Bacterial Cellulose Production in Co-Culture Systems: Opportunities, Challenges, and Future Directions
Abstract
1. Introduction
2. The Co-Culture Paradigm: A Strategy for Enhanced Yield and Functionalization of BC
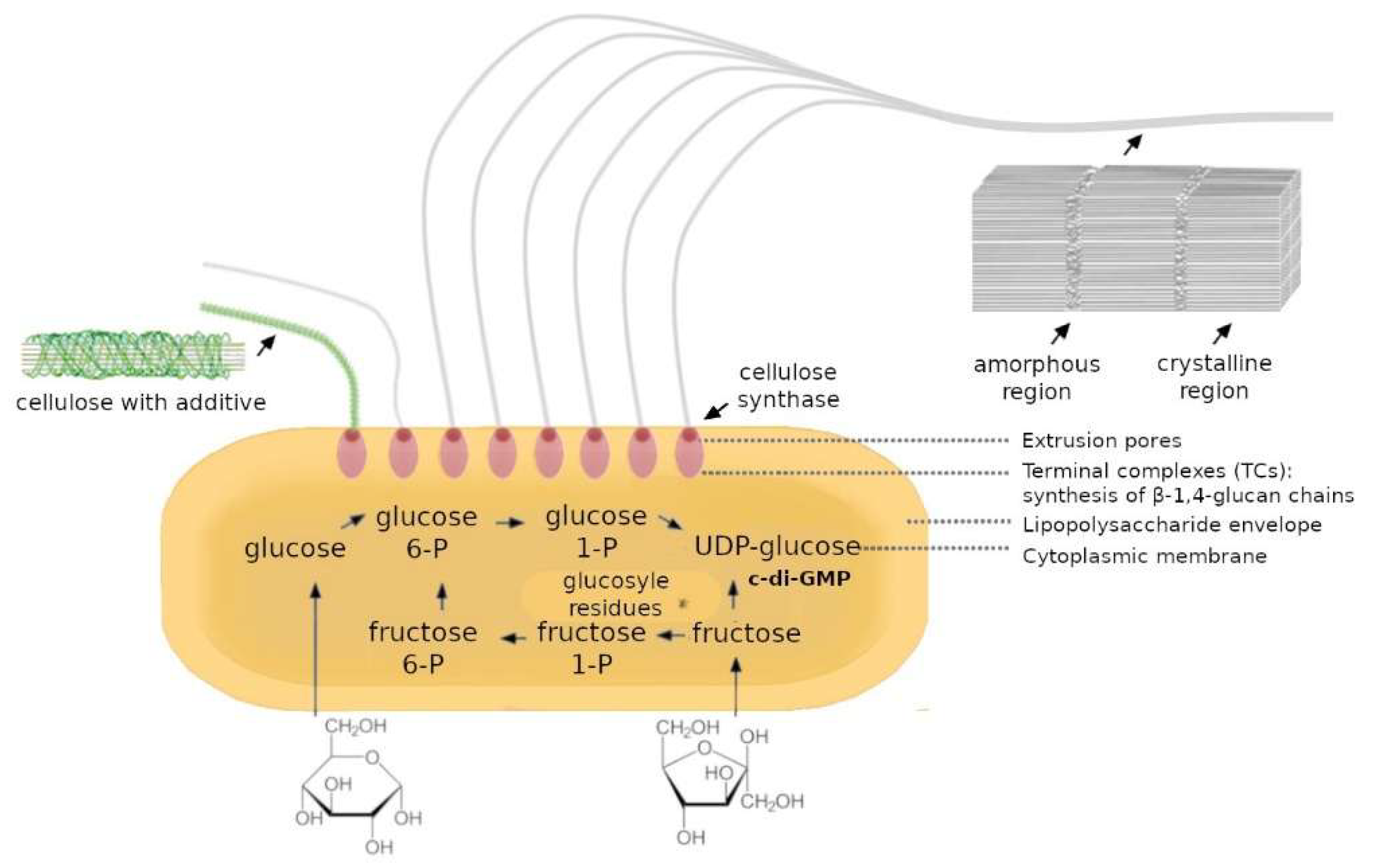
2.1. Overcoming BC Monoculture Limitations Through Co-Culture Strategies
2.2. Bioreactor Configurations for Co-Culture-Based BC Fermentation
- Static Fermentation
- 2.
- Agitation Fermentation
- 3.
- Airlift Bioreactor
- 4.
- Rotating Biological Contactor (RBC)
- 5.
- Dual-Vessel Co-Culture Reactors
- 6.
