Development of Fermented Teff-Based Probiotic Beverage and Its Process Monitoring Using Two-Dimensional Fluorescence Spectroscopy †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Starter Culture
2.2. Off-Line Measurement of Cell Count and Fermentation Condition
2.3. Off-Line Measurement of Glucose and Lactic Acid
2.4. On-Line Measurement Using 2D Fluorescence Spectroscopy
3. Results and Discussion
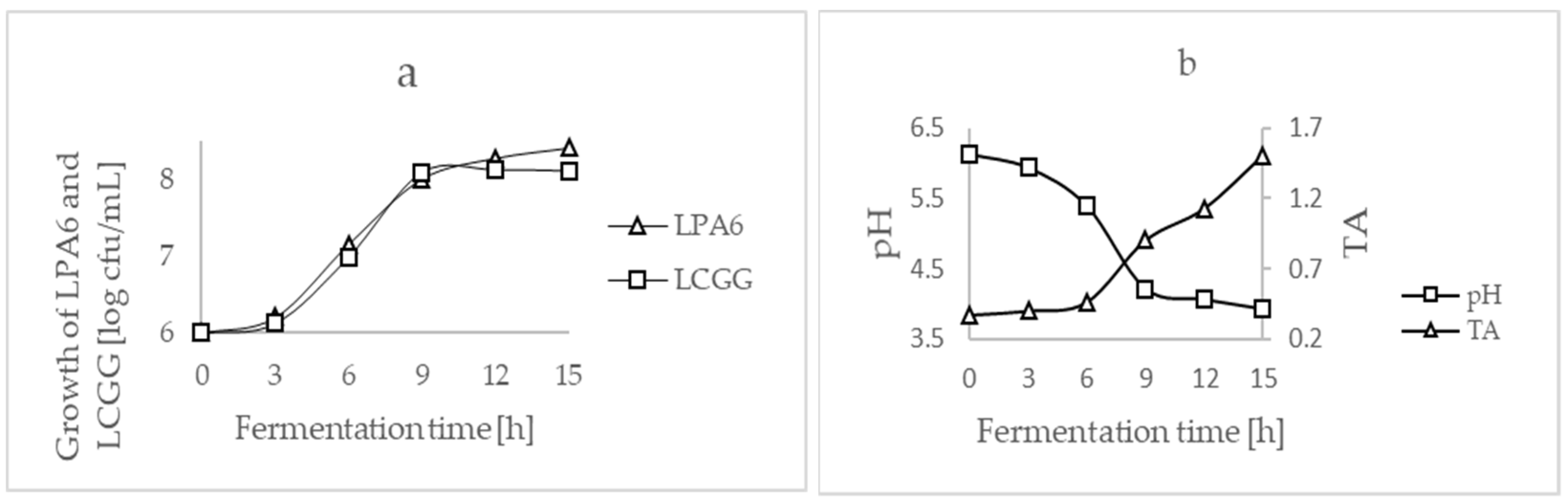
3.1. Optimization of Fermentation Process
3.2. Co-Culture Strains Fermentation
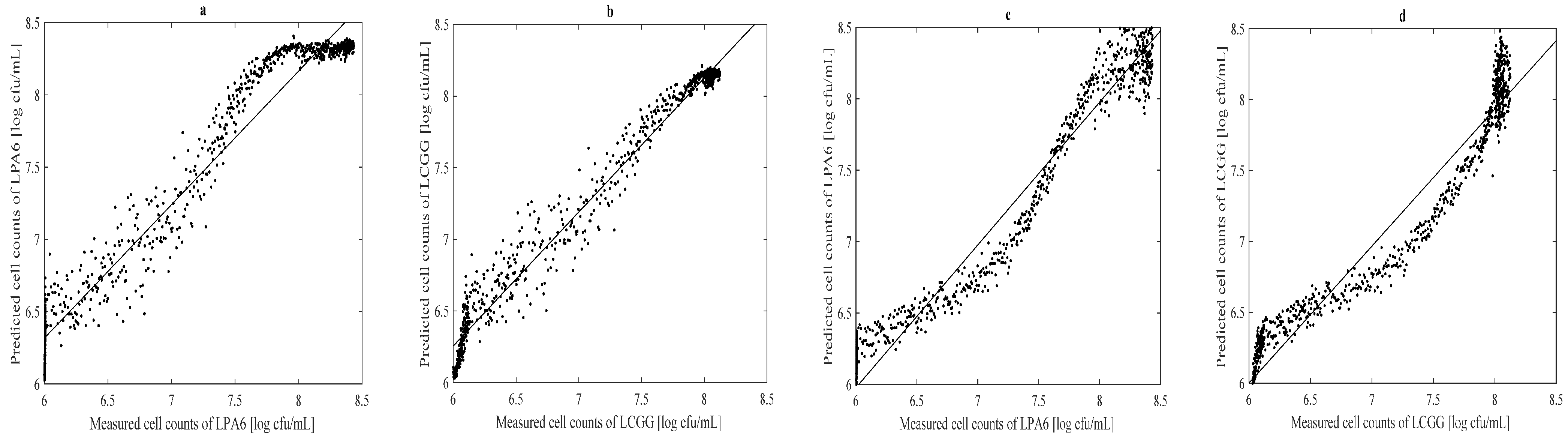
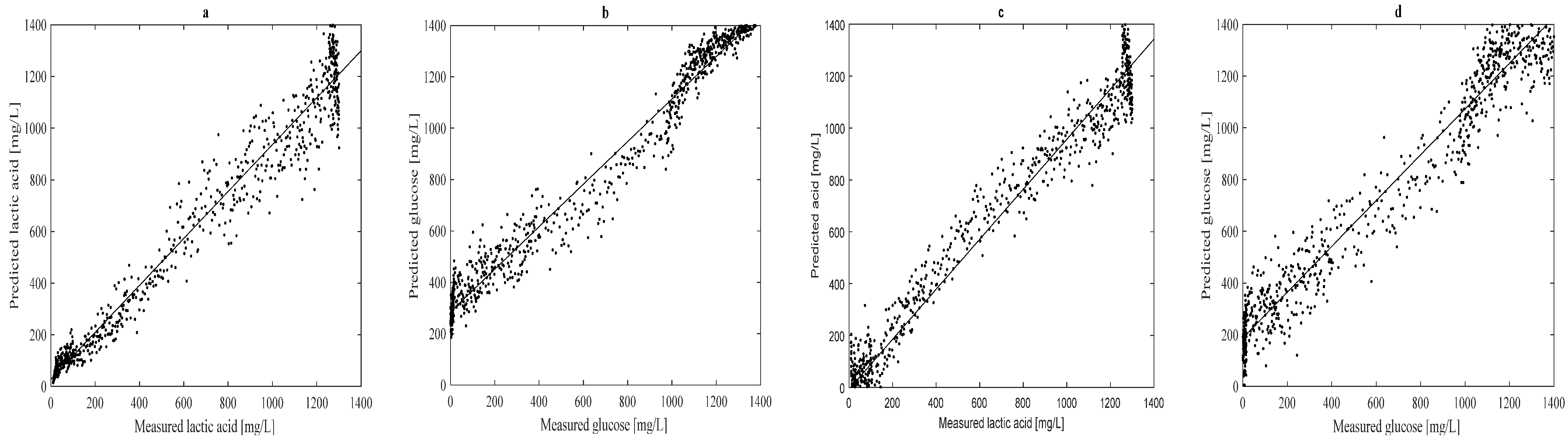
3.3. Prediction of Lactic Acid, Glucose and Cell Counts
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Küçükgöz, K.; Trząskowska, M. Nondairy Probiotic Products: Functional Foods That Require More Attention. Nutrients 2022, 14, 753. [Google Scholar] [CrossRef] [PubMed]
- Angelov, A.; Yaneva-Marinova, T.; Gotcheva, V. Oats as a matrix of choice for developing fermented functional beverages. J. Food Sci. Technol. 2018, 55, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Corbo, M.R.; Bevilacqua, A.; Petruzzi, L.; Casanova, F.P.; Sinigaglia, M. Functional beverages: The emerging side of functional foods: Commercial trends, research, and health implications. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1192–1206. [Google Scholar] [CrossRef]
- Waters, D.M.; Mauch, A.; Coffey, A.; Arendt, E.K.; Zannini, E. Lactic acid bacteria as a cell factory for the delivery of functional biomolecules and ingredients in cereal-based beverages: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 503–520. [Google Scholar] [CrossRef] [PubMed]
