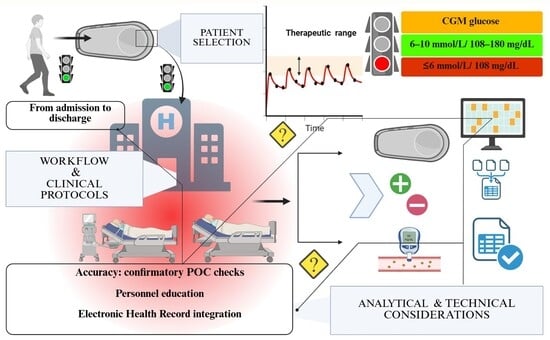
The Evolving Role of Continuous Glucose Monitoring in Hospital Settings: Bridging the Analytical and Clinical Needs
Abstract
1. Introduction
2. Methods
3. The Rationale for In-Hospital CGM Use
4. Analytical Accuracy and Clinical Performance of CGM Devices
5. Glycemic Discrepancies Between CGM and POC Measurements: CGM Lag Time and MARD Doping
Methodological Considerations and Limitations of Current CGM Evidence
6. Review of Current Recommendations, Evidence, and Clinical Applications by Settings
- a.
- CGM In Non-ICU Settings
- b.
- CGM in the Intraoperative Setting
- c.
- CGM in the ICU Settings
- d.
- CGM In Patients with Diabetic Ketoacidosis (DKA)
7. CGM-Derived Glycemic Targets and CGM Alarm Settings for In-Patients
The In-Patient Clinical Protocols
8. Implementation and Future Directions
8.1. Practical Limitations and Constraints of In-Patient CGM Use
8.1.1. Technical and Analytical Limitations
8.1.2. Operational and Implementation Challenges
8.2. Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zelada, H.; Perez-Guzman, M.C.; Chernavvsky, D.R.; Galindo, R.J. Continuous Glucose Monitoring for Inpatient Diabetes Management: An Update on Current Evidence and Practice. Endocr. Connect. 2023, 12, e230180. [Google Scholar] [CrossRef]
- Ajjan, R.A.; Battelino, T.; Cos, X.; Del Prato, S.; Philips, J.-C.; Meyer, L.; Seufert, J.; Seidu, S. Continuous Glucose Monitoring for the Routine Care of Type 2 Diabetes Mellitus. Nat. Rev. Endocrinol. 2024, 20, 426–440. [Google Scholar] [CrossRef]
- Klupa, T.; Czupryniak, L.; Dzida, G.; Fichna, P.; Jarosz-Chobot, P.; Gumprecht, J.; Mysliwiec, M.; Szadkowska, A.; Bomba-Opon, D.; Czajkowski, K.; et al. Expanding the Role of Continuous Glucose Monitoring in Modern Diabetes Care Beyond Type 1 Disease. Diabetes Ther. Res. Treat. Educ. Diabetes Relat. Disord. 2023, 14, 1241–1266. [Google Scholar] [CrossRef]
- Battelino, T.; Lalic, N.; Hussain, S.; Ceriello, A.; Klobucar, S.; Davies, S.J.; Topsever, P.; Heverly, J.; Ulivi, F.; Brady, K.; et al. The Use of Continuous Glucose Monitoring in People Living with Obesity, Intermediate Hyperglycemia or Type 2 Diabetes. Diabetes Res. Clin. Pract. 2025, 223, 112111. [Google Scholar] [CrossRef]
- Spanakis, E.K.; Cook, C.B.; Kulasa, K.; Aloi, J.A.; Bally, L.; Davis, G.; Dungan, K.M.; Galindo, R.J.; Mendez, C.E.; Pasquel, F.J.; et al. A Consensus Statement for Continuous Glucose Monitoring Metrics for Inpatient Clinical Trials. J. Diabetes Sci. Technol. 2023, 17, 1527–1552. [Google Scholar] [CrossRef]
- Korytkowski, M.T.; Muniyappa, R.; Antinori-Lent, K.; Donihi, A.C.; Drincic, A.T.; Hirsch, I.B.; Luger, A.; McDonnell, M.E.; Murad, M.H.; Nielsen, C.; et al. Management of Hyperglycemia in Hospitalized Adult Patients in Non-Critical Care Settings: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2022, 107, 2101–2128. [Google Scholar] [CrossRef] [PubMed]
