Polymeric Micelles Co-Loaded with Cannabidiol, Celecoxib, and Temozolomide—Early-Stage Assessment of Anti-Glioma Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Polymeric Micelles
2.2.1. Surfactant Selection
2.2.2. Optimization of Micelle Preparation Methods
2.2.3. Incorporation of Active Substances
2.3. Characterization of Micelles
2.3.1. Particle Size, Polydispersity Index
2.3.2. Zeta Potential
2.3.3. Stability Study
2.3.4. Encapsulation Efficiency and Loading Capacity
2.3.5. In Vitro Drug Release Tests from Loaded Micelles
2.4. Biological Activities
2.4.1. Cell Line Culture
2.4.2. MTT Viability Assay
2.4.3. Cytosolic and Nuclear Fraction Extraction
2.4.4. Apoptosis Analysis
2.4.5. Western Blot Analysis
2.4.6. Statistical Analysis
3. Results
3.1. Optimization of Micellar Formulations and Selection of the Final System
3.2. Physicochemical Characterization and Stability Testing of Micelles Containing Active Substances
3.3. Assessment of Encapsulation Efficiency (EE%) and Loading Capacity (DL%) of Selected Drugs
3.4. In Vitro Release Profile of Active Substances from Polymeric Micelles
3.5. MTT Cytotoxicity Assay on Optimized Micelles
3.6. Apoptosis Assessment in U-87 MG and U-138 MG Cells
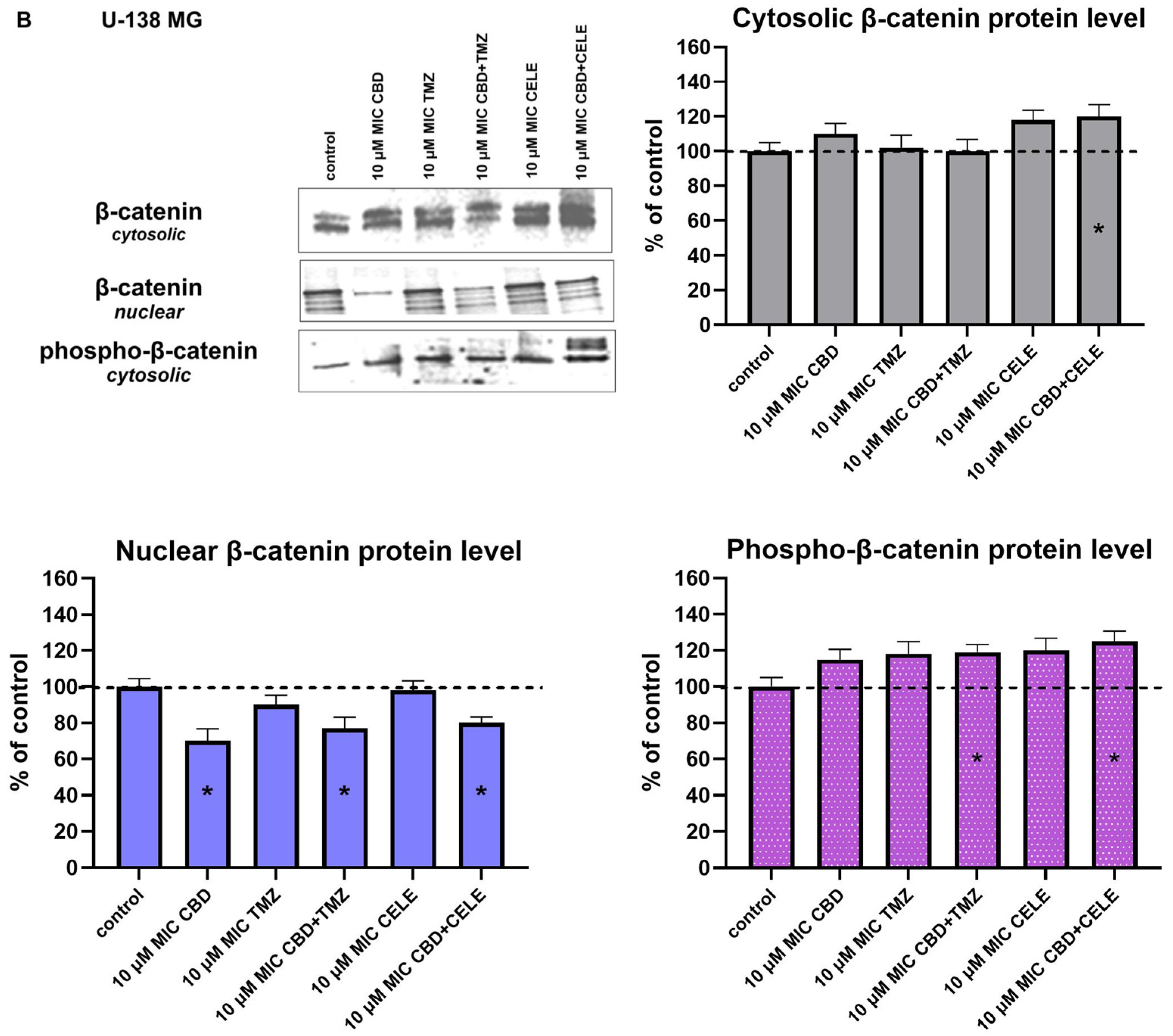
3.7. CBD + TMZ Co-Loaded Micelles Attenuate Wnt/β-Catenin Signaling in Malignant Glioma Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gilard, V.; Tebani, A.; Dabaj, I.; Laquerrière, A.; Fontanilles, M.; Derrey, S.; Marret, S.; Bekri, S. Diagnosis and Management of Glioblastoma: A Comprehensive Perspective. J. Pers. Med. 2021, 11, 258. [Google Scholar] [CrossRef]
- Pouyan, A.; Ghorbanlo, M.; Eslami, M.; Jahanshahi, M.; Ziaei, E.; Salami, A.; Mokhtari, K.; Shahpasand, K.; Farahani, N.; Meybodi, T.E.; et al. Glioblastoma multiforme: Insights into pathogenesis, key signaling pathways, and therapeutic strategies. Mol. Cancer 2025, 24, 58. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A Summary. Neuro-Oncol. 2021, 23, 1231–1251. [Google Scholar] [PubMed]
- Boylan, J.; Byers, E.; Kelly, D.F. The Glioblastoma Landscape: Hallmarks of Disease, Therapeutic Resistance, and Treatment Opportunities. Med. Res. Arch. 2023, 11, 10. [Google Scholar] [CrossRef]
- Wiwatchaitawee, K.; Quarterman, J.C.; Geary, S.M.; Salem, A.K. Enhancement of Therapies for Glioblastoma (GBM) Using Nanoparticle-Based Delivery Systems. AAPS PharmSciTech 2021, 22, 71. [Google Scholar] [CrossRef]
- Janjua, T.I.; Rewatkar, P.; Ahmed-Cox, A.; Saeed, I.; Mansfeld, F.M.; Kulshreshtha, R.; Kumeria, T.; Ziegler, D.S.; Kavallaris, M.; Mazzieri, R.; et al. Frontiers in the Treatment of Glioblastoma: Past, Present and Emerging. Adv. Drug Deliv. Rev. 2021, 171, 108–138. [Google Scholar] [CrossRef]
