Metal–Porphyrin-Based Covalent Organic Framework Composite Membrane for Salts and Dyes Separation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
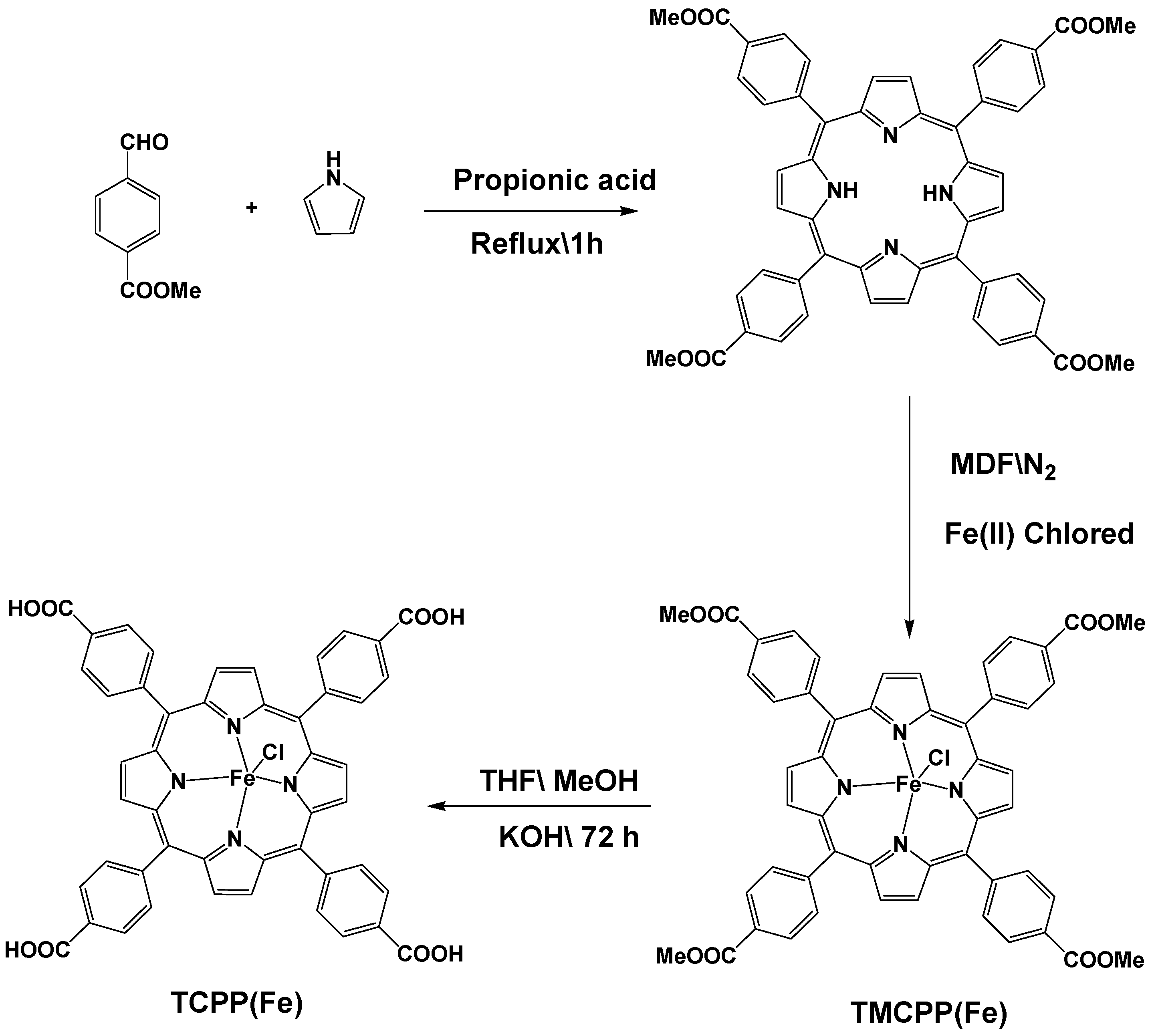
2.2. Synthesis of TMCPP (5,10,15,20–Tetrakis (4-Methoxycorbonyl Phenyl) Porphyrinate)
2.3. Synthesis of TMCPP (5,10,15,20–Tetrakis (4-Methoxycorbonyl Phenyl) Porphyrinate) with Fe (II)
2.4. Synthesis of TCPP (Fe (III))
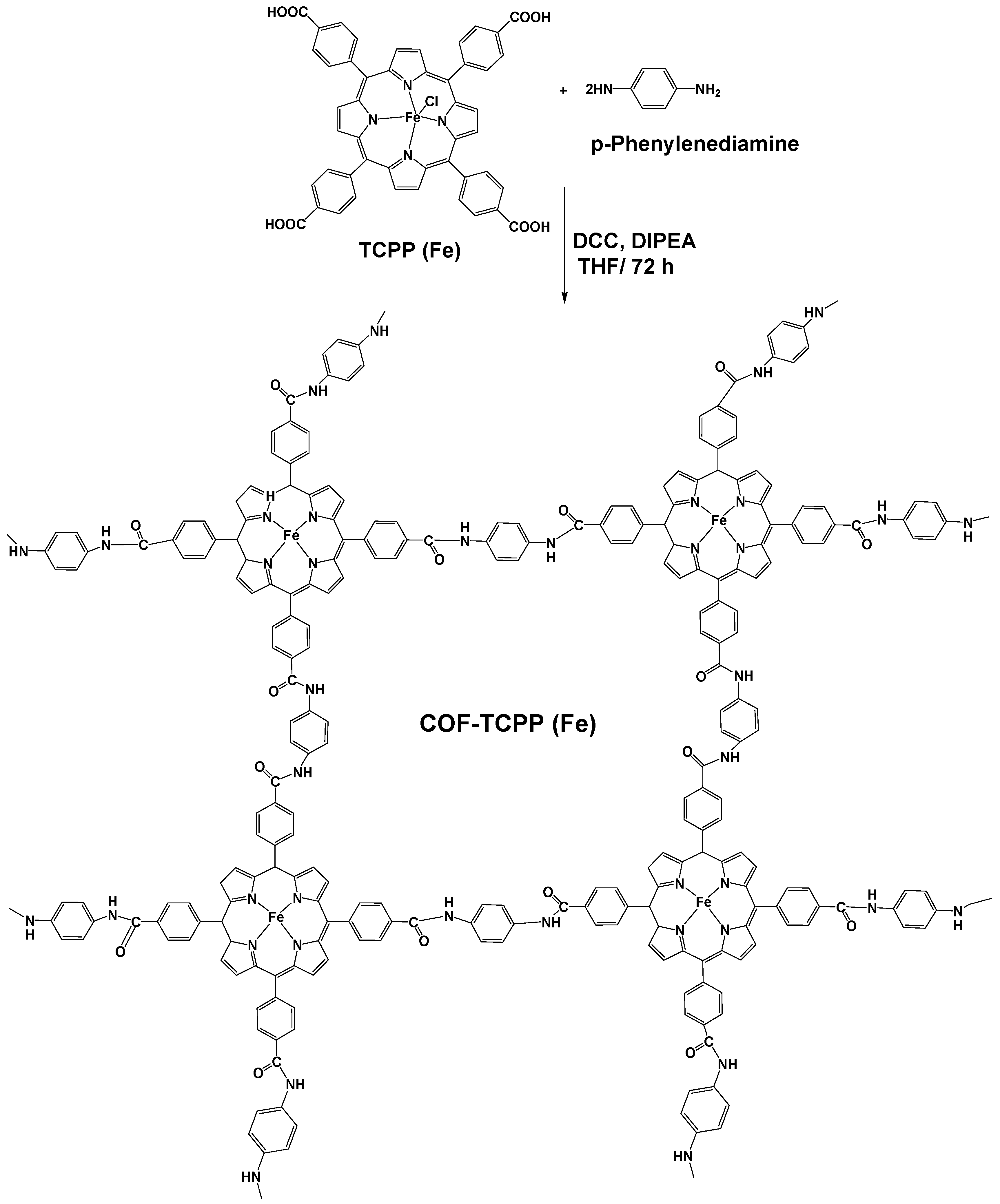
2.5. Synthesis of COF-TCPP (Fe)
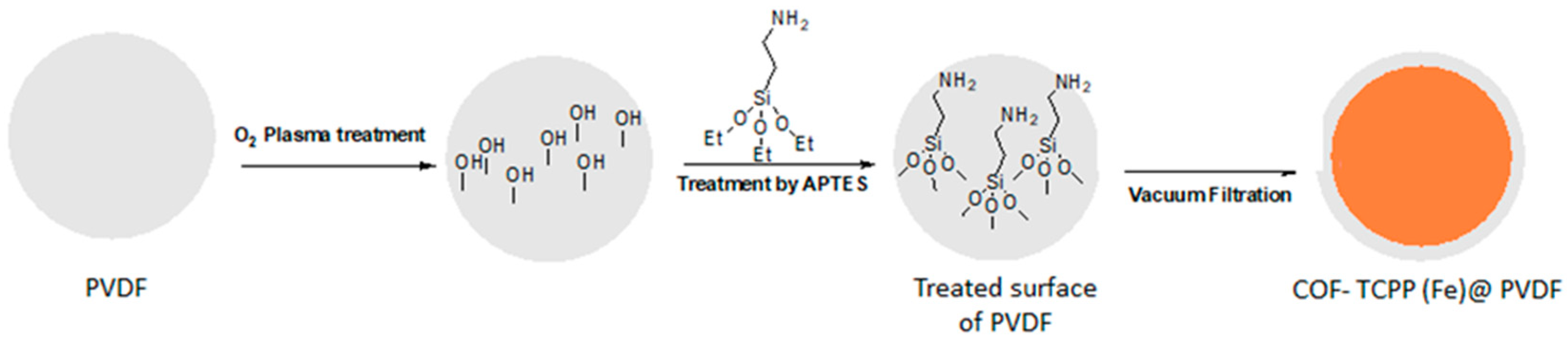
2.6. Membrane Pre-Treatment
2.7. Preparing COF-TCPP (Fe)@ PVDF Membrane
2.8. COFs and Membranes Characterization
2.9. Nanofiltration Experiments
3. Results and Discussion
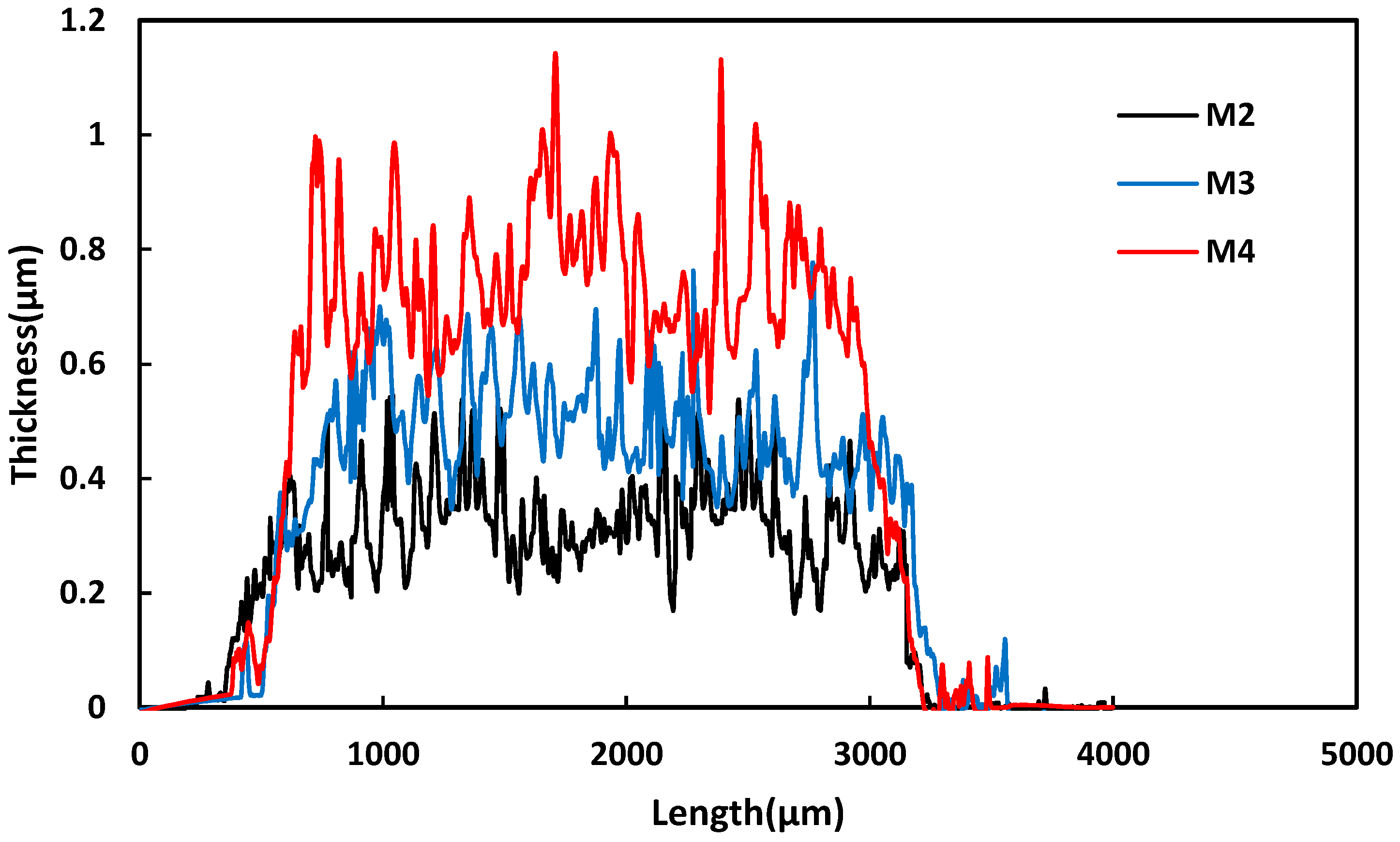
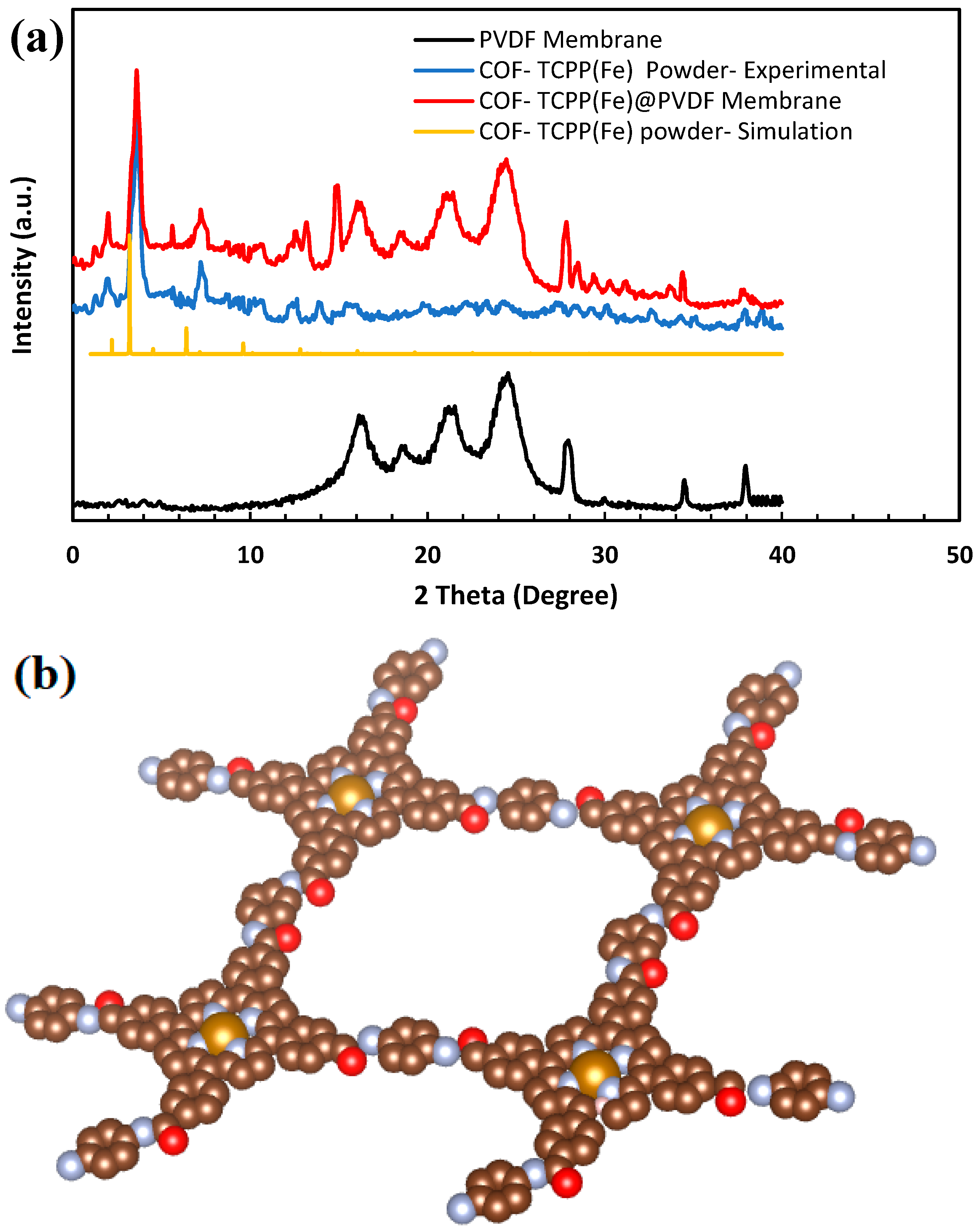
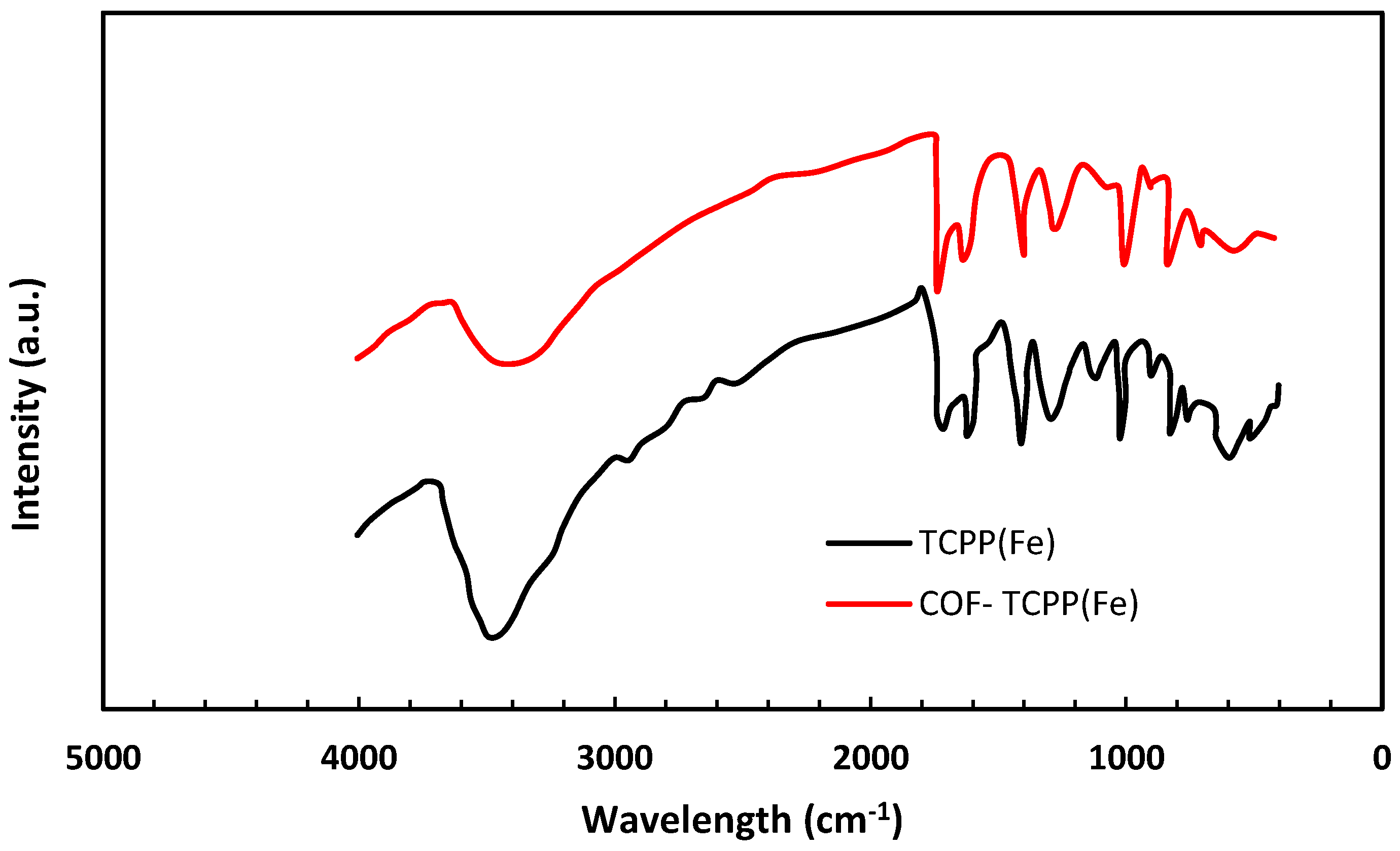
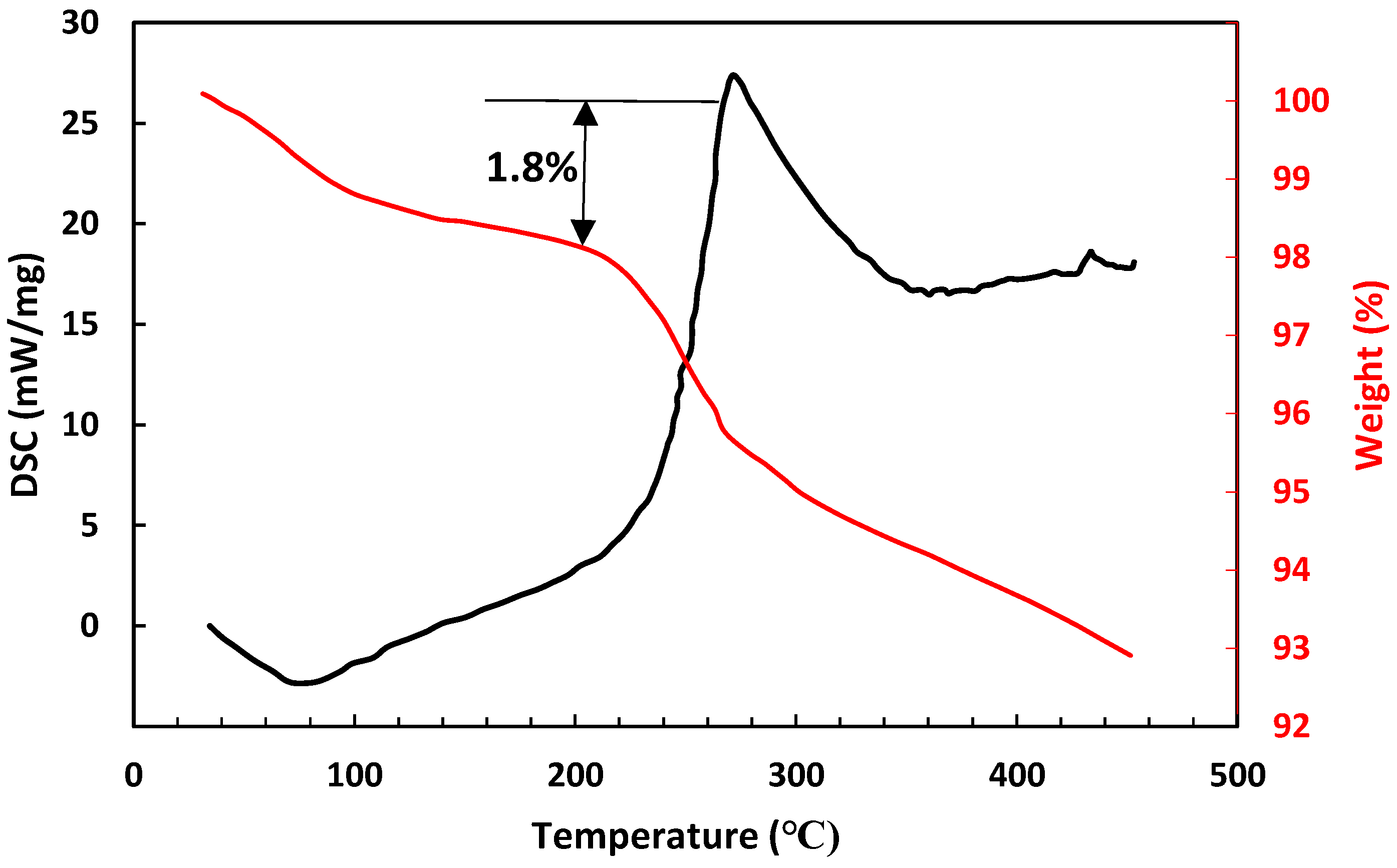
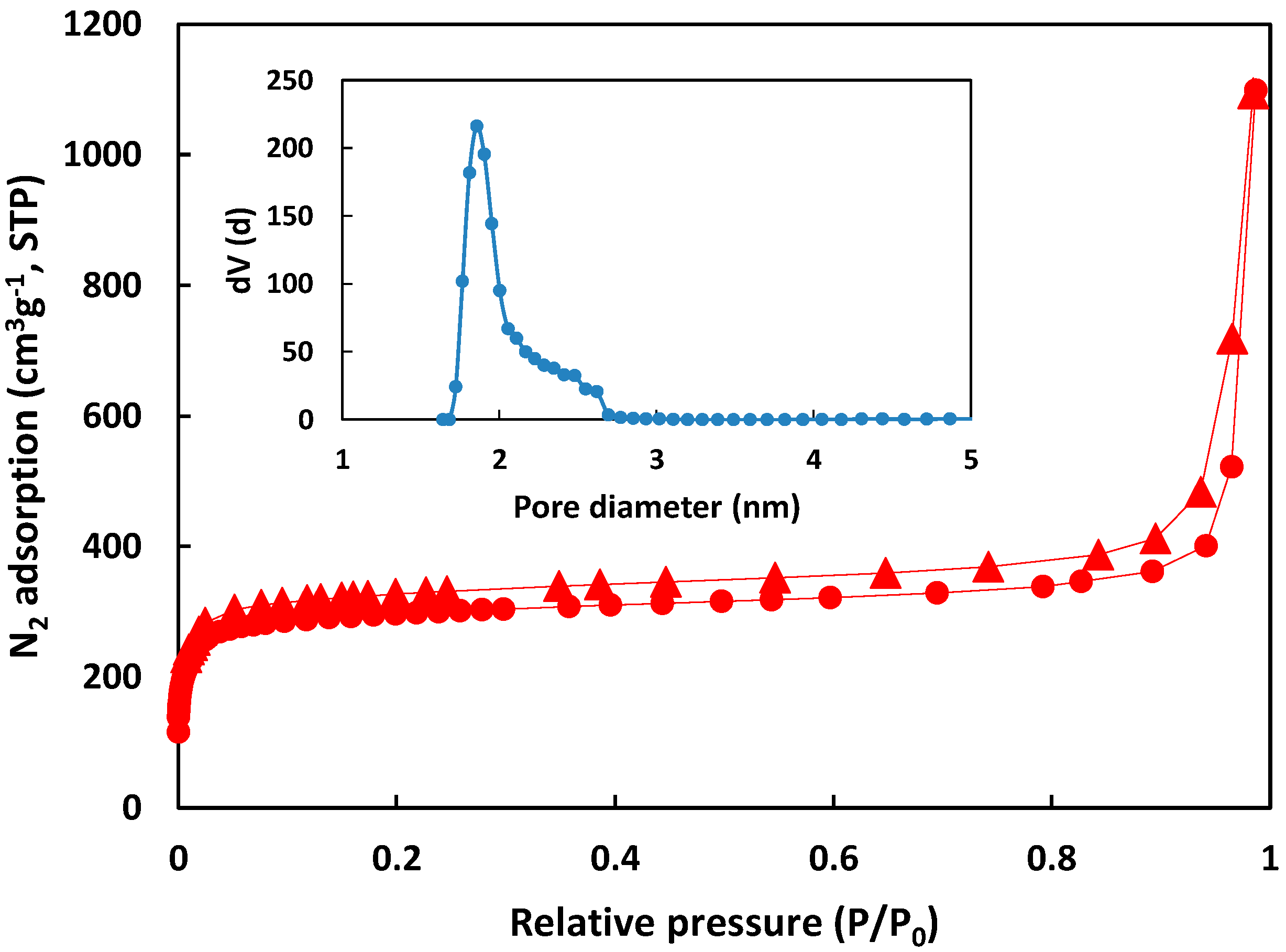
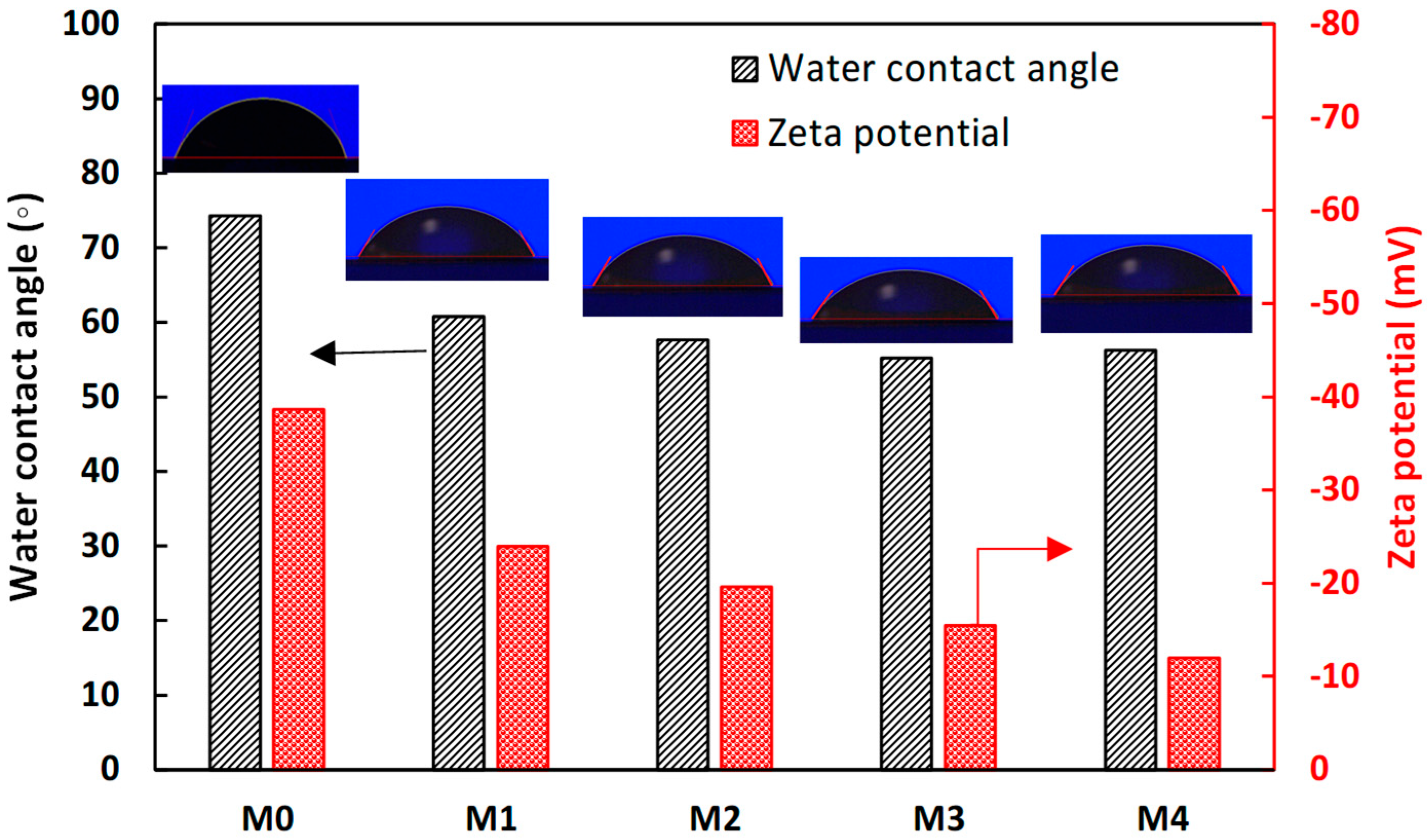
3.1. Membrane Characterization
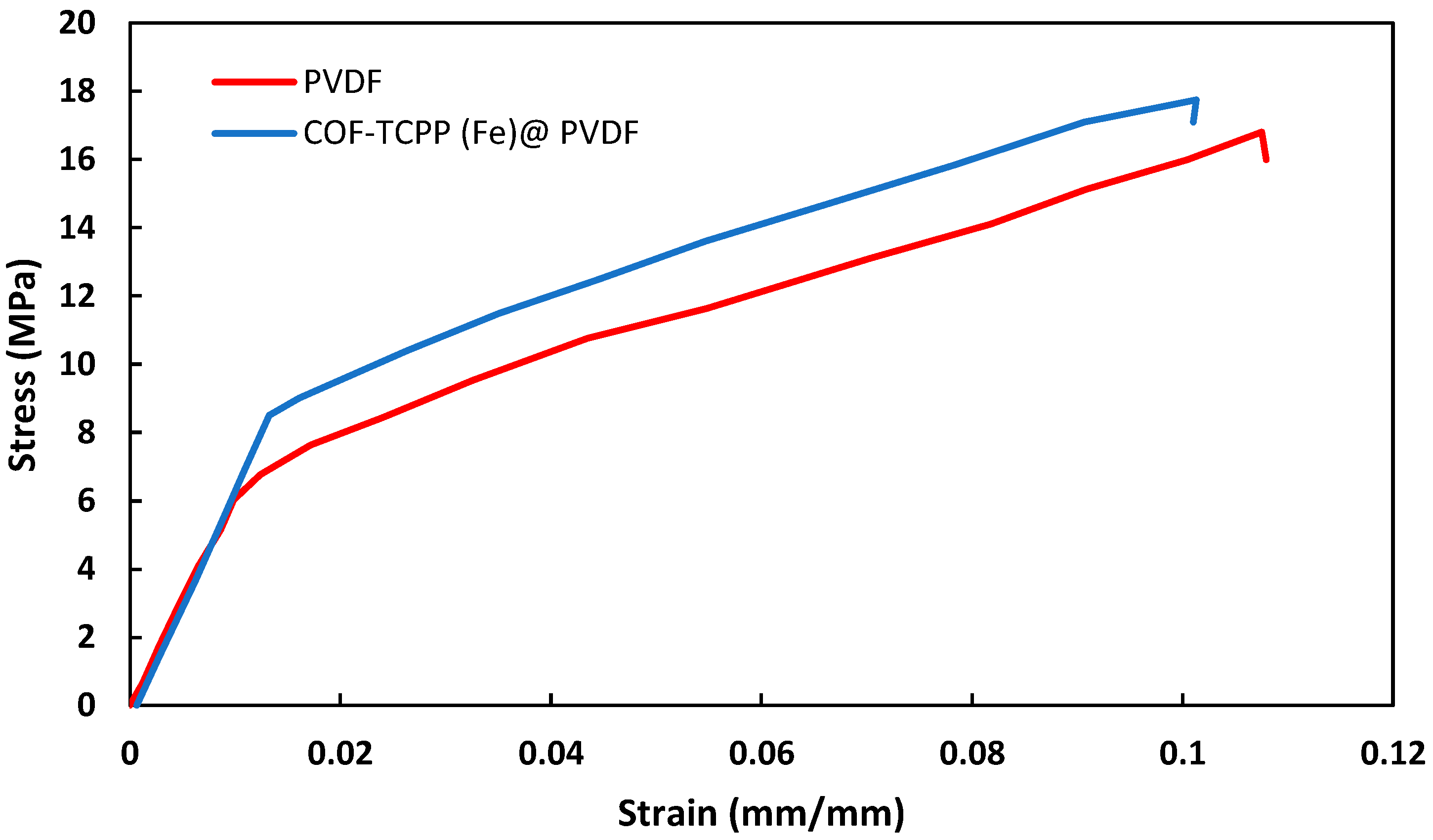
3.2. Membrane Surface and Mechanical Properties
3.3. Performance of COF-TCPP (Fe) @ PVDF Membrane
3.3.1. Permeability/Selectivity Performance
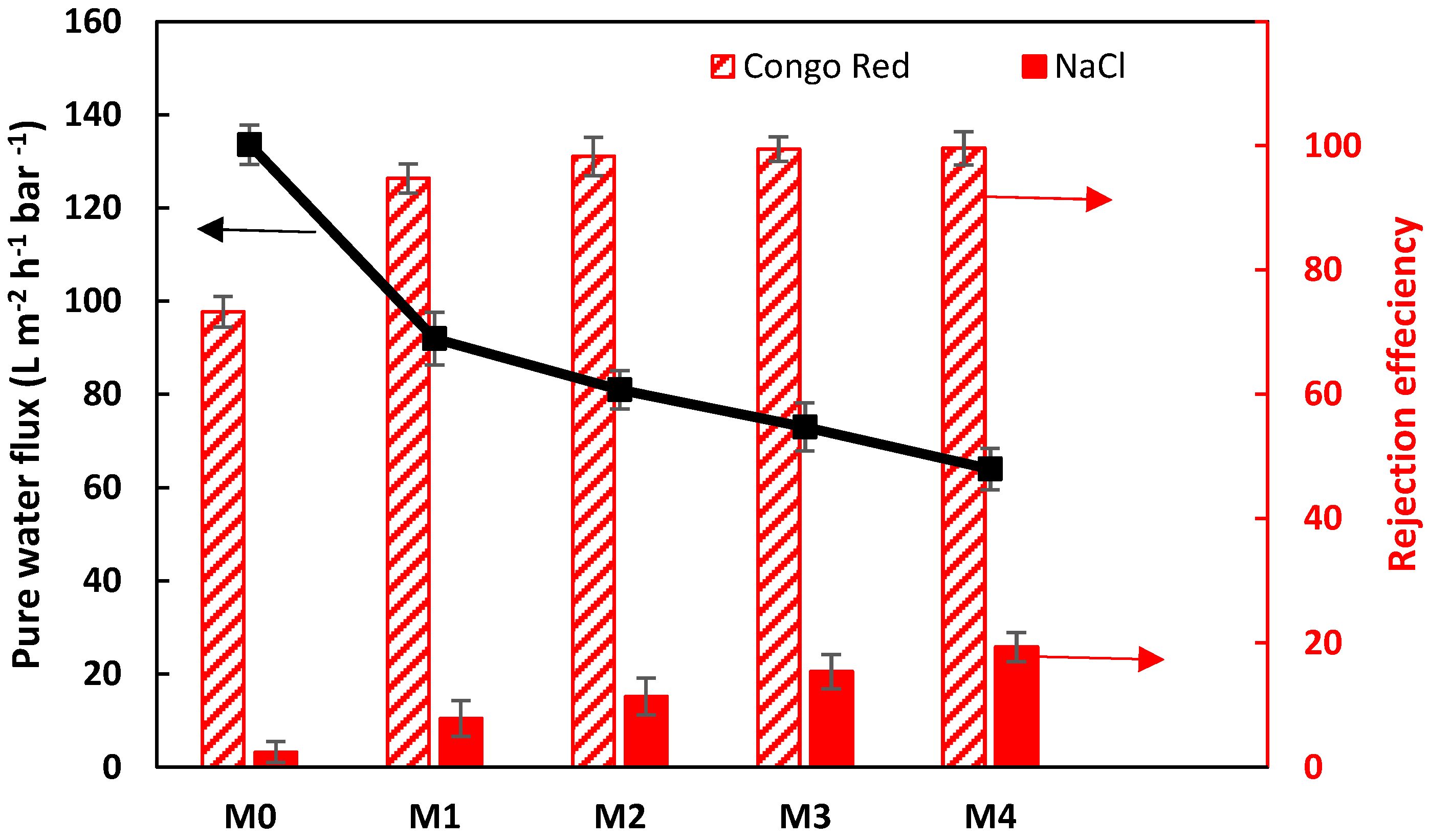
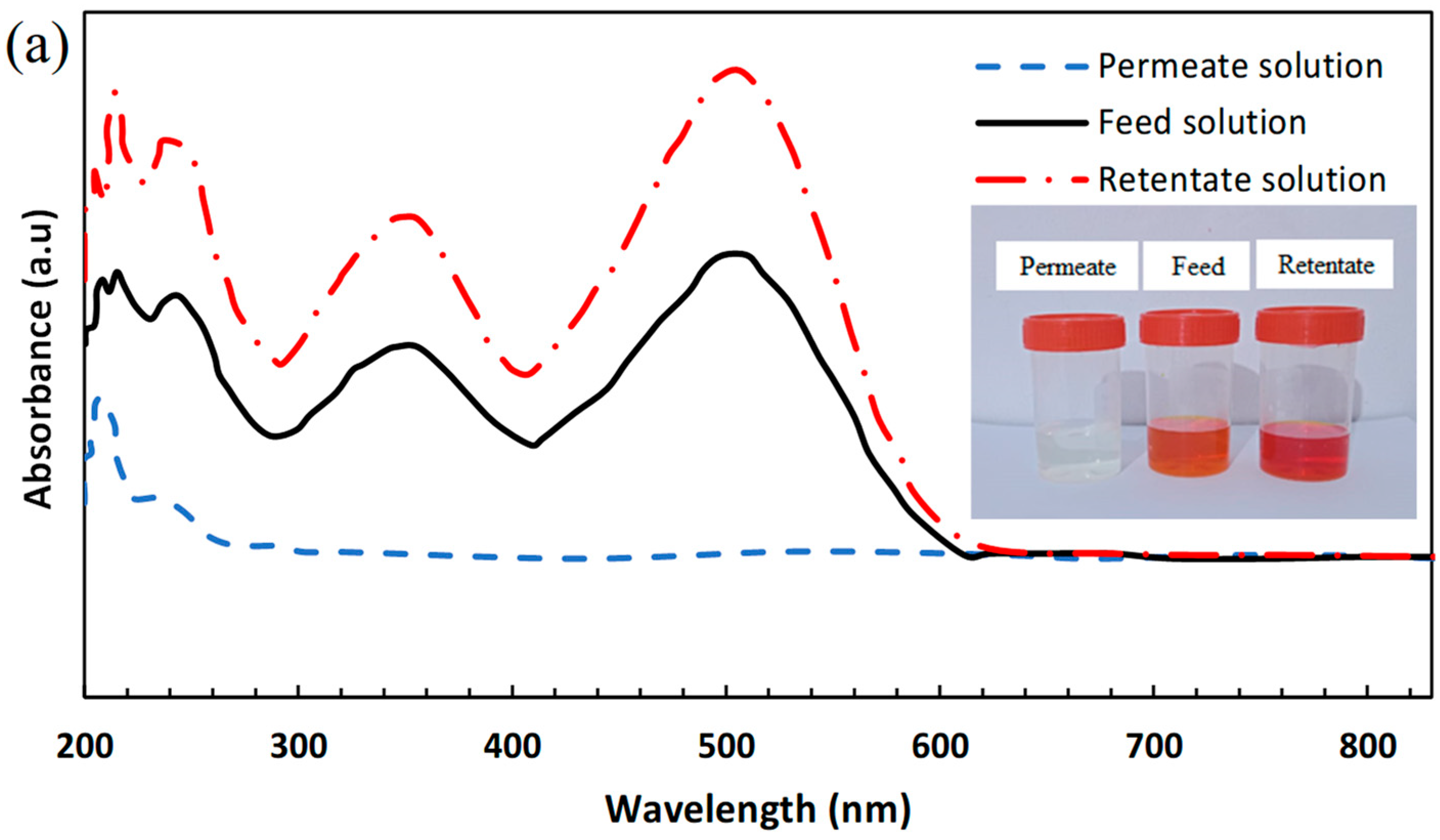
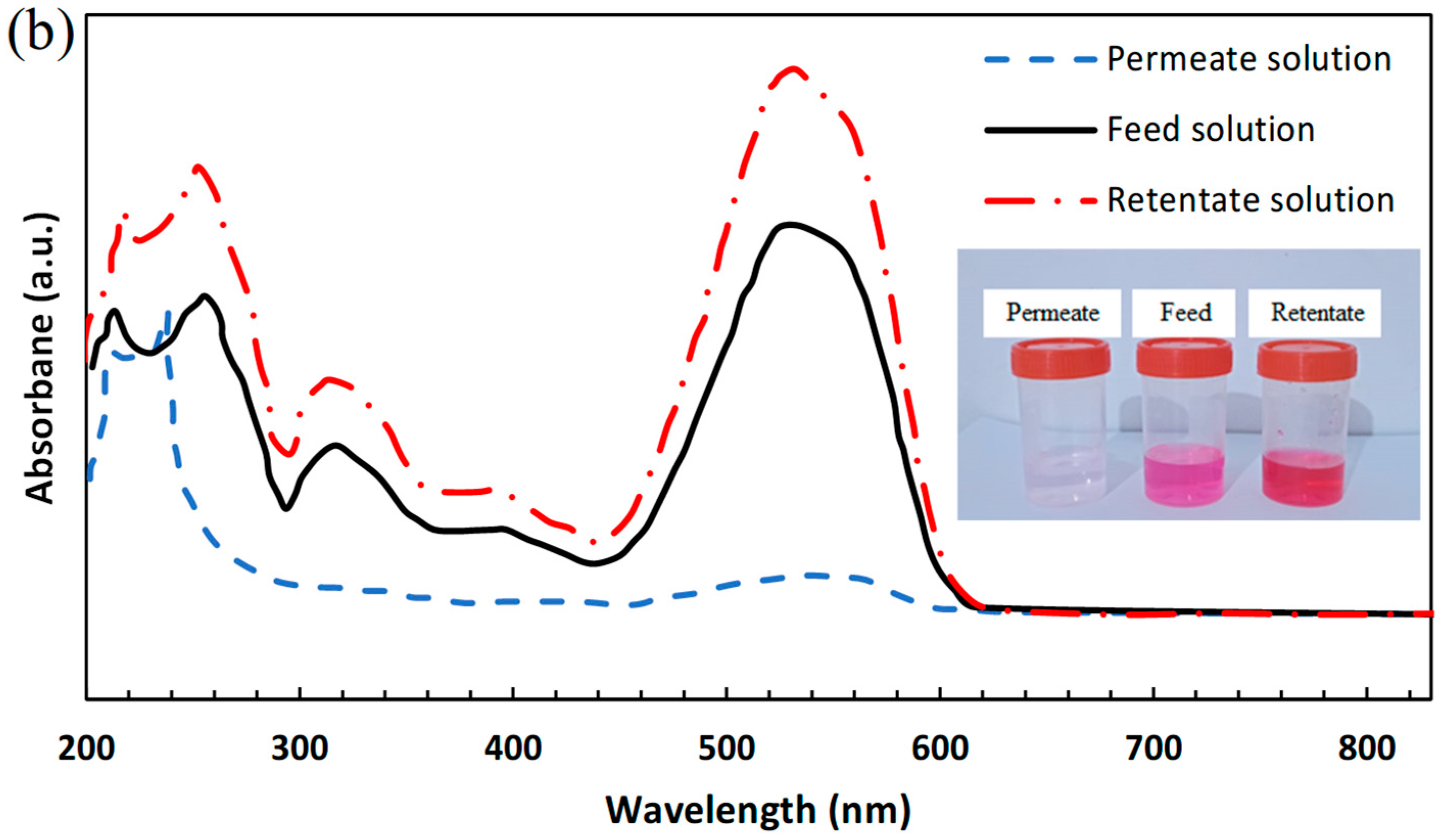
3.3.2. Single Salt/Dye Separation
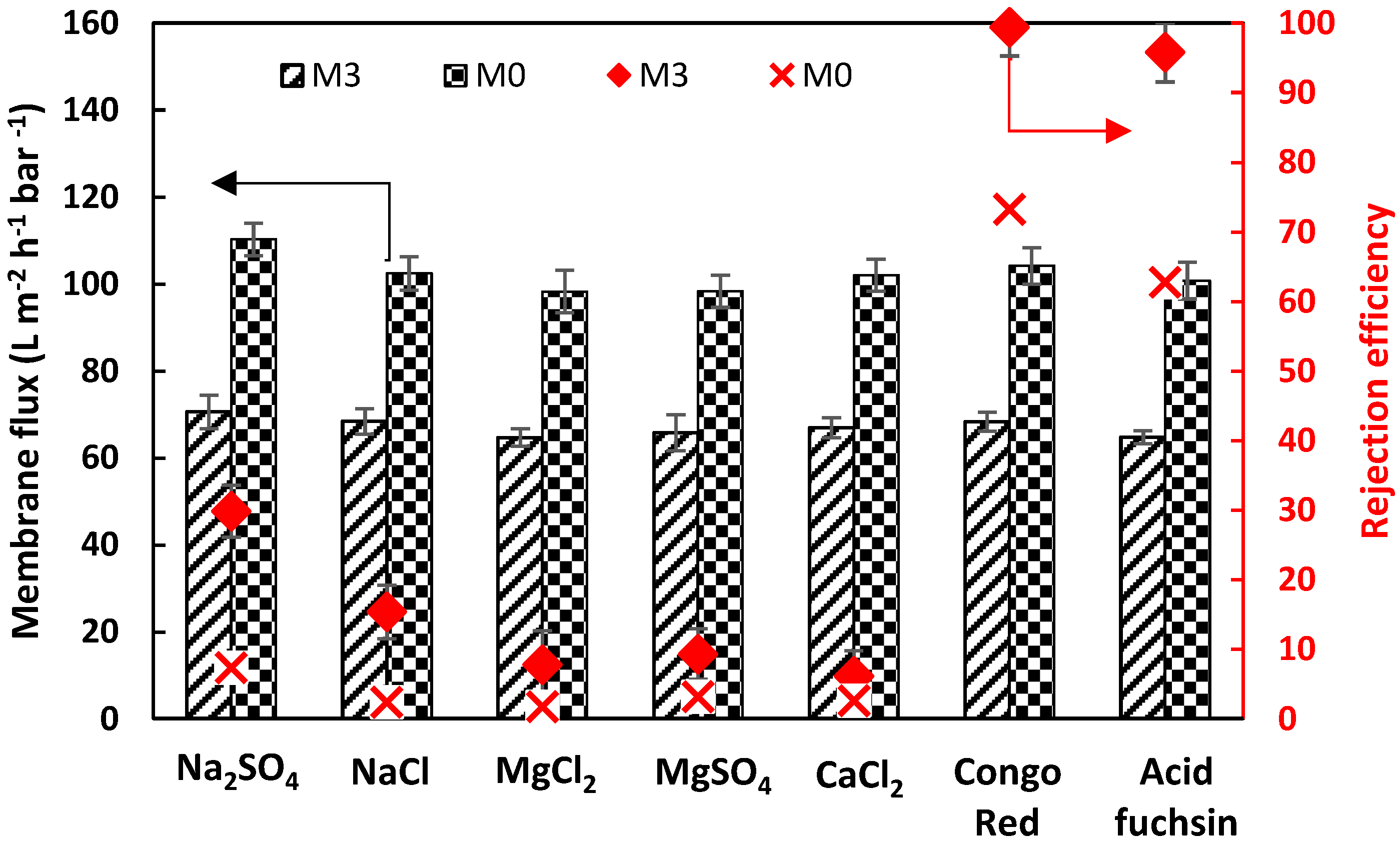
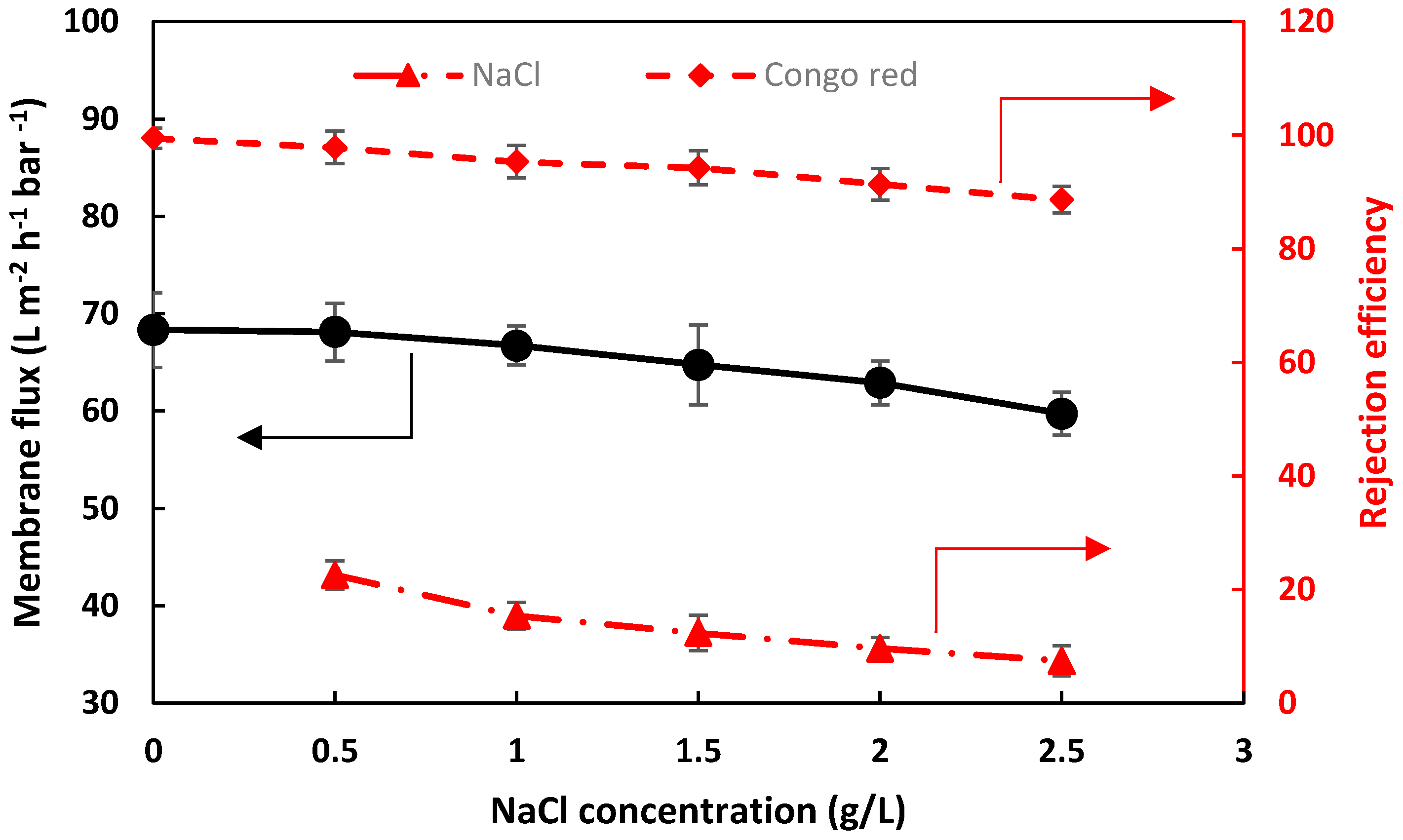
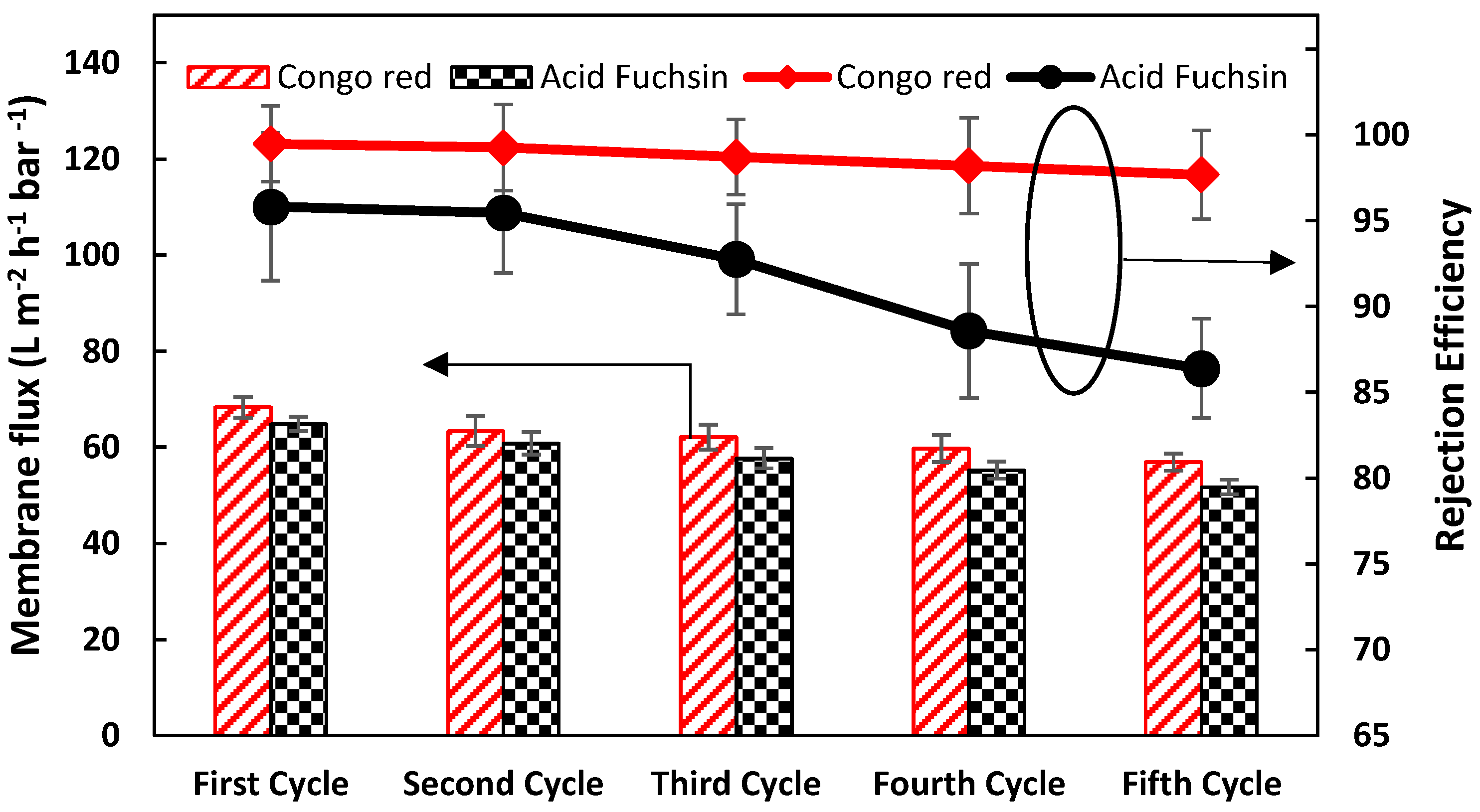
3.3.3. Salt/Dye Mixture Separation
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zango, Z.U.; Binzowaimil, A.M.; Aldaghri, O.A.; Eisa, M.H.; Garba, A.; Ahmed, N.M.; Lim, J.W.; Ng, H.-S.; Daud, H.; Jumbri, K.; et al. Applications of covalent organic frameworks for the elimination of dyes from wastewater: A state-of-the-arts review. Chemosphere 2023, 343, 140223. [Google Scholar] [CrossRef]
- Altaf, A.; Baig, N.; Sohail, M.; Sher, M.; Ul-Hamid, A.; Altaf, M. Covalent organic frameworks: Advances in synthesis and applications. Mater. Today Commun. 2021, 28, 102612. [Google Scholar] [CrossRef]
- Bagheri, A.R.; Aramesh, N.; Sher, F.; Bilal, M. Covalent organic frameworks as robust materials for mitigation of environmental pollutants. Chemosphere 2021, 270, 129523. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Nie, M.; Cheng, S.; Li, R.; Wang, Y.; Ali, A.S.; Han, X.; Sun, Y. Voltage-Gated Pillararene-Intercalated MXene membranes for adjusting water purification. Sep. Purif. Technol. 2025, 365, 132667. [Google Scholar] [CrossRef]
- Zhang, Q.; Ni, C.; Deng, N.; Huang, X. Tailoring Electronic and Morphology Features of Iron-Doped Ni2P Nanoflowers for Enhanced Ammonia Electrosynthesis in Solid Electrolyte Reactors. Adv. Energy Mater. 2025, 15, 2405442. [Google Scholar] [CrossRef]
- Rasheed, T. Covalent organic frameworks as promising adsorbent paradigm for environmental pollutants from aqueous matrices: Perspective and challenges. Sci. Total Environ. 2022, 833, 155279. [Google Scholar] [CrossRef]
