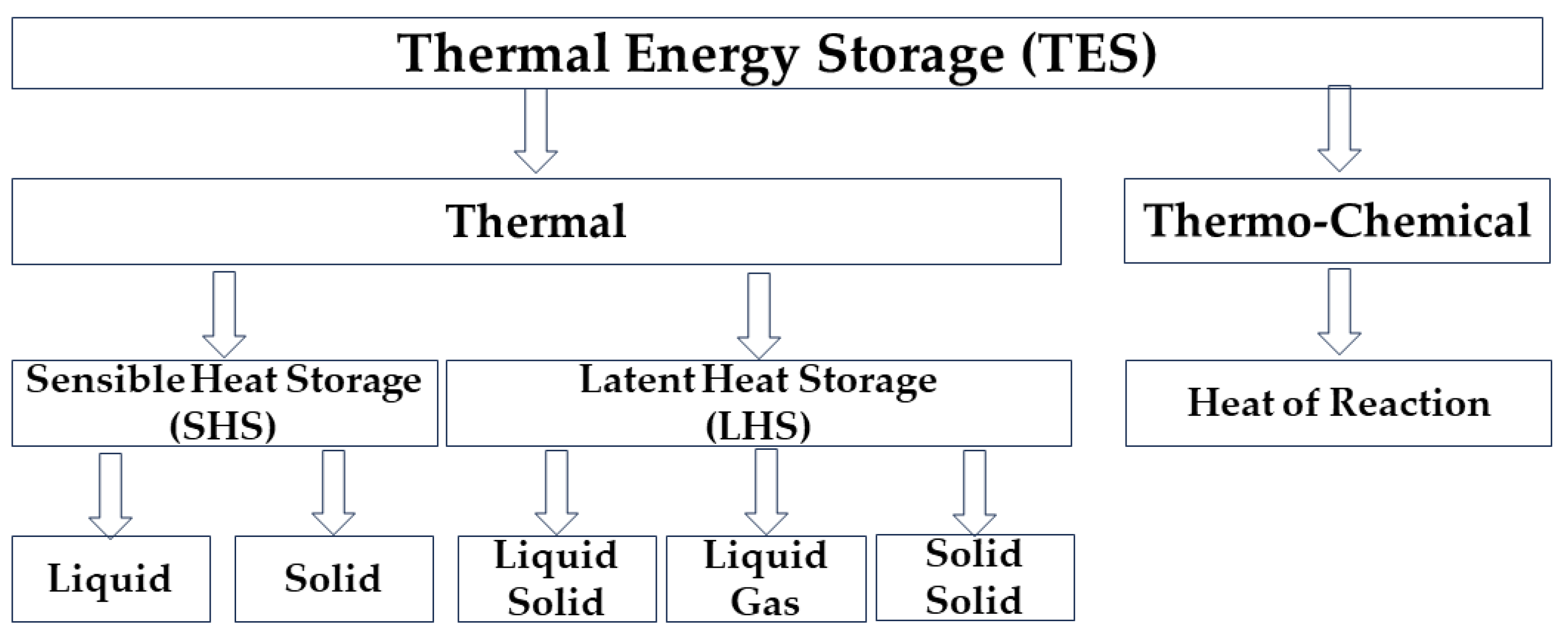
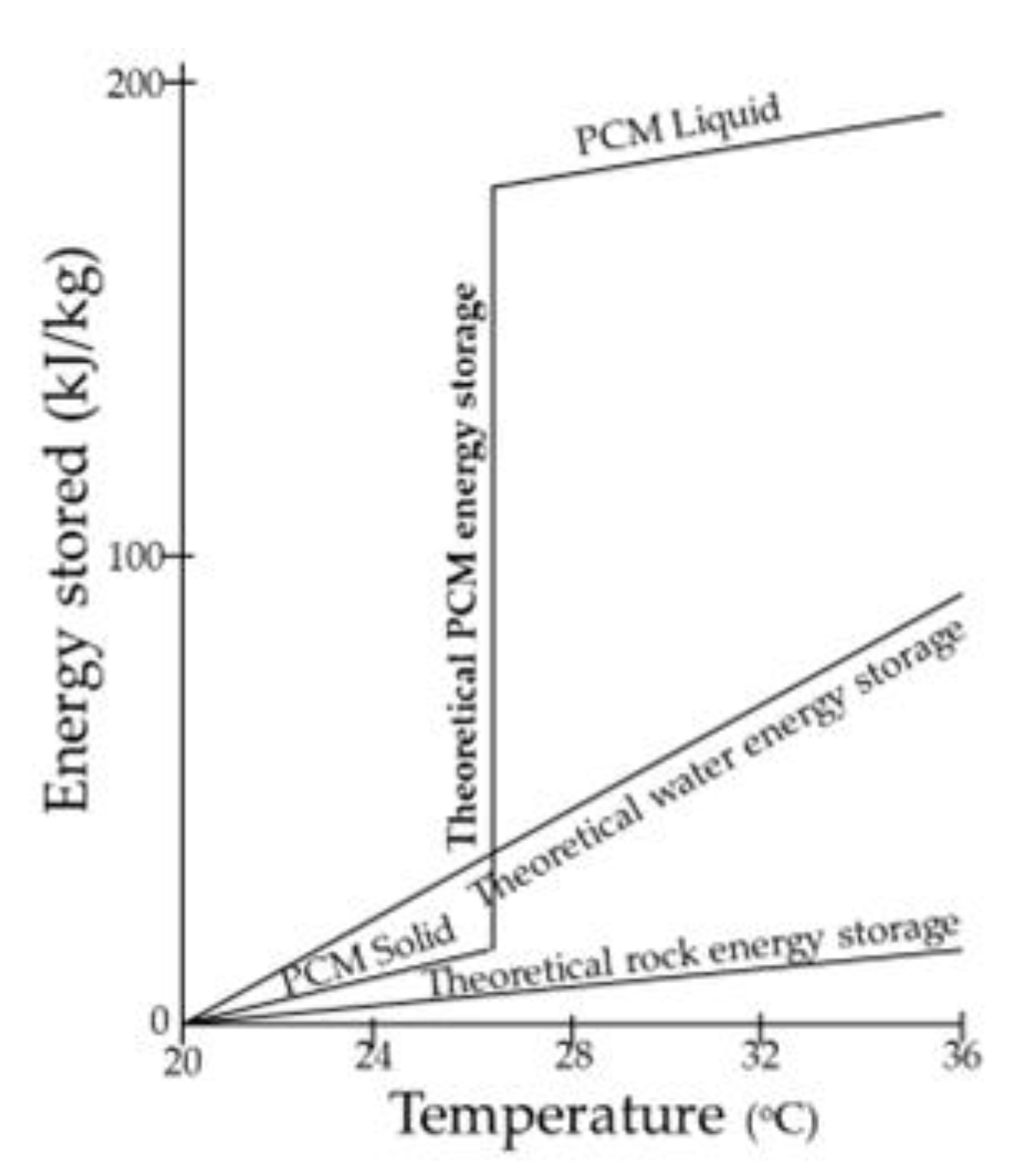
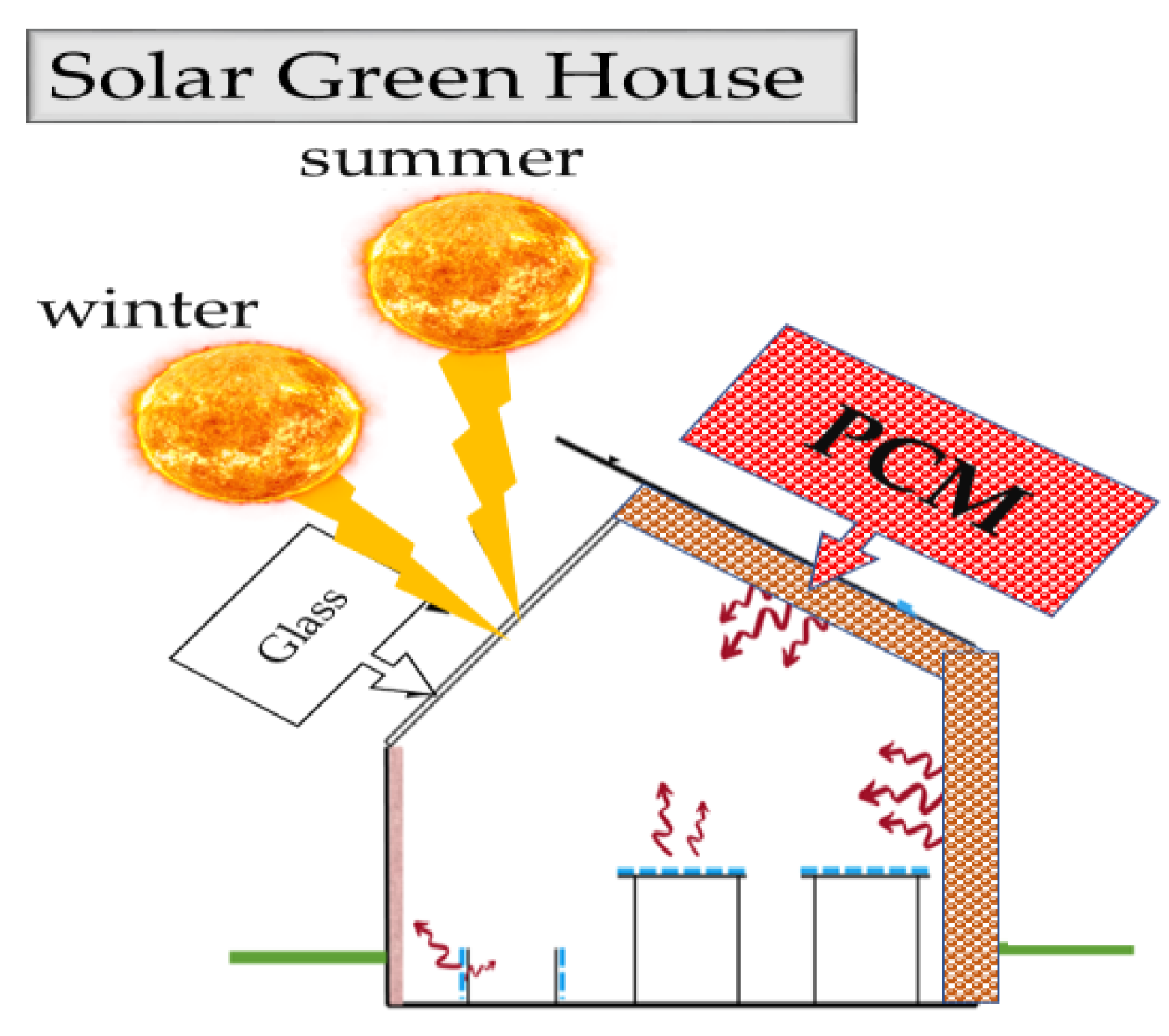
1. Introduction
The rehabilitation of oil and natural gas in countries that do not have such energy resources and the reduction in pollution due to combustion has led to the research and development of new technologies for the absorption and storage of solar energy [
1]. We know that energy is not created or destroyed but preserved [
2]. This principle is the basis of the technology of storing and exploiting solar energy from a period of low demand to a period of high demand. Renewable energy sources and storage technologies offer solutions to replace some fuels and make them life-saving solutions for the future. Energy sources, such as wind, solar, etc., will only apply if ways of storing the produced energy are found simultaneously. The generated energy can be stored with phase change materials (PCMs) [
3,
4,
5]. The application of PCMs is essential where electricity is expensive or in areas where the electricity supply could be more practical.
PCMs change phases at a specific temperature achieved through internal energy transfer as a heat transfer that we call “latent heat”. Liquid-dissolved PCMs create another category of materials we call phase change slurries (PCSs), enclosed in heat transfer circuits [
6]. PCS can contain up to 40% PCMs with a melting temperature of 60 °C and have a lower physical coefficient of thermal permeability compared to water. One application is water heaters, which in recent times are now on the market under operating conditions. Recent review publications present such applications [
7,