- Longo, R.R.; Elias, H.; Khan, M.; Seley, J.J. Use and Accuracy of Inpatient CGM During the COVID-19 Pandemic: An Observational Study of General Medicine and ICU Patients. J. Diabetes Sci. Technol. 2022, 16, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association Professional Practice Committee. 7. Diabetes Technology: Standards of Care in Diabetes—2026. Diabetes Care 2026, 49, S150–S165. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association Professional Practice Committee for Diabetes. 16. Diabetes Care in the Hospital: Standards of Care in Diabetes—2026. Diabetes Care 2026, 49, S339–S355. [Google Scholar] [CrossRef]
- Wilson, L.M.; Herzig, S.J.; Marcantonio, E.R.; Steinman, M.A.; Schonberg, M.A.; Wang, B.X.; Hileman-Kaplan, E.; Anderson, T.S. Management of Diabetes and Hyperglycemia in the Hospital: A Systematic Review of Clinical Practice Guidelines. Diabetes Care 2025, 48, 655–664. [Google Scholar] [CrossRef]
- Avari, P.; Choudhary, P.; Lumb, A.; Misra, S.; Rayman, G.; Flanagan, D.; Dhatariya, K.; Joint British Diabetes Societies for Inpatient Care (JBDS-IP) group. Using Technology to Support Diabetes Care in Hospital: Guidelines from the Joint British Diabetes Societies for Inpatient Care (JBDS-IP) Group and Diabetes Technology Network (DTN) UK. Diabet. Med. J. Br. Diabet. Assoc. 2025, 42, e15452. [Google Scholar] [CrossRef]
- Seisa, M.O.; Saadi, S.; Nayfeh, T.; Muthusamy, K.; Shah, S.H.; Firwana, M.; Hasan, B.; Jawaid, T.; Abd-Rabu, R.; Korytkowski, M.T.; et al. A Systematic Review Supporting the Endocrine Society Clinical Practice Guideline for the Management of Hyperglycemia in Adults Hospitalized for Noncritical Illness or Undergoing Elective Surgical Procedures. J. Clin. Endocrinol. Metab. 2022, 107, 2139–2147. [Google Scholar] [CrossRef]
- Irace, C.; Coluzzi, S.; Di Cianni, G.; Forte, E.; Landi, F.; Rizzo, M.R.; Sesti, G.; Succurro, E.; Consoli, A. Continuous Glucose Monitoring (CGM) in a Non-Icu Hospital Setting: The Patient’s Journey. Nutr. Metab. Cardiovasc. Dis. 2023, 33, 2107–2118. [Google Scholar] [CrossRef]
- Bellido, V.; Freckman, G.; Pérez, A.; Galindo, R.J. Accuracy and Potential Interferences of Continuous Glucose Monitoring Sensors in the Hospital. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2023, 29, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Perez-Guzman, M.C.; Duggan, E.; Gibanica, S.; Cardona, S.; Corujo-Rodriguez, A.; Faloye, A.; Halkos, M.; Umpierrez, G.E.; Peng, L.; Davis, G.M.; et al. Continuous Glucose Monitoring in the Operating Room and Cardiac Intensive Care Unit. Diabetes Care 2021, 44, e50–e52. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.M.; Faulds, E.; Walker, T.; Vigliotti, D.; Rabinovich, M.; Hester, J.; Peng, L.; McLean, B.; Hannon, P.; Poindexter, N.; et al. Remote Continuous Glucose Monitoring with a Computerized Insulin Infusion Protocol for Critically Ill Patients in a COVID-19 Medical ICU: Proof of Concept. Diabetes Care 2021, 44, 1055–1058. [Google Scholar] [CrossRef]
- Putzu, A.; Grange, E.; Schorer, R.; Schiffer, E.; Gariani, K. Continuous Peri-Operative Glucose Monitoring in Noncardiac Surgery: A Systematic Review. Eur. J. Anaesthesiol. 2025, 42, 162–171. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Davis, G.M.; ElSayed, N.A.; Fadini, G.P.; Galindo, R.J.; Hirsch, I.B.; Klonoff, D.C.; McCoy, R.G.; Misra, S.; Gabbay, R.A.; et al. Hyperglycaemic Crises in Adults with Diabetes: A Consensus Report. Diabetologia 2024, 67, 1455–1479. [Google Scholar] [CrossRef]