- Norollahi, S.E.; Yousefi, B.; Nejatifar, F.; Yousefzadeh-Chabok, S.; Rashidy-Pour, A.; Samadani, A.A. Practical immunomodulatory landscape of glioblastoma multiforme (GBM) therapy. J. Egypt. Natl. Cancer Inst. 2024, 36, 33. [Google Scholar] [CrossRef] [PubMed]
- Di Filippo, L.D.; Duarte, J.L.; Luiz, M.T.; de Araújo, J.T.C.; Chorilli, M. Drug Delivery Nanosystems in Glioblastoma Multiforme Treatment: Current State of the Art. Curr. Neuropharmacol. 2021, 19, 787–812. [Google Scholar] [CrossRef]
- Tomar, V.S.; Patil, V.; Somasundaram, K. Temozolomide induces activation of Wnt/β-catenin signaling in glioma cells via PI3K/Akt pathway: Implications in glioma therapy. Cell Biol. Toxicol. 2020, 36, 273–278. [Google Scholar]
- Gao, X.Y.; Zang, J.; Zheng, M.H.; Zhang, Y.F.; Yue, K.Y.; Cao, X.L.; Cao, Y.; Li, X.X.; Han, H.; Jiang, X.F.; et al. Temozolomide Treatment Induces HMGB1 to Promote the Formation of Glioma Stem Cells via the TLR2/NEAT1/Wnt Pathway in Glioblastoma. Front. Cell Dev. Biol. 2021, 9, 620883. [Google Scholar] [CrossRef]
- Rybarczyk, A.; Majchrzak-Celińska, A.; Krajka-Kuźniak, V. Synergistic combination of cannabidiol and celecoxib or 2,5-dimethylcelecoxib exerts oxidative stress-mediated cytotoxicity and mitigates glioblastoma invasiveness. Acta Biochim. Pol. 2025, 72, 15062. [Google Scholar] [CrossRef]
- Majchrzak-Celińska, A.; Misiorek, J.O.; Kruhlenia, N.; Przybyl, L.; Kleszcz, R.; Rolle, K.; Krajka-Kuźniak, V. COXIBs and 2,5-dimethylcelecoxib counteract the hyperactivated Wnt/β-catenin pathway and COX-2/PGE2/EP4 signaling in glioblastoma cells. BMC Cancer 2021, 21, 493. [Google Scholar] [CrossRef]
- Rybarczyk, A.; Majchrzak-Celińska, A.; Piwowarczyk, L.; Krajka-Kuźniak, V. The Anti-Glioblastoma Effects of Novel Liposomal Formulations Loaded with Cannabidiol, Celecoxib, and 2,5-Dimethylcelecoxib. Pharmaceutics 2025, 17, 1031. [Google Scholar] [CrossRef]
- Mendes, M.; Branco, F.; Vitorino, R.; Sousa, J.; Pais, A.; Vitorino, C. A Two-Pronged Approach against Glioblastoma: Drug Repurposing and Nanoformulation Design for in Situ-Controlled Release. Drug Deliv. Transl. Res. 2023, 13, 3169–3191. [Google Scholar] [CrossRef]
- Dinur, E.; Goldenberg, H.; Robinson, E.; Naggan, L.; Kozela, E.; Yirmiya, R. A Novel Anti-Inflammatory Formulation Comprising Celecoxib and Cannabidiol Exerts Antidepressant and Anxiolytic Effects. Cannabis Cannabinoid Res. 2022, 9, 561–580. [Google Scholar] [CrossRef] [PubMed]