8]. This publication examines phase change memory (PCM) materials encapsulated into containers to improve the performance of devices. Encapsulating PCMs in nanocontainers is a one-way solution for many applications. PCMs on a paraffin basis dissolve on objects that are applied, e.g., electronic devices, thereby corroding these devices. Their isolation in a container is indicated to isolate paraffin from the environment and use its function as an energy storage/cooling material. Another application concerns the slurries where the systems need to be protected from the solution environment to act for a long time as a kind of heat capacity enhancement of this solution. These shells guarantee the long-term operation of the PCM materials. Another severe problem solved by encapsulating the PCMs into containers is the reduction in temperature gradients during heat transfer inside or outside the material during heat transfer in and out of the material. In the last few years, many works have concerned the development of silica shells [
1], styrene-methyl methacrylate copolymer [
2,
3], urea-formaldehyde (UF) [
4,
5], polystyrene (PS) [
6], melamine formaldehyde (MF) [
7], polymethyl methacrylate (PMMA) [
8,
9,
10,
11], polycarbonate (PC) [
3,
12], and polyurethane (PU) [
13,
14,
15]. We refer to the corresponding literature to produce these capsules. This tremendous amount of work is the subject of this review, and suggestions will be made on how these technologies can be applied to practical applications. The reader will see more applications in this review where the development of PCM@shells is necessary for many applications.
In addition, double-wall nanocontainers incorporating solar energy absorption coatings are reported to enhance spinel coatings' absorption efficiency with light microtraps [
9]. In the end, there is a literature review on producing electricity using nanocontainer nanotechnology from the sun.
Figure 1 shows the applications that employ this publication.
