Resurgence of Electron Quantum Tunneling Sensors
Abstract
1. Introduction to Electron Quantum Tunneling
2. Electron Quantum Tunneling Sensors—Generalized Working Principle, Beginnings and a Brief Survey
3. Nanogap Electrodes for Molecular Electronics
4. Advancement of IoT and the Need of Ultra-Low-Power Sensor Networks
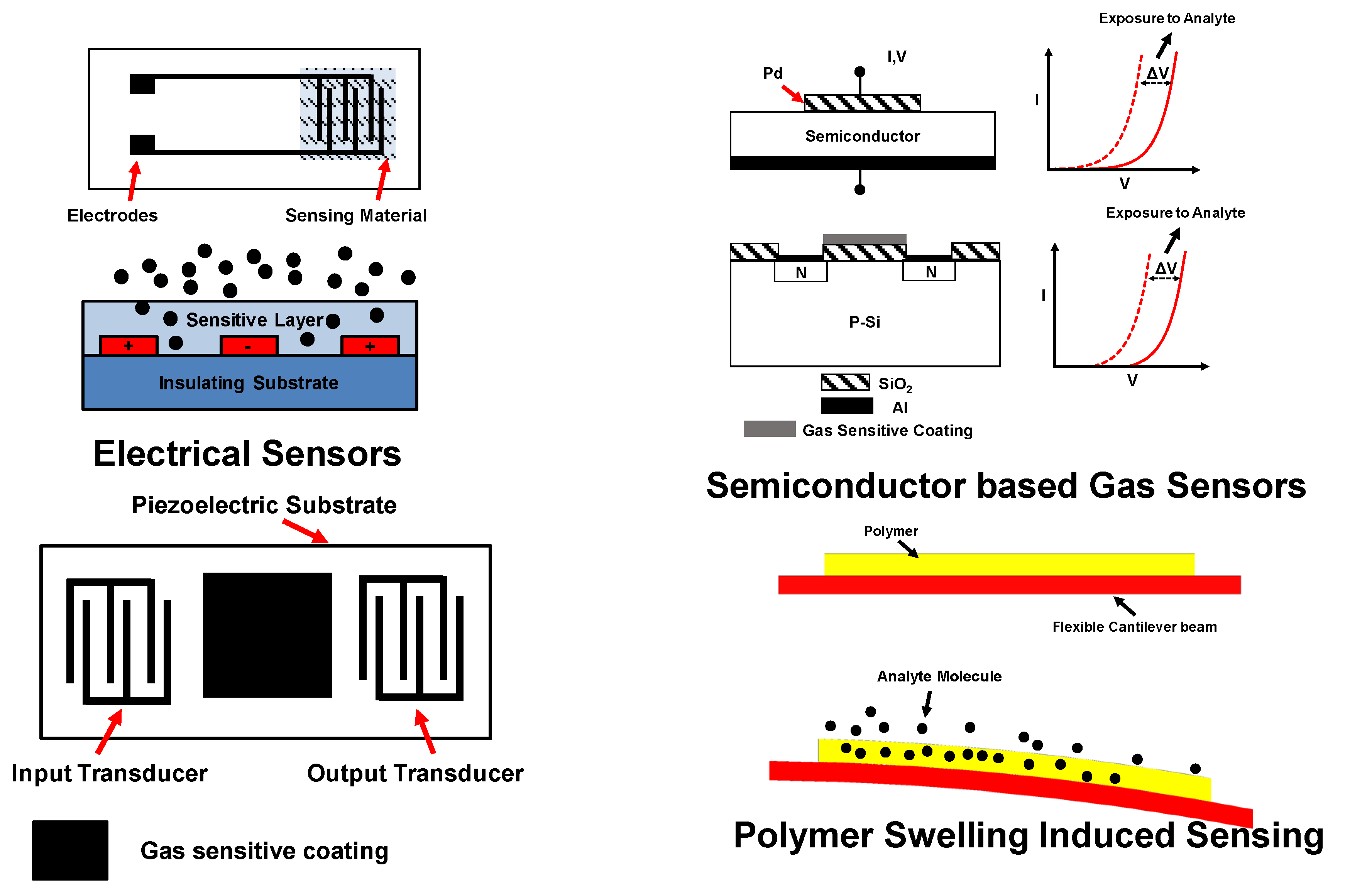
5. Current Sensor Technology and Their Drawbacks of w.r.t IoT Applications
6. Previous Efforts in Developing Sensor Technology for IoT Applications
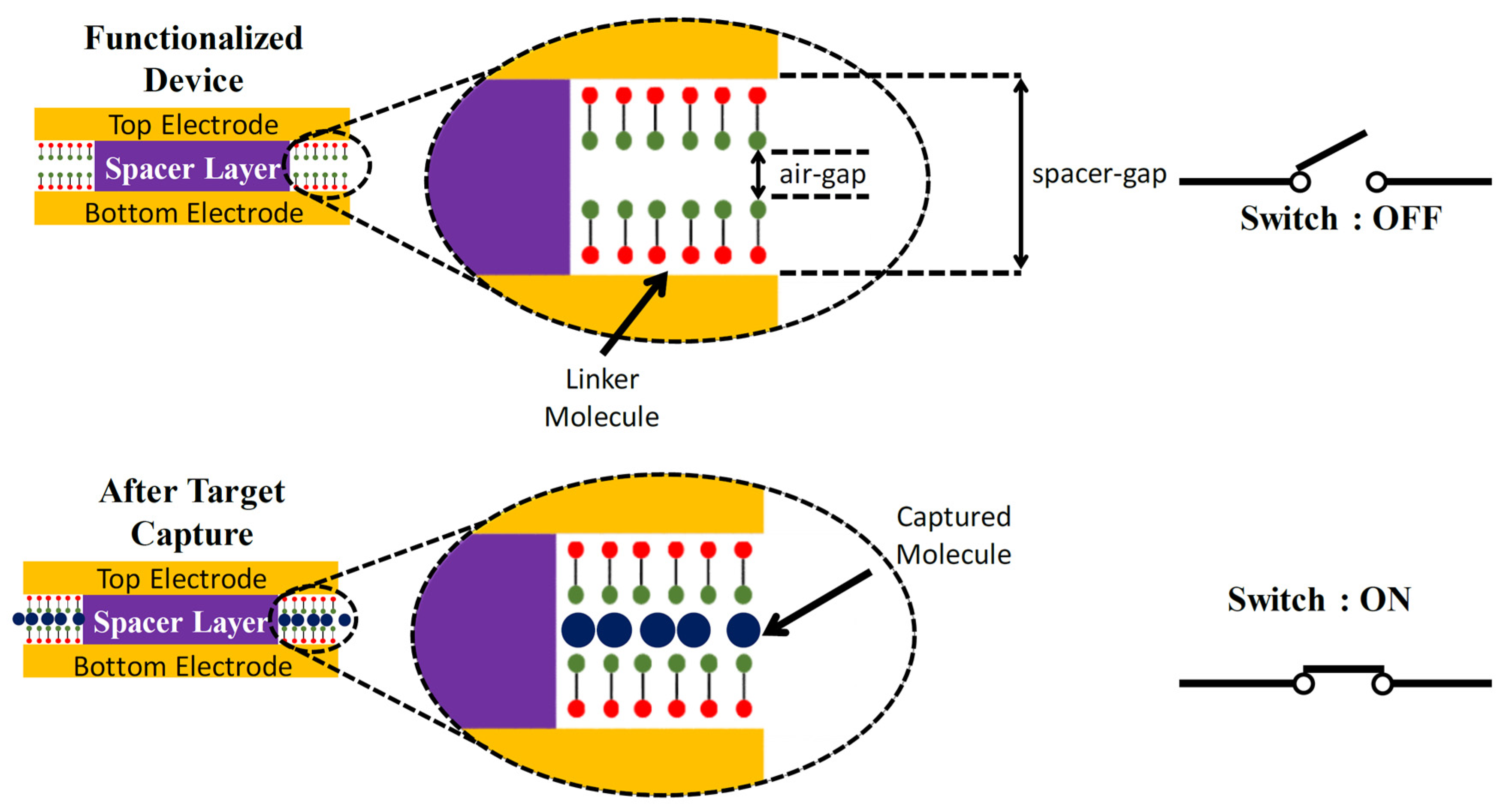
7. Nanogap Electrode-Based Quantum Tunneling Gas Sensors
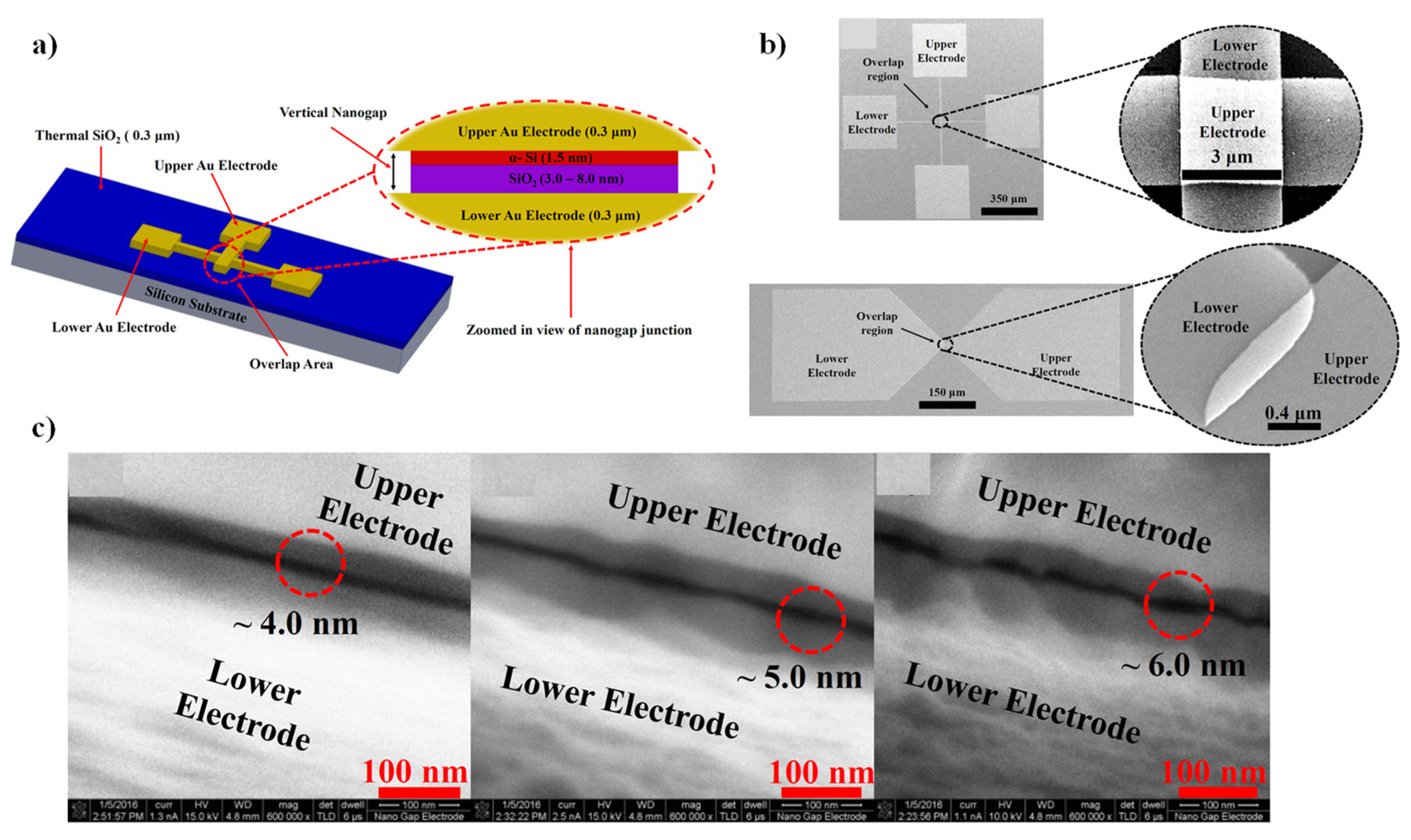
7.1. Batch Fabrication of Nanogap Tunneling Junctions: Device Structure, Design and Fabrication
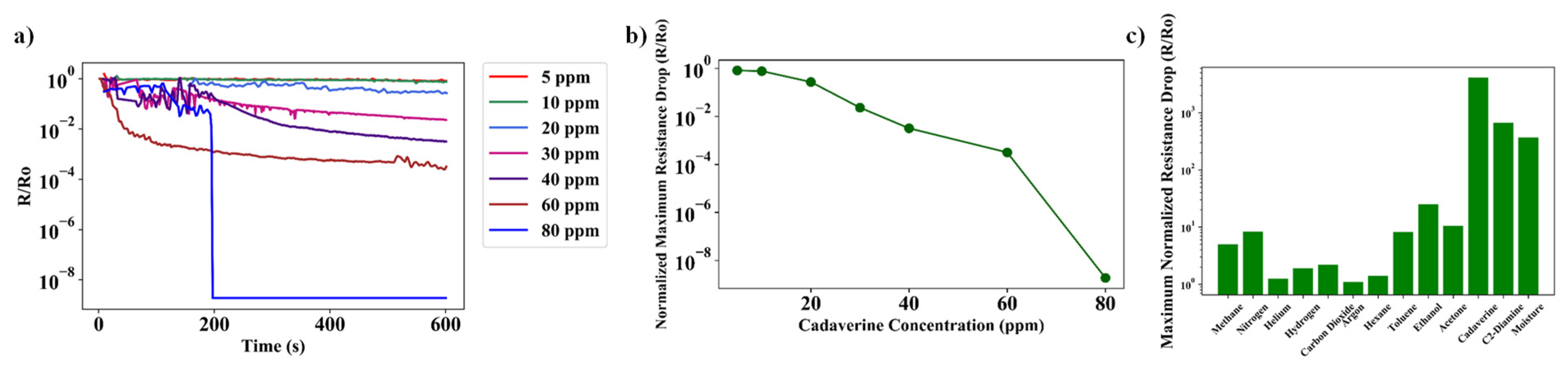
7.2. Sensor Action
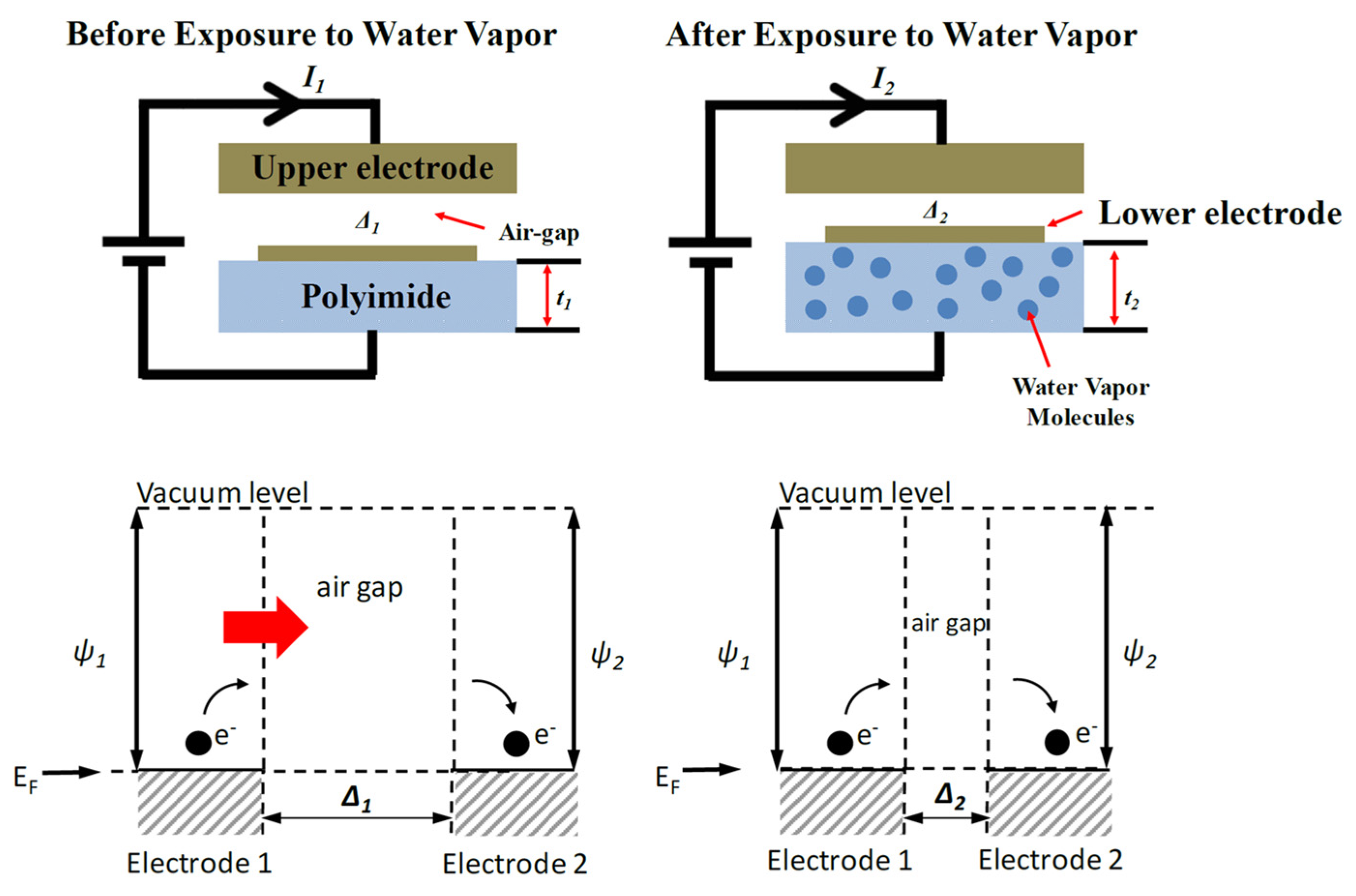
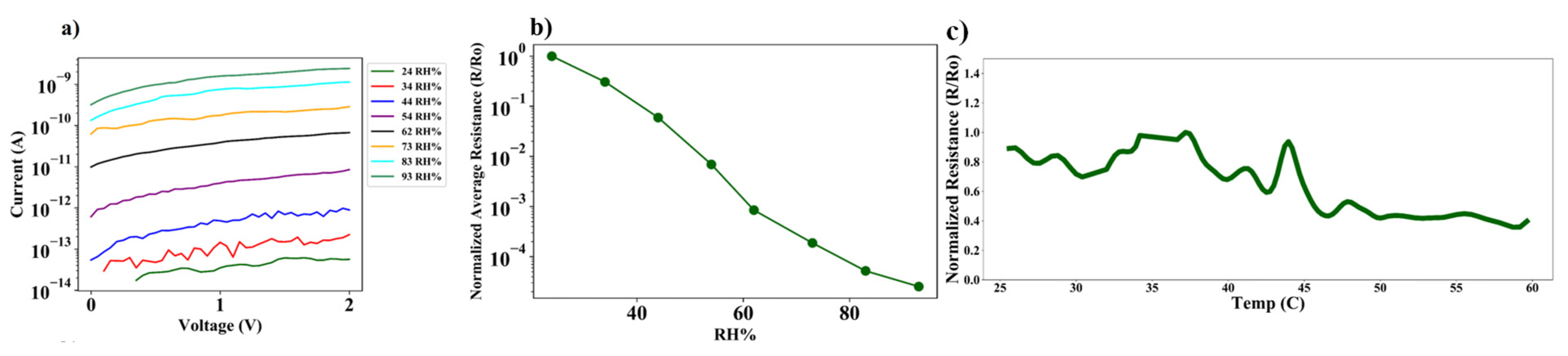
7.3. Temperature Compensated Quantum Tunneling Hygrometer
8. Roadblocks
9. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Razavy, M. Quantum Theory of Tunneling, 1st ed.; World Scientific Publishing Company: Hackensack, NJ, USA, 2003. [Google Scholar]
- Rother, F. Über den Austritt von Elektronen aus kalten Metallen. Ann. Phys. 1926, 386, 317–372. [Google Scholar] [CrossRef]
- Hund, F. Zur Deutung der Molekelspektren. I. Z. Phys. 1927, 40, 742–764. [Google Scholar] [CrossRef]