- Perumal, S.; Atchudan, R.; Lee, W. A Review of Polymeric Micelles and Their Applications. Polymers 2022, 14, 2510. [Google Scholar] [CrossRef]
- Grzegorzewski, J.; Michalak, M.; Wołoszczuk, M.; Bulicz, M.; Majchrzak-Celińska, A. Nanotherapy of Glioblastoma—Where Hope Grows. Int. J. Mol. Sci. 2025, 26, 1814. [Google Scholar] [CrossRef]
- Wang, N.; Qing, Q.; Xue, Y.; Cai, S.; Zheng, M.; Zhang, D.; Ismail, M. Enhancing lipid nanoparticles-mediated RNA delivery to glioblastoma via targeted strategies. J. Control. Release 2025, 389, 114472. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Wang, Y.; Li, Y.; Liu, J.; Zheng, M.; Zou, Y. Stimuli-Responsive Polymeric Nanocarriers Accelerate On-Demand Drug Release to Combat Glioblastoma. Biomacromolecules 2024, 25, 6250–6282. [Google Scholar] [PubMed]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric micelles in drug delivery: An insight of the techniques for their characterization and assessment in biorelevant conditions. J. Control. Release 2021, 332, 312–336. [Google Scholar] [CrossRef]
- Lin, S.; Yu, Y.; Wu, E.; Ding, T.; Chu, Y.; Pan, F.; Yang, Y.; Zhan, C. Reexamining in vivo fate of paclitaxel-loaded polymeric micelles. Nano Today 2024, 56, 102255. [Google Scholar] [CrossRef]
- Hwang, D.; Ramsey, J.D.; Kabanov, A.V. Polymeric micelles for the delivery of poorly soluble drugs: From nanoformulation to clinical approval. Adv. Drug Deliv. Rev. 2020, 156, 80–118. [Google Scholar] [CrossRef]
- Sofias, A.M.; Dunne, M.; Storm, G.; Allen, C. The battle of “nano” paclitaxel. Adv. Drug Deliv. Rev. 2017, 122, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Raval, N.; Maheshwari, R.; Shukla, H.; Kalia, K.; Torchilin, V.P.; Tekade, R.K. Multifunctional polymeric micellar nanomedicine in the diagnosis and treatment of cancer. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 126, 112186. [Google Scholar] [PubMed]
- Smith, A.; Taylor, M.; In-Young, C.; Lee, P.; Kelly, E.; Aroa, T.; Waitsman, F. 450 Eight weeks of creatine monohydrate supplementation is feasible and associated with increased brain creatine in patients with AD. J. Clin. Transl. Sci. 2025, 9, 133. [Google Scholar] [CrossRef]
- Dasgupta, A.; May, J.N.; Klinkenberg, G.; Besse, H.C.; Buhl, E.M.; Moeckel, D.; Mihyar, R.; Peña, Q.; Shalmani, A.A.; Hark, C.; et al. Multidrug micelles and sonopermeation for chemotherapy co-delivery to brain tumors. J. Control. Release 2025, 380, 818–828. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, R.; Yu, Y.; Liu, J.; Luo, T.; Fan, F. Glioblastoma treatment modalities besides surgery. J. Cancer 2019, 10, 4793–4806. [Google Scholar] [CrossRef]