3. Solar Water Heating Systems
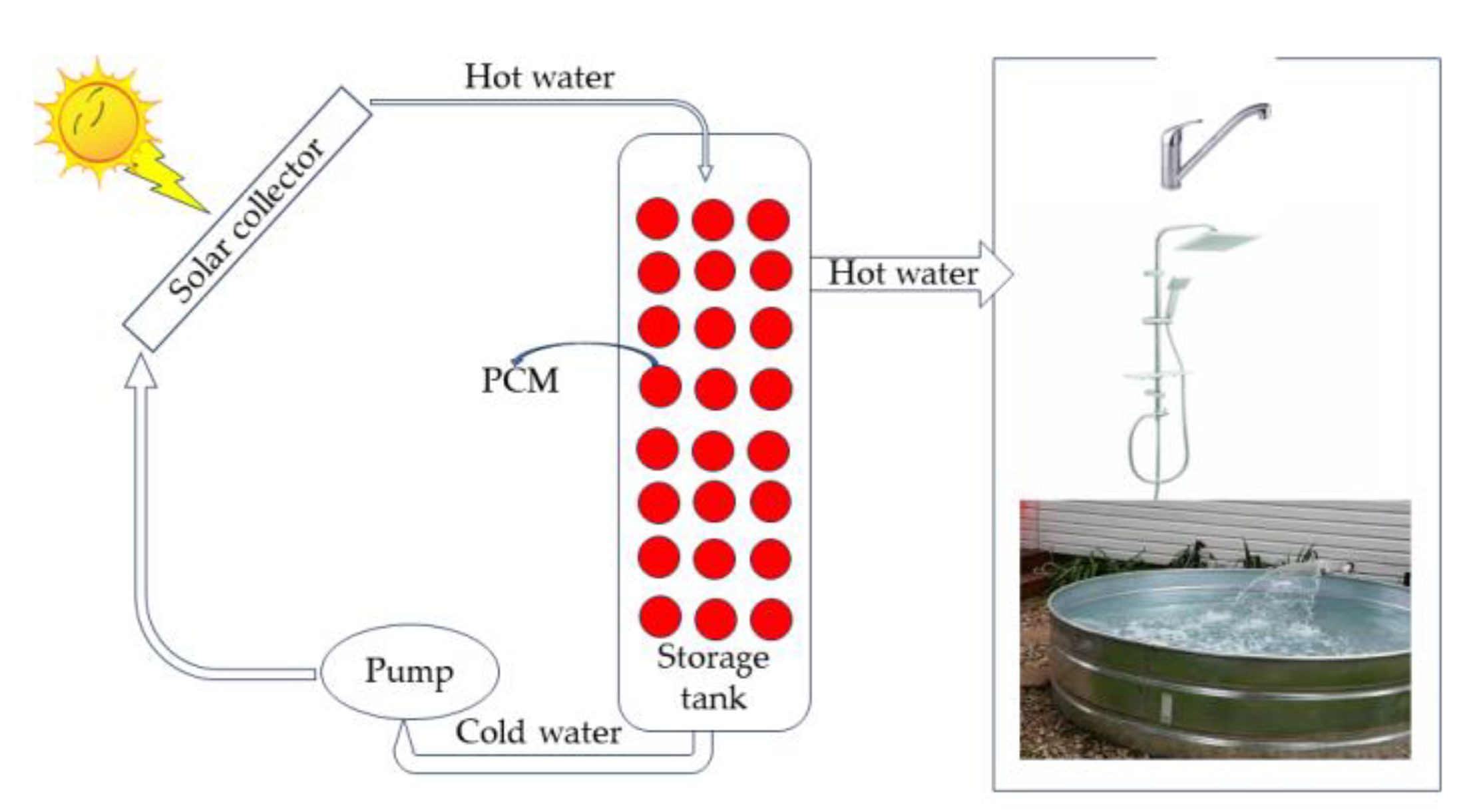
A solar water heater is a case where PCMs can be applied since they are relatively cheap materials and do not raise costs but only increase the efficiency of the water heater and insulation compared to polyurethane. During the hours of sunshine, the water heats until the heat is transferred to the PCM when the temperature reaches the material’s melting point. The PCM gathers energy in the form of latent heat and melts. During long hours of sunshine, hot water is withdrawn and replaced by cold water, which gains power from PCM.
The PCM’s energy in its phases changes from liquid to solid. Therefore, constructing a water heater unit with PCM will be integrated into its thermal insulation system. The hot water can be transferred from the water heaters to a heat storage system located in the basement of a house that will return to where it is needed in sunless hours.
An isolated sizeable cylindrical storage tank was used to experimentally investigate the material storage performance of paraffin wax in small spherical aluminum containers. The aluminum containers are commercially available. It was found that a PCM-loaded storage device has an advantage of 13 to 10 4 °C in hot water storage compared to a layout without PCMs. During a 24 h trial, the stored water temperature remained 30 °C higher than the ambient temperature. One can use such devices for one’s everyday routine [
71].
In an exciting work, the absorbent plate of a solar water heater was painted with paraffin wax PCM and compared with a corresponding plate without paraffin paint. The solar water heater with a storage absorption plate painted with paraffin performs better than the solar collector without paraffin wax. The experimental performance was 70.98% for the paraffin layout, and for the numerical simulation, it was 67.73%. These experiments showed that the absorption with paraffin increased significantly in the water heater system due to the collector’s increased ability to absorb the sun’s rays [
72].
Emphasis has been placed in the literature on improving the efficiency of commercial solar water systems by using PCM throughout the heating season [
73]. PCM particles from 1 to 3 mm have been used in thermal energy storage, and analysis showed that the application was beneficial inside a water tank with a significant drop-in heat transfer rate [
74,
75]. Such a system is believed to be more flexible and responsive for energy storage at a reduced volume [
74]. It was found that the temperature difference required to transfer the heat coefficient by natural convection is reduced by quite a large percentage [
73].
Solar water heating in a solar collector system can significantly improve efficiency if the water tank is structured with phase change materials. One experiment used 2.5 kg of paraffin wax cut into a cylindrical aluminum casing with a total % PCM volume of 3%. The outer diameters of the cylindrical geometry were at a height of 0.3 m, and the inner diameter was 0.127 m. The temperature distribution along the size of the storage tank the energy and energy efficiency of the charge and the collector's efficiency were finally calculated and compared with CFD software. The system’s energy efficiency improved by up to 36.48% due to improved heat transfer between PCM and water [
76].
The lack of stability of PCM storage materials inside the systems mentioned above creates a problem and limits the development of PCM applications. This instability has aroused scientists’ interest in incorporating them into containers to isolate them from their environment to determine their alterations [
77]. The freezing and melting of PCM, 50–80% entrapped in polyester spherical containers studied in the literature, found their lifespan [
78,
79]. BASF developed microencapsulated PCMs for commercial use in solar water heaters [
80].
Figure 8 shows a typical heat transfer unit consisting of a large, medium container, into which spheres of a few centimeters containing paraffin are incorporated and organized in a class. Among these spheres is a slurry consisting of nanospheres, which may be a kind of paraffin, encapsulated silica spheres, which flow undisturbed into the large spheres and exit the end of this arrangement. The thermal properties of such an installation depend on the type of pools and fluid we use, the size of the facility, i.e., cross-sectional area and length, the slurry flow rate, and the temperature of the slurry inlet [
81]. The device stores thermal energy during the day with sunshine and returns to the heating and cooling system of buildings on demand, where a second circuit is activated when the ambient temperature drops to the point that requires the commissioning of this device [
7,
71]. Depending on the thermal energy storage system, one can use these facilities for heating swimming pools, buildings, and other household appliances, depending on the system’s architectural design.
