The Clinical Impact of Precisely Defining Mantle Cell Lymphoma: Contributions of Elaine Jaffe
Abstract
:1. Introduction
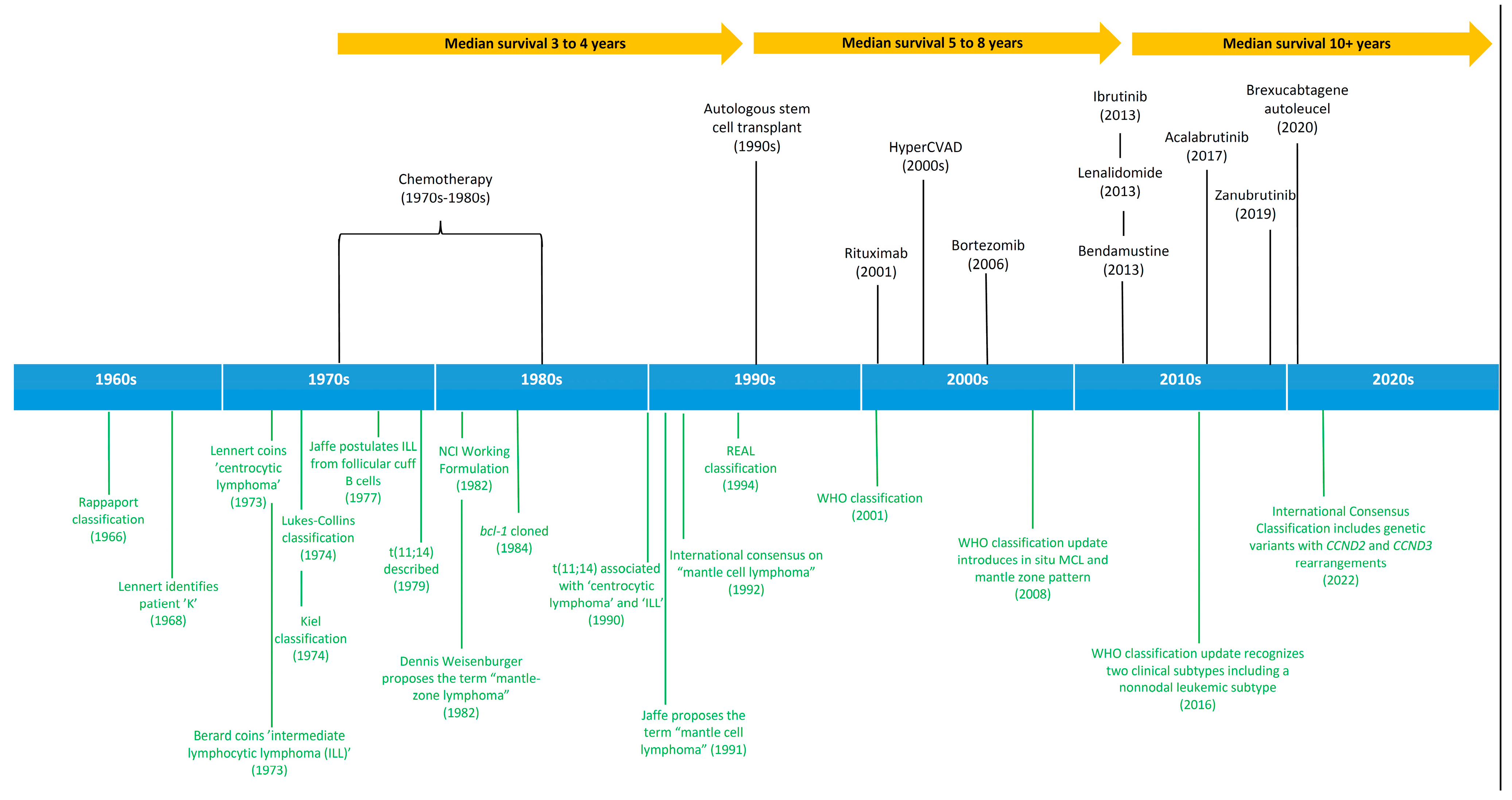
2. History and Evolution of Lymphoid Malignancies Classification
3. Mantle Cell Lymphoma as a Distinct Entity
“The cells of nodular lymphomas are neoplastic counterparts of follicular B lymphocytes whereas well-differentiated lymphocytic lymphoma (WDL) cells are more closely related to medullary-cord B cells. Lymphoma of intermediate differentiation type may derive from B cells of the lymphoid cuff at the margins of follicles and thus exhibit features at the interface between nodular lymphomas and WDL”. Jaffe et al., Cancer Treat Rep. 1977;61(6):953–962.