- Desprez, P.Y.; Murase, R.; Limbad, C.; Woo, R.W.L.; Adrados, I.; Weitenthaler, K.; Soroceanu, L.; Salomonis, N.; McAllister, S.D. Cannabidiol treatment results in a common gene expression response across aggressive cancer cells from various origins. Cannabis Cannabinoid Res. 2021, 6, 148–155. [Google Scholar] [CrossRef]
- Cásedas, G.; Yarza-Sancho, M.d.; López, V. Cannabidiol (CBD): A Systematic Review of Clinical and Preclinical Evidence in the Treatment of Pain. Pharmaceuticals 2024, 17, 1438. [Google Scholar] [CrossRef]
- Ahsan, H.; Malik, S.I.; Shah, F.A.; El-Serehy, H.A.; Ullah, A.; Shah, Z.A. Celecoxib Suppresses NF-κB p65 (RelA) and TNFα Expression Signaling in Glioblastoma. J. Clin. Med. 2023, 12, 6683. [Google Scholar]
- Inggas, M.A.M.; Patel, U.; Wijaya, J.H.; Otinashvili, N.; Menon, V.R.; Iyer, A.K.; Turjman, T.; Dadwal, S.; Gadaevi, M.; Ismayilova, A.; et al. The role of temozolomide as adjuvant therapy in glioblastoma management: A systematic review and meta-analysis. BMC Cancer 2025, 25, 399. [Google Scholar]
- Román-Vargas, Y.; Porras-Arguello, J.D.; Blandón-Naranjo, L.; Pérez-Pérez, L.D.; Benjumea, D.M. Evaluation of the Analgesic Effect of High-Cannabidiol-Content Cannabis Extracts in Different Pain Models by Using Polymeric Micelles as Vehicles. Molecules 2023, 28, 4299. [Google Scholar] [CrossRef]
- Aslan, T.N. Cationic Micelle-like Nanoparticles as the Carrier of Methotrexate for Glioblastoma Treatment. Molecules 2024, 29, 5977. [Google Scholar] [CrossRef]
- Krishnan, S.; Lee, S.; Amoozgar, Z.; Subudhi, S.; Kumar, A.S.S.; Posada, J.M.; Lindeman, N.; Lei, P.; Duquette, M.; Steinbuch, S.; et al. Wnt inhibition alleviates resistance to anti-PD1 therapy and improves antitumor immunity in glioblastoma. Proc. Natl. Acad. Sci. USA 2025, 122, 2414941122. [Google Scholar]
- Latour, M.; Her, N.-G.; Kesari, S.; Nurmemmedov, E. WNT Signaling as a Therapeutic Target for Glioblastoma. Int. J. Mol. Sci. 2021, 22, 8428. [Google Scholar] [CrossRef] [PubMed]
- Zhan, P.; Tan, Y.; Wang, H.; Liu, J.; Shao, L.; Wu, Z. Effects of Non-Ionic Surfactant Tween 80 on Enzymatic Saccharification of Avicel and Steam-Exploded Poplar at High Solid Loading. Processes 2025, 13, 2960. [Google Scholar] [CrossRef]
- Hirun, N.; Kraisit, P.; Tantishaiyakul, V. Thermosensitive Polymer Blend Composed of Poloxamer 407, Poloxamer 188 and Polycarbophil for the Use as Mucoadhesive In Situ Gel. Polymers 2022, 14, 1836. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.N.; Liew, K.B.; Lim, Y.M.; Chew, Y.-L.; Chua, A.-L.; Yang, S.-B.; Lee, S.-K. Development and Characterization of EGCG-Loaded TPGS/Poloxamer 407 Micelles with Evaluation of In Vitro Drug Release and In Vivo Pharmacokinetics and Tolerability Observations Following Oral Administration. Pharmaceutics 2025, 17, 1441. [Google Scholar] [CrossRef]