4. Solar Cookers
Solar cookers are one of the significant applications of PCMs that can be used for cooking when there is no sun (
Figure 9). The kitchen will use PCM with latent heat and storage materials in a solar cooker-type frame to cook food late at night. Solar cookers are limited to lots of sunshine and equipped with heat storage systems above 100 °C. PCMs can be magnesium nitrate hexahydrate (Mg(NO
3)
2-6H
2O) as a PCM for heat storage [
85].
PCM is charged faster when the charging temperature increases. Decent as it was found, the heat discharge time increases with the degree of overheating of the PCM. The kitchen’s performance was four times that of other steam sun appliances for indoor cooking. It was observed that when the PCM was magnesium chloride hexahydrate with a melting temperature of 110.7 °C, the device achieved temperatures higher than 100 °C. A solar cooker was developed to collect energy, store it during the day, and use it for cooking food at night using acetamide latent heat storage materials. This cooker was compared to a typical solar cooker, proving that cooking food is possible in the evenings [
86]. Solar concentration-type cookers are believed to have a significant rate of return. Theoretical studies were carried out on this where heat losses from the empty food containers were considered. High temperatures will be achieved when these containers are constructed adequately with the appropriate devices, including PCM. These structures are sensitive to wind speed, where in the end, it will take longer average cooking time with increasing wind speed [
87]. High temperatures of up to 200 °C were obtained in a solar system using vacuum collectors with built-in long heat pipes for frying and baking in a short time using water as a working liquid. The performance of such a system is excellent, and instabilities in the thermal conductivity of lines under different load conditions are not observed [
88]. Liquid suspensions (slurries) containing particles 1 to 100 nm in size are called nanofluids and have received considerable attention for amplifying these liquids in enhanced heat transfer applications. A slurry containing indium nanoparticles and indium polyalphaolefin (PAO)-coated nanoparticles exhibiting a melting temperature of 157 °C exhibits increased thermal conductivity and specific heat of conventional fluids [
89]. A single-phase liquid with a particular heat transfer capacity is improved by twice as much if phase change nanoparticles are added, which can discard heat energy during solid–liquid phase changes. These experiments were carried out using two types of slurry, naked indium, and silicon-coated indium, for high-temperature applications of 150 to 180 °C. This system can operate at a high temperature of 150 to 180 °C. Experiments showed that the heat transfer coefficient reached 47,000 W/m
2 K at a flow rate of 3.5 mL/s (velocity 0.28 m/s). Thermal cycle tests were performed 5000 times, proving the stability of the slurry [
89,
90].
Solar cookers are feasible in countries of plentiful sunshine under three conditions: the solar concentrate, which must be a pipe or parabolic arrangement, and the appropriate backing stove with good insulation, as shown in
Figure 9. Appropriate nanofluids and PCMs must also be investigated for reducing cooking time and optimizing the temperature the device can achieve.
6. Buildings
PCMs have been developed for thermal storage in buildings. PCM is integrated into walls, plasterboard, shutters, insulation, and underfloor heating systems as part of the building for heating and cooling applications [
46,
95,
97,
98]. The purpose is to shift the peak load and harness solar energy. The implementation of PCMs in the building can have two different objectives. They first used natural heat as solar energy for heating or cooling. Second, they used artificial heat or complex sources. In any case, storage of heat or cold is necessary to match availability and demand over time. PCMs will be used in wall construction, building elements other than the walls, storage plants, plasterboard, bricks, and paints (
Figure 11).
In recent years, buildings have developed efficient materials and technologies to meet these cooling and heating needs. Building envelopes lose more energy and are integrated with heat storage materials to regulate the internal temperature and improve the energy efficiency of buildings.
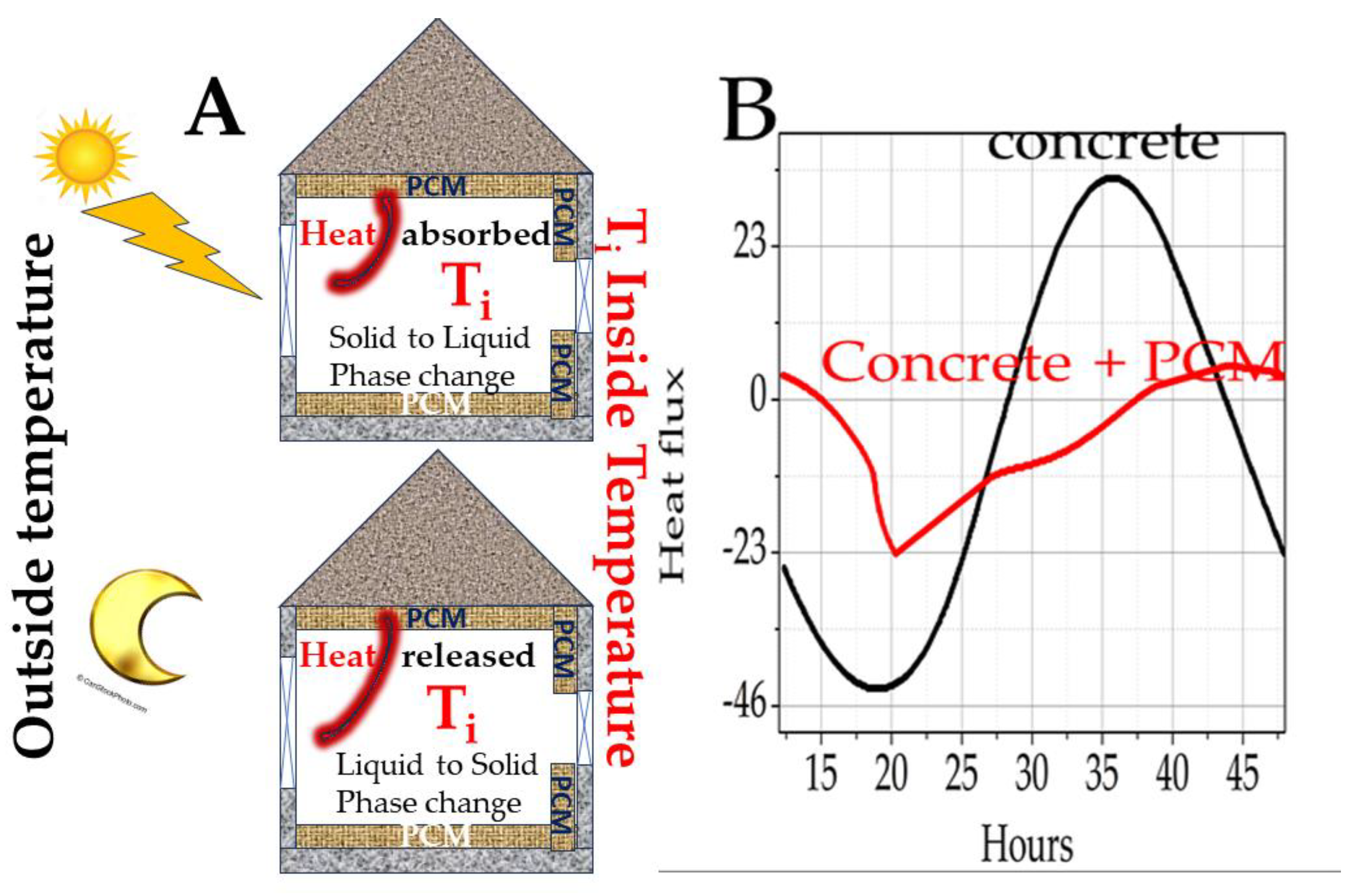
Figure 12 shows the external change in ambient temperature and the reduction in this oscillation within the building if surrounded by PCM walls [
13].