- Haas, N.L.; Ang, L.; Esfandiari, N.H.; Khan, A.M.; Cranford, J.A.; Cohen, A.; Sell, J.; Abdel-Hamid, M.; Romanchik, K.E.; Korley, F.K. Analytical Accuracy of a Continuous Glucose Monitor in Adult Diabetic Ketoacidosis. CHEST Crit. Care 2025, 3, 100109. [Google Scholar] [CrossRef]
- Wang, M.; Singh, L.G.; Spanakis, E.K. Advancing the Use of CGM Devices in a Non-ICU Setting. J. Diabetes Sci. Technol. 2019, 13, 674–681. [Google Scholar] [CrossRef] [PubMed]
- van den Berghe, G.; Wouters, P.; Weekers, F.; Verwaest, C.; Bruyninckx, F.; Schetz, M.; Vlasselaers, D.; Ferdinande, P.; Lauwers, P.; Bouillon, R. Intensive Insulin Therapy in Critically Ill Patients. N. Engl. J. Med. 2001, 345, 1359–1367. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Isaacs, S.D.; Bazargan, N.; You, X.; Thaler, L.M.; Kitabchi, A.E. Hyperglycemia: An Independent Marker of in-Hospital Mortality in Patients with Undiagnosed Diabetes. J. Clin. Endocrinol. Metab. 2002, 87, 978–982. [Google Scholar] [CrossRef]
- Kotagal, M.; Symons, R.G.; Hirsch, I.B.; Umpierrez, G.E.; Dellinger, E.P.; Farrokhi, E.T.; Flum, D.R.; SCOAP-CERTAIN Collaborative. Perioperative Hyperglycemia and Risk of Adverse Events among Patients with and without Diabetes. Ann. Surg. 2015, 261, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Frisch, A.; Chandra, P.; Smiley, D.; Peng, L.; Rizzo, M.; Gatcliffe, C.; Hudson, M.; Mendoza, J.; Johnson, R.; Lin, E.; et al. Prevalence and Clinical Outcome of Hyperglycemia in the Perioperative Period in Noncardiac Surgery. Diabetes Care 2010, 33, 1783–1788. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.E.; Graham, L.A.; Morris, M.S.; Richman, J.S.; Hollis, R.H.; Wahl, T.S.; Copeland, L.A.; Burns, E.A.; Itani, K.M.F.; Hawn, M.T. Association Between Preoperative Hemoglobin A1c Levels, Postoperative Hyperglycemia, and Readmissions Following Gastrointestinal Surgery. JAMA Surg. 2017, 152, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Thabit, H.; Schofield, J. Technology in the Management of Diabetes in Hospitalised Adults. Diabetologia 2024, 67, 2114–2128. [Google Scholar] [CrossRef]
- Colunga-Lozano, L.E.; Gonzalez Torres, F.J.; Delgado-Figueroa, N.; Gonzalez-Padilla, D.A.; Hernandez, A.V.; Roman, Y.; Cuello-García, C.A. Sliding Scale Insulin for Non-Critically Ill Hospitalised Adults with Diabetes Mellitus. Cochrane Database Syst. Rev. 2018, 11, CD011296. [Google Scholar] [CrossRef]
- Lee, M.Y.; Seav, S.M.; Ongwela, L.; Lee, J.J.; Aubyrn, R.; Cao, F.Y.; Kalinsky, A.; Aparicio Ramos, O.; Gu, Y.; Kingston, K.; et al. Empowering Hospitalized Patients with Diabetes: Implementation of a Hospital-Wide CGM Policy With EHR-Integrated Validation for Dosing Insulin. Diabetes Care 2024, 47, 1838–1845. [Google Scholar] [CrossRef]