- Abdellah, A.R.; Abdelhamid, H.N.; El-Adasy, A.-B.A.A.M.; Atalla, A.A.; Aly, K.I. One-pot synthesis of hierarchical porous covalent organic frameworks and two-dimensional nanomaterials for selective removal of anionic dyes. J. Environ. Chem. Eng. 2020, 8, 104054. [Google Scholar] [CrossRef]
- Dong, L.; Li, M.; Zhang, S.; Si, X.; Bai, Y.; Zhang, C. NH2-Fe3O4-regulated graphene oxide membranes with well-defined laminar nanochannels for desalination of dye solutions. Desalination 2020, 476, 114227. [Google Scholar] [CrossRef]
- Solayman, H.M.; Hossen, M.A.; Abd Aziz, A.; Yahya, N.Y.; Leong, K.H.; Sim, L.C.; Monir, M.U.; Zoh, K.-D. Performance evaluation of dye wastewater treatment technologies: A review. J. Environ. Chem. Eng. 2023, 11, 109610. [Google Scholar] [CrossRef]
- Sukhatskiy, Y.; Shepida, M.; Sozanskyi, M.; Znak, Z.; Gogate, P.R. Periodate-based advanced oxidation processes for wastewater treatment: A review. Sep. Purif. Technol. 2023, 304, 122305. [Google Scholar] [CrossRef]
- Guo, S.; Wan, Y.; Chen, X.; Luo, J. Loose nanofiltration membrane custom-tailored for resource recovery. Chem. Eng. J. 2021, 409, 127376. [Google Scholar] [CrossRef]
- Lin, J.; Ye, W.; Zeng, H.; Yang, H.; Shen, J.; Darvishmanesh, S.; Luis, P.; Sotto, A.; Van der Bruggen, B. Fractionation of direct dyes and salts in aqueous solution using loose nanofiltration membranes. J. Membr. Sci. 2015, 477, 183–193. [Google Scholar] [CrossRef]
- Du, C.; Li, P.; Zhuang, Z.; Fang, Z.; He, S.; Feng, L.; Chen, W. Highly porous nanostructures: Rational fabrication and promising application in energy electrocatalysis. Coord. Chem. Rev. 2022, 466, 214604. [Google Scholar] [CrossRef]
- Albdoor, A.K.; Ma, Z.; Cooper, P.; Al-Ghazzawi, F.; Liu, J.; Richardson, C.; Wagner, P. Air-to-air enthalpy exchangers: Membrane modification using metal-organic frameworks, characterisation and performance assessment. J. Clean. Prod. 2021, 293, 126157. [Google Scholar] [CrossRef]
- Al-Ghazzawi, F.; Conte, L.; Potts, M.W.; Richardson, C.; Wagner, P. Reactive Extrusion Printing of Zeolitic Imidazolate Framework Films. ACS Appl. Mater. Interfaces 2024, 16, 44270–44277. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, X.; Yang, B.; Xiao, K.; Duan, H.; Zhao, H. The chemical stability of metal-organic frameworks in water treatments: Fundamentals, effect of water matrix and judging methods. Chem. Eng. J. 2022, 450, 138215. [Google Scholar] [CrossRef]
- Kumar, R.; Zhang, C.; Itta, A.K.; Koros, W.J. Highly permeable carbon molecular sieve membranes for efficient CO2/N2 separation at ambient and subambient temperatures. J. Membr. Sci. 2019, 583, 9–15. [Google Scholar] [CrossRef]