- Alemneh, S.T.; Emire, S.A.; Hitzmann, B.; Zettel, V. Comparative Study of Chemical Composition, Pasting, Thermal and Functional properties of Teff (Eragrostis tef) Flours Grown in Ethiopia and South Africa. Int. J. Food Prop. 2022, 25, 144–158. [Google Scholar] [CrossRef]
- Lourenço, N.; Lopes, J.; Almeida, C.; Sarraguça, M.; Pinheiro, H. Bioreactor monitoring with spectroscopy and chemometrics: A review. Anal. Bioanal. Chem. 2012, 404, 1211–1237. [Google Scholar] [CrossRef] [PubMed]
- Assawajaruwan, S.; Reinalter, J.; Hitzmann, B. Comparison of methods for wavelength combination selection from multi-wavelength fluorescence spectra for on-line monitoring of yeast cultivations. Anal. Bioanal. Chem. 2017, 409, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Claßen, J.; Aupert, F.; Reardon, K.F.; Solle, D.; Scheper, T. Spectroscopic sensors for in-line bioprocess monitoring in research and pharmaceutical industrial application. Anal. Bioanal. Chem. 2017, 409, 651–666. [Google Scholar] [CrossRef] [PubMed]
- Alemneh, S.T.; Emire, S.A.; Hitzmann, B. Teff-Based Probiotic Functional Beverage Fermented with Lactobacillus rhamnosus and Lactobacillus plantarum. Foods 2021, 10, 2333. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.T.; Guyot, J.-P.; Icard-Vernière, C.; Rochette, I.; Loiseau, G. Effect of high pressure homogenisation on the capacity of Lactobacillus plantarum A6 to ferment rice/soybean slurries to prepare high energy density complementary food. Food Chem. 2007, 102, 1288–1295. [Google Scholar] [CrossRef]