4. Therapeutic Advances in Mantle Cell Lymphoma
5. Trainees and Mentees
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jaffe, E.S.; Shevach, E.M.; Frank, M.M.; Berard, C.W.; Green, I. Nodular lymphoma—Evidence for origin from follicular B lymphocytes. N. Engl. J. Med. 1974, 290, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, E.S.; Braylan, R.C.; Nanba, K.; Frank, M.M.; Berard, C.W. Functional markers: A new perspective on malignant lymphomas. Cancer Treat. Rep. 1977, 61, 953–962. [Google Scholar] [PubMed]
- Berard, C.W.; Jaffe, E.S.; Braylan, R.C.; Mann, R.B.; Nanba, K. Immunologic aspects and pathology of the malignant lymphomas. Cancer 1978, 42, 911–921. [Google Scholar] [CrossRef]
- Raffeld, M.; Jaffe, E.S. bcl-1, t(11;14), and mantle cell-derived lymphomas. Blood 1991, 78, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.O.; Longo, D.L. Mantle-Cell Lymphoma. N. Engl. J. Med. 2022, 386, 2495–2506. [Google Scholar] [CrossRef] [PubMed]
- Rappaport, H. Tumors of the Hematopoietic System; Armed Forces Institute of Pathology: Washington, DC, USA, 1966. [Google Scholar]
- Hicks, E.B.; Rappaport, H.; Winter, W.J. Follicular lymphoma; A re-evaluation of its position in the scheme of malignant lymphoma, based on a survey of 253 cases. Cancer 1956, 9, 792–821. [Google Scholar]
- Lennert, K. Malignant Lymphomas Other than Hodgkin’s Disease: Histology, Cytology, Ultrastructure, Immunology; Springer: Berlin/Heidelberg, Germany, 1978. [Google Scholar]
- Lukes, R.J.; Collins, R.D. Immunologic characterization of human malignant lymphomas. Cancer 1974, 34 (Suppl. 4), 1488–1503. [Google Scholar] [CrossRef]
- National Cancer Institute sponsored study of classifications of non-Hodgkin’s lymphomas: Summary and description of a working formulation for clinical usage. The Non-Hodgkin’s Lymphoma Pathologic Classification Project. Cancer 1982, 49, 2112–2135. [CrossRef]
- Harris, N.L.; Jaffe, E.S.; Stein, H.; Banks, P.M.; Chan, J.K.; Cleary, M.L.; Delsol, G.; De Wolf-Peeters, C.; Falini, B.; Gatter, K.C.; et al. A revised European-American classification of lymphoid neoplasms: A proposal from the International Lymphoma Study Group. Blood 1994, 84, 1361–1392. [Google Scholar] [CrossRef]
- A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. The Non-Hodgkin’s Lymphoma Classification Project. Blood 1997, 89, 3909–3918. [CrossRef]
- Armitage, J.O.; Weisenburger, D.D. New approach to classifying non-Hodgkin’s lymphomas: Clinical features of the major histologic subtypes. Non-Hodgkin’s Lymphoma Classification Project. J. Clin. Oncol. 1998, 16, 2780–2795. [Google Scholar] [CrossRef] [PubMed]
- Harris, N.L.; Jaffe, E.S.; Diebold, J.; Flandrin, G.; Muller-Hermelink, H.K.; Vardiman, J. Lymphoma classification—From controversy to consensus: The R.E.A.L. and WHO Classification of lymphoid neoplasms. Ann. Oncol. 2000, 11, S3–S10. [Google Scholar] [CrossRef]
- Jaffe, E.S.; Harris, N.L.; Stein, H.; Vardiman, J. Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues; IARC Press: Lyon, France, 2001; Volume 3. [Google Scholar]
- Campo, E.; Swerdlow, S.H.; Harris, N.L.; Pileri, S.; Stein, H.; Jaffe, E.S. The 2008 WHO classification of lymphoid neoplasms and beyond: Evolving concepts and practical applications. Blood 2011, 117, 5019–5032. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; Jaffe, E.S. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef]