- Zuluaga, S.; Fuentes-Fernandez, E.M.; Tan, K.; Xu, F.; Li, J.; Chabal, Y.J.; Thonhauser, T. Understanding and controlling water stability of MOF-74. J. Mater. Chem. A 2016, 4, 5176–5183. [Google Scholar] [CrossRef]
- Zhivulin, V.E.; Trofimov, E.A.; Zaitseva, O.V.; Sherstyuk, D.P.; Cherkasova, N.A.; Taskaev, S.V.; Vinnik, D.A.; Alekhina, Y.A.; Perov, N.S.; Naidu, K.C. Preparation, phase stability, and magnetization behavior of high entropy hexaferrites. iScience 2023, 26, 107077. [Google Scholar] [CrossRef]
- Trukhanov, S.; Trukhanov, A.; Panina, L.; Kostishyn, V.; Turchenko, V.; Trukhanova, E.; Trukhanov, A.V.; Zubar, T.; Ivanov, V.; Tishkevich, D. Temperature evolution of the structure parameters and exchange interactions in BaFe12−xInxO19. J. Magn. Magn. Mater. 2018, 466, 393–405. [Google Scholar] [CrossRef]
- Trukhanov, S.; Trukhanov, A.; Kostishyn, V.; Zabeivorota, N.; Panina, L.; Trukhanov, A.V.; Turchenko, V.; Trukhanova, E.; Oleynik, V.; Yakovenko, O. High-frequency absorption properties of gallium weakly doped barium hexaferrites. Philos. Mag. 2019, 99, 585–605. [Google Scholar] [CrossRef]
- Wang, J.; Zhuang, S. Covalent organic frameworks (COFs) for environmental applications. Coord. Chem. Rev. 2019, 400, 213046. [Google Scholar] [CrossRef]
- Zhu, R.; Zhang, P.; Zhang, X.; Yang, M.; Zhao, R.; Liu, W.; Li, Z. Fabrication of synergistic sites on an oxygen-rich covalent organic framework for efficient removal of Cd (II) and Pb (II) from water. J. Hazard. Mater. 2022, 424, 127301. [Google Scholar] [CrossRef]
- Sheng, F.; Li, X.; Li, Y.; Afsar, N.U.; Zhao, Z.; Ge, L.; Xu, T. Cationic covalent organic framework membranes for efficient dye/salt separation. J. Membr. Sci. 2022, 644, 120118. [Google Scholar] [CrossRef]
- Zhang, Y.; Ye, H.; Chen, D.; Li, N.; Xu, Q.; Li, H.; He, J.; Lu, J. In situ assembly of a covalent organic framework composite membrane for dye separation. J. Membr. Sci. 2021, 628, 119216. [Google Scholar] [CrossRef]
- Miao, Q.; Wang, Y.; Chen, D.; Cao, N.; Pang, J. Development of novel ionic covalent organic frameworks composite nanofiltration membranes for dye/salt separation. J. Hazard. Mater. 2024, 465, 133049. [Google Scholar] [CrossRef]
- Dai, J.-Y.; Fang, Y.-X.; Xu, Z.-L.; Pandaya, D.; Liang, J.; Yan, H.-F.; Tang, Y.-J. Robust covalent organic frameworks membranes for ultrafast dye/salt separation in harsh environments. Desalination 2023, 568, 117025. [Google Scholar] [CrossRef]
- Alduraiei, F.; Kumar, S.; Liu, J.; Nunes, S.P.; Szekely, G. Rapid fabrication of fluorinated covalent organic polymer membranes for organic solvent nanofiltration. J. Membr. Sci. 2022, 648, 120345. [Google Scholar] [CrossRef]
- Wang, G.; Chen, Y.; Pan, C.; Chen, H.; Ding, S.; Chen, X. Rapid synthesis of self-standing covalent organic frameworks membrane via polyethylene glycol-assisted space-confined strategy. J. Membr. Sci. 2022, 652, 120494. [Google Scholar] [CrossRef]