- Pantanetti, P.; Biondini, F.; Mancin, S.; Sguanci, M.; Masini, A.; Panella, M.; Palomares, S.M.; Ferrara, G.; Petrelli, F.; Cangelosi, G. Sleep Quality and Glycemic Control in Type 1 Diabetes: A Retrospective Cohort Study Using Advanced Technological Devices. Diabetology 2025, 6, 21. [Google Scholar] [CrossRef]
- Van den Berghe, G.; Wilmer, A.; Hermans, G.; Meersseman, W.; Wouters, P.J.; Milants, I.; Van Wijngaerden, E.; Bobbaers, H.; Bouillon, R. Intensive Insulin Therapy in the Medical ICU. N. Engl. J. Med. 2006, 354, 449–461. [Google Scholar] [CrossRef]
- NICE-SUGAR Study Investigators; Finfer, S.; Chittock, D.R.; Su, S.Y.-S.; Blair, D.; Foster, D.; Dhingra, V.; Bellomo, R.; Cook, D.; Dodek, P.; et al. Intensive versus Conventional Glucose Control in Critically Ill Patients. N. Engl. J. Med. 2009, 360, 1283–1297. [Google Scholar] [CrossRef] [PubMed]
- Umpierrez, G.; Cardona, S.; Pasquel, F.; Jacobs, S.; Peng, L.; Unigwe, M.; Newton, C.A.; Smiley-Byrd, D.; Vellanki, P.; Halkos, M.; et al. Randomized Controlled Trial of Intensive Versus Conservative Glucose Control in Patients Undergoing Coronary Artery Bypass Graft Surgery: GLUCO-CABG Trial. Diabetes Care 2015, 38, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.; Kaoutzanis, C.; Welch, K.B.; Vandewarker, J.F.; Winter, S.; Krapohl, G.; Lampman, R.M.; Franz, M.G.; Cleary, R.K. Postoperative Hyperglycemia and Adverse Outcomes in Patients Undergoing Colorectal Surgery: Results from the Michigan Surgical Quality Collaborative Database. Int. J. Color. Dis. 2015, 30, 1515–1523. [Google Scholar] [CrossRef]
- Peacock, T.S. Perioperative Hyperglycemia: A Literature Review. AORN J. 2019, 109, 80–86. [Google Scholar] [CrossRef]
- Omar, A.S.; Salama, A.; Allam, M.; Elgohary, Y.; Mohammed, S.; Tuli, A.K.; Singh, R. Association of Time in Blood Glucose Range with Outcomes Following Cardiac Surgery. BMC Anesthesiol. 2015, 15, 14. [Google Scholar] [CrossRef]
- Bláha, J.; Mráz, M.; Kopecký, P.; Stříteský, M.; Lipš, M.; Matias, M.; Kunstýř, J.; Pořízka, M.; Kotulák, T.; Kolníková, I.; et al. Perioperative Tight Glucose Control Reduces Postoperative Adverse Events in Nondiabetic Cardiac Surgery Patients. J. Clin. Endocrinol. Metab. 2015, 100, 3081–3089. [Google Scholar] [CrossRef] [PubMed]
- Shanks, A.M.; Woodrum, D.T.; Kumar, S.S.; Campbell, D.A.; Kheterpal, S. Intraoperative Hyperglycemia Is Independently Associated with Infectious Complications after Non-Cardiac Surgery. BMC Anesthesiol. 2018, 18, 90. [Google Scholar] [CrossRef]
- Rosenstock, J.; Bajaj, H.S.; Janež, A.; Silver, R.; Begtrup, K.; Hansen, M.V.; Jia, T.; Goldenberg, R. NN1436-4383 Investigators Once-Weekly Insulin for Type 2 Diabetes without Previous Insulin Treatment. N. Engl. J. Med. 2020, 383, 2107–2116. [Google Scholar] [CrossRef]
- Janez, A.; Muzurovic, E.; Bogdanski, P.; Czupryniak, L.; Fabryova, L.; Fras, Z.; Guja, C.; Haluzik, M.; Kempler, P.; Lalic, N.; et al. Modern Management of Cardiometabolic Continuum: From Overweight/Obesity to Prediabetes/Type 2 Diabetes Mellitus. Recommendations from the Eastern and Southern Europe Diabetes and Obesity Expert Group. Diabetes Ther. 2024, 15, 1865–1892. [Google Scholar] [CrossRef]