The choice of PCM incorporated into buildings should suit indoor and human comfort temperatures. Thermal comfort for people indoors is 23.5 °C to 25.5 °C in summer and 21 °C to 23 °C in winter. Based on these preferences, PCMs for buildings must have operating temperatures between 18 °C and 30 °C. According to these criteria, the melting point of PCM is selected. Latent heat fusion determines the amount of material we will use in buildings. The density of PCMs also plays a vital role in the selection so as not to occupy a large volume in buildings. The reader from an industry interested in using PCMs for buildings can refer to the literature to find the appropriate material for the structure considering the region’s climate [
8,
95].
Table 1,
Table 2 and
Table 3 present a few building materials.
The PCMs incorporated into containers do not adversely affect the building material’s operation. The dimensions of the small encapsulated PCMs are so small that no effects on the properties of materials are observed. The enclave is a simple and economical technique of integrating materials into compatible building materials, and there are currently products from companies called Micronal1PCM. The melting point of this material is 26 to 8C, and the phase change enthalpy is 110 kJ/kg. PCMs were incorporated into concrete and reached a compressive strength of 25 MPa and a tensile strength of over 6 MPa (after 28 days) [
99]. Thermal analyses of small, encapsulated paraffin were performed, and it was found that their geometric size was kept constant for up to 1000 cycles. A high storage and release capacity of energy amounting to 20–90 J/g, depending on the composition ratios, was also found [
100].
Two rooms were full-size in one experiment, one with panels with PCM and the others without PCMs. The panels encapsulating capric acid were installed on the outer surface and encapsulated a 1-dodecanol (CADE) with a melting point of 26.5 °C. The experiment showed first the stability of the average temperature at 26 °C and the reduction in temperature fluctuations inside the room to 2 ◦C. In contrast, the external fluctuations were up to 32 °C [
47].
7. Textiles
An essential application in the future can be in fabrics that contain phase change materials and immediately mitigate external temperature changes and maintain a constant body temperature [
101]. Encapsulating PCM in capsules and incorporating these into textile products provides tremendous energy storage benefits and protects the body from temperature changes. These can be used to irrigate bed linen, and in various clothing technologies, constant temperatures are necessary [
101]. The PCM capsules are planned to be incorporated into acrylic fiber yarns that, with some processing, will be subjected to standard textile machinery. When the outside temperature rises, the microcapsules absorb heat and store it as liquefaction of phase change materials. The opposite happens when the temperature drops; worse phase change materials solidify again, releasing temperature. These possibilities can be realized with capsules containing PCMs incorporated into garments. The microcapsules must be incorporated into the fibers of the fabrics directly to be woven into the final product. Gelatin/sodium alginate/clay capsules encapsulated n-eicosane and examined whether they can improve fabrics’ thermal comfort and flame-retardant properties. Electron microscopy (SEM) confirmed that the produced n-eicosane/capsule system was spherical, averaging 1.5 μm. Thermal analysis showed that they could store 97–114 J/g and release 82–105 J/g latent thermal energy, quantities sufficient for their application in the textile industry. The experiments showed that this n-eicosane/capsule system significantly slows flame in fabrics [
102]. Commercial microcapsules of type CENTER FINISH 164/01 M.T. produced by COLOR CENTER (Tarrasa, Spain) were incorporated into textile fibers. The shell of the capsules was melamine formalin, and the capsules contained a mint aroma. Their size was about 5–10 μm. This study is unique because it aimed to mitigate odor in the body, which was proven after this technology. The debate is that goals can be made with a dual mission, such as stabilizing the temperature through a PCM in the container and avoiding odors with substances incorporated into the capsules [
103]. A technique was developed based on containers manufactured on the in-situ polymerization process to be used in various applications in the textile industry. The thermal performance of the resulting fabrics was tested for given cold weather conditions and a person’s physical activity using multilayer fabrics. They can operate at −2 °C for about an hour. The microcapsules were widely distributed, as demonstrated by SEM studies [
104].
8. Nano- and Micro-Encapsulation of PCM
Caprylic acid is a PCM that can find use in sectors such as construction, textiles, and agriculture because it has a melting point of about 16 °C and a latent heat storage capacity of 158 J/g. It is in liquid form at room temperature, and under such conditions, it is not easy to use. Encapsulating caprylic acid in capsules is the one-way solution for its use as a PCM [
49]. This is why the caprylic acid (octanoic acid) was incorporated into various wall materials, such as urea-formaldehyde resin, melamine-formaldehyde resin, and urea-formaldehyde resin. The PCM compound was hardened with formaldehyde. The size of the capsules was measured by scanning electron microscopy (SEM) and found to be between 200 nm and 1.5 μm. This research found that the shell prepared from the urea-formaldehyde resin was the best for the caprylic acid goal. The thermal analysis determined that PCM’s melting phase change enthalpy and freezing were 93.9 J/g and 106.1 J/g, respectively [
49].
The sol-gel method encapsulated paraffin in TiO
2 as thermal energy storage materials. Paraffin in the TiO
2 was encapsulated in the TiO
2 according to FT-IR, XRD, and SEM measurements. The encapsulated paraffin melts at 58.8 °C and has a latent heat of 161.1 kJ/kg and solidifies at 56.5 °C with a latent heat of 144.6 kJ/kg, as the microencapsulation ratio is 85.5%. These results show that micro-encapsulated paraffin and TiO
2 may have major thermal energy storage potential. The literature describes how one can manufacture TiO
2 and encapsulate paraffin [
105].
Stearic acid (S.A.) was imprisoned in silicon dioxide (SiO2) containers to create PCMs using the sol-gel method. Silica stearic acid solidifies at 52.6 °C with a latent heat of 162.0 kJ/kg and melts at 53.5 °C with a latent heat of 171.0 kJ/kg when the S.A. encapsulation ratio is 90.7%. The analysis showed that silica shells improve their thermal stability with stearic acid (S.A.), possibly using a thermal energy storage medium in buildings. Chemical preparations were described in the literature [
106].
One paper used emulsion polymerization to characterize the nano-micro-encapsulated capric, lauric, and myristic acids with polystyrene (P.S.). The thermal analysis showed that the feathers melt and freeze at 22–48 °C and 19–49 °C as they store and release latent heat in the 87–98 J/g range and 84–96 J/g range. They found a minor alteration of properties after 5000 thermal cycles. The results of these ratings suggest that these materials can be used for thermal regulation, solar heat pumps, and many space heating applications in buildings [
107].
Magnetic microcapsules were prepared using an n-eicosane core and Fe
3O
4/SiO
2 shell, a dual-functional phase change material. After a study, the magnetic microcapsules exhibited magnetic qualities and extremely low magnetic retention and compulsion. The dual property makes them useful for innovative applications in fiber fabrics and many other military uses that require double armor properties [
108].