- Fan, H.; Gu, J.; Meng, H.; Knebel, A.; Caro, J. High-flux membranes based on the covalent organic framework COF-LZU1 for selective dye separation by nanofiltration. Angew. Chem. Int. Ed. 2018, 57, 4083–4087. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, J.; Han, G.; Bai, Y.; Ge, Q.; Ma, J.; Lau, C.H.; Shao, L. Molecularly soldered covalent organic frameworks for ultrafast precision sieving. Sci. Adv. 2021, 7, eabe8706. [Google Scholar] [CrossRef]
- He, Y.; Lin, X. Fabricating compact covalent organic framework membranes with superior performance in dye separation. J. Membr. Sci. 2021, 637, 119667. [Google Scholar] [CrossRef]
- Mokhtari, N.; Dinari, M.; Fashandi, H. Developing polysulfone-based mixed matrix membrane containing hydrazone-linked covalent organic frameworks towards dye wastewater purification. Chem. Eng. J. 2022, 446, 137456. [Google Scholar] [CrossRef]
- Wang, R.; Shi, X.; Zhang, Z.; Xiao, A.; Sun, S.-P.; Cui, Z.; Wang, Y. Unidirectional diffusion synthesis of covalent organic frameworks (COFs) on polymeric substrates for dye separation. J. Membr. Sci. 2019, 586, 274–280. [Google Scholar] [CrossRef]
- Kandambeth, S.; Biswal, B.P.; Chaudhari, H.D.; Rout, K.C.; Mitra, S.; Karak, S.; Das, A.; Mukherjee, R.; Kharul, U.K.; Banerjee, R. Selective molecular sieving in self-standing porous covalent-organic-framework membranes. Adv. Mater. 2017, 29, 1603945. [Google Scholar] [CrossRef]
- Khan, N.A.; Zhang, R.; Wu, H.; Shen, J.; Yuan, J.; Fan, C.; Cao, L.; Olson, M.A.; Jiang, Z. Solid–vapor interface engineered covalent organic framework membranes for molecular separation. J. Am. Chem. Soc. 2020, 142, 13450–13458. [Google Scholar] [CrossRef]
- Wang, H.; Wang, M.; Liang, X.; Yuan, J.; Yang, H.; Wang, S.; Ren, Y.; Wu, H.; Pan, F.; Jiang, Z. Organic molecular sieve membranes for chemical separations. Chem. Soc. Rev. 2021, 50, 5468–5516. [Google Scholar] [CrossRef]
- He, G.; Zhang, R.; Jiang, Z. Engineering covalent organic framework membranes. Acc. Mater. Res. 2021, 2, 630–643. [Google Scholar] [CrossRef]
- Kong, F.-X.; Yue, L.; Yang, Z.; Sun, G.; Chen, J.-F. Cross-linked covalent organic framework-based membranes with trimesoyl chloride for enhanced desalination. ACS Appl. Mater. Interfaces 2021, 13, 21379–21389. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Rao, J.; Lin, X.; Huang, Y.; He, Y. Congo red stitched covalent organic framework membrane for dye separation. Sep. Purif. Technol. 2025, 354, 129021. [Google Scholar] [CrossRef]
- Li, X.; Qiao, J.; Chee, S.W.; Xu, H.-S.; Zhao, X.; Choi, H.S.; Yu, W.; Quek, S.Y.; Mirsaidov, U.; Loh, K.P. Rapid, Scalable Construction of Highly Crystalline Acylhydrazone Two-Dimensional Covalent Organic Frameworks via Dipole-Induced Antiparallel Stacking. J. Am. Chem. Soc. 2020, 142, 4932–4943. [Google Scholar] [CrossRef]