- Haluzik, M.; Taybani, Z.; Araszkiewicz, A.; Cerghizan, A.; Mankovsky, B.; Zuhdi, A.; Malecki, M. Expert Opinion on Optimising Type 2 Diabetes Treatment Using Fixed-Ratio Combination of Basal Insulin and GLP-1 RA for Treatment Intensification and Simplification. Diabetes Ther. Res. Treat. Educ. Diabetes Relat. Disord. 2024, 15, 1673–1685. [Google Scholar] [CrossRef] [PubMed]
- Pantanetti, P.; Cangelosi, G.; Morales Palomares, S.; Ferrara, G.; Biondini, F.; Mancin, S.; Caggianelli, G.; Parozzi, M.; Sguanci, M.; Petrelli, F. Real-World Life Analysis of a Continuous Glucose Monitoring and Smart Insulin Pen System in Type 1 Diabetes: A Cohort Study. Diabetology 2025, 6, 7. [Google Scholar] [CrossRef]
- Bender, C.; Vestergaard, P.; Cichosz, S.L. The History, Evolution and Future of Continuous Glucose Monitoring (CGM). Diabetology 2025, 6, 17. [Google Scholar] [CrossRef]
- Sanchiz, M.S.T.; Navas-Moreno, V.; Valles, F.S.; López, J.J.R.; La Ganga, C.S.; López, E.C.; Castañar, S.G.; Amar, S.; Vargas, M.L.; Martín, J.A.A.; et al. Time in Tight Range in AHCL Systems: Propensity-Score-Matched Analysis of MiniMed 780G and Control-IQ. Diabetology 2025, 6, 69. [Google Scholar] [CrossRef]
- Klonoff, D.C.; Gabbay, M.; Moon, S.J.; Wilmot, E.G. Importance of FDA-Integrated Continuous Glucose Monitors to Ensure Accuracy of Continuous Glucose Monitoring. J. Diabetes Sci. Technol. 2025, 19, 1392–1399. [Google Scholar] [CrossRef] [PubMed]
- Janez, A.; Battelino, T.; Klupa, T.; Kocsis, G.; Kuricová, M.; Lalić, N.; Stoian, A.P.; Prázný, M.; Rahelić, D.; Šoupal, J.; et al. Hybrid Closed-Loop Systems for the Treatment of Type 1 Diabetes: A Collaborative, Expert Group Position Statement for Clinical Use in Central and Eastern Europe. Diabetes Ther. Res. Treat. Educ. Diabetes Relat. Disord. 2021, 12, 3107–3135. [Google Scholar] [CrossRef]
- Nuzzo, M.G.; Schettino, M. Advanced Technology (Continuous Glucose Monitoring and Advanced Hybrid Closed-Loop Systems) in Diabetes from the Perspective of Gender Differences. Diabetology 2023, 4, 519–526. [Google Scholar] [CrossRef]
- Kim, S.J.; Hirsch, I.B. Intersystem Accuracy in Continuous Glucose Monitoring: When Does This Matter? Diabetes Care 2025, 48, 1161–1163. [Google Scholar] [CrossRef]
- Freckmann, G.; Wehrstedt, S.; Eichenlaub, M.; Pleus, S.; Link, M.; Jendrike, N.; Öter, S.; Brandt, D.; Haug, C.; Waldenmaier, D. A Comparative Analysis of Glycemic Metrics Derived from Three Continuous Glucose Monitoring Systems. Diabetes Care 2025, 48, 1213–1217. [Google Scholar] [CrossRef]
- Dávila-Ruales, V.; Gilón, L.F.; Gómez, A.M.; Muñoz, O.M.; Serrano, M.N.; Henao, D.C. Evaluating the Precision and Reliability of Real-Time Continuous Glucose Monitoring Systems in Ambulatory Settings: A Systematic Review. Ther. Adv. Endocrinol. Metab. 2024, 15, 20420188241304459. [Google Scholar] [CrossRef] [PubMed]
- Cruz, P.; McKee, A.M.; Chiang, H.-H.; McGill, J.B.; Hirsch, I.B.; Ringenberg, K.; Wildes, T.S. Perioperative Care of Patients Using Wearable Diabetes Devices. Anesth. Analg. 2025, 140, 2–12. [Google Scholar] [CrossRef]