- Klapper, W.; Koch, K.; Mechler, U.; Borck, C.; Fuhry, E.; Siebert, R. Lymphoma ‘type K.’-in memory of Karl Lennert (1921–2012). Leukemia 2013, 27, 519–521. [Google Scholar] [CrossRef] [PubMed]
- Tolksdorf, G.; Stein, H.; Lennert, K. Morphological and immunological definition of a malignant lymphoma derived from germinal-centre cells with cleaved nuclei (centrocytes). Br. J. Cancer 1980, 41, 168–182. [Google Scholar] [CrossRef]
- Berard, C.W.; Dorfman, R.F. Histopathology of malignant lymphomas. Clin. Haematol. 1974, 3, 39–76. [Google Scholar] [CrossRef]
- Weisenburger, D.D.; Kim, H.; Rappaport, H. Mantle-zone lymphoma: A follicular variant of intermediate lymphocytic lymphoma. Cancer 1982, 49, 1429–1438. [Google Scholar] [CrossRef]
- Weisenburger, D.D.; Nathwani, B.N.; Diamond, L.W.; Winberg, C.D.; Rappaport, H. Malignant lymphoma, intermediate lymphocytic type: A clinicopathologic study of 42 cases. Cancer 1981, 48, 1415–1425. [Google Scholar] [CrossRef]
- Jaffe, E.S.; Bookman, M.A.; Longo, D.L. Lymphocytic lymphoma of intermediate differentiation--mantle zone lymphoma: A distinct subtype of B-cell lymphoma. Hum. Pathol. 1987, 18, 877–880. [Google Scholar] [CrossRef]
- Nanba, K.; Jaffe, E.S.; Braylan, R.C.; Soban, E.J.; Berard, C.W. Alkaline phosphatase-positive malignant lymphoma. A subtype of B-cell lymphomas. Am. J. Clin. Pathol. 1977, 68, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Van Den Berghe, H.; Parloir, C.; David, G.; Michaux, J.L.; Sokal, G. A new characteristic karyotypic anomaly in lymphoproliferative disorders. Cancer 1979, 44, 188–195. [Google Scholar] [CrossRef]
- Yunis, J.J.; Oken, M.M.; Kaplan, M.E.; Ensrud, K.M.; Howe, R.R.; Theologides, A. Distinctive chromosomal abnormalities in histologic subtypes of non-Hodgkin’s lymphoma. N. Engl. J. Med. 1982, 307, 1231–1236. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, Y.; Yunis, J.; Onorato-Showe, L.; Erikson, J.; Nowell, P.C.; Croce, C.M. Molecular cloning of the chromosomal breakpoint of B-cell lymphomas and leukemias with the t(11;14) chromosome translocation. Science 1984, 224, 1403–1406. [Google Scholar] [CrossRef]
- Weisenburger, D.D.; Sanger, W.G.; Armitage, J.O.; Purtilo, D.T. Intermediate lymphocytic lymphoma: Immunophenotypic and cytogenetic findings. Blood 1987, 69, 1617–1621. [Google Scholar] [CrossRef]
- Rimokh, R.; Berger, F.; Cornillet, P.; Wahbi, K.; Rouault, J.P.; Ffrench, M.; Bryon, P.A.; Gadoux, M.; Gentilhomme, O.; Germain, D.; et al. Break in the BCL1 locus is closely associated with intermediate lymphocytic lymphoma subtype. Genes Chromosomes Cancer 1990, 2, 223–226. [Google Scholar] [CrossRef]
- Williams, M.E.; Westermann, C.D.; Swerdlow, S.H. Genotypic characterization of centrocytic lymphoma: Frequent rearrangement of the chromosome 11 bcl-1 locus. Blood 1990, 76, 1387–1391. [Google Scholar] [CrossRef]
- Medeiros, L.J.; Van Krieken, J.H.; Jaffe, E.S.; Raffeld, M. Association of bcl-1 rearrangements with lymphocytic lymphoma of intermediate differentiation. Blood 1990, 76, 2086–2090. [Google Scholar] [CrossRef]