- Ralph, H.F.; Nordheim, L. Electron emission in intense electric fields. Proc. R. Soc. Lond. Ser. A Contain. Pap. A Math. Phys. Character 1928, 119, 173–181. [Google Scholar] [CrossRef]
- Merzbacher, E. The Early History of Quantum Tunneling. Phys. Today 2002, 55, 44–49. [Google Scholar] [CrossRef]
- Esaki, L. New Phenomenon in Narrow Germanium p − n Junctions. Phys. Rev. 1958, 109, 603–604. [Google Scholar] [CrossRef]
- Simmons, J.G. Generalized Formula for the Electric Tunnel Effect between Similar Electrodes Separated by a Thin Insulating Film. J. Appl. Phys. 1963, 34, 1793–1803. [Google Scholar] [CrossRef]
- Binnig, G.; Rohrer, H. Scanning Tunneling Microscopy. Surf. Sci. 1983, 126, 236–244. [Google Scholar] [CrossRef]
- Kenny, T.W.; Reynolds, J.K.; Podosek, J.A.; Vote, E.C.; Miller, L.M.; Rockstad, H.K.; Kaiser, W.J. Micromachined infrared sensors using tunneling displacement transducers. Rev. Sci. Instrum. 1996, 67, 112–128. [Google Scholar] [CrossRef]
- Kenny, T.W.; Kaiser, W.J.; Podosek, J.A.; Rockstad, H.K.; Reynolds, J.K.; Vote, E.C. Micromachined tunneling displacement transducers for physical sensors. J. Vac. Sci. Technol. A Vac. Surf. Film. 1993, 11, 797–802. [Google Scholar] [CrossRef]