- Mesh Dispenser Vessel (MDV) Bioreactor
3. Advancements in BC Production: From Natural Consortia to Co-Culture Fermentation Systems
3.1. Key Advances in Co-Culture BC Production by Komagataeibacter
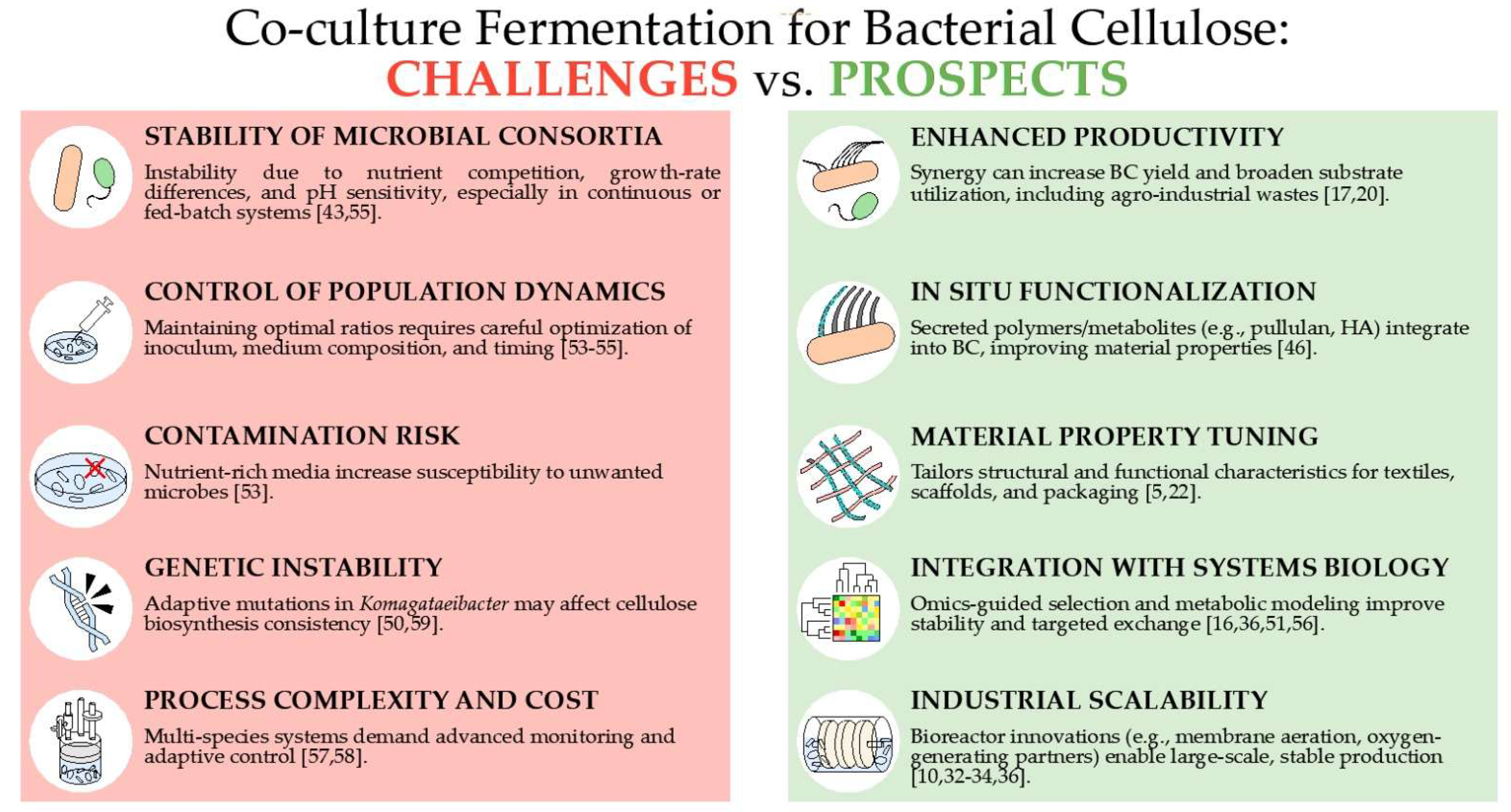
3.2. Key Challenge of Co-Culture Fermentation-Based BC Production
4. Industrial Co-Culture Fermentation: Lessons and Comparison with BC System
5. Techno-Economic Analysis of BC Production: Monoculture vs. Co-Culture
5.1. Cost Considerations in BC Production: Monoculture vs. Co-Culture
5.2. Techno-Economic Analysis and Industrial Feasibility of Co-Culture-Based BC Production
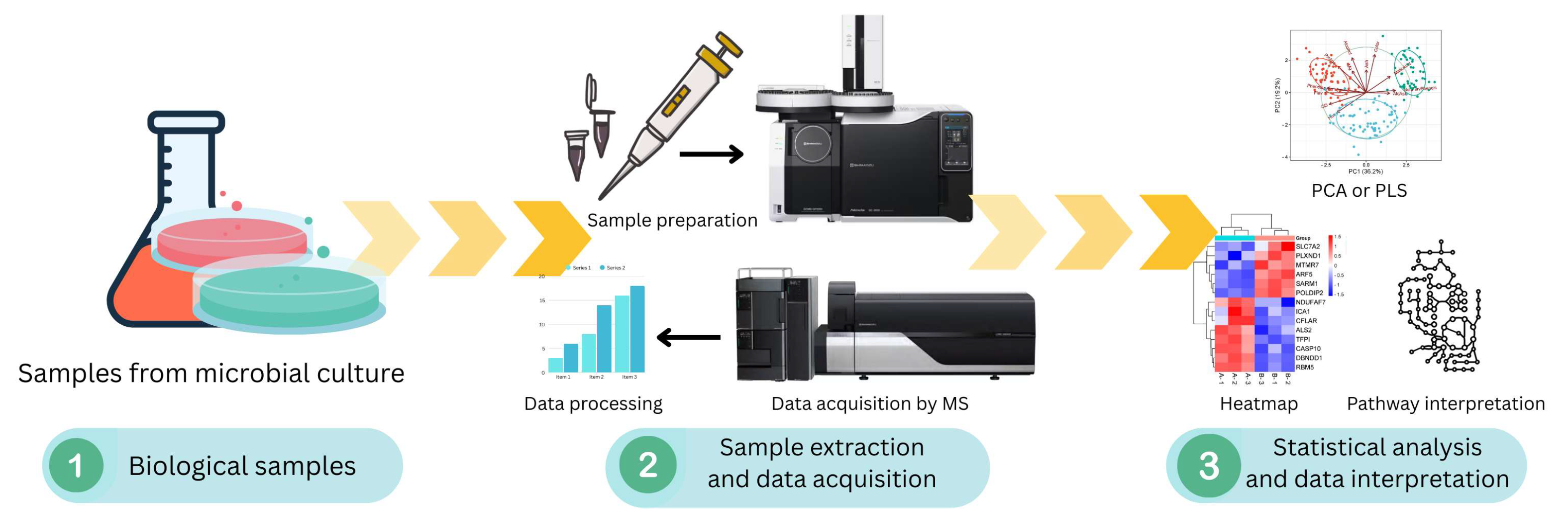
6. Omics Integration for Co-Culture Engineering
7. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Girard, V.-D.; Chaussé, J.; Vermette, P. Bacterial Cellulose: A Review of Production, Applications and Perspectives. J. Appl. Polym. Sci. 2024, 141, e55163. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Flanagan, B.; Gidley, M.J.; Dykes, G.A. Characterization of Cellulose Production by a Gluconacetobacter xylinus Strain from Kombucha. Curr. Microbiol. 2008, 57, 449–453. [Google Scholar] [CrossRef]
- Sozcu, S.; Frajova, J.; Wiener, J.; Venkataraman, M.; Tomkova, B.; Militky, J. Synthesis of Acetobacter xylinum Bacterial Cellulose Aerogels and Their Effect on the Selected Properties. Gels 2025, 11, 272. [Google Scholar] [CrossRef] [PubMed]
- Jozala, A.F.; Pértile, R.A.N.; dos Santos, C.A.; Santos-Ebinuma, V.C.; Seckler, M.M.; Gama, F.M.; Pessoa, A., Jr. Bacterial Cellulose Production by Gluconacetobacter xylinus Employing Alternative Culture Media. Appl. Microbiol. Biotechnol. 2015, 99, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Melro, L.; Alves, C.; Fernandes, M.; Rocha, S.; Mehravani, B.; Ribeiro, A.I.; Azevedo, S.; Cardoso, V.F.; Carvalho, Ó.; Dourado, N.; et al. Bacterial Nanocellulose as a Versatile Scaffold for Biomedical Applications: Synthesis, Functionalization, and Future Prospects. Appl. Mater. Today 2025, 46, 102858. [Google Scholar] [CrossRef]
- Mohamad, N.; Mohd Amin, M.C.; Pandey, M.; Ahmad, N.; Rajab, N.F. Bacterial Cellulose/Acrylic Acid Hydrogel Synthesized via Electron Beam Irradiation: Accelerated Burn Wound Healing in an Animal Model. Carbohydr. Polym. 2014, 114, 312–320. [Google Scholar] [CrossRef]
- Figueiredo, A.R.P.; Figueiredo, A.G.P.R.; Silva, N.H.C.S.; Barros-Timmons, A.; Almeida, A.; Silvestre, A.J.D.; Freire, C.S.R. Antimicrobial Bacterial Cellulose Nanocomposites Prepared by In Situ Polymerization of 2-Aminoethyl Methacrylate. Carbohydr. Polym. 2015, 123, 443–453. [Google Scholar] [CrossRef]
- Fernandes, I.A.A.; Pedro, A.C.; Ribeiro, V.R.; Bortolini, D.G.; Ozaki, M.S.C.; Maciel, G.M.; Haminiuk, C.W.I. Bacterial Cellulose: From Production Optimization to New Applications. Int. J. Biol. Macromol. 2020, 164, 2598–2611. [Google Scholar] [CrossRef]
- Singhania, R.R.; Patel, A.K.; Tsai, M.-L.; Chen, C.-W.; Dong, C.D. Genetic Modification for Enhancing Bacterial Cellulose Production and Its Applications. Bioengineered 2021, 12, 6793–6807. [Google Scholar] [CrossRef]
- Cruz, M.A.; Flor-Unda, O.; Avila, A.; Garcia, M.D.; Cerda-Mejía, L. Advances in Bacterial Cellulose Production: A Scoping Review. Coatings 2024, 14, 1401. [Google Scholar] [CrossRef]
- Öz, Y.E.; Bingül, N.D.; Morçimen, Z.G.; Şendemir, A.; Hameş, E.E. Fabrication of porous bone scaffolds using degradable and mouldable bacterial cellulose. Cellulose 2024, 31, 2921–2935. [Google Scholar] [CrossRef]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M., Jr. The future prospects of microbial cellulose in biomedical applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef]
- Hur, D.H.; Choi, W.S.; Kim, T.Y.; Lee, S.Y.; Park, J.H.; Jeong, K.J. Enhanced Production of Bacterial Cellulose in Komagataeibacter xylinus via Tuning of Biosynthesis Genes with Synthetic RBS. J. Microbiol. Biotechnol. 2020, 30, 1430–1435. [Google Scholar] [CrossRef]
- Zhong, C. Industrial-scale production and applications of bacterial cellulose. Front. Bioeng. Biotechnol. 2020, 8, 605374. [Google Scholar] [CrossRef]