- Li, X.; Xu, H.-S.; Leng, K.; Chee, S.W.; Zhao, X.; Jain, N.; Xu, H.; Qiao, J.; Gao, Q.; Park, I.-H.; et al. Partitioning the interlayer space of covalent organic frameworks by embedding pseudorotaxanes in their backbones. Nat. Chem. 2020, 12, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.-Q.; Liu, Y.; Sun, Y. Macrocycle-based metal–organic and covalent organic framework membranes. Coord. Chem. Rev. 2025, 534, 216559. [Google Scholar] [CrossRef]
- Zhang, X.; Tu, B.; Cao, Z.; Fang, M.; Zhang, G.; Yang, J.; Ying, Y.; Sun, Z.; Hou, J.; Fang, Q. Anomalous mechanical and electrical interplay in a covalent organic framework monolayer membrane. J. Am. Chem. Soc. 2023, 145, 17786–17794. [Google Scholar] [CrossRef]
- Hussain, S.; Peng, X. Ultra-fast photothermal-responsive Fe-TCPP-based thin-film nanocomposite membranes for ON/OFF switchable nanofiltration. Sep. Purif. Technol. 2021, 278, 119528. [Google Scholar] [CrossRef]
- Albdoor, A.K.; Ma, Z.; Cooper, P. Moisture diffusion measurement and evaluation for porous membranes used in enthalpy exchangers. Energy Procedia 2019, 160, 499–506. [Google Scholar] [CrossRef]
- Zhao, X.; Yuan, L.; Zhang, Z.-Q.; Wang, Y.-S.; Yu, Q.; Li, J. Synthetic Methodology for the Fabrication of Porous Porphyrin Materials with Metal–Organic–Polymer Aerogels. Inorg. Chem. 2016, 55, 5287–5296. [Google Scholar] [CrossRef] [PubMed]
- Lovo de Carvalho, A.; Ferreira, B.F.; Martins, C.H.G.; Nassar, E.J.; Nakagaki, S.; Machado, G.S.; Rives, V.; Trujillano, R.; Vicente, M.A.; Gil, A.; et al. Tetracarboxyphenylporphyrin–Kaolinite Hybrid Materials as Efficient Catalysts and Antibacterial Agents. J. Phys. Chem. C 2014, 118, 24562–24574. [Google Scholar] [CrossRef]
- Liao, H.; Wang, H.; Ding, H.; Meng, X.; Xu, H.; Wang, B.; Ai, X.; Wang, C. A 2D porous porphyrin-based covalent organic framework for sulfur storage in lithium–sulfur batteries. J. Mater. Chem. A 2016, 4, 7416–7421. [Google Scholar] [CrossRef]
- Mahajan, P.G.; Dige, N.C.; Vanjare, B.D.; Phull, A.R.; Kim, S.J.; Hong, S.-K.; Lee, K.H. Synthesis, Photophysical Properties and Application of New Porphyrin Derivatives for Use in Photodynamic Therapy and Cell Imaging. J. Fluoresc. 2018, 28, 871–882. [Google Scholar] [CrossRef]
- Fang, Y.; Zhu, C.-Y.; Yang, H.-C.; Zhang, C.; Xu, Z.-K. Polyamide nanofiltration membranes by vacuum-assisted interfacial polymerization: Broad universality of Substrate, wide window of monomer concentration and high reproducibility of performance. J. Colloid Interface Sci. 2024, 655, 327–334. [Google Scholar] [CrossRef]
- Liu, S.; Kang, L.; Hu, J.; Jung, E.; Zhang, J.; Jun, S.C.; Yamauchi, Y. Unlocking the Potential of Oxygen-Deficient Copper-Doped Co3O4 Nanocrystals Confined in Carbon as an Advanced Electrode for Flexible Solid-State Supercapacitors. ACS Energy Lett. 2021, 6, 3011–3019. [Google Scholar] [CrossRef]