- Williams, M.E.; Meeker, T.C.; Swerdlow, S.H. Rearrangement of the chromosome 11 bcl-1 locus in centrocytic lymphoma: Analysis with multiple breakpoint probes. Blood 1991, 78, 493–498. [Google Scholar] [CrossRef]
- Motokura, T.; Bloom, T.; Kim, H.G.; Juppner, H.; Ruderman, J.V.; Kronenberg, H.M.; Arnold, A. A novel cyclin encoded by a bcl1-linked candidate oncogene. Nature 1991, 350, 512–515. [Google Scholar] [CrossRef]
- Bosch, F.; Jares, P.; Campo, E.; Lopez-Guillermo, A.; Piris, M.A.; Villamor, N.; Tassies, D.; Jaffe, E.S.; Monteserrat, E.; Rozman, C.; et al. PRAD-1/cyclin D1 gene overexpression in chronic lymphoproliferative disorders: A highly specific marker of mantle cell lymphoma. Blood 1994, 84, 2726–2732. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, C.L.; Wong, E.; Petty, E.M.; Bale, A.; Tsujimoto, Y.; Harris, N.L.; Arnold, A. PRAD1, a candidate BCL1 oncogene: Mapping and expression in centrocytic lymphoma. Proc. Natl. Acad. Sci. USA 1991, 88, 9638–9642. [Google Scholar] [CrossRef] [PubMed]
- Rimokh, R.; Berger, F.; Delsol, G.; Charrin, C.; Bertheas, M.F.; Ffrench, M.; Garoscio, M.; Felman, P.; Coiffier, B.; Bryon, P.A.; et al. Rearrangement and overexpression of the BCL-1/PRAD-1 gene in intermediate lymphocytic lymphomas and in t(11q13)-bearing leukemias. Blood 1993, 81, 3063–3067. [Google Scholar] [CrossRef] [PubMed]
- Banks, P.M.; Chan, J.; Cleary, M.L.; Delsol, G.; De Wolf-Peeters, C.; Gatter, K.; Grogan, T.M.; Harris, N.L.; Isaacson, P.G.; Jaffe, E.S.; et al. Mantle cell lymphoma. A proposal for unification of morphologic, immunologic, and molecular data. Am. J. Surg. Pathol. 1992, 16, 637–640. [Google Scholar] [CrossRef]
- Rosenwald, A.; Wright, G.; Wiestner, A.; Chan, W.C.; Connors, J.M.; Campo, E.; Gascoyne, R.D.; Grogan, T.M.; Muller-Hermelink, H.K.; Smeland, E.B.; et al. The proliferation gene expression signature is a quantitative integrator of oncogenic events that predicts survival in mantle cell lymphoma. Cancer Cell. 2003, 3, 185–197. [Google Scholar] [CrossRef]
- Fu, K.; Weisenburger, D.D.; Greiner, T.C.; Dave, S.; Wright, G.; Rosenwald, A.; Chiorazzi, M.; Iqbal, J.; Gesk, S.; Siebert, R.; et al. Cyclin D1-negative mantle cell lymphoma: A clinicopathologic study based on gene expression profiling. Blood 2005, 106, 4315–4321. [Google Scholar] [CrossRef]
- Carvajal-Cuenca, A.; Sua, L.F.; Silva, N.M.; Pittaluga, S.; Royo, C.; Song, J.Y.; Sargent, R.L.; Espinet, B.; Climent, F.; Jacobs, S.A.; et al. In situ mantle cell lymphoma: Clinical implications of an incidental finding with indolent clinical behavior. Haematologica 2012, 97, 270–278. [Google Scholar] [CrossRef]
- Navarro, A.; Clot, G.; Royo, C.; Jares, P.; Hadzidimitriou, A.; Agathangelidis, A.; Bikos, V.; Darzentas, N.; Papadaki, T.; Salaverria, I.; et al. Molecular subsets of mantle cell lymphoma defined by the IGHV mutational status and SOX11 expression have distinct biologic and clinical features. Cancer Res. 2012, 72, 5307–5316. [Google Scholar] [CrossRef]
- Fernandez, V.; Salamero, O.; Espinet, B.; Sole, F.; Royo, C.; Navarro, A.; Carnacho, F.; Bea, S.; Hartmann, E.; Amador, V.; et al. Genomic and gene expression profiling defines indolent forms of mantle cell lymphoma. Cancer Res. 2010, 70, 1408–1418. [Google Scholar] [CrossRef]