Nanoencapsulated phase change materials and their high mechanical stability are used in thermal energy storage and heat transfer systems. But they have two main drawbacks; the first is poor thermal conductivity, and the second is supercooling, leaving the latent heat almost intact. To solve these problems, silicon oxide shells were prepared in a graphene microemulsion. The measurements showed an increase in thermal conductivity of 132.9%. In pure water, thermal conductivity increased by 9.6%, and the photo-thermal conversion efficiency improved from 31% to 70%. The new technology allows for unthinkable applications in intelligent fabrics, thermal energy storage, and heat transfer fluids [
108].
Microcapsules were synthesized using the microemulsion polymerization method and studied to find the best composition conditions that remained stable at high temperatures. These are n-octadecane in poly (styrene-co-divinylbenzene-co-acrylamide) shells encapsulating n-octadecane as PCM. The materials’ melting and freezing onset temperatures were slightly lower than the phase transition temperatures of n-octadecane and the enthalpy values. With this preparation technique, the capsules are stable up to 148 °C. The high-temperature stability makes them useful for temperatures above 100 °C [
109].
A eutectic mixture of myristic acid – palmitic acid (MA-PA) was incorporated in polymethylmethacrylate (PMMA) containers. Their size was determined to have distributions between 0.1 and 300 μm. The melting temperature was 38 °C, while the storage capacity was 100.5 J/g. TGA measurements showed that the capsule withstood up to 250 °C. The capsules have excellent thermal and chemical resistance because they withstand 5000 cooling and heating cycles. These results are promising for many practical applications [
110].
Paraffin was incorporated into SiO
2 capsules using methods known from the literature to store heat [
111]. The capsules were enriched with various amounts of paraffin up to 80%, showing a latent heat of up to ∼156 J/g. The size of the spheres ranged between 550 and 700 nm. The paraffin/SiO
2 system does not show agglomeration, making it promising for heat storage applications [
23].
The mechanical properties of the shell prepared were studied using melamine-formaldehyde resin as the shell material and found to possess a yield point of about 1.1·105 Pa, showing that the capsules exhibited plastic behavior. The capsules incorporated lauryl alcohol as phase change material with a melting point of 24 °C, and the quantity of heat included in the phase transition was 225.5 J/g. The diameter of the microcapsules varies from 5 to 10 μm, and they were spherical and smooth. The purpose of creating these capsules was to be used indoors to save energy [
112].
Shells were prepared with different fractions of fatty acids to find the maximum concentration of fatty acids in PMMA that acts as a supporting material. The fatty acids were stearic acid (S.A.), palmitic acid (P.A.), myristic acid (M.A.), and lauric acid (L.A.), which are promising materials for use as PCMs. Mixtures of fatty acid/PMMA equal to 80/20 wt.% gallery do not show leakage of the fatty acids from the shell but are also thermally stable. The stable form of these pellets is required for applications on floors and plasterboard to eliminate the thermal resistance caused by the shell [
113].
In one paper, the authors prepared binary thin and paraffin mixtures such as n-heptadecane (C17), n-octane-cane (C18), n-nonadecane (C19), n-eicosane (C20), and n-tetracosane (C24), which were encapsulated in Polymethyl methacrylate (PMMA). This resulted in PMMA/(C17–C24), PMMA/(C19–C18), PMMA/(C19–C24), PMMA/(C20–C24), and PMMA/(C20–C24) micro/nano-capsules, which were characterized to find their physicochemical properties. The melting temperature range was 20–36 °C, and the latent heat capacity ranged between 86 and 169 J/g. They underwent several freezing–melting cycles to measure their stability, and it was found that they withstood even 5000 cycles. They also had good thermal conductivity and revocable freezing–melting points [
114].
Various techniques characterized n-heptadecane in an n-heptadecane shell. Their diameters were found to be 14–0.40 μm. Their properties were stable after 5000 repeated thermal cycles. TGA measurements proved that they have excellent thermal conductivity and stability. The analysis results showed that they have good energy storage capabilities [
115].
Sodium phosphate dodecahydrate (DSP) was encapsulated in methylene diisocyanate (MDI). The study dealt in detail with the production conditions, the capsule’s morphology, and the water-soluble materials’ efficiency. The increase in the quantity of MDI promoted urethane bonds, whereas they decreased with the increase in the amount of cellulose acetate butyrate (CAB). The result showed that the best MDI-to-CAB ratio was 0.4 to 1 [
116].
Poly(diallyl dimethyl ammonium chloride) sodium salt (PDADMAC) and Poly(4-styrene sulfonic acid) sodium salt (PSS) were used to manufacture multilayer shells in emulsified octadecane droplets using either bovine serum albumin (BSA) or sodium dodecyl sulfate (SDS) as surfactant. The size of the sphere was 500 microns. A high degree of loading equal to 91.3% by mass was also measured, demonstrating thermal stability after multiple cycles and heat treatments [
117].
The encapsulation of N-octadoctan in a polyurea nanocontainer was accomplished, and analyses followed to find its properties. The best conditions are obtained due to a shell core weight ratio of 70 to 30, making these materials suitable for PCM products [
118].
Alumina shell capsules were filled in with copper spheres about 3 mm in diameter from an opening in the alumina capsule, which was finally sealed after a CuAl
2O
4 compound creation process. XRD and SEM confirmed the sealing process. The result showed that copper pellets containing 5–10% aluminum created a system that can store heat and operate above 1000 °C. In the future, such methodical production methods can be used for other PCMs in different temperatures for different devices.
Figure 13 shows how these advanced PCMs are produced and operated [
119].
Copper spheres were laid in a nickel-chromium bilayer with an electrical conversion method, and studies were carried out to find the latent heat density of the capsules, which was found to be 75% of the theoretical value of 71 J/g at the melting temperature of 1077 °C. They showed no capsule oxidation and remained stable after a long charge and discharge cycle. It turned out that these capsules can be used for storing energy at high temperatures [
120].
9. Heat Generation
Water heaters use solar selective coatings to convert solar energy into thermal energy. These coatings must meet three criteria to be helpful: showing high absorption, α, in the spectral range from 0.4 to 2.5 μm, having a low emission, ε, in the infrared range from 2.5 to 50 μm, and demonstrating high selectivity, α/ε, at 100 °C. The ideal selective coating shows reflection with R = 0 in the visible region and R = 1 in the infrared, i.e., the black body spectra. Tabor produced the first commercially valuable absorbent coating by electro-galvanization 1995 [
121]. Chrome has become the typical product in the marketplace [
122]. Today’s commercial absorbent layers are produced with vacuum deposition techniques with excellent results. Alanod launched on the market various commercial products such as mirotherm Control
®, mirotherm
®, eta plus
®, and mirosol
® with α
s and ε
T up to 97% and 4% (
b/
w = 24.25), with the PVD technique [
123]. In recent years, scientists have manufactured solar absorbent coatings with the sol-gel process to reduce the cost of the production facilities of these returns with great success [
124]. Based on CoCuMnO
x spinels with SiO
x coatings, coatings CuO@SiO
2 were prepared with values α
s = 0.85–0.91 and ε
T below 0.036 (α/ε = 17–18) [
125,
126]. In coatings with CuCoMnO
x produced on an aluminum substrate, CuO@SiO
2 micro-containers were incorporated into these coatings of 980 μm ± 30 nm. CuO micro-containers 715 μm ± 15 nm in size were first prepared. The TEM measurement demonstrates that the CuO micro-containers are internally empty [
9]. CuO micro-containers are coated with SiO
2 to produce CuO@SiO
2 double-shell micro-containers [
9]. The concentration of CuO@SiO
2 micro-containers in the spinel coatings varied from 0 to 1 wt.%. The resulting α/ε ratio was measured in these samples [
9].
