3.1. Effect of IL Mixing Ratio on the Photophysical Behavior of Eu(D-facam)3 in 1-butanol
Upon the addition of EMImOAc to the Eu(
D-facam)
3 butanol solution, a slight increase in ligand absorbance at approximately 310 nm was observed in the absorption spectra, while the CD spectra also changed, including a decrease and increase in the Cotton effect in the shorter- and longer-wavelength regions, respectively (
Figure S1). These results suggest that the local coordination symmetry around the Eu
3+ ion and the chirality of the complex are affected by the interactions between the Eu complex and IL.
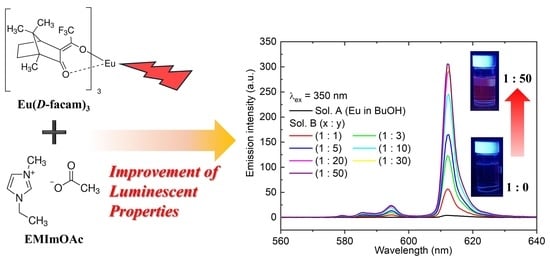
Figure 2a shows the emission spectra of Eu(
D-facam)
3 in 1-butanol upon excitation at 350 nm. The emission intensity was markedly enhanced with the addition of EMImOAc, exhibiting up to a 74-fold increase (
Figure 2b). To examine whether the observed enhancement in emission intensity originates from changes in light absorption, the optical densities at the excitation wavelength (350 nm) were evaluated for representative samples. The optical density at 350 nm was 0.062 for Sol. A (Eu in BuOH) and 0.075 for Sol. B (1:50), indicating only a minor difference in absorption upon the addition of EMImOAc. All emission measurements were performed under strictly identical instrumental conditions, including the excitation light intensity, detector sensitivity, optical slit widths, and integration time, without any normalization of the excitation intensity. Therefore, the pronounced enhancement in emission intensity cannot be attributed to increased absorption of the excitation light or changes in measurement conditions.
Figure 2c shows the relative intensity (
Irel) of the electric-dipole transition at approximately 613 nm normalized to the magnetic-dipole transition at approximately 590 nm for each mixing ratio.
Irel is widely used as an indicator of the local coordination environment of lanthanide ions, since the magnetic-dipole transition is essentially insensitive to the coordination environment, whereas the electric-dipole transition is highly sensitive to the local coordination symmetry around the Eu
3+ ion.
Irel increased sharply upon the addition of five equivalents of EMImOAc and continued to gradually increase at higher concentrations. These results indicate a reduction in the local coordination symmetry around the Eu
3+ ion, which increases the electric-dipole contribution to the f–f transitions, thereby enhancing the transition probability of these parity-forbidden transitions under higher symmetry. An increase in the transition probability enhances the luminescence intensity of the Eu(III) complex. These structural changes induced by the addition of the IL are consistent with the findings by Puntus et al. [
22]. They demonstrated that the luminescence behavior of Eu(III) complexes in ILs is strongly influenced by the strength of the cation–anion interactions, as well as by the molecular size and coordinating ability of the anion. EMImOAc contains a small and strongly coordinating carboxylate anion, which can alter the local coordination environment around Eu
3+ and suppress vibrational quenching caused by –OH groups in 1-butanol. These effects are likely responsible for the observed enhancement in emission intensity.
To obtain complementary evidence for structural association between Eu(
D-facam)
3 and EMImOAc, we also examined the system by ESI-MS (
Figure S3). Spectroscopic identification of subtle coordination changes by IR spectroscopy was considered; however, acetate-related vibrational shifts associated with weak or partial interactions are difficult to resolve in solution, and vibrational modes involving Eu–O coordination are expected to appear in the low-frequency region (around 500 cm
−1), which is beyond the accessible range of our IR setup.
As an alternative and complementary approach, ESI-MS was therefore employed. In the negative-ion mode of Sol. A (Eu in BuOH), the spectrum is dominated by signals attributable to the D-facam ligand and related fragment ions. In contrast, Sol. B (1:50) exhibits an additional peak assignable to a Eu(D-facam)3·OAc− species, indicating association between the Eu(III) complex and acetate anions.
Furthermore, in the positive-ion mode of Sol. B (1:50), signals corresponding to Eu(D-facam)3·2EMIm+·OAc- adduct species were observed, whereas such features were absent in Sol. A. These results suggest that Eu(D-facam)3 is capable of forming associated species with acetate anions and EMIm+ cations in solution.