- Bosch, F.; Lopez-Guillermo, A.; Campo, E.; Ribera, J.M.; Conde, E.; Piris, M.A.; Vallespi, T.; Woessner, S.; Montserrat, E. Mantle cell lymphoma: Presenting features, response to therapy, and prognostic factors. Cancer 1998, 82, 567–575. [Google Scholar] [CrossRef]
- Vose, J.M.; Bierman, P.J.; Weisenburger, D.D.; Lynch, J.C.; Bociek, Y.; Chan, W.C.; Greiner, T.C.; Armitage, J.O. Autologous hematopoietic stem cell transplantation for mantle cell lymphoma. Biol. Blood Marrow Transpl. 2000, 6, 640–645. [Google Scholar] [CrossRef]
- Vandenberghe, E.; Ruiz de Elvira, C.; Loberiza, F.R.; Conde, E.; Lopez-Guillermo, A.; Gisselbrecht, C.; Guihot, F.; Vose, J.M.; van Biesen, K.; Rizzo, J.D.; et al. Outcome of autologous transplantation for mantle cell lymphoma: A study by the European Blood and Bone Marrow Transplant and Autologous Blood and Marrow Transplant Registries. Br. J. Haematol. 2003, 120, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Khouri, I.F.; Romaguera, J.; Kantarjian, H.; Palmer, J.L.; Pugh, W.C.; Korbling, M.; Hagemeister, F.; Samuels, B.; Rodrigeuz, A.; Giralt, S.; et al. Hyper-CVAD and high-dose methotrexate/cytarabine followed by stem-cell transplantation: An active regimen for aggressive mantle-cell lymphoma. J. Clin. Oncol. 1998, 16, 3803–3809. [Google Scholar] [CrossRef] [PubMed]
- Howard, O.M.; Gribben, J.G.; Neuberg, D.S.; Grossbard, M.; Poor, C.; Janicek, M.J.; Shipp, M.A. Rituximab and CHOP induction therapy for newly diagnosed mantle-cell lymphoma: Molecular complete responses are not predictive of progression-free survival. J. Clin. Oncol. 2002, 20, 1288–1294. [Google Scholar] [CrossRef] [PubMed]
- LaCasce, A.S.; Vandergrift, J.L.; Rodriguez, M.A.; Abel, G.A.; Crosby, A.L.; Czuczman, M.S.; Nademanee, A.P.; Blayney, D.W.; Gordon, L.I.; Millenson, M.; et al. Comparative outcome of initial therapy for younger patients with mantle cell lymphoma: An analysis from the NCCN NHL Database. Blood 2012, 119, 2093–2099. [Google Scholar] [CrossRef] [PubMed]
- Geisler, C.H.; Kolstad, A.; Laurell, A.; Anderson, N.S.; Pederson, L.B.; Jerkeman, M.; Eriksson, M.; Nordstrom, M.; Kimby, E.; Boesen, A.M.; et al. Long-term progression-free survival of mantle cell lymphoma after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: A nonrandomized phase 2 multicenter study by the Nordic Lymphoma Group. Blood 2008, 112, 2687–2693. [Google Scholar] [CrossRef]
- Romaguera, J.E.; Fayad, L.; Rodriguez, M.A.; Broglio, K.R.; Hagermeister, F.B.; Pro, B.; McLaughlin, P.; Younes, A.; Samaniego, F.; Goy, A.; et al. High rate of durable remissions after treatment of newly diagnosed aggressive mantle-cell lymphoma with rituximab plus hyper-CVAD alternating with rituximab plus high-dose methotrexate and cytarabine. J. Clin. Oncol. 2005, 23, 7013–7023. [Google Scholar] [CrossRef]
- Hermine, O.; Hoster, E.; Walewski, J.; Bosly, A.; Stilgenbauer, S.; Thieblemont, C.; Szymczyk, M.; Bouabdallah, R.; Kneba, M.; Hallek, M.; et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): A randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma Network. Lancet 2016, 388, 565–575. [Google Scholar]
- Le Gouill, S.; Thieblemont, C.; Oberic, L.; Moreau, A.; Bouabdallah, K.; Dartigeas, C.; Damaj, G.; Gastinne, T.; Ribrag, V.; Feugier, P.; et al. Rituximab after Autologous Stem-Cell Transplantation in Mantle-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 1250–1260. [Google Scholar] [CrossRef]