- El-Gendi, H.; El-Sayed, E.S.R.; Hassan, M.L.; Hebeish, A. Recent advances in bacterial cellulose: A low-cost effective production media, optimization strategies and applications. Cellulose 2022, 29, 7495–7533. [Google Scholar] [CrossRef]
- Shi, S.; Wei, Y.; Lin, X.; Liang, H.; Zhang, S.; Chen, Y.; Dong, L.; Ji, C. Microbial metabolic transformation and antioxidant activity evaluation of polyphenols in Kombucha. Food Biosci. 2023, 51, 102287. [Google Scholar] [CrossRef]
- Li, W.; Huang, X.; Liu, H.; Lian, H.; Xu, B.; Zhang, W.; Sun, X.; Wang, W.; Jia, S.; Zhong, C. Improvement in Bacterial Cellulose Production by Co-Culturing Bacillus cereus and Komagataeibacter xylinus. Carbohydr. Polym. 2023, 313, 120892. [Google Scholar] [CrossRef]
- Jiang, H.; Song, Z.; Hao, Y.; Hu, X.; Lin, X.; Liu, S.; Li, C. Effect of co-culture of Komagataeibacter nataicola and selected Lactobacillus fermentum on the production and characterization of bacterial cellulose. LWT Food Sci. Technol. 2023, 173, 114224. [Google Scholar] [CrossRef]
- Lin, X.; Song, Z.; Jiang, H.; Hao, Y.; Hu, X.; Liu, S.; Li, C. Production of bacterial cellulose in the medium with yeasts pre-fermented coconut water or with addition of selected amino acids. Foods 2022, 11, 3627. [Google Scholar] [CrossRef]
- Seto, A.; Saito, Y.; Matsushige, M.; Kobayashi, H.; Sasaki, Y.; Tonouchi, N.; Tsuchida, T.; Yoshinaga, F.; Ueda, K.; Beppu, T. Effective cellulose production by a coculture of Gluconacetobacter xylinus and Lactobacillus mali. Appl. Microbiol. Biotechnol. 2006, 73, 915–921. [Google Scholar] [CrossRef]
- Quijano, L.; Rodrigues, R.; Fischer, D.; Tovar-Castro, J.D.; Payne, A.; Navone, L.; Hui, Y.; Yan, H.; Pinmanee, P.; Poon, E.; et al. Bacterial Cellulose Cookbook: A Systematic Review on Sustainable and Cost-Effective Substrates. J. Bioresour. Bioprod. 2024, 9, 379–409. [Google Scholar] [CrossRef]
- Fernandes, M.; Souto, A.P.; Dourado, F.; Gama, M. Application of bacterial cellulose in the textile and shoe industry: Development of biocomposites. Polysaccharides 2021, 2, 566–581. [Google Scholar] [CrossRef]
- Absharina, D.; Padri, M.; Veres, C.; Vágvölgyi, C. Bacterial cellulose: From biofabrication to applications in sustainable fashion and vegan leather. Fermentation 2025, 11, 23. [Google Scholar] [CrossRef]
- Paronyan, M.; Saghatelyan, L.; Avetisyan, S.; Koloyan, H.; Kinosyan, M.; Bagiyan, V.; Hovhannisyan, S.; Akopian, O.; Hovsepyan, A. Co-Cultivation of Komagataeibacter xylinus MS2530 with Various Yeast Strains: Production and Characterization of Bacterial Cellulose Films. Carbohydr. Polym. Technol. Appl. 2025, 11, 100840. [Google Scholar] [CrossRef]
- Peng, X.-Y.; Wu, J.-T.; Shao, C.-L.; Li, Z.-Y.; Chen, M.; Wang, C.-Y. Co-culture: Stimulate the metabolic potential and explore the molecular diversity of natural products from microorganisms. Mar. Life Sci. Technol. 2021, 3, 363–374. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, J.; Liu, D.; Xia, M.; Song, J.; Liang, K.; Li, C.; Zheng, Y.; Wang, M. Microbial metabolic interaction in fermentation ecosystem and cooperation in flavor compounds formation of Chinese cereal vinegar. Food Sci. Hum. Wellness 2024, 13, 3472–3481. [Google Scholar] [CrossRef]
- Selegato, D.M.; Castro-Gamboa, I. Enhancing chemical and biological diversity by co-cultivation. Front. Microbiol. 2023, 14, 1117559. [Google Scholar] [CrossRef]
- Stanisławska, A.; Szkodo, M.; Staroszczyk, H.; Dawidowska, K.; Kołaczkowska, M.; Siondalski, P. Effect of the ex situ physical and in situ chemical modification of bacterial nanocellulose on mechanical properties in the context of its potential applications in heart valve design. Int. J. Biol. Macromol. 2024, 269, 131951. [Google Scholar] [CrossRef]
- Sundaram, M.K.; Nehru, G.; Tadi, S.R.R.; Katsuno, N.; Nishizu, T.; Sivaprakasam, S. Bacterial Cellulose Production by Komagataeibacter hansenii Utilizing Agro-Industrial Residues and Its Application in Coffee Milk Stabilization. Biomass Convers. Biorefin. 2023, 13, 7971–7981. [Google Scholar] [CrossRef]
- Hung, C.H.; Cheng, C.H.; Guan, D.W.; Wang, S.T.; Hsu, S.C.; Liang, C.M.; Lin, C.Y. Interactions between Clostridium sp. and other facultative anaerobes in a self-formed granular sludge hydrogen-producing bioreactor. Int. J. Hydrogen Energy 2011, 36, 8704–8711. [Google Scholar] [CrossRef]