Although these measurements were performed in an alcohol-based medium and do not directly establish the coordination structure in neat EMImOAc, the observed adduct ions support the presence of specific interactions between Eu(D-facam)3 and EMImOAc components, consistent with the proposed perturbation of the local coordination environment around the Eu3+ ion.
Figure 3 shows the emission decay profiles of Eu(
D-facam)
3 in 1-butanol for the
5D
0 →
7F
2 transition (612 nm). Upon the addition of EMImOAc, the decay behavior changed markedly, with the short-lived component being suppressed, allowing the decay curves to be well fitted by a single-exponential function with a longer lifetime. This behavior suggests that the luminescent environment of the Eu(III) complexes in solution became more homogeneous [
23,
24]. Furthermore, the average emission lifetime was significantly prolonged by the addition of EMImOAc. Specifically, the average emission lifetime of the Eu(
D-facam)
3 solution increased from 66.0 μs in the absence of the IL to 112 μs when 50 equivalents of EMImOAc were added. This indicates a suppression of nonradiative deactivation processes from the
5D
0 excited state of the Eu(III) ion. It is likely that electrostatic interactions between EMImOAc and Eu(
D-facam)
3 lead to the formation of a protective environment around the complex, thereby suppressing vibrational quenching induced by the –OH groups of 1-butanol [
25,
26].
Figure 4 shows the CPL spectra of Eu(
D-facam)
3 in 1-butanol with varying EMImOAc concentrations. Although no CPL signal was observed for Eu(
D-facam)
3 alone, the addition of EMImOAc induced a clear CPL response. This emergence of CPL is consistent with the changes observed in the CD spectra and suggests that the introduction of EMImOAc alters the coordination environment and chirality of the Eu(III) complex. The sensitivity of the CPL to subtle electronic and geometric perturbations within the coordination sphere has been demonstrated in related Eu(III) systems. Hasegawa et al. [
10] reported that even slight variations in ligand or counter-ion interactions can modify the electric and magnetic transition dipole moments through LMCT mixing, resulting in enhanced g
CPL values of up to −1.54. This mechanism is directly relevant to the present system, as the involvement of EMImOAc is expected to induce analogous perturbations around the Eu
3+ center. In line with this mechanism, the luminescence dissymmetry factor (g
CPL), an intensity-independent parameter for CPL, reached −0.28 for the
5D
0 →
7F
1 transition, indicating pronounced chiroptical emission properties. As discussed above, the changes in
Irel and CD spectra indicate a reorganization of the local coordination environment around the Eu
3+ ion induced by interactions with EMImOAc. In our previous study, we found that the interaction between alkylammonium acetate and Eu(
D-facam)
3 in 1-butanol solutions leads to an enhancement of the luminescence properties [
17]. In the present system, similar interactions and associated changes in the local coordination environment are considered to occur. The activation of CPL is likely caused by such local structural reorganization, which modulates the chiral coordination environment of the complex.
3.2. Comparison of the Photophysical Properties of Sol. A (Eu in BuOH), Sol. B (1:50 in BuOH), and Sol. C (Eu in IL)
We prepared the Eu(D-facam)3 solution by directly dissolving it into EMImOAc (Sol. C (Eu in IL)) and compared its photophysical properties with those of Sol. A (Eu in BuOH) and Sol. B (1:50). This comparison allowed us to elucidate how the solvent and IL interacted with the Eu(III) complex and influenced its overall behavior.
The absorption band of Eu(
D-facam)
3 was slightly red-shifted in the IL matrix, reflecting differences in the matrix polarity and the microenvironment around the Eu(III) complexes. The shapes of the CD spectra of Sol. C (Eu in IL) were different from those of Sol. B (
Figure S2). These results suggest that the structure and chirality of the complex are affected by its interactions with the surrounding solvent.