- Rivero-Gutiérrez, B.; Anzola, A.; Martínez-Augustin, O.; de Medina, F.S. Stain-Free Detection as Loading Control Alternative to Ponceau and Housekeeping Protein Immunodetection in Western Blotting. Anal. Biochem. 2014, 467, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]
- Larsson, M.; Hill, A.; Duffy, J. Suspension Stability; Why Particle Size, Zeta Potential and Rheology Are Important. Ann. Trans. Nord. Rheol. Soc. 2012, 20, 209–214. [Google Scholar]
- Sunoqrot, S.; Alsadi, A.; Tarawneh, O.; Hamed, R. Polymer type and molecular weight dictate the encapsulation efficiency and release of Quercetin from polymeric micelles. Colloid Polym. Sci. 2017, 295, 2051–2059. [Google Scholar] [CrossRef]
- Peeri, H.; Koltai, H. Cannabis Biomolecule Effects on Cancer Cells and Cancer Stem Cells: Cytotoxic, Anti-Proliferative, and Anti-Migratory Activities. Biomolecules 2022, 12, 491. [Google Scholar] [CrossRef] [PubMed]
- Ali, R.; Qamar, W.; Kalam, M.A.; Binkhathlan, Z. Soluplus-TPGS Mixed Micelles as a Delivery System for Brigatinib: Characterization and In Vitro Evaluation. ACS Omega 2024, 9, 41830–41840. [Google Scholar] [CrossRef]
- Polat, H.; Eren, M.C.; Polat, M.; Koss, K.M.; Polat, O.K. Comparative Stability of Synthetic and Natural Polymeric Micelles in Physiological Environments: Implications for Drug Delivery. Pharmaceutics 2025, 17, 1439. [Google Scholar] [CrossRef]
- Pereira-Silva, M.; Diaz-Gomez, L.; Blanco-Fernandez, B.; Ferreirós, A.; Veiga, F.; Concheiro, A.; Paiva-Santos, A.C.; Alvarez-Lorenzo, C. Cancer cell membrane-modified Soluplus® micelles for gemcitabine delivery to pancreatic cancer using a prodrug approach. Int. J. Pharm. 2024, 662, 124529. [Google Scholar] [CrossRef] [PubMed]
- Nabissi, M.; Morelli, M.B.; Santoni, M.; Santoni, G. Triggering of the TRPV2 channel by cannabidiol sensitizes glioblastoma cells to cytotoxic chemotherapeutic agents. Carcinogenesis 2013, 34, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Al-Husein, B.A.; Al-Azayzih, A.; Redh, F.S. Sorafenib combination with imatinib enhances apoptotic and cell cycle arrest effects with no effect on viability and BCR-ABL expression of K-562 cells. J. Appl. Pharm. Sci. 2024, 14, 190–195. [Google Scholar] [CrossRef]
- Pu, J.; Yuan, K.; Tao, J.; Qin, Y.; Li, Y.; Fu, J.; Li, Z.; Zhou, H.; Tang, Z.; Li, L.; et al. Glioblastoma multiforme: An updated overview of temozolomide resistance mechanisms and strategies to overcome resistance. Discov. Oncol. 2025, 16, 731. [Google Scholar] [CrossRef]