- Islam, M.U.; Ullah, M.W.; Khan, S.; Shah, N.; Park, J.K. Strategies for Cost-Effective and Enhanced Production of Bacterial Cellulose. Int. J. Biol. Macromol. 2017, 102, 1166–1173. [Google Scholar] [CrossRef]
- Wu, S.C.; Li, M.H. Production of Bacterial Cellulose Membranes in a Modified Airlift Bioreactor by Gluconacetobacter xylinus. J. Biosci. Bioeng. 2015, 120, 444–449. [Google Scholar] [CrossRef]
- Sharma, C.; Bhardwaj, N.K.; Pathak, P. Rotary Disc Bioreactor-Based Approach for Bacterial Nanocellulose Production Using Gluconacetobacter xylinus NCIM 2526 Strain. Cellulose 2022, 29, 7177–7191. [Google Scholar] [CrossRef]
- Pilafidis, S.; Vardaxi, A.; Kourmentza, K.; Pispas, S.; Dimopoulou, M.; Tsouko, E. From Bread Waste to Bacterial Cellulose Nanostructures: Development of a Novel Rotating Disk Bioreactor. Int. J. Biol. Macromol. 2025, 314, 144374. [Google Scholar] [CrossRef]
- Lin, D.; Lopez-Sanchez, P.; Li, R.; Li, Z. Production of Bacterial Cellulose by Gluconacetobacter hansenii CGMCC 3917 Using Only Waste Beer Yeast as Nutrient Source. Bioresour. Technol. 2013, 151, 113–119. [Google Scholar] [CrossRef]
- Loh, J.; Arnardottir, T.; Gilmour, K.; Muratoglu, M.; Ebrahim, M.; Fernandez, J.; Bennet, P.; Shen, H.; Zhang, L.; Tait, S. Enhanced Production of Bacterial Cellulose with a Mesh Dispenser Vessel-Based Bioreactor. Cellulose 2025, 32, 2209–2226. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Jin, C.; Wu, Y. Synthetic biology-powered microbial co-culture strategy and application of bacterial cellulose-based composite materials. Carbohydr. Polym. 2022, 283, 119171. [Google Scholar] [CrossRef]
- Brugnoli, M.; Mazzini, I.; La China, S.; De Vero, L.; Gullo, M. A microbial co-culturing system for producing cellulose-hyaluronic acid composites. Microorganisms 2023, 11, 1504. [Google Scholar] [CrossRef]
- Wang, J.; Tavakoli, J.; Tang, Y. Bacterial Cellulose Production, Properties and Applications with Different Culture Methods—A Review. Carbohydr. Polym. 2019, 219, 63–76. [Google Scholar] [CrossRef]
- Zhang, T.Z.; Liu, L.P.; Ye, L.; Li, W.C.; Xin, B.; Xie, Y.Y.; Jia, S.R.; Wang, T.F.; Zhong, C. The production of bacterial cellulose in Gluconacetobacter xylinus regulated by luxR overexpression of quorum sensing system. Appl. Microbiol. Biotechnol. 2021, 105, 7801–7811. [Google Scholar] [CrossRef]
- Stephens, K.; Pozo, M.; Tsao, C.Y.; Hauk, P.; Bentley, W.E. Bacterial co-culture with cell signaling translator and growth controller modules for autonomously regulated culture composition. Nat. Commun. 2019, 10, 4129. [Google Scholar] [CrossRef]
- Lin, L.; Ma, D.; Wang, Q.; Chen, X.; Zhang, H.; Chen, C. Bottom-up synthetic ecology study of microbial consortia to enhance lignocellulose bioconversion. Biotechnol. Biofuels Bioprod. 2022, 15, 14. [Google Scholar] [CrossRef]
- Fei, S.; Yang, X.; Xu, W.; Zhang, J. Insights into proteomics reveal mechanisms of ethanol-enhanced bacterial cellulose biosynthesis by Komagataeibacter nataicola. Fermentation 2023, 9, 575. [Google Scholar] [CrossRef]
- Rodrigues, D.M.; da Silva, M.F.; Almeida, F.L.C.; de Mélo, A.H.F.; Forte, M.B.S.; Martín, C.; Barud, H.S.; Baudel, H.M.; Goldbeck, R. A Green Approach to Biomass Residue Valorization: Bacterial Nanocellulose Production from Agro-Industrial Waste. Biocatal. Agric. Biotechnol. 2024, 56, 103036. [Google Scholar] [CrossRef]
- Hu, H.; Catchmark, J.M.; Demirci, A. Effects of pullulan additive and co-culture of Aureobasidium pullulans on bacterial cellulose produced by Komagataeibacter hansenii. Bioprocess Biosyst. Eng. 2022, 45, 573–587. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Catchmark, J.M. Bacterial cellulose/hyaluronic acid nanocomposites production through co-culturing Gluconacetobacter hansenii and Lactococcus lactis under different initial pH values of fermentation media. Cellulose 2020, 27, 2529–2540. [Google Scholar] [CrossRef]
- Liu, K.; Catchmark, J.M. Bacterial cellulose/hyaluronic acid nanocomposites production through co-culturing Gluconacetobacter hansenii and Lactococcus lactis in a two-vessel circulating system. Bioresour. Technol. 2019, 290, 121715. [Google Scholar] [CrossRef]