- Kandambeth, S.; Shinde, D.B.; Panda, M.K.; Lukose, B.; Heine, T.; Banerjee, R. Enhancement of Chemical Stability and Crystallinity in Porphyrin-Containing Covalent Organic Frameworks by Intramolecular Hydrogen Bonds. Angew. Chem. Int. Ed. 2013, 52, 13052–13056. [Google Scholar] [CrossRef]
- Xu, C.; Yang, Z.; Zhang, S.; Gao, X.; Bao, L.; Meng, Z. Porphyrin-based covalent organic frameworks with synergistic active centers for enhanced peroxidase-like activity as a colorimetric biosensing platform. Microchem. J. 2025, 210, 112918. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, J.; Wang, D.; Xu, C.; Sheng, S.; Cheng, J.; Bao, C.; Li, Y.; Tian, H. Fe-TCPP@CS nanoparticles as photodynamic and photothermal agents for efficient antimicrobial therapy. Biomater. Sci. 2020, 8, 6526–6532. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Huang, X.; Deng, N. Short-Chain Carboxylic Acids Influencing Mineralization Mechanisms of Ferrihydrite Transformation to Hematite and Goethite. Environ. Sci. Technol. 2025, 59, 12910–12919. [Google Scholar] [CrossRef]
- Huang, G.; Su, T.-M.; Zeng, K.; Guo, Y.-A.; Zhao, S.-K.; Wei, S.-J. Mesoporous chitosan-immobilized iron tetrakis(4-carboxyphenyl)porphyrin as a model of cytochrome P-450 enzyme for oxidation of ethylbenzene. Appl. Organomet. Chem. 2018, 32, e4140. [Google Scholar] [CrossRef]
- Shan, Z.; Sun, Y.; Wu, M.; Zhou, Y.; Wang, J.; Chen, S.; Wang, R.; Zhang, G. Metal-porphyrin-based three-dimensional covalent organic frameworks for electrocatalytic nitrogen reduction. Appl. Catal. B Environ. 2024, 342, 123418. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, R.; Ma, Y.; Chen, R.; Sun, X.; Sun, X. A novel one-dimensional porphyrin-based covalent organic framework. Molecules 2019, 24, 3361. [Google Scholar] [CrossRef]
- Li, X.; Chen, Y.; Hu, X.; Zhang, Y.; Hu, L. Desalination of dye solution utilizing PVA/PVDF hollow fiber composite membrane modified with TiO2 nanoparticles. J. Membr. Sci. 2014, 471, 118–129. [Google Scholar] [CrossRef]
- Zhu, J.; Tian, M.; Hou, J.; Wang, J.; Lin, J.; Zhang, Y.; Liu, J.; Van der Bruggen, B. Surface zwitterionic functionalized graphene oxide for a novel loose nanofiltration membrane. J. Mater. Chem. A 2016, 4, 1980–1990. [Google Scholar] [CrossRef]
- Zhang, X.; Li, H.; Wang, J.; Peng, D.; Liu, J.; Zhang, Y. In-situ grown covalent organic framework nanosheets on graphene for membrane-based dye/salt separation. J. Membr. Sci. 2019, 581, 321–330. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, L.; Xiao, X.; Wang, X.; Cao, J.; Ye, H.; Li, H.; Zhang, Y. Two-dimensional Cu-FeTCPP nanozyme incorporated loose nanofiltration membrane with self-cleaning property for dye/salt separation. Sep. Purif. Technol. 2025, 359, 130551. [Google Scholar] [CrossRef]