- Kluin-Nelemans, H.C.; Hoster, E.; Hermine, O.; Walewski, J.; Trney, M.; Geisler, C.H.; Stilgenbauer, S.; Thieblemont, C.; Vehling-Kaiser, U.; Doorduijn, J.K.; et al. Treatment of older patients with mantle-cell lymphoma. N. Engl. J. Med. 2012, 367, 520–531. [Google Scholar] [CrossRef]
- Romaguera, J.E.; Fayad, L.E.; Feng, L.; Hartig, K.; Weaver, P.; Rodriguez, M.A.; Hagemeister, F.B.; Pro, B.; McLaughlin, P.; Younes, A.; et al. Ten-year follow-up after intense chemoimmunotherapy with Rituximab-HyperCVAD alternating with Rituximab-high dose methotrexate/cytarabine (R-MA) and without stem cell transplantation in patients with untreated aggressive mantle cell lymphoma. Br. J. Haematol. 2010, 150, 200–208. [Google Scholar] [CrossRef]
- Griffiths, R.; Mikhael, J.; Gleeson, M.; Danese, M.; Dreyling, M. Addition of rituximab to chemotherapy alone as first-line therapy improves overall survival in elderly patients with mantle cell lymphoma. Blood 2011, 118, 4808–4816. [Google Scholar] [CrossRef]
- Eskelund, C.W.; Kolstad, A.; Jerkeman, M.; Raty, R.; Laurell, A.; Eloranta, S.; Smedby, K.E.; Husby, S.; Pedersen, L.B.; Andersen, N.S.; et al. 15-year follow-up of the Second Nordic Mantle Cell Lymphoma trial (MCL2): Prolonged remissions without survival plateau. Br. J. Haematol. 2016, 175, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Cohen, J.B.; Wang, M.; Kumar, A.; Hill, B.; Villa, D.; Switchenko, J.M.; Kahl, B.; Maddocks, K.; Grover, N.S.; et al. Treatment Outcomes and Roles of Transplantation and Maintenance Rituximab in Patients With Previously Untreated Mantle Cell Lymphoma: Results From Large Real-World Cohorts. J. Clin. Oncol 2022, JCO2102698. [Google Scholar] [CrossRef] [PubMed]
- Goy, A.; Younes, A.; McLaughlin, P.; Pro, B.; Romaguera, J.E.; Hagermeister, F.; Fayad, L.; Dang, N.H.; Samaniego, F.; Wang, M.; et al. Phase II study of proteasome inhibitor bortezomib in relapsed or refractory B-cell non-Hodgkin’s lymphoma. J. Clin. Oncol. 2005, 23, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Robak, T.; Huang, H.; Jin, J.; Zhu, J.; Liu, T.; Samoilova, O.; Pylypenko, H.; Verhoef, G.; Siritanaratkul, N.; Osmanov, E.; et al. Bortezomib-based therapy for newly diagnosed mantle-cell lymphoma. N. Engl. J. Med. 2015, 372, 944–953. [Google Scholar] [CrossRef]
- Ruan, J.; Martin, P.; Shah, B.; Schuster, S.J.; Smith, S.M.; Furman, R.R.; Christos, P.; Rodriguez, A.; Svoboda, J.; Lewis, J.; et al. Lenalidomide plus Rituximab as Initial Treatment for Mantle-Cell Lymphoma. N. Engl. J. Med. 2015, 373, 1835–1844. [Google Scholar] [CrossRef]
- Ladetto, M.; Cortelazzo, S.; Ferrero, S.; Evangelista, A.; Mian, M.; Tavarozzi, R.; Zanni, M.; Cavallo, F.; Di Rocco, A.; Stefoni, V.; et al. Lenalidomide maintenance after autologous haematopoietic stem-cell transplantation in mantle cell lymphoma: Results of a Fondazione Italiana Linfomi (FIL) multicentre, randomised, phase 3 trial. Lancet Haematol. 2021, 8, e34–e44. [Google Scholar] [CrossRef]