- Liu, K.; Catchmark, J.M. Enhanced mechanical properties of bacterial cellulose nanocomposites produced by co-culturing Gluconacetobacter hansenii and Escherichia coli under static conditions. Carbohydr. Polym. 2019, 219, 12–20. [Google Scholar] [CrossRef]
- Yu, K.; Chua, S.T.; Smith, A.; Smith, A.G.; Ellis, T.; Vignolini, S. Cultivating future materials: Artificial symbiosis for bulk production of bacterial cellulose composites. bioRxiv 2025. [Google Scholar] [CrossRef]
- Li, X.; Chen, Z.; Wang, J.; Mu, J.; Ma, Q.; Lu, X. Symbiosis of acetic acid bacteria and yeast isolated from black tea fungus mimicking the kombucha environment in bacterial cellulose synthesis. Int. Food Res. J. 2023, 30, 1504–1518. [Google Scholar] [CrossRef]
- Hidalgo, C.; Vega, R.; García, A.; Romero, J. Metabolic dynamics of the kombucha consortium during fermentation. Int. J. Food Microbiol. 2013, 165, 7–13. [Google Scholar] [CrossRef]
- Tu, C.; Li, L.; Wang, Y.; Liu, Y.; Zhou, J.; Liu, X.; Zhu, X.; Zhang, J. Dynamics of Microbial Communities, Flavor, and Physicochemical Properties of Kombucha-Fermented Sargassum fusiforme Beverage during Fermentation. LWT 2024, 192, 115729. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhao, B.; Kang, J.; He, C.; Fei, S.; Liu, S.; Qin, X.; Li, C. Lactiplantibacillus plantarum Causes the abnormal fermentation of bacterial cellulose by Komagataeibacter nataicola during nata de coco production. Food Biosci. 2024, 61, 104603. [Google Scholar] [CrossRef]
- Fei, S.; Fu, M.; Kang, J.; Luo, J.; Wang, Y.; Jia, J.; Liu, S.; Li, C. Enhancing bacterial cellulose production of Komagataeibacter nataicola through fermented coconut water by Saccharomyces cerevisiae: A metabonomics approach. Curr. Res. Food Sci. 2024, 8, 100761. [Google Scholar] [CrossRef]
- Anguluri, K.; La China, S.; Brugnoli, M.; Cassanelli, S.; Gullo, M. Better under stress: Improving bacterial cellulose production by Komagataeibacter xylinus K2G30 (UMCC 2756) using adaptive laboratory evolution. Front. Microbiol. 2022, 13, 994097. [Google Scholar] [CrossRef]
- Krystynowicz, A.; Czaja, W.; Wiktorowska-Jeżewska, A.; Gonçalves-Monge, C.; Wronski, T.; Bielecki, S. Factors Affecting the Yield and Properties of Bacterial Cellulose. Pol. J. Environ. Stud. 2002, 11, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, T.P.; Nugroho, D.A. Monitoring bacterial cellulose growth during fermentation with various carbon sources by applying real-time image processing. IOP Conf. Ser. Earth Environ. Sci. 2023, 1183, 012031. [Google Scholar] [CrossRef]
- Nugroho, D.A.; Sutiarso, L.; Rahayu, E.S.; Masithoh, R.E. Utilizing real-time image processing for monitoring bacterial cellulose formation during fermentation. agriTECH 2020, 40, 118–123. [Google Scholar] [CrossRef]
- Moradi, M.; Jacek, P.; Farhangfar, A.; Guimarães, J.T.; Forough, M. The role of genetic manipulation and in situ modifications on production of bacterial nanocellulose: A review. Int. J. Biol. Macromol. 2021, 183, 635–650. [Google Scholar] [CrossRef]
- Sayah, I.; Gervasi, C.; Achour, S.; Gervasi, T. Fermentation Techniques and Biotechnological Applications of Modified Bacterial Cellulose: An Up-to-Date Overview. Fermentation 2024, 10, 100. [Google Scholar] [CrossRef]
- Behera, B.; Laavanya, D.; Balasubramanian, P. Techno-Economic Feasibility Assessment of Bacterial Cellulose Biofilm Production during the Kombucha Fermentation Process. Bioresour. Technol. 2022, 346, 126659. [Google Scholar] [CrossRef]
- Kao, P.M.; Hsu, B.M.; Chang, T.Y.; Chiu, Y.C.; Tsai, S.H.; Huang, Y.L.; Chang, C.M. Biohydrogen production by Clostridium butyricum and Rhodopseudomonas palustris in co-cultures. Int. J. Green Energy 2016, 13, 715–719. [Google Scholar] [CrossRef]
- Kang, Y.; Xiao, J.; Ding, R.; Xu, K.; Zhang, T.; Tremblay, P.L. A Two-Stage Process for the Autotrophic and Mixotrophic Conversion of C1 Gases into Bacterial Cellulose. Bioresour. Technol. 2022, 361, 127711. [Google Scholar] [CrossRef] [PubMed]