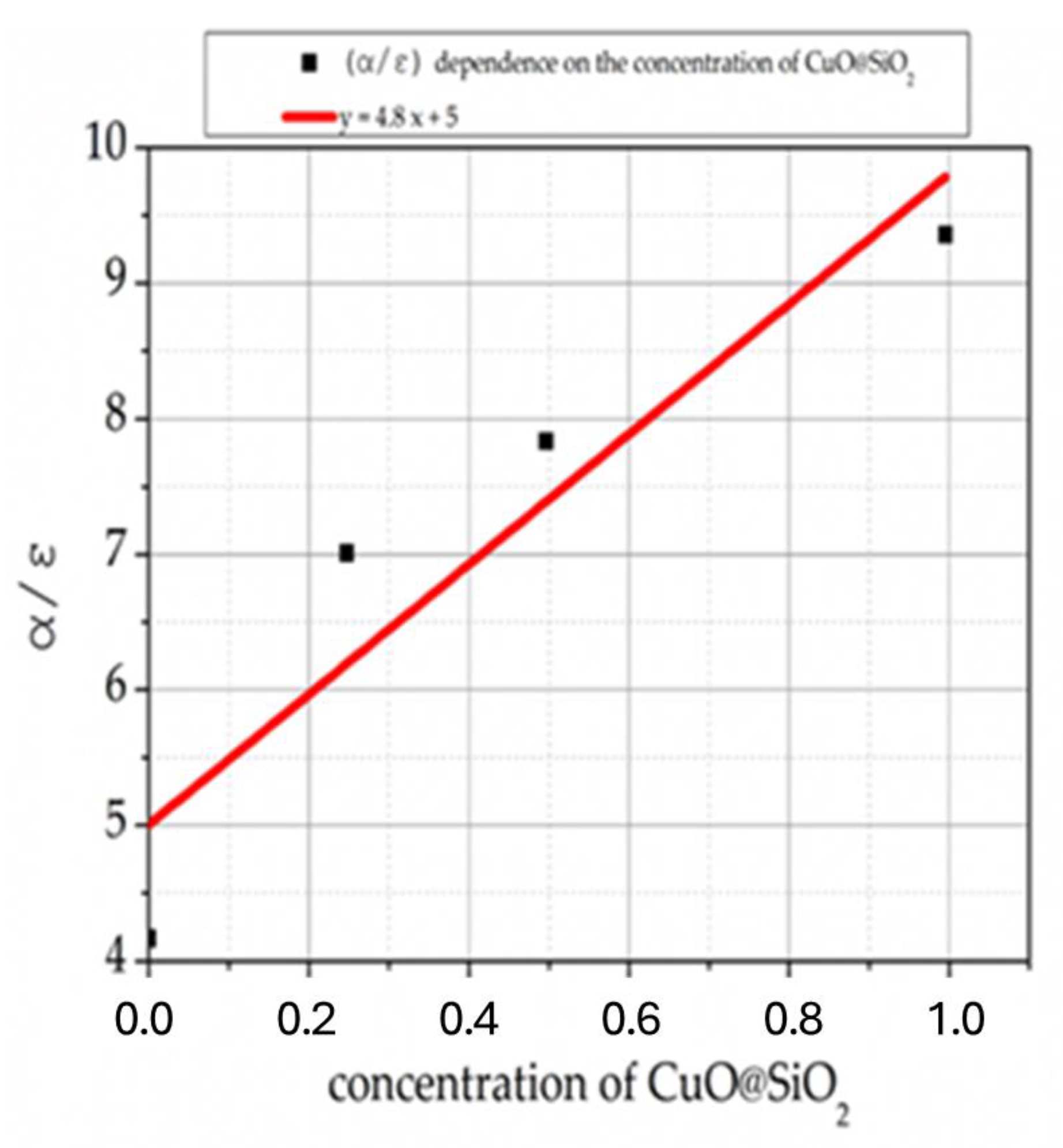
Figure 14 shows the results of α/ε ratio as a function of concentration. The α/ε ratio varies linearly with the concentration from 5 to 10 for 0 to 1 CuO@SiO
2, respectively.
The additional light absorption is generated by increasing the α/ε ratio by the CuO@SiO
2 micro-containers. The CuO@SiO
2 micro-containers act in the coatings as “light microtraps” [
9].
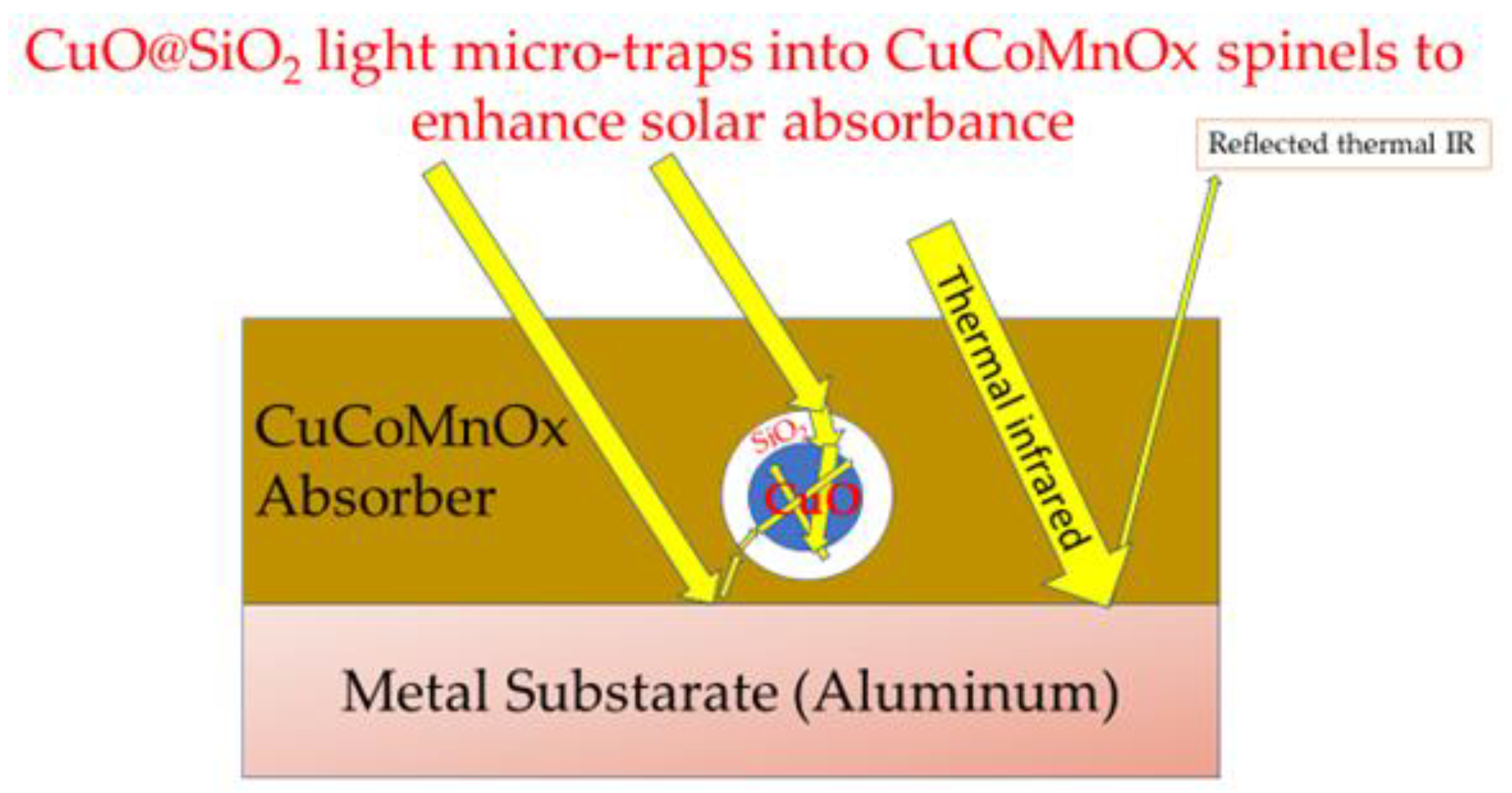
Figure 15 describes the mechanism of light trapping in the CuO@SiO
2 graphically and comprehensively described in the literature [
9].