- Wei, X.; Kong, X.; Sun, C.; Chen, J. Characterization and application of a thin-film composite nanofiltration hollow fiber membrane for dye desalination and concentration. Chem. Eng. J. 2013, 223, 172–182. [Google Scholar] [CrossRef]
- Wang, R.; Shi, X.; Xiao, A.; Zhou, W.; Wang, Y. Interfacial polymerization of covalent organic frameworks (COFs) on polymeric substrates for molecular separations. J. Membr. Sci. 2018, 566, 197–204. [Google Scholar] [CrossRef]
- Wang, T.; Wu, H.; Zhao, S.; Zhang, W.; Tahir, M.; Wang, Z.; Wang, J. Interfacial polymerized and pore-variable covalent organic framework composite membrane for dye separation. Chem. Eng. J. 2020, 384, 123347. [Google Scholar] [CrossRef]
- Wang, Z.; Si, Z.; Cai, D.; Shufeng Li, G.L.; Qin, P. Synthesis of stable COF-300 nanofiltration membrane via in-situ growth with ultrahigh flux for selective dye separation. J. Membr. Sci. 2020, 615, 118466. [Google Scholar] [CrossRef]
- Basel, N.; Liu, Q.; Fan, L.; Wang, Q.; Xu, N.; Wan, Y.; Dong, Q.; Huang, Z.; Guo, T. Surface charge enhanced synthesis of TpEB-based covalent organic framework (COF) membrane for dye separation with three typical charge properties. Sep. Purif. Technol. 2022, 303, 122243. [Google Scholar] [CrossRef]
- Fang, Y.-X.; Lin, Y.-F.; Xu, Z.-L.; Mo, J.-W.; Li, P.-P. A novel clover-like COFs membrane fabricated via one-step interfacial polymerization for dye/salt separation. J. Membr. Sci. 2023, 673, 121470. [Google Scholar] [CrossRef]
- Zhao, S.; Di, N.; Lei, R.; Wang, J.; Wang, Z. Triphenylamine-based COFs composite membrane fabricated through oligomer-triggered interfacial polymerization. J. Membr. Sci. 2023, 672, 121424. [Google Scholar] [CrossRef]
- Wang, L.; Fang, M.; Liu, J.; He, J.; Deng, L.; Li, J.; Lei, J. The influence of dispersed phases on polyamide/ZIF-8 nanofiltration membranes for dye removal from water. RSC Adv. 2015, 5, 50942–50954. [Google Scholar] [CrossRef]
- Liu, M.; Zhou, C.; Dong, B.; Wu, Z.; Wang, L.; Yu, S.; Gao, C. Enhancing the permselectivity of thin-film composite poly (vinyl alcohol)(PVA) nanofiltration membrane by incorporating poly (sodium-p-styrene-sulfonate)(PSSNa). J. Membr. Sci. 2014, 463, 173–182. [Google Scholar] [CrossRef]
- Zhang, C.; Wei, K.; Zhang, W.; Bai, Y.; Sun, Y.; Gu, J. Graphene oxide quantum dots incorporated into a thin film nanocomposite membrane with high flux and antifouling properties for low-pressure nanofiltration. ACS Appl. Mater. Interfaces 2017, 9, 11082–11094. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, Z.; Yang, L.; Zhao, S.; Song, P.; Khan, B. A novel loose-NF membrane based on the phosphorylation and cross-linking of polyethyleneimine layer on porous PAN UF membranes. J. Membr. Sci. 2018, 555, 56–68. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, H.; Wang, Z.; Song, P.; Ban, M.; Song, X. A loose hybrid nanofiltration membrane fabricated via chelating-assisted in-situ growth of Co/Ni LDHs for dye wastewater treatment. Chem. Eng. J. 2018, 353, 460–471. [Google Scholar] [CrossRef]