- Prado, M.R.; Blandón, L.M.; Vandenberghe, L.P.S.; Rodrigues, C.; Castro, G.R.; Thomaz-Soccol, V.; Soccol, C.R. Milk kefir: Composition, microbial cultures, biological activities, and related products. Front. Microbiol. 2015, 6, 1177. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, S. Yogurt Production. Methods Mol. Biol. 2019, 1887, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Harrison, K.; Curtin, C. Microbial Composition of SCOBY Starter Cultures Used by Commercial Kombucha Brewers in North America. Microorganisms 2021, 9, 1060. [Google Scholar] [CrossRef]
- Costa, A.F.S.; Almeida, F.C.G.; Vinhas, G.M.; Sarubbo, L.A. Production of Bacterial Cellulose by Gluconacetobacter hansenii Using Corn Steep Liquor as Nutrient Sources. Front. Microbiol. 2017, 8, 2027. [Google Scholar] [CrossRef]
- Yang, Y.; Jia, J.; Xing, J.; Chen, J.; Lu, S. Isolation and Characteristics Analysis of a Novel High Bacterial Cellulose Producing Strain Gluconacetobacter intermedius CIs26. Carbohydr. Polym. 2013, 92, 2012–2017. [Google Scholar] [CrossRef]
- Souza, E.F.; Furtado, M.R.; Carvalho, C.W.P.; Freitas-Silva, O.; Gottschalk, L.M.F. Production and Characterization of Gluconacetobacter xylinus Bacterial Cellulose Using Cashew Apple Juice and Soybean Molasses. Int. J. Biol. Macromol. 2020, 146, 285–289. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Guo, S.; Liu, J.; Zhu, P. Preparation and Characterization of Bacterial Cellulose Synthesized by Kombucha from Vinegar Residue. Int. J. Biol. Macromol. 2024, 258, 128939. [Google Scholar] [CrossRef]
- Dourado, F.; Fontão, A.; Leal, M.; Rodrigues, A.C.; Gama, M. Process Modeling and Techno-Economic Evaluation of an Industrial Bacterial Nanocellulose Fermentation Process. In Bacterial Nanocellulose: From Biotechnology to Bio-Economy; Gama, M., Dourado, F., Bielecki, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Henry, S.; Dhital, S.; Sumer, H.; Butardo, V., Jr. Solid-State Fermentation of Cereal Waste Improves the Bioavailability and Yield of Bacterial Cellulose Production by a Novacetimonas sp. Isolate. Foods 2024, 13, 3052. [Google Scholar] [CrossRef] [PubMed]
- Nóvak, I.C.; Segat, B.; Garcia, M.C.F.; Pezzin, A.P.T.; Schneider, A.L.S. Alternative Production of Bacterial Cellulose by Komagataeibacter hansenii and Microbial Consortium. Polímeros 2024, 34, e20240021. [Google Scholar] [CrossRef]
- Wang, S.S.; Han, Y.H.; Chen, J.L.; Zhang, D.C.; Shi, X.X.; Ye, Y.X.; Chen, D.L.; Li, M. Insights into bacterial cellulose biosynthesis from different carbon sources and the associated biochemical transformation pathways in Komagataeibacter sp. W1. Polymers 2018, 10, 963. [Google Scholar] [CrossRef] [PubMed]
- Ryngajłło, M.; Kubiak, K.; Jędrzejczak-Krzepkowska, M.; Jacek, P.; Bielecki, S. Comparative genomics of the Komagataeibacter genus and insights into bacterial cellulose biosynthesis. Microbiol. Open 2019, 8, e00625. [Google Scholar] [CrossRef]
- Liu, H.; Cao, Y.; Guo, J.; Xu, X.; Long, Q.; Song, L.; Xian, M. Study on the isoprene-producing co-culture system of Synechococcus elongatus–Escherichia coli through omics analysis. Microb. Cell Fact. 2021, 20, 6. [Google Scholar] [CrossRef]
- Chang, H.; Zheng, Z.; Li, H.; Xu, Y.; Zhen, G.; Zhang, Y.; Ren, X.; Liu, X.; Zhu, D. Multi-omics investigation of high-transglutaminase production mechanisms in Streptomyces mobaraensis and co-culture-enhanced fermentation strategies. Front. Microbiol. 2025, 16, 1525673. [Google Scholar] [CrossRef]
- Acharya, D.; Miller, I.; Cui, Y.; Braun, D.R.; Berres, M.E.; Styles, M.J.; Li, L.; Kwan, J.; Rajski, S.R.; Blackwell, H.E.; et al. Omics technologies to understand activation of a biosynthetic gene cluster in Micromonospora sp. WMMB235: Deciphering keyicin biosynthesis. ACS Chem. Biol. 2019, 14, 1260–1270. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Xin, J.; Chen, X.; Xu, T.; He, J.; Pan, Z.; Zhang, C. Metabolomic profiles of the liquid state fermentation in co-culture of Eurotium amstelodami and Bacillus licheniformis. Front. Microbiol. 2023, 14, 1080743. [Google Scholar] [CrossRef]
- Liu, H.; Xian, M.; Cao, Y.; Guo, J.; Kan, L.; Xu, X. Omics integration for in-depth understanding of the low-carbon co-culture platform system of Chlorella vulgaris–Escherichia coli. Algal Res. 2023, 75, 103252. [Google Scholar] [CrossRef]

| Phase | Conceptual Overview | Key Studies | Ref. |
|---|---|---|---|