Alanod manufactures absorbent coatings that convert sunlight into heat, which all companies use today. Production is accomplished with extensive facilities, which are highly reliable but expensive to realize. The sol-gel technology proposed here has the advantage that these absorption coatings can be manufactured by a polymer chemist with low investment cost, demonstrating high repeatability, provided, of course, that the appropriate coating developed by this method is competitive (α and ε) with the current ones produced by the Alanod company. Once this has been achieved, the light traps added to the outstanding coatings will further enhance their performance compared to these coatings, and they are expected to surpass those of Alanod coatings. This is likely to happen soon, and the potential of this technology will exceed all expectations.
10. Electricity Generation
In a past study, it was shown that light could be trapped in ZnO micro-containers. These ZnO-type hollow light traps are beautiful as photovoltaic sun solar cells due to their high surface area for absorbing incident light, high electron mobility, and low production costs. In one study, the ZnO spheres exhibited a 400–500 nm diameter, with a power conversion efficiency of 4.33% and a short-circuit current density of 9.56 mAcm
−2 [
127]. To improve the coefficient of efficiency of ZnO microspheres, multi-shell spheres with well-defined structures were produced with a defined number of inner shells and controlled distances between them through the production process. This research showed that hollow ZnO microspheres with different shell structures exhibit various energy conversion efficiency factors. The quadruple shell microstructure is one in which sunlight reflects internally multiple times, losing its energy internally, raising its performance to 5.6% [
128]. The corresponding spectrum of U.V./vis diffuse reflectance behaves accordingly. The multi-shelled ZnO hollow microspheres (M.S. ZnO HMS) were decorated with TiO
2 nano-tree (N.T.) [
129]. The TiO
2 decoration significantly enhances light scattering and increases the specific area of ZnO HMS. The I–V tests show a significant enhancement of short-circuit current density (Jsc) by controlling M.S. Combining ZnO HMS shell numbers and M.S. ZnO HMS with TiO
2 NT reaches a power conversion efficiency (PCE) of 7.40% [
129]. In addition, combining T.S. ZnO HMS with TiO
2 NT increases light-collecting efficiency and extends electron lifetime. Another approach was made using CdS and CdSe quantum dots to decorate the surface of the ZnO core/shell hollow microspheres to increase the light scattering of the ZnO hollow structure [
130]. The new system improved power conversion efficiency by 76.22% and 21.74%, higher than in ZnO N.P.s and ZnO HMS [
130].
Figure 16 describes the multiple reflections of sunlight on the inner walls of containers, which absorb more sunlight; in this way, the sun’s energy is converted into electricity [
129,
130].
The ZnO and CuO@SiO2 micro-containers entrap the sunlight to generate electricity and thermal energy. Therefore, these microstructures will be helpful soon for producing power from sunlight, which we call from now on “micro-traps of light”.
n-eicosane@SiO
2/CdS can be used not only for solar energy storage but also in the solar photocatalytic discharge of industrial wastewater, solar photochemical cleaning of organic water pollutants, etc. The capsules showed high solar photocatalytic activity in organic dyes under natural sunlight. The microcapsules also exhibit fluorescent function due to the outer CdS cortex [
131]. With the capsules having multiple properties for many applications simultaneously, this development opens their use in many different technologies.
11. Conclusions
The text was written to approach young scientists who want to start their careers in the field of encapsulated PCMs and company engineers to understand the possible commercial potential of the encapsulated PCMs. The encapsulation of PCMs involves scientists with deep knowledge of polymer chemistry synthesizing and improving existing shells to cope with the repeated operation cycles of these systems. We also require the involvement of experienced technologists from research centers, universities, and industry to develop and improve these systems with a view to their commercial exploitation. The potential of this technology is enormous, and one can solve many problems related to the absorption and storage of solar energy. From this literature review, we are at the beginning of such an activity, which must continue with a growing dynamic to solve the energy problems that generate developed and even developing countries. More is needed for these ideas to remain at the level of research study, but the industry must immediately invest in commercial exploitation through state funding. The benefits will be enormous if you consider a building with solar collectors on its roof. In the basement, a storage device with PCM is encapsulated in containers to store the excess thermal energy produced during a sunny day and return the heat to the building at night to maintain a constant temperature. One can think of cooking stoves so that using energy-intensive appliances in houses is minimized in their service to reduce a home’s energy costs. Buildings with PCMs are of great benefit because the internal temperature will stabilize at an average temperature comfortable to humans, and there will no longer be a need for heating or cooling these buildings in countries at the level of current energy needs. The future impact of this technology is enormous, and this article aims to raise awareness in industry and propose ways to collaborate with scientists utilizing containers to protect PCMs so that all these ideas can find practical application soon. Because the scientific field has many shortcomings, one urgently needs to establish the requirements and specifications of these technologies so that an industrialist can give some guarantees for their product. The new prospects of developing light traps to absorb solar energy and turn it into heat are in their infancy and need to be improved. Here, the sunlight absorption coating incorporating light traps must possess absorbance at values above 95% to be enhanced for these coatings to be commercially valuable. Also, converting solar energy into electricity is a promising new technology with potential for application; however, it needs further intensive work to improve its performance. The coming years will be a giant field of action that young scientists must enter with many new ideas for developing new materials and seeking further applications. We also expect more support from theoretical scientists to study the behavior of PCMs inside a single container as a function of its size, wall thickness, and materials to be used, as well as to optimize the devices under construction to supplement the experimental data and reduce the amount of practical work. A theorist can incorporate PCMs in various materials such as plasterboard and bricks and determine the benefit of incorporating PCMs in these structural components. This way, they will shorten the development time for new innovative materials. Such cooperation between theorists and experimentalists will dramatically reduce the development time of new products and technologies.