The photophysical parameters of each solution are summarized in
Table 1 (calculations of these parameters and the emission decay profile of Sol. C (Eu in IL) are provided in the
Supporting Information (Figure S4)). The luminescence properties of the pristine Eu(
D-facam)
3 complex have been reported previously in the literature. The luminous properties observed for Sol. A (Eu in BuOH) in the present study are largely consistent with those reported for Eu(
D-facam)
3 in alcohol solution [
16,
17]. The radiative rate (
kr) of the Eu(III) complexes increased upon interaction with the IL, indicating a decrease in the local coordination symmetry around the Eu
3+ ion, as was observed in
Figure 2. The average emission lifetimes were 66.0, 112, and 429 μs for Sol. A, Sol. B, and Sol. C, respectively, indicating an obvious extension of the lifetime in the IL medium (Sol. C). In addition, the luminescent quantum yields of the Eu(III) complexes increased upon interaction with the IL. At high EMImOAc contents, substantial changes in the photophysical parameters, including kr and knr, were observed. To examine whether these changes could be associated with aggregation or association of the Eu(
D-facam)
3 complex, absorption spectra were measured while varying the concentration of Eu(
D-facam)
3 at a fixed EMImOAc content (
Figure S5). As a result, no significant changes in the spectral shape of the ligand-centered absorption bands were observed, and the absorbance increased linearly with concentration, following the Lambert–Beer law (
Figure S5). These results suggest that, within the concentration range studied, Eu(
D-facam)
3 does not form aggregated species that significantly affect its photophysical properties. Taken together, these results suggest that the nonradiative rate constant (
knr) was significantly suppressed, most likely owing to the inhibition of vibrational quenching in the absence of –OH groups from 1-butanol [
25,
26]. Moreover, the ligand-to-Eu
3+ ion energy-transfer efficiency (
ηsens) was also enhanced, indicating that EMImOAc contributes not only to the luminescent process from the excited state of Eu
3+ ions but also to the overall energy-transfer process of the Eu(III) complex. Recent investigations employing time-resolved PL spectroscopy and femtosecond transient absorption spectroscopy have demonstrated that suppression of solvent coordination enhances both the intersystem crossing (ISC) efficiency and the ligand-to-Eu
3+ ion energy-transfer efficiency [
27]. Furthermore, solvent exclusion and the formation of rigid Eu(III) complexes by interaction with organic cations have been reported [
10].
In our system, a similar effect is likely operative. In 1-butanol, the interaction with EMImOAc partially suppresses solvent coordination, whereas in neat EMImOAc, the coordinating solvents are completely excluded. These factors collectively promote the formation of a more rigid coordination environment around the Eu
3+ center, which may in turn enhance the ISC efficiency and the subsequent energy-transfer process. Taken together, these results indicate that the enhancement of the emission intensity observed in
Figure 2 is supported not only by the reduction in the local coordination symmetry around the Eu
3+ ion but also by photophysical factors, such as suppressed nonradiative relaxation and improved energy-transfer efficiency. It has also been reported that ILs can suppress ligand-centered nonradiative processes, thereby stabilizing the intramolecular energy-transfer pathway in other Eu(III) complex systems [
28].
Figure 5 shows the CPL spectrum of Sol. C (Eu in IL). Even in the IL environment, the g
CPL value for the
5D
0 →
7F
1 transition (~593 nm) reached −0.22, which, although slightly lower than the value obtained for Sol. B (−0.28), still demonstrates a pronounced chiral photoluminescent property. This result suggests that Eu(
D-facam)
3 maintains a chiral local environment even in EMImOAc, indicating that the chiral structure of the complex remains stable in the IL medium.
In previously reported Eu-based CPL systems, exceptionally large dissymmetry factors have been achieved by carefully designed chiral coordination environments. For example, camphor-based Eu(III) complexes bearing alkylammonium cations have been reported to exhibit very large |g
CPL| values, reaching up to −1.54, which is among the highest values reported for Eu(III) complexes [
10]. In these systems, the luminescence quantum yields are comparable to those obtained in the present study.
In contrast, although the |gCPL| value of Sol. C (Eu in IL) is smaller than those record values, the present system offers distinct advantages in terms of simplicity and operational robustness. Strong luminescence and CPL activity are achieved simply by mixing Eu(D-facam)3 with EMImOAc, without the need for elaborate molecular design or rigid host matrices. Moreover, the absence of volatile molecular solvents and the low-volatility nature of the ionic liquid enables stable operation over a wide temperature range. These features place the Eu(D-facam)3/EMImOAc system among competitive Eu-based CPL platforms, particularly for applications requiring thermal robustness and facile processability.