- Tang, H.; Ji, S.; Gong, L.; Guo, H.; Zhang, G. Tubular ceramic-based multilayer separation membranes using spray layer-by-layer assembly. Polym. Chem. 2013, 4, 5621–5628. [Google Scholar] [CrossRef]
- Mu, M.; Graham, N.; Yu, W.; Sun, K.; Xu, X.; Liu, T. Optimal cross-linking of MXene-based membranes for high rejection and low adsorption with long-term stability for water treatment. J. Membr. Sci. 2024, 701, 122738. [Google Scholar] [CrossRef]



| COF | Substrate | Dye | Rejection Efficiency | Membrane Flux (L m−2h−1bar−1) | Reference |
|---|---|---|---|---|---|
| LZU1 | Al2O3 | Methyl blue | 99.2 | 76 | [30] |
| TpPa | PSF | Congo red | 99.5 | 50 | [65] |
| TpBd | PSF | Congo red | 99.5 | 33.6 | [66] |
| TpTGCL | PAN | Methylene blue | 99 | 24 | [24] |
| COF-300 | Al2O3 | Chrome black T | 97.4 | 79 | [67] |
| TBDH | Nylon | Congo red | 99 | 439.4 | [32] |
| Hz-TFPTZ | PSF/PVP | Basic blue 41 | 92 | 94.7 | [33] |
| TpEB | PAN | Congo red | 99 | 32.3 | [68] |
| MPD-TEB | Nylon | Congo red | 98.6 | 94.4 | [69] |
| Tp-TAPA | PAN | Congo red | 99 | 68.1 | [70] |
| Tp-TTA | mPSF | Chrome black T | 98.1 | 36.5 | [25] |
| ZIF-8 | PA | Congo red | 99.98 | 2.26 | [71] |
| PVA | PSSNa-PSf | Congo red | 99.68 | 7.7 | [72] |
| TA | GOQDs-PAN | Congo red | 99.8 | 11.7 | [73] |
| Fe(III)-phos-(PEI) | HPAN | Acid fuchsin | 98.99 | 5.7 | [74] |
| LDHs | PEI-M | Acid fuchsin | 97.5 | 19.86 | [75] |
| PEI | PAA | Acid fuchsin | 99 | 2 | [76] |
| GO/NH2-Fe3O4-8 | PVDF | Congo red | 94 | 78 | [8] |
| GO/COF-1 | PAN | Congo red | 99.62 | 31.09 | [62] |
| GO | PAN | Congo red | 98.54 | 6.73 | [62] |
| DA/PEG-MXene | PVDF | Methylene Blue | 94.11 | 70.235 | [77] |
| COF-TCPP (Fe) | PVDF | Congo red | 99.5 | 68.4 | Present study |
| COF-TCPP (Fe) | PVDF | Acid fuchsin | 95.8 | 64.8 | Present study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Ghazzawi, F.; Al-Mossawi, M.M.; Allayeith, H.K. Metal–Porphyrin-Based Covalent Organic Framework Composite Membrane for Salts and Dyes Separation. Compounds 2025, 5, 34. https://doi.org/10.3390/compounds5030034
Al-Ghazzawi F, Al-Mossawi MM, Allayeith HK. Metal–Porphyrin-Based Covalent Organic Framework Composite Membrane for Salts and Dyes Separation. Compounds. 2025; 5(3):34. https://doi.org/10.3390/compounds5030034
Chicago/Turabian StyleAl-Ghazzawi, Fatimah, Mohammed Mahdi Al-Mossawi, and Hadeel K. Allayeith. 2025. "Metal–Porphyrin-Based Covalent Organic Framework Composite Membrane for Salts and Dyes Separation" Compounds 5, no. 3: 34. https://doi.org/10.3390/compounds5030034
APA StyleAl-Ghazzawi, F., Al-Mossawi, M. M., & Allayeith, H. K. (2025). Metal–Porphyrin-Based Covalent Organic Framework Composite Membrane for Salts and Dyes Separation. Compounds, 5(3), 34. https://doi.org/10.3390/compounds5030034