| Natural Consortia | Traditional fermented foods such as kombucha and nata de coco relying on undefined microbial communities (yeasts and bacteria) that interact synergistically to produce BC. | Kombucha: yeast + Komagataeibacter → ethanol → BC; Nata de coco: K. nataicola + Lactobacillus increased BC production. | [16,18] |
| Defined Dual-Microbe Co-Culture Strategies (2006–2020) | Strategic pairing of BC producers with mutualistic partner microbes (e.g., ethanol producers, acid regulators) to enhance and stabilize yields. | G. xylinus + L. mali yielded 3× BC yield compared to monoculture; other examples include BC–HA and BC–PHB composite production. | [20,38] |
| Engineered Co-Cultures and Omics-Guided Design (>2020) | Integration of synthetic biology, signal-responsive gene regulation, and omics-based optimization for fine control of microbial interactions and BC production. | Examples include quorum-sensing-based luxR overexpression in G. xylinus, co-cultures with synthetic signaling modules, and omics-informed pathway engineering in K. nataicola and Komagataeibacter spp. | [39,40,41,42,43] |
| Primary Producer (BC) | Co-Culture Partner(s) | Fermentation Type | Yield | Core Finding (Properties) | Mechanism | Ref. |
|---|---|---|---|---|---|---|
| K. xylinus | B. cereus | Corn stover enzymatic hydrolysate | Fourfold increase in BC yield with lignocellulosic hydrolysate | Robust BC formation under nutrient-rich cultivation | Acetoin and 2,3-butanediol from B. cereus enhance BC synthesis | [17] |
| K. nataicola Q2 | L. fermentum | Coconut water-based medium | BC yield increased by up to 59.5% | Improved crystallinity, reduced thermal degradation, enhanced water retention and mechanical performance | LAB-derived acids activate Krebs cycle; enhanced β-1,4-glucan formation and cell co-aggregation support fiber assembly | [18] |
| G. xylinus | L. mali | Static | 3-fold increase in BC production | Not analyzed | LAB secretes growth-promoting metabolites | [20] |
| K. xylinus MS2530 | Various yeasts | Sterilized brewing waste | BC production improved 4–5-fold compared to monoculture; 2–2.5-fold increase with solely brewing waste | Industrial waste enhances productivity and reduces cost | Yeasts provide ethanol and CO2 that stimulate BC synthesis; waste stream offers nutrients | [24] |
| Komagataeibacter sp. | Lactocaseibacillus sp. | Static | Higher BC yield | Reduced crystallinity, increased fiber size and WHC | In situ incorporation of hyaluronic acid (HA) | [38] |
| K. hansenii | A. pullulans | Molasses medium | Increase of 22.4% over monoculture | Improved Young’s modulus and tensile strength | In situ integration of pullulan into BC matrix | [45] |
| G. hansenii | L. lactis | Static (pH-controlled) | Yield varies with pH; best mechanics at pH 4.0 | Young’s modulus improved to 5029 MPa, altered ribbon width-tuned HA secretion affects BC/HA matrix architecture | Lower pH showed favorable synergy between G. hansenii and HA secretion from L. lactis | [46] |
| G. hansenii | L. lactis (HA+) | Two-vessel circulation | Enhanced yield with HA integration | Controlled HA production, improved crystallinity and mechanical strength | Engineered HA secretion and stirrer bioreactor to optimize lactic acid production of LAB circulating into BC vessel | [47] |
| G. hansenii ATCC 23769 | E. coli ATCC 700728 | Static | Yield increased by 10.8% compared to monoculture | Enhanced mechanical properties | In situ incorporation of mannose-rich exopolysaccharide | [48] |
| K. hansenii | C. reinhardtii | Static | ~20% increase | Enhanced 3D BC architecture formation, overcoming oxygen limitation | In situ generation O2 generation by photosynthetic microalgae | [49] |
| K. intermedius | B. bruxellensis, Z. bisporus | Inoculum ratio optimization (1:10:10) | Maximum dry weight yield of 5.51 g/L under optimized inoculum ratios | Co-culture promoted efficient substrate conversion and BC assembly | Yeasts produced ethanol and growth factors; optimal inoculum ratio also critical for microbes’ interaction | [50] |
| Aspect | Key Challenges | Prior Studies | Potential Mitigation Strategies | Ref. |
|---|---|---|---|---|