- Rathore, S.; Salmerón, I.; Pandiella, S.S. Production of potentially probiotic beverages using single and mixed cereal substrates fermented with lactic acid bacteria cultures. Food Microbiol. 2012, 30, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Hamid, N.; Gutierrez-Maddox, N.; Kantono, K.; Kitundu, E. Development of a probiotic beverage using breadfruit flour as a substrate. Foods 2019, 8, 214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alemneh, S.T.; Emire, S.A.; Jekle, M.; Paquet-Durand, O.; Hitzmann, B. Development of Fermented Teff-Based Probiotic Beverage and Its Process Monitoring Using Two-Dimensional Fluorescence Spectroscopy. Eng. Proc. 2022, 19, 37. https://doi.org/10.3390/ECP2022-12650
Alemneh ST, Emire SA, Jekle M, Paquet-Durand O, Hitzmann B. Development of Fermented Teff-Based Probiotic Beverage and Its Process Monitoring Using Two-Dimensional Fluorescence Spectroscopy. Engineering Proceedings. 2022; 19(1):37. https://doi.org/10.3390/ECP2022-12650
Chicago/Turabian StyleAlemneh, Sendeku Takele, Shimelis Admassu Emire, Mario Jekle, Olivier Paquet-Durand, and Bernd Hitzmann. 2022. "Development of Fermented Teff-Based Probiotic Beverage and Its Process Monitoring Using Two-Dimensional Fluorescence Spectroscopy" Engineering Proceedings 19, no. 1: 37. https://doi.org/10.3390/ECP2022-12650
APA StyleAlemneh, S. T., Emire, S. A., Jekle, M., Paquet-Durand, O., & Hitzmann, B. (2022). Development of Fermented Teff-Based Probiotic Beverage and Its Process Monitoring Using Two-Dimensional Fluorescence Spectroscopy. Engineering Proceedings, 19(1), 37. https://doi.org/10.3390/ECP2022-12650