- Smerdi, D.; Moutafi, M.; Kotsantis, I.; Stavrinou, L.C.; Psyrri, A. Overcoming Resistance to Temozolomide in Glioblastoma: A Scoping Review of Preclinical and Clinical Data. Life 2024, 14, 673. [Google Scholar] [CrossRef]
- Soroceanu, L.; Singer, E.; Dighe, P.; Sidorov, M.; Limbad, C.; Rodriquez-Brotons, A.; Rix, P.; Woo, R.W.L.; Dickinson, L.; Desprez, P.Y.; et al. Cannabidiol inhibits RAD51 and sensitizes glioblastoma to temozolomide in multiple orthotopic tumor models. Neuro-Oncol. Adv. 2022, 4, vdac019. [Google Scholar] [CrossRef]
- Ismail, M.; Yang, W.; Li, Y.; Chai, T.; Zhang, D.; Du, Q.; Muhammad, P.; Hanif, S.; Zheng, M.; Shi, B. Targeted liposomes for combined delivery of artesunate and temozolomide to resistant glioblastoma. Biomaterials 2022, 287, 121608. [Google Scholar] [CrossRef]
- Sen, P.; Sadat, S.; Ebisumoto, K.; Al-Msari, R.; Miyauchi, S.; Roy, S.; Mohammadzadeh, P.; Lips, K.; Nakagawa, T.; Saddawi-Konefka, R.; et al. CBD promotes antitumor activity by modulating tumor immune microenvironment in HPV associated head and neck squamous cell carcinoma. Front. Immunol. 2025, 16, 1528520. [Google Scholar] [CrossRef] [PubMed]
- Wen, B.; Wei, Y.T.; Mu, L.L.; Wen, G.R.; Zhao, K. The molecular mechanisms of celecoxib in tumor development. Medicine 2020, 99, 22544. [Google Scholar] [CrossRef] [PubMed]
- Twelves, C.; Sabel, M.; Checketts, D.; Miller, S.; Tayo, B.; Jove, M.; Brazil, L.; Short, S.C. A Phase1b Randomised, Placebo-Controlled Trial of Nabiximols Cannabinoid Oromucosal Spray with Temozolomide in Patients with Recurrent Glioblastoma. Br. J. Cancer 2021, 124, 1379–1387. [Google Scholar] [CrossRef] [PubMed]






| Surfactant | Type of Compound | Structure |
|---|---|---|
| Tween 20 | Alkyl esters |  |
| Tween 80 | Alkyl esters |  |
| Poloxamer 188 | Block copolymers |  |
| Poloxamer 407 | Block copolymers | 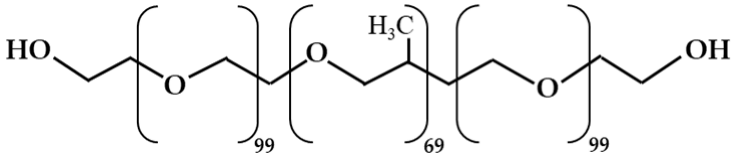 |
| Name | Surfactant | Solvent | Technique of Obtaining | Ultrasound Time |
|---|---|---|---|---|
| Type of Surfactant | ||||
| MIC1 | Tween 20 | PBS | Sonication | 30 min |
| MIC2 | Tween 80 | |||
| MIC3 | Poloxamer 188 | |||
| MIC4 | Poloxamer 407 | |||
| MIC5 | Tween 20/Tween 80 | |||
| MIC6 | Poloxamer 188/Pluronic F127 | |||
| MIC7 | Tween 20/Poloxamer 188 | |||
| MIC8 | Tween 20/Pluronic F127 | |||
| Type of solvent | ||||
| MIC5 | Tween 20/Tween 80 | PBS | Sonication | 30 min |
| MIC9 | Water | |||
| Technique of obtaining | ||||
| MIC5 | Tween 20/Tween 80 | PBS | Sonication | 30 min |