- Kenny, T.W.; Kaiser, W.J.; Waltman, S.B.; Reynolds, J.K. Novel infrared detector based on a tunneling displacement transducer. Appl. Phys. Lett. 1991, 59, 1820–1822. [Google Scholar] [CrossRef]
- VanZandt, T.R.; Kenny, T.W.; Kaiser, W.J. Novel position sensor technologies for microaccelerometers. In Sensors and Sensor Systems for Guidance and Navigation II; SPIE: Orlando, FL, USA, 1992; pp. 165–172. [Google Scholar] [CrossRef]
- DiLella, D.; Whitman, L.J.; Colton, R.J.; Kenny, T.W.; Kaiser, W.J.; Vote, E.C.; Podosek, J.A.; Miller, L.M. A micromachined magnetic-field sensor based on an electron tunneling displacement transducer. Sens. Actuators A Phys. 2000, 86, 8–20. [Google Scholar] [CrossRef]
- Reed, M.A.; Zhou, C.; Muller, C.J.; Burgin, T.P.; Tour, J.M. Conductance of a Molecular Junction. Science 1979, 278, 252–254. [Google Scholar] [CrossRef]
- Dolan, G.J. Offset masks for lift-off photoprocessing. Appl. Phys. Lett. 1977, 31, 337–339. [Google Scholar] [CrossRef]
- Valladares, L.D.L.S.; Felix, L.L.; Dominguez, A.B.; Mitrelias, T.; Sfigakis, F.; Khondaker, S.I.; Barnes, C.H.; Majima, Y. Controlled electroplating and electromigration in nickel electrodes for nanogap formation. Nanotechnology 2010, 21, 445304. [Google Scholar] [CrossRef]
- Hatzor, A.; Weiss, P.S. Molecular Rulers for Scaling Down Nanostructures. Science 2001, 291, 1019–1020. [Google Scholar] [CrossRef]
