolecules 2016, 17, 1347–1357. [Google Scholar] [CrossRef]
- Borodina, T.; Markvicheva, E.; Kunizhev, S.; Möhwald, H.; Sukhorukov, G.B.; Kreft, O. Controlled Release of DNA from Self-Degrading Microcapsules. Macromol. Rapid Commun. 2007, 28, 1894–1899. [Google Scholar] [CrossRef]
- Kim, B.-S.; Choi, J.-W. Polyelectrolyte multilayer microcapsules: Self-assembly and toward biomedical applications. Biotechnol. Bioprocess Eng. 2007, 12, 323–332. [Google Scholar] [CrossRef]
- Johnston, A.; Cortez, C.; Angelatos, A.S.; Caruso, F. Layer-by-layer engineered capsules and their applications. Curr. Opin. Colloid Interface Sci. 2006, 11, 203–209. [Google Scholar] [CrossRef]
- Santos, J.L.; Nouri, A.; Fernandes, T.; Rodrigues, J.; Tomás, H. Gene delivery using biodegradable polyelectrolyte microcapsules prepared through the layer-by-layer technique. Biotechnol. Prog. 2012, 28, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Zheludkevich, M.L.; Shchukin, D.G.; Yasakau, K.A.; Möhwald, H.; Ferreira, M.G.S. Anticorrosion Coatings with Self-Healing Effect Based on Nanocontainers Impregnated with Corrosion Inhibitor. Chem. Mater. 2007, 19, 402–411. [Google Scholar] [CrossRef]
- Kartsonakis, I.A.; Danilidis, I.L.; Pappas, G.S.; Kordas, G.C. Encapsulation and release of corrosion inhibitors into titania nanocontainers. J. Nanosci. Nanotechnol. 2010, 10, 5912–5920. [Google Scholar] [CrossRef]
- Feng, Y.; Cheng, Y.F. An intelligent coating doped with inhibitor-encapsulated nanocontainers for corrosion protection of pipeline steel. Chem. Eng. J. 2017, 315, 537–551. [Google Scholar] [CrossRef]
- Shchukin, D.G.; Lamaka, S.V.; Yasakau, K.A.; Zheludkevich, M.L.; Ferreira, M.G.S.; Möhwald, H. Active Anticorrosion Coatings with Halloysite Nanocontainers. J. Phys. Chem. C 2008, 112, 958–964. [Google Scholar] [CrossRef]
- Lvov, Y.M.; Shchukin, D.; Moehwald, H.; Price, R.R. Halloysite Clay Nanotubes for Controlled Release of Protective Agents. ACS Nano 2008, 2, 814–820. [Google Scholar] [CrossRef]
- Balaskas, A.; Kartsonakis, I.; Tziveleka, L.A.; Kordas, G. Improvement of anti-corrosive properties of epoxy-coated AA 2024-T3 with TiO2 nanocontainers loaded with 8-hydroxyquinoline. Prog. Org. Coat. 2012, 74, 418–426. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, J. Encapsulation of the Herbicide Picloram by Using Polyelectrolyte Biopolymers as Layer-by-Layer Materials. J. Agric. Food Chem. 2013, 61, 3789–3796. [Google Scholar] [CrossRef] [PubMed]

| The Initiator of Substance Release | Microcapsule’s Constitution (First Layer/Second Layer)Number of Repeats | Released Substance | Citation |
|---|---|---|---|
| Acidity of the solution (pH) | BSA/(PAH/PSS)5 | Doxorubicin | [14] |
| Ionic strength of the solution | (PSS/PAH)9/PSS | Fluorescein | [15] |
| Glucose | (PAD/PSS)6 | FITC-BSA | [16] |
| (Mannan/PAH-BOH)7 | TRITC-BSA | [17] | |
| Light | (PSS/PDADMAC)5/PSS/DDAB/SiO2-TiO2 | Phenol red | [18] |
| MF/(PSS/PAH)4PSS | FITC-dextran | [19] | |
| (PAH-BP/PSS)4 | Rhodamine B | [20] | |
| (PSS/PAH)2-Ag-(PSS/PAH)2 | - | [21] | |
| Bt-SiO2/PEI/PSS/PEI/PSS Bt-TiO2/PEI/PSS/ PEI/PSS | Benzotriazole | [22] | |
| (PDADMAC/Au/PSS)4 | Alexa Fluor 555 dextran | [23] | |
| (PAH/PMA)4 /NR | Nimbin and Doxorubicin | [24] | |
| Ultrasound | (PSS/PAH)2/Ag/(PSS/PAH)2 (PSS/PAH)2/Ag/(PSS/PAH)8 | FITC-PAH | [25] |
| (PAH/DS)3/AgNPs/(PAH/DS) | FITC-dextran | [26] | |
| (PSS/PAH)4 (AuNP/PAH)4 | FITC-dextran | [27] | |
| (PAH/PSS)2(PAH/Fe3O4)4(PAH/PSS)2 | FITC-dextran | [11] | |
| Magnetic field | (PAH/ Fe3O4 NPs)4 | Insulin | [28] |
| (PSS/PAH)5/Pd | Phenol red | [29] | |
| (PDADMAC/PSS)6 | FITC-dextran | [30] | |
| (PSS/PAH)/(PSS/P(Am-DDA)/ NPs/ (PAH)(PSS/PAH)2 | Cascade Blue-labelled dextran | [31] | |
| Redox | (PVPON/PMASH)4 | FITC-transferrin | [32] |
| (PSS-Rh/PAH)2/(PSS/PAH)3 (PSS-Rh/PAH)2/(PSS/PAH)3/PSS (PSS-Rh/PAH)2/(PGA/PLL)4 (PSS-Rh/PAH)2/(PGA/PLL)4/PGA | 9-amino-acid peptide KP9 | [33] | |
| Enzymatic digestion | (PSS/P(HPMA-DMAE))4 | FITC-dextran | [34] |
| (DS/PLA)4 | Rhodamine isothiocyanate | [35] | |
| (CT/DS)3 | FITC-albumin | [36] | |
| (CHI/ALG)5 | Indomethacin | [37] | |
| Osmotic pressure | (Dex-HEMA-DMAEMA)/(PSS/PAH)3 | FITC-dextran | [38] |
| (Dex-HEMA-DMAEMA)/(PSS/PAH)3 | TRITC-dextran | [39] | |
| Spore germination | (spore forms of Bacillus subtilis)/(PSS/PAH)3 | FITC-dextran | [12] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musin, E.V.; Kim, A.L.; Tikhonenko, S.A. Substance Release from Polyelectrolyte Microcapsules. Encyclopedia 2022, 2, 428-440. https://doi.org/10.3390/encyclopedia2010026
Musin EV, Kim AL, Tikhonenko SA. Substance Release from Polyelectrolyte Microcapsules. Encyclopedia. 2022; 2(1):428-440. https://doi.org/10.3390/encyclopedia2010026
Chicago/Turabian StyleMusin, Egor V., Aleksandr L. Kim, and Sergey A. Tikhonenko. 2022. "Substance Release from Polyelectrolyte Microcapsules" Encyclopedia 2, no. 1: 428-440. https://doi.org/10.3390/encyclopedia2010026
APA StyleMusin, E. V., Kim, A. L., & Tikhonenko, S. A. (2022). Substance Release from Polyelectrolyte Microcapsules. Encyclopedia, 2(1), 428-440. https://doi.org/10.3390/encyclopedia2010026