3.3. Temperature Dependence of Luminescent Properties
Next, the temperature dependences of the luminescence intensity and lifetime were evaluated. Because the emission intensity of Sol. A (Eu in BuOH) was quite low, a detailed discussion of its emission properties was difficult. Therefore, we focused on the luminescent properties of Sol. B (1:50) and Sol. C (Eu in IL).
Figure 6 shows the temperature-dependent changes in maximum emission intensity for the two solutions over the range from 20 to 100 °C. For Sol. B (1:50), the emission intensity decreased drastically with increasing temperature, reaching only 39% and 2.1% of its initial value at 30 and 60 °C, respectively, indicating that maintaining luminescence performance under high-temperature conditions is difficult in this system. This luminescence quenching with increasing temperature is attributed to the accelerated vibrational deactivation from the excited state of the Eu
3+ center and the reverse energy transfer from the excited Eu
3+ state to the T
1 state of the ligand [
29]. In contrast, Sol. C (Eu in IL) retained 50% and 28% of its initial intensity at 70 and 100 °C, respectively, showing a much more gradual decrease. These results demonstrate that EMImOAc serves as an effective medium for enhancing the thermal stability of Eu(
D-facam)
3, and the high emission retention of Sol. C makes it a promising candidate for luminescent devices requiring thermal robustness. In addition, although systematic thermal cycling tests were not conducted, the emission intensity of Sol. B (1:50) and Sol. C (Eu in IL) were found to be largely reversible over several heating–cooling cycles between 20 and 100 °C (
Figure S6), indicating good thermal cycling stability of the luminescence.
Figure 7 shows the temperature dependence of the emission lifetimes for the two solutions, measured from 20 to 100 °C (up to 90 °C for Sol. B (1:50)). The emission lifetimes of Sol. C (Eu in IL) decreased with increasing temperature, and this decrease generally corresponded with a decrease in emission intensity. It retained approximately 30% of its room-temperature lifetime even at 100 °C, indicating that the complex maintains a relatively long lifetime under high-temperature conditions. Sol. B (1:50) also exhibited a decrease in the emission lifetime with increasing temperature; however, the extent of this reduction was not significantly different from that observed for Sol. C. Notably, while Sol. B exhibited a remarkable decrease in emission intensity at high temperatures—for example, retaining only approximately 2% of its room-temperature intensity at 60 °C (
Figure 6)—the emission lifetime remained comparatively less affected, maintaining approximately 35% of its room-temperature value at the same temperature. This suggests that the pronounced decrease in the emission intensity of Sol. B is not primarily due to an increase in the nonradiative decay process from the
5D
0 excited state of the Eu
3+ ion, but rather because of a significant reduction in the efficiency of energy transfer from the ligand excited states to the Eu
3+ center.
Temperature-dependent luminescence quenching of lanthanide complexes is commonly analyzed using the Arrhenius analysis, in which thermally activated nonradiative processes are described by an activation energy. This approach has been widely applied to evaluate both emission intensity and lifetime variations with temperature in Eu(III) and other lanthanide-based luminescent systems [
29,
30].
Figure 8 shows the Arrhenius plots constructed from the temperature dependence of the inverse of emission intensity (1/
Itot) of Eu(
D-facam)
3. Linear regression analysis of the Arrhenius plots showed good linearity within the analyzed temperature ranges (20–100 °C), with correlation coefficients (R
2) of 0.998 for Sol. B (1:50) and 0.963 for Sol. C (Eu in IL). In Sol. B, the emission intensity decreased sharply with increasing temperature, indicating that the overall nonradiative relaxation pathways of the complex were strongly promoted at high temperatures. In contrast, the slope of Sol. C was much smaller, suggesting that the increase in nonradiative processes was significantly suppressed with increasing temperature, resulting in stable emission behavior against temperature variations. These results demonstrate that EMImOAc is an effective medium for suppressing the temperature dependence of nonradiative relaxation in Eu complexes. Moreover, the stable thermal luminescence behavior of Sol. C (Eu in IL) highlighted its potential application as a temperature-responsive luminescent material.