| MIC10 | High-shear homogenization | |||
| MIC11 | Sonication/High-shear homogenization | |||
| Ultrasound time | ||||
| MIC5 | Tween 20/Tween 80 | PBS | Sonication | 30 min |
| MIC12 | 45 min | |||
| MIC13 | 60 min | |||
| MIC14 | 90 min | |||
| Name | Measurement Date | Z-Ave [nm] ± SD | PDI [%] ± SD | ZP [mV] ± SD |
|---|---|---|---|---|
| MIC1 | 1 day | 210.8 ± 1.6 | 0.25 ± 0.003 | −22.16 ± 0.46 |
| 7 days | 227.2 ± 2.1 | 0.26 ± 0.005 | −22.92 ± 0.84 | |
| 14 days | 234.6 ± 0.9 | 0.26 ± 0.004 | −21.54 ± 0.16 | |
| MIC2 | 1 day | 324.3 ± 2.6 | 0.317 ± 0.002 | −26.99 ± 0.34 |
| 7 days | 352.2 ± 1.5 | 0.337 ± 0.004 | −26.60 ± 0.12 | |
| 14 days | 331.5 ± 0.8 | 0.388 ± 0.05 | −25.88 ± 0.12 | |
| MIC3 | 1 day | 374.2 ± 1.4 | 0.437 ± 0.02 | −23.28 ± 0.29 |
| 7 days | 380.8 ± 3.5 | 0.446 ± 0.06 | −22.48 ± 0.53 | |
| 14 days | 385.8 ± 1.5 | 0.470 ± 0.04 | −22.15 ± 0.39 | |
| MIC4 | 1 day | 385.8 ± 2.5 | 0.375 ± 0.05 | −20.73 ± 0.60 |
| 7 days | 351.8 ± 2.2 | 0.383 ± 0.06 | −19.38 ± 0.11 | |
| 14 days | 364.8 ± 1.3 | 0.437 ± 0.05 | −20.17 ± 0.50 | |
| MIC5 | 1 day | 152.8 ± 0.3 | 0.186 ± 0.002 | −31.31 ± 0.11 |
| 7 days | 155.6 ± 0.1 | 0.204 ± 0.002 | −32.02 ± 0.47 | |
| 14 days | 162.7 ± 0.3 | 0.221 ± 0.01 | −30.96 ± 0.36 | |
| MIC6 | 1 day | 264.7 ± 3.3 | 0.326 ± 0.005 | −17.80 ± 0.59 |
| 7 days | 271.8 ± 2.5 | 0.354 ± 0.03 | −16.88 ± 0.28 | |
| 14 days | 276.8 ± 2.3 | 0.427 ± 0.01 | −15.96 ± 0.41 | |
| MIC7 | 1 day | 202.8 ± 1.5 | 0.323 ± 0.005 | −24.57 ± 0.20 |
| 7 days | 315.0 ± 1.3 | 0.343 ± 0.03 | −23.60 ± 0.40 | |
| 14 days | 325.0 ± 1.7 | 0.357 ± 0.01 | −23.17 ± 0.44 | |
| MIC8 | 1 day | 365.8 ± 1.4 | 0.320 ± 0.004 | −26.48 ± 0.14 |
| 7 days | 372.8 ± 1.5 | 0.372 ± 0.02 | −25.60 ± 0.20 | |
| 14 days | 387.9 ± 2.1 | 0.388 ± 0.02 | −23.30 ± 0.41 |
| Name | Stability Condition | Sampling Interval (Day) | Z-Ave [nm] ± SD | PDI [%] ± SD | ZP [mV] ± SD |
|---|---|---|---|---|---|
| MIC control | 2 ± 2 °C | 1 | 154.2 ± 1.2 | 0.197 ± 0.021 | −29.55 ± 0.42 |
| 14 | 166.8 ± 1.8 | 0.247 ± 0.024 | −28.87 ± 0.38 | ||
| 25 ± 2 °C | 1 | 152.2 ± 0.3 | 0.186 ± 0.002 | −31.31 ± 0.11 | |
| 14 | 162.7 ± 0.3 | 0.211 ± 0.01 | −30.96 ± 0.36 | ||
| MIC CBD | 2 ± 2 °C | 1 | 159.1 ± 1.4 | 0.254 ± 0.022 | −33.54 ± 1.11 |
| 14 | 165.4 ± 1.8 | 0.267 ± 0.031 | −31.54 ± 1.06 | ||
| 25 ± 2 °C | 1 | 157.6 ± 0.7 | 0.214 ± 0.002 | −37.29 ± 0.14 | |
| 14 | 163.1 ± 0.5 | 0.218 ± 0.002 | −36.59 ± 0.31 | ||
| MIC CELE | 2 ± 2 °C | 1 | 160.1 ± 1.5 | 0.246 ± 0.051 | −31.62 ± 0.41 |
| 14 | 165.8 ± 1.6 | 0.254 ± 0.067 | −30.11 ± 1.62 | ||
| 25 ± 2 °C | 1 | 155.7 ± 0.2 | 0.209 ± 0.005 | −33.28 ± 0.29 | |