- Li, T.; Hu, W.; Zhu, D. Nanogap Electrodes. Adv. Mater. 2010, 22, 286–300. [Google Scholar] [CrossRef]
- Ashwell, G.J.; Wierzchowiec, P.; Bartlett, C.J.; Buckle, P.D. Molecular electronics: Connection across nano-sized electrode gaps. Chem. Commun. 2007, 12, 1254–1256. [Google Scholar] [CrossRef]
- Tyagi, P.; Li, D.; Holmes, S.M.; Hinds, B.J. Molecular Electrodes at the Exposed Edge of Metal/Insulator/Metal Trilayer Structures. J. Am. Chem. Soc. 2007, 129, 4929–4938. [Google Scholar] [CrossRef]
- Howell, S.W.; Dirk, S.M.; Childs, K.; Pang, H.; Blain, M.; Simonson, R.J.; Tour, J.M.; Wheeler, D.R. Mass-fabricated one-dimensional silicon nanogaps for hybrid organic/nanoparticle arrays. Nanotechnology 2005, 16, 754–758. [Google Scholar] [CrossRef]
- Nevill, J.T.; di Carlo, D.; Liu, P.; Jeong, K.H.; Lee, L.P. Detection of protein conformational changes with a nanogap biosensor. In Proceedings of the 13th International Conference on Solid-State Sensors, Actuators and Microsystems, 2005. Digest of Technical Papers. TRANSDUCERS ‘05, Seoul, Republic of Korea, 5–9 June 2005. [Google Scholar]
- Lee, L.P.; Oh, S.; Choi, Y.; Lee, J.S.; Sensor, B.; Center, A. Molecular Nanoelectronics for Label Free DNA Analysis. In Proceedings of the 2002 International Microprocesses and Nanotechnology Conference, 2002. Digest of Papers, Tokyo, Japan, 6–8 November 2002. [Google Scholar]