Figure 9 shows the Arrhenius plots constructed from the inverse of the temperature-dependent emission lifetimes (1/
τave). Linear regression analysis of the Arrhenius plots based on the emission lifetimes showed good linearity within the analyzed temperature range (Sol. B: 20–90 °C; Sol. C: 20–100 °C), with correlation coefficients (R
2) of 0.988 for Sol. B (1:50) and 0.956 for Sol. C (Eu in IL). It should be noted that the lifetime-based analysis reflects only the deactivation processes occurring from the
5D
0 excited state of Eu
3+ ions and therefore does not include any temperature-dependent changes occurring in the ligand-centered excited states or in the ligand-to-Eu
3+ energy-transfer processes. For both solutions, the variation in lifetime with increasing temperature was relatively small, indicating that the temperature dependence of nonradiative relaxation was weaker than that derived from the emission-intensity-based analysis (
Figure 8). In particular, even in 1-butanol, complexation with EMImOAc effectively suppressed the activation of nonradiative transitions from the Eu(III) excited state and stabilized the temperature dependence.
The activation energies of the nonradiative processes were determined from the temperature dependence of the emission intensity and lifetime based on the Arrhenius equations ((2) for emission intensity and (3) for emission lifetime), and the differences in photophysical properties between the two solutions were examined.
Assuming that the absorption of the ligand is temperature-independent, the inverse of total emission intensity (1/
Itot) can be approximated by the emission quantum yield (
Ftot) and expressed as the product of the radiative rate constant (
kr), emission lifetime (
τave), and the energy-transfer efficiency from the ligand to the Eu
3+ ion (
ηsens), as shown in Equation (4).
The inverse of the emission lifetime (1/
τave) is represented by the following kinetic relationship.
Because the radiative rate constant (
kr) is known to be less temperature-dependent [
30], the activation energy obtained from the Arrhenius analysis of the emission lifetime using Equation (3) mainly reflects the temperature dependence of the nonradiative decay rate constant (
knr) corresponding to the local relaxation pathways from the
5D
0 level of Eu(III) to the ground state. In contrast, the analysis based on emission intensity (Equation (2)) reflects not only the temperature dependence of
knr but also the overall energy-transfer processes within the complex, including radiative and nonradiative transitions from the ligand excited states (S
1 and T
1) and the subsequent energy transfer from the ligand to the Eu
3+ ion. These combined contributions reflect the temperature dependence of the overall energy-transfer dynamics within the system. The difference between the activation energies obtained using Equations (2) and (3) are therefore considered to primarily reflect the temperature dependence of the rate constants associated with the processes that contribute to the energy-transfer efficiency (
ηsens). Accordingly, a comparison of these two Arrhenius analyses allows the evaluation of how the energy-transfer efficiency between the ligand and Eu
3+ ion varies with temperature.
Table 2 lists the activation energies of Sol. B (1:50) and Sol. C (Eu in IL) calculated from the emission intensity (
Ea) and emission lifetime (
E’a) dependence.
In Sol. B (1:50), the activation energy determined from the emission lifetime was relatively low (18.9 kJ mol−1), whereas that derived from the emission intensity was remarkably high (77.8 kJ mol−1). This large discrepancy suggests a significant decrease in the energy-transfer efficiency (ηsens) from the ligand to the Eu3+ ion with increasing temperature. In contrast, in Sol. C (Eu in IL), the activation energies derived from both emission lifetime (13.7 kJ mol−1) and emission intensity (13.5 kJ mol−1) were nearly identical, indicating that the ligand-related nonradiative processes exhibit minimal temperature dependence. The marked difference in temperature dependence of ηsens between Sol. B and Sol. C can likely be attributed to the distinct coordination environments created by the two solvent systems. In particular, the IL medium provided a rigid complexation environment that stabilized the ligand excited states and suppressed nonradiative deactivation pathways at higher temperatures. This behavior contrasts with that in 1-butanol, in which solvent coordination cannot be completely eliminated. These results clearly demonstrate that ILs offer a thermally robust environment that maintains highly efficient ligand-to-Eu3+ ion energy transfer.
From a temperature-sensing perspective, photophysical parameters other than emission intensity are more suitable for reliable readout. In particular, the long emission lifetime characteristic of trivalent lanthanide ions enables time-gated detection, allowing efficient discrimination of the Eu3+ emission from background fluorescence. In addition, circularly polarized luminescence provides an orthogonal optical parameter that facilitates discrimination from other emissive components, such as unpolarized or linearly polarized light, while both the emission lifetime and the dissymmetry factor (gCPL) are intrinsically independent of excitation intensity, making them promising metrics for stable and robust temperature sensing.