- Rummel, M.J.; Niederle, N.; Maschmeyer, G.; Banat, G.A.; von Grunhagen, U.; Losem, C.; Kofahl-Krause, D.; Heil, G.; Welslau, M.; Balser, C.; et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: An open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet 2013, 381, 1203–1210. [Google Scholar] [CrossRef]
- Chen, R.W.; Li, H.; Bernstein, S.H.; Kahwash, S.; Rimsza, L.M.; Forman, S.J.; Constine, L.; Shea, T.C.; Cashen, A.F.; Blum, K.A.; et al. RB but not R-HCVAD is a feasible induction regimen prior to auto-HCT in frontline MCL: Results of SWOG Study S1106. Br. J. Haematol. 2017, 176, 759–769. [Google Scholar] [CrossRef]
- Ruan, J.; Martin, P.; Christos, P.; Cerchietti, L.; Tam, W.; Shah, B.; Schuster, S.J.; Rodriguez, A.; Hyman, D.; Calvo-Vidal, M.N.; et al. Five-year follow-up of lenalidomide plus rituximab as initial treatment for mantle cell lymphoma. Blood 2018, 132, 2016–2025. [Google Scholar] [CrossRef] [PubMed]
- Gine, E.; de la Cruz, F.; Jimenez Ubieto, A.; Lopez Jimenez, J.; Martin Garcia-Sancho, A.; Terol, M.J.; Gonzalez Barca, E.; Casanova, M.; de la Fuente, A.; Marin-Niebla, A.; et al. Ibrutinib in Combination With Rituximab for Indolent Clinical Forms of Mantle Cell Lymphoma (IMCL-2015): A Multicenter, Open-Label, Single-Arm, Phase II Trial. J. Clin. Oncol. 2022, 40, 1196–1205. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Jurczak, W.; Jerkeman, M.; Trotman, J.; Zinzani, P.; Belada, D.; Boccomini, C.; Flinn, I.W.; Giri, P.; Goy, A.; et al. Ibrutinib plus Bendamustine and Rituximab in Untreated Mantle-Cell Lymphoma. N. Engl. J. Med. 2022, 386, 2482–2494. [Google Scholar] [CrossRef]
- Wang, M.; Munoz, J.; Goy, A.; Locke, F.L.; Jacobson, C.A.; Hill, B.T.; Timmerman, J.M.; Holmes, H.; Jaglowski, S.; Flinn, I.W.; et al. KTE-X19 CAR T-Cell Therapy in Relapsed or Refractory Mantle-Cell Lymphoma. N. Engl. J. Med. 2020, 382, 1331–1342. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Munoz, J.; Goy, A.; Locke, F.L.; Jacobson, C.A.; Hill, B.T.; Timmerman, J.M.; Holmes, H.; Jaglowski, S.; Flinn, I.W.; et al. Three-Year Follow-Up of KTE-X19 in Patients With Relapsed/Refractory Mantle Cell Lymphoma, Including High-Risk Subgroups, in the ZUMA-2 Study. J. Clin. Oncol. 2022, JCO2102370. [Google Scholar] [CrossRef]
- Castellino, A.; Wang, Y.; Larson, M.C.; Maurer, M.J.; Link, B.K.; Farooq, U.; Feldman, A.L.; Syrbu, S.I.; Habermann, T.M.; Paludo, J.; et al. Evolving frontline immunochemotherapy for mantle cell lymphoma and the impact on survival outcomes. Blood Adv. 2022, 6, 1350–1360. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roschewski, M.; Longo, D.L. The Clinical Impact of Precisely Defining Mantle Cell Lymphoma: Contributions of Elaine Jaffe. Hemato 2022, 3, 508-517. https://doi.org/10.3390/hemato3030035
Roschewski M, Longo DL. The Clinical Impact of Precisely Defining Mantle Cell Lymphoma: Contributions of Elaine Jaffe. Hemato. 2022; 3(3):508-517. https://doi.org/10.3390/hemato3030035
Chicago/Turabian StyleRoschewski, Mark, and Dan L. Longo. 2022. "The Clinical Impact of Precisely Defining Mantle Cell Lymphoma: Contributions of Elaine Jaffe" Hemato 3, no. 3: 508-517. https://doi.org/10.3390/hemato3030035
APA StyleRoschewski, M., & Longo, D. L. (2022). The Clinical Impact of Precisely Defining Mantle Cell Lymphoma: Contributions of Elaine Jaffe. Hemato, 3(3), 508-517. https://doi.org/10.3390/hemato3030035