- Porath, D.; Bezryadin, A.; De Vries, S.; Dekker, C. Direct measurement of electrical transport through DNAmolecules. Nature 2000, 403, 635. [Google Scholar] [CrossRef]
- Choi, Y.-K.; Lee, J.S.; Zhu, J.; Somorjai, G.A.; Lee, L.P.; Bokor, J. Sublithographic nanofabrication technology for nanocatalysts and DNA chips. J. Vac. Sci. Technol. B Microelectron. Nanometer Struct. 2003, 21, 2951. [Google Scholar] [CrossRef]
- Schlecht, U.; Malavé, A.; Gronewold, T.M.A.; Tewes, M.; Löhndorf, M. Detection of Rev peptides with impedance-sensors-Comparison of device-geometries. Biosens. Bioelectron. 2007, 22, 2337–2340. [Google Scholar] [CrossRef] [PubMed]
- Schlecht, U.; Malavé, A.; Gronewold, T.; Tewes, M.; Löhndorf, M. Comparison of antibody and aptamer receptors for the specific detection of thrombin with a nanometer gap-sized impedance biosensor. Anal. Chim. Acta 2006, 573–574, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Tiwari, R. A review paper on ‘IOT’ & It’s Smart Applications. IJSETR 2016, 5, 472–477. [Google Scholar]
- Lafond, A. 4 Barriers to Building an IoT Solution—And How to Overcome Them! Available online: https://foobot.io/resources/hvac-pro-blog/4-barriers-to-building-an-iot-solution-and-how-to-overcome-them/ (accessed on 2 November 2022).