| 14 | 161.3 ± 0.3 | 0.217 ± 0.007 | −32.48 ± 0.24 | ||
| MIC TMZ | 2 ± 2 °C | 1 | 157.4 ± 1.3 | 0.277 ± 0.031 | −35.44 ± 1.74 |
| 14 | 160.1 ± 1.4 | 0.279 ± 0.110 | −34.89 ± 1.63 | ||
| 25 ± 2 °C | 1 | 150.8 ± 0.7 | 0.207 ± 0.004 | −39.39 ± 0.15 | |
| 14 | 154.3 ± 0.4 | 0.208 ± 0.005 | −39.50 ± 0.22 | ||
| MIC CBD + CELE | 2 ± 2 °C | 1 | 154.3 ± 0.5 | 0.210 ± 0.022 | −35.34 ± 0.15 |
| 14 | 158.7 ± 0.6 | 0.223 ± 0.032 | −34.33 ± 0.52 | ||
| 25 ± 2 °C | 1 | 149.2 ± 0.06 | 0.206 ± 0.004 | −38.31 ± 0.11 | |
| 14 | 152.7 ± 0.3 | 0.204 ± 0.003 | −37.96 ± 0.46 | ||
| MIC CBD + TMZ | 2 ± 2 °C | 1 | 151.21 ± 0.2 | 0.199 ± 0.015 | 38.17 ± 1.72 |
| 14 | 159.1 ± 0.3 | 0.223 ± 0.017 | 37.76 ± 1.34 | ||
| 25 ± 2 °C | 1 | 147.8 ± 0.7 | 0.187 ± 0.005 | −41.82 ± 0.61 | |
| 14 | 153.8 ± 0.4 | 0.215 ± 0.004 | −41.29 ± 0.49 |
| Formulation | %EE (% ± SD) | %DL (% ± SD) |
|---|---|---|
| MIC CBD | 79.42 ± 4.43 | 10.51 ± 0.24 |
| MIC CELE | 76.21 ± 2.11 | 8.25 ± 0.31 |
| MIC TMZ | 72.62 ± 4.32 | 10.23 ± 0.12 |
| MIC CBD + CELE | 82.12 ± 1.68 | 12.32 ± 0.12 |
| MIC CBD + TMZ | 81.55 ± 2.25 | 10.02 ± 0.14 |
| IC50 ± SEM [µM] | ||
|---|---|---|
| U-87 MG | U-138 MG | |
| MIC control | >50 | >50 |
| MIC CBD | 24.5 ± 0.7 | 17.5 ± 1.0 |
| MIC CELE | >50 | 18.9 ± 0.6 |
| MIC TMZ | >50 | 44.5 ± 2.9 |
| MIC CBD + CELE | >50 | 33.3 ± 0.9 |
| MIC CBD + TMZ | >50 | 23.5 ± 1.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Musielak, E.; Feliczak-Guzik, A.; Majchrzak-Celińska, A.; Rybarczyk, A.; Krajka-Kuźniak, V. Polymeric Micelles Co-Loaded with Cannabidiol, Celecoxib, and Temozolomide—Early-Stage Assessment of Anti-Glioma Properties. Micro 2026, 6, 11. https://doi.org/10.3390/micro6010011
Musielak E, Feliczak-Guzik A, Majchrzak-Celińska A, Rybarczyk A, Krajka-Kuźniak V. Polymeric Micelles Co-Loaded with Cannabidiol, Celecoxib, and Temozolomide—Early-Stage Assessment of Anti-Glioma Properties. Micro. 2026; 6(1):11. https://doi.org/10.3390/micro6010011
Chicago/Turabian StyleMusielak, Ewelina, Agnieszka Feliczak-Guzik, Aleksandra Majchrzak-Celińska, Anna Rybarczyk, and Violetta Krajka-Kuźniak. 2026. "Polymeric Micelles Co-Loaded with Cannabidiol, Celecoxib, and Temozolomide—Early-Stage Assessment of Anti-Glioma Properties" Micro 6, no. 1: 11. https://doi.org/10.3390/micro6010011
APA StyleMusielak, E., Feliczak-Guzik, A., Majchrzak-Celińska, A., Rybarczyk, A., & Krajka-Kuźniak, V. (2026). Polymeric Micelles Co-Loaded with Cannabidiol, Celecoxib, and Temozolomide—Early-Stage Assessment of Anti-Glioma Properties. Micro, 6(1), 11. https://doi.org/10.3390/micro6010011