| Yield of BC and Material Characteristics | Enhancing BC yield may compromise essential material characteristics such as crystallinity. | K. nataicola + L. fermentum SR improved yield but reduced mechanical performance. | Real-time monitoring of bacterial cellulose formation combined with genetic modification of strains to tailor material characteristics. | [18,19,57,58,59] |
| Stability and Yield Predictability | Co-cultures often lack stability, especially under industrial-scale or prolonged fermentations. | Pre-fermented coconut water became unstable, especially with more than three species. | Designing obligate mutualistic systems and using adaptive laboratory evolution (ALE) to enhance stability. | [19,55] |
| Contamination and Metabolite Interference | Co-culture partners may produce inhibitory metabolites or alter BC structure. | HA-producing LAB enhanced BC yield but reduced crystallinity. | Comprehensive chemical and structural characterization to meet regulatory standards for food and biomedical applications. | [38,46,47] |
| Operational Complexity and Cost | Multispecies cultures require advanced infrastructure and pose economic challenges. | Complex systems increase contamination risk and operational costs. | Using simplified two-member systems, employing real-time multispecies sensing, and integrating economic modeling. | [60,61] |
| Mutualism vs Competition | Microbial interactions may be antagonistic or neutral, not always beneficial. | Some strains outcompete others, leading to reduced BC yield. | Conducting detailed strain screening and utilizing omics-guided metabolic modeling to select true mutualists. | [50,53,54] |
| Genetic Instability | Komagataeibacter spp. exhibit adaptive genetic variation in response to environmental stress and consortial conditions. | Cellulose synthase operon mutations have been observed, affecting production consistency. | Engineering robust Komagataeibacter strains through genomic stabilization and enhanced stress-response regulation. | [55,59] |
| Process Controllability | It is difficult to control population dynamics and metabolic flux in real time. | Population ratios shift dynamically; substrate pulsing helps but is not universal. | Engineering of inducible gene circuits, integrated with feedback control systems utilizing real-time sensor data. | [57,58] |
| (a) Production cost comparison (USD/kg) | |||
|---|---|---|---|
| Parameter | Kombucha-based BC | Beet molasses-based BC | |
| Capital Investment (USD) | 13.72 | 13 | |
| Operating Cost (USD/year) | 3.8 | 7.4 | |
| Production Cost (USD/kg) | 63.8 | 14.8 | |
| ROI (%) | 23.64 | - | |
| IRR (%) | 16.48 | - | |
| Payback Period (years) | 4.2 | - | |
| Net Profit (M USD/year) | - | 3.3 | |
| (b) Substrate cost comparison (USD/L) | |||
| Substrate | Cost (USD/L) | ||
| HS medium | 1.36 | ||
| Black tea + sugar | 0.1 | ||
| Enzyme-treated substrate | 5.58 | ||
| (c) SSF vs. HS medium cost (AUD/g) | |||
| Medium type | Cost (AUD/g) | ||
| HS medium | 17.08 | ||
| SSF-treated RB and cereal dust | 2.51 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Absharina, D.; Putra, F.J.N.; Ogino, C.; Kocsubé, S.; Veres, C.; Vágvölgyi, C. Bacterial Cellulose Production in Co-Culture Systems: Opportunities, Challenges, and Future Directions. Appl. Microbiol. 2025, 5, 92. https://doi.org/10.3390/applmicrobiol5030092
Absharina D, Putra FJN, Ogino C, Kocsubé S, Veres C, Vágvölgyi C. Bacterial Cellulose Production in Co-Culture Systems: Opportunities, Challenges, and Future Directions. Applied Microbiology. 2025; 5(3):92. https://doi.org/10.3390/applmicrobiol5030092
Chicago/Turabian StyleAbsharina, Dheanda, Filemon Jalu Nusantara Putra, Chiaki Ogino, Sándor Kocsubé, Csilla Veres, and Csaba Vágvölgyi. 2025. "Bacterial Cellulose Production in Co-Culture Systems: Opportunities, Challenges, and Future Directions" Applied Microbiology 5, no. 3: 92. https://doi.org/10.3390/applmicrobiol5030092
APA StyleAbsharina, D., Putra, F. J. N., Ogino, C., Kocsubé, S., Veres, C., & Vágvölgyi, C. (2025). Bacterial Cellulose Production in Co-Culture Systems: Opportunities, Challenges, and Future Directions. Applied Microbiology, 5(3), 92. https://doi.org/10.3390/applmicrobiol5030092