- Muñoz, B.C.; Steinthal, G.; Sunshine, S. Conductive polymer-carbon black composites-based sensor arrays for use in an electronic nose. Sens. Rev. 1999, 19, 300–305. [Google Scholar] [CrossRef]
- Pearce, T.C.; Schiffman, S.S.; Nagle, H.T.; Gardner, J.W. Handbook of Machine Olfaction: Electronic Nose Technology; Wiley: Hoboken, NJ, USA, 2006. [Google Scholar]
- Heeger, A.J. Semiconducting and Metallic Polymers: The Fourth Generation of Polymeric Materials. J. Phys. Chem. B 2001, 105, 8475–8491. [Google Scholar] [CrossRef]
- Hatfield, J.V.; Covington, J.A.; Gardner, J.W. GasFETs incorporating conducting polymers as gate materials. Sens. Actuators B Chem. 2000, 65, 253–256. [Google Scholar] [CrossRef]
- Nagle, H.T.; Gutierrez-Osuna, R.; Schiffman, S.S. The how and why of electronic noses. IEEE Spectr. 1998, 35, 22–31. [Google Scholar] [CrossRef]
- Grattan, K.T.V.; Sun, T. Fiber optic sensor technology: An overview. Sens. Actuators A Phys. 2000, 82, 40–61. [Google Scholar] [CrossRef]
- Arshak, K.; Moore, E.; Lyons, G.M.; Harris, J.; Clifford, S. A review of gas sensors employed in electronic nose applications. Sens. Rev. 2004, 24, 181–198. [Google Scholar] [CrossRef]
- Khlebarov, Z.P.; Stoyanova, A.I.; Topalova, D.I. Surface acoustic wave gas sensors. Sens. Actuators B Chem. 1992, 8, 33–40. [Google Scholar] [CrossRef]
- Schaller, E.; Bosset, J.O.; Escher, F. ‘Electronic Noses’ and Their Application to Food. LWT Food Sci. Technol. 1998, 31, 305–316. [Google Scholar] [CrossRef]
- Lee, C.-Y.; Lee, G.-B. MEMS-based humidity sensors with integrated temperature sensors for signal drift compensation. In Proceedings of the IEEE Sensors 2003 (IEEE Cat. No.03CH37498), Toronto, ON, Canada, 22–24 October 2003; pp. 384–388. [Google Scholar] [CrossRef]
- Lange, D.; Brand, O.; Baltes, H. CMOS Cantilever Sensor Systems; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar] [CrossRef]
- Banerjee, A.; Ghosh, C.; Khan, S.U.H.; Deshpande, A.; Pourshaban, E.; Karkhanis, M.U.; Noh, S.; Kim, H.; Mastranoelo, C.H. Chemocapacitive Detection of Ethylene Using Potassium Permanganate/Polyimide Composite Thin-Films. In Proceedings of the 2021 21st International Conference on Solid-State Sensors, Actuators and Microsystems (Transducers), Orlando, FL, USA, 20–24 June 2021; pp. 851–854. [Google Scholar] [CrossRef]
- Likhite, R.; Banerjee, A.; Majumder, A.; Karkhanis, M.; Kim, H.; Mastrangelo, C.H. VOC sensing using batch-fabricated temperature compensated self-leveling microstructures. Sens. Actuators B Chem. 2020, 311, 127817. [Google Scholar] [CrossRef]
- Likhite, R.; Banerjee, A.; Majumder, A.; Karkhanis, M.; Kim, H.; Mastrangelo, C.H. Parametrically Amplified Low-Power MEMS Capacitive Humidity Sensor. Sensors 2019, 19, 3954. [Google Scholar] [CrossRef]
- Likhite, R.; Banerjee, A.; Kim, H.; Mastrangelo, C.H. Self-Leveling Micromechanical Gas Sensors. In Proceedings of the 2018 IEEE SENSORS, New Delhi, India, 28–31 October 2018; pp. 1–4. [Google Scholar] [CrossRef]
- Likhite, R.; Pandey, S.S.; Banerjee, A.; Kim, H.; Mastrangelo, C.H. Amplified chemomechanical comb gas sensor. In Proceedings of the 2016 IEEE SENSORS, Orlando, FL, USA, 30 October–3 November 2016; pp. 1–3. [Google Scholar] [CrossRef]
- Pandey, S.S.; Banerjee, N.; Banerjee, A.; Hasan, N.; Kim, H.; Mastrangelo, C.H. High-sensitivity parametrically amplified chemo-mechanical vapor sensors. In Proceedings of the 2015 IEEE SENSORS, Busan, Republic of Korea, 1–4 November 2015; pp. 1–4. [Google Scholar] [CrossRef]
- Top 15 Sensor Types Being Used in IoT. Available online: https://www.finoit.com/blog/top-15-sensor-types-used-iot (accessed on 12 October 2021).
- Importance of Sensors in the Internet of Things. Available online: https://www.electronicsforu.com/technology-trends/tech-focus/iot-sensors (accessed on 28 May 2018).
- Laubhan, K.; Talaat, K.; Riehl, S.; Aman, M.S.; Abdelgawad, A.; Yelamarthi, K. A low-power IoT framework: From sensors to the cloud. In Proceedings of the 2016 IEEE International Conference on Electro Information Technology (EIT), Grand Forks, ND, USA, 19–21 May 2016; pp. 0648–0652. [Google Scholar] [CrossRef]
- Banerjee, N.; Banerjee, A.; Hasan, N.; Pandey, S.S.; Gogoi, B.P.; Mastrangelo, C.H. A Monolithically Integrated Multisensor Platform. IEEE Sens. J. 2016, 16, 8854–8860. [Google Scholar] [CrossRef]