- O’Connor, M.Y.; Flint, K.L.; Sabean, A.; Ashley, A.; Zheng, H.; Yan, J.; Steiner, B.A.; Anandakugan, N.; Calverley, M.; Bartholomew, R.; et al. Accuracy of Continuous Glucose Monitoring in the Hospital Setting: An Observational Study. Diabetologia 2024, 67, 2650–2659. [Google Scholar] [CrossRef] [PubMed]
- Migdal, A.L.; Spanakis, E.K.; Galindo, R.J.; Davis, G.; Singh, L.G.; Satyarengga, M.; Scott, W.H.; Fayfman, M.; Pasquel, F.J.; Albury, B.; et al. Accuracy and Precision of Continuous Glucose Monitoring in Hospitalized Patients Undergoing Radiology Procedures. J. Diabetes Sci. Technol. 2020, 14, 1135–1136. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.M.; Spanakis, E.K.; Migdal, A.L.; Singh, L.G.; Albury, B.; Urrutia, M.A.; Zamudio-Coronado, K.W.; Scott, W.H.; Doerfler, R.; Lizama, S.; et al. Accuracy of Dexcom G6 Continuous Glucose Monitoring in Non–Critically Ill Hospitalized Patients with Diabetes. Diabetes Care 2021, 44, 1641–1646. [Google Scholar] [CrossRef]
- Bann, S.A.; Hercus, J.C.; Atkins, P.; Alkhairy, A.; Loyal, J.P.; Sekhon, M.; Thompson, D.J. Accuracy of a Continuous Glucose Monitor in the Intensive Care Unit: A Proposed Accuracy Standard and Calibration Protocol for Inpatient Use. Diabetes Technol. Ther. 2024, 26, 797–805. [Google Scholar] [CrossRef]
- Pemberton, J.S.; Adolfsson, P.; Wilmot, E.G.; Choudhary, P.; Moser, O. Urgent Need for Standardization in CGM Performance Assessment. Diabetes Care 2025, 48, e126–e127. [Google Scholar] [CrossRef]
- Ghosh, M.; Bora, V.R. Evolution in Blood Glucose Monitoring: A Comprehensive Review of Invasive to Non-Invasive Devices and Sensors. Discov. Med. 2025, 2, 74. [Google Scholar] [CrossRef]
- Ang, L.; Lin, Y.K.; Schroeder, L.F.; Huang, Y.; DeGeorge, C.A.; Arnold, P.; Akanbi, F.; Knotts, S.; DuBois, E.; Desbrough, N.; et al. Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit. J. Diabetes Sci. Technol. 2024, 18, 562–569. [Google Scholar] [CrossRef]
- Avari, P.; Pushparatnam, R.; Leelarathna, L.; Tan, T.; Frankel, A.H.; Oliver, N.; Reddy, M. Accuracy of the Dexcom G7 Continuous Glucose Monitoring Sensors in People with Diabetes Undergoing Hemodialysis (ALPHA-2 Study). Diabetes Technol. Ther. 2025, 27, 402–406. [Google Scholar] [CrossRef]
- Aziz, Q.-U.-A.; Batra, K.; Fatima, S.; Splinter, J.; Champion, A.L.; Kumar, A.M.; Izuora, K.E. Clinical Accuracy of Continuous Glucose Monitoring Immediately After Kidney Transplant in Patients with Type 2 Diabetes. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2025, 31, 858–866. [Google Scholar] [CrossRef]
- Baker, M.; Lauterwasser, S.; Valenti, C.; Kallenberger, M.; Stolte, H. Evaluation of a Hybrid Protocol Using Continuous Glucose Monitoring and Point-of-Care Testing in Non-Critically Ill Patients in a Community Hospital. Am. J. Health-Syst. Pharm. AJHP Off. J. Am. Soc. Health-Syst. Pharm. 2024, 81, e261–e267. [Google Scholar] [CrossRef]
- Chen, A.X.; Radhakutty, A.; Zimmermann, A.; Stranks, S.N.; Thompson, C.H.; Burt, M.G. The Performance of Freestyle Libre Pro Flash Continuous Glucose Monitoring in Hospitalized Patients