- Banerjee, N.; Banerjee, A.; Hasan, N.; Pandey, S.S.; Gogoi, B.P.; Mastrangelo, C.H. A monolithically integrated multi-sensor platform. In Proceedings of the 2015 IEEE SENSORS, Busan, Republic of Korea, 1–4 November 2015; pp. 1–4. [Google Scholar] [CrossRef]
- Chikkadi, K.; Muoth, M.; Maiwald, V.; Roman, C.; Hierold, C. Ultra-low power operation of self-heated, suspended carbon nanotube gas sensors. Appl. Phys. Lett. 2013, 103, 223109. [Google Scholar] [CrossRef]
- Choi, S.-W.; Katoch, A.; Kim, J.-H.; Kim, S.S. Prominent Reducing Gas-Sensing Performances of n -SnO 2 Nanowires by Local Creation of p − n Heterojunctions by Functionalization with p -Cr 2 O 3 Nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 17723–17729. [Google Scholar] [CrossRef]
- Park, S.; Sun, G.-J.; Jin, C.; Kim, H.W.; Lee, S.; Lee, C. Synergistic Effects of a Combination of Cr 2 O 3 -Functionalization and UV-Irradiation Techniques on the Ethanol Gas Sensing Performance of ZnO Nanorod Gas Sensors. ACS Appl. Mater. Interfaces 2016, 8, 2805–2811. [Google Scholar] [CrossRef]
- Woo, H.-S.; Na, C.W.; Kim, I.-D.; Lee, J.-H. Highly sensitive and selective trimethylamine sensor using one-dimensional ZnO–Cr 2 O 3 hetero-nanostructures. Nanotechnology 2012, 23, 245501. [Google Scholar] [CrossRef]
- Shim, Y.S.; Jeong, H.Y.; Kim, Y.H.; Nahm, S.H.; Kang, C.Y.; Kim, J.S.; Lee, W.; Jang, H.W. Utilization of both-side metal decoration in close-packed SnO2 nanodome arrays for ultrasensitive gas sensing. Sens. Actuators B Chem. 2015, 213, 314–321. [Google Scholar] [CrossRef]
- Banerjee, A.; Khan, S.U.H.; Broadbent, S.; Likhite, R.; Looper, R.; Kim, H.; Mastrangelo, C.H. Batch-Fabricated α-Si Assisted Nanogap Tunneling Junctions. Nanomaterials 2019, 9, 727. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Khan, S.U.H.; Broadbent, S.; Bulbul, A.; Kim, K.H.; Noh, S.; Looper, R.; Mastrangelo, C.H.; Kim, H. Molecular bridge-mediated ultralow-power gas sensing. Microsyst. Nanoeng. 2021, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Farhoudi, N.; Ghosh, C.; Mastrangelo, C.H.; Kim, H.; Broadbent, S.J.; Looper, R. Picowatt gas sensing and resistance switching in tunneling nano-gap electrodes. In Proceedings of the 2016 IEEE SENSORS, Orlando, FL, USA, 30 October–3 November 2016; pp. 1–3. [Google Scholar] [CrossRef]
- Ghosh, C.; Khan, S.H.; Broadbent, S.J.; Hsieh, H.C.; Noh, S.; Banerjee, A.; Farhoudi, N.; Mastrangelo, C.H.; Looper, R.; Kim, H. Nano-gap vapor sensor. In Proceedings of the 2017 IEEE SENSORS, Glasgow, UK, 29 October–1 November 2017; pp. 1–3. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Banerjee, A.; Broadbent, S.; Kairy, P.D.; Kim, K.H.; Mastrangelo, C.H.; Looper, R.; Kim, H. Molecular Length Based Target Identification using a Nano-Gap Sensor. In Proceedings of the 2019 IEEE 32nd International Conference on Micro Electro Mechanical Systems (MEMS), Seoul, Republic of Korea, 27–31 January 2019; pp. 460–463. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Banerjee, A.; Broadbent, S.; Bulbul, A.; Simmons, M.C.; Kim, K.H.; Mastrangelo, C.H.; Looper, R.; Kim, H. Statistics-Based Gas Sensor. In Proceedings of the 2019 IEEE 32nd International Conference on Micro Electro Mechanical Systems (MEMS), Seoul, Republic of Korea, 27–31 January 2019; pp. 137–140. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Banerjee, A.; Kim, K.H.; Salvant, J.; Looper, R.; Mastrangelo, C.H.; Kim, H. Threshold Point Modulation of a Wake-Up Nano-Gap Gas Sensor. In Proceedings of the 2020 IEEE 33rd International Conference on Micro Electro Mechanical Systems (MEMS), Vancouver, BC, Canada, 18–22 January 2020; pp. 733–736. [Google Scholar] [CrossRef]
- Truong, S.K.M.; Kim, K.H.; Khan, S.U.H.; Salvant, J.; Banerjee, A.; Looper, R.; Mastrangelo, C.H.; Kim, H. Demonstration of 155. In 1 μW Wake-Up Gas Sensor Node Toward 8 Month Lifetime. In Proceedings of the 2020 IEEE 33rd International Conference on Micro Electro Mechanical Systems (MEMS), Vancouver, BC, Canada, 18–22 January 2020; pp. 622–625. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Banerjee, A.; Broadbent, S.; Noh, S.; Kim, K.H.; Bulbul, A.; Looper, R.E.; Mastrangelo, C.H.; Kim, H. Characterization of a Wake-Up Nano-Gap Gas Sensor for Ultra Low Power Operation. J. Microelectromechanical Syst. 2022, 31, 791–801. [Google Scholar] [CrossRef]
- Khan, S.H.; Jung, Y.J.; Hsieh, H.C.; Wu, T.; Mastrangelo, C.H.; Kim, H.; Broadbent, S.J.; Looper, R. Ultra-low-power chemical sensor node. In Proceedings of the GOMAC Tech, Washington, DC, USA, 9 January 2018. [Google Scholar]
- Banerjee, A.; Ghosh, T.; Likhite, R.; Hasan, N.; Kim, H.; Mastrangelo, C.G. Electrical detection of proteins using batch-fabricated vertical metal nanogap break-junctions. In Proceedings of the 20th International Conference on Miniaturized Systems for Chemistry and Life Sciences, MicroTAS, Dublin, Ireland, 9–13 October 2016. [Google Scholar]
- Banerjee, A.; Likhite, R.; Kim, H.; Mastrangelo, C.H. Quantum Tunneling Hygrometer with Temperature Stabilized Nanometer Gap. Sci. Rep. 2020, 10, 4440. [Google Scholar] [CrossRef] [PubMed]
- Schroth, A.; Sager, K.; Gerlach, G.; Häberli, A.; Boltshauser, T.; Baltes, H. A resonant poliyimide-based humidity sensor. Sens. Actuators B Chem. 1996, 34, 301–304. [Google Scholar] [CrossRef]
- Banerjee, A.; Likhite, R.; Kim, H.; Mastrangelo, C. An ultra-low power highly-sensitive vapor sensor based on quantum tunneling. In Proceedings of the The 23rd International Conference onMiniaturized Systems for Chemistry and Life Sciences MicroTAS, Basel, Switzerland, 27–31 October 2019. [Google Scholar]
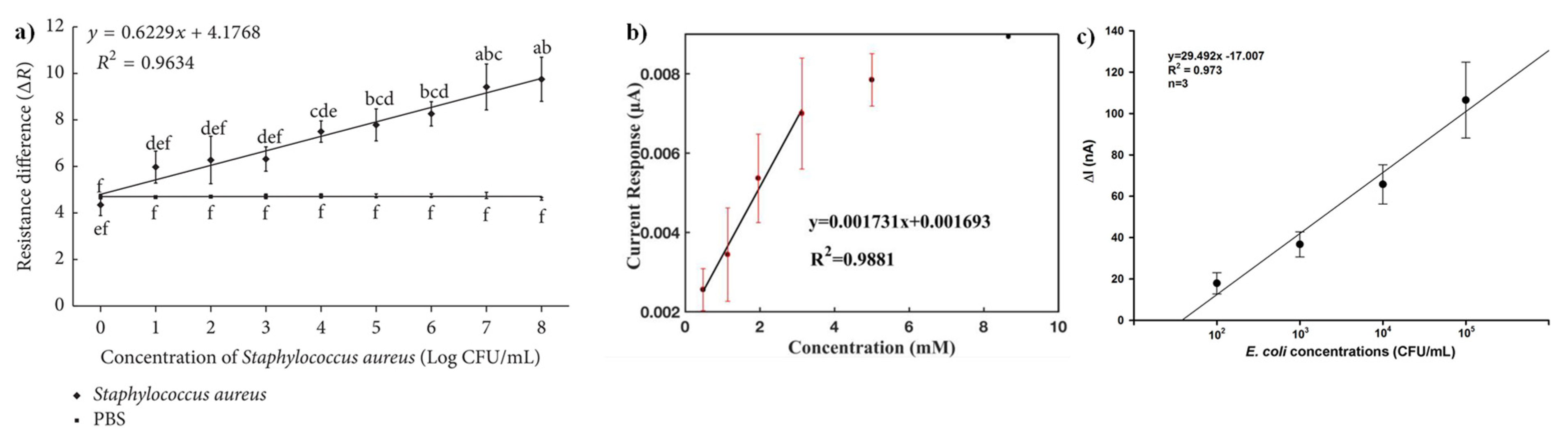
- Banerjee, A.; Maity, S.; Mastrangelo, C.H. Nanostructures for Biosensing, with a Brief Overview on Cancer Detection, IoT, and the Role of Machine Learning in Smart Biosensors. Sensors 2021, 21, 1253. [Google Scholar] [CrossRef]
- Choi, H.-K.; Lee, J.; Park, M.-K.; Oh, J.-H. Development of Single-Walled Carbon Nanotube-Based Biosensor for the Detection of Staphylococcus aureus. J. Food Qual. 2017, 2017, 5239487. [Google Scholar] [CrossRef]
- Ermini, M.L.; Song, X.C.; Špringer, T.; Homola, J. Peptide Functionalization of Gold Nanoparticles for the Detection of Carcinoembryonic Antigen in Blood Plasma via SPR-Based Biosensor. Front. Chem. 2019, 7, 40. [Google Scholar] [CrossRef]
- Yamada, K.; Kim, C.T.; Kim, J.H.; Chung, J.H.; Lee, H.G.; Jun, S. Single walled carbon nanotube-based junction biosensor for detection of escherichia coli. PLoS ONE 2014, 9, e105767. [Google Scholar] [CrossRef]
- Luo, X.; Shi, W.; Yu, H.; Xie, Z.; Li, K.; Cui, Y. Wearable carbon nanotube-based biosensors on gloves for lactate. Sensors 2018, 18, 3398. [Google Scholar] [CrossRef]
- Ah, C.S.; Yun, Y.J.; Lee, J.S.; Park, H.J.; Ha, D.H.; Yun, W.S. Fabrication of integrated nanogap electrodes by surface-catalyzed chemical deposition. Appl. Phys. Lett. 2006, 88, 133116. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Tope, S.; Dolpati, R.; Kim, K.H.; Noh, S.; Bulbul, A.; Mural, R.V.; Banerjee, A.; Schnable, J.C.; Ji, M.; et al. Development of a Gas Sensor for Green Leaf Volatile Detection. In Proceedings of the 2021 21st International Conference on Solid-State Sensors, Actuators and Microsystems (Transducers), Orlando, FL, USA, 20–24 June 2021; pp. 250–253. [Google Scholar] [CrossRef]
- Khan, S.U.H.; Karkhanis, M.; Hatasaka, B.; Tope, S.; Noh, S.; Bulbul, A.; Banerjee, A.; Ji, M.; Mastrangelo, C.H.; Kim, H.; et al. Field Deployment of A Nanogap Gas Sensor For Crop Damage Detection. In Proceedings of the 2022 IEEE 35th International Conference on Micro Electro Mechanical Systems Conference (MEMS), Tokyo, Japan, 9–13 January 2022; pp. 720–723. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banerjee, A.; Mastrangelo, C.H. Resurgence of Electron Quantum Tunneling Sensors. Micro 2022, 2, 679-698. https://doi.org/10.3390/micro2040046
Banerjee A, Mastrangelo CH. Resurgence of Electron Quantum Tunneling Sensors. Micro. 2022; 2(4):679-698. https://doi.org/10.3390/micro2040046
Chicago/Turabian StyleBanerjee, Aishwaryadev, and Carlos H. Mastrangelo. 2022. "Resurgence of Electron Quantum Tunneling Sensors" Micro 2, no. 4: 679-698. https://doi.org/10.3390/micro2040046
APA StyleBanerjee, A., & Mastrangelo, C. H. (2022). Resurgence of Electron Quantum Tunneling Sensors. Micro, 2(4), 679-698. https://doi.org/10.3390/micro2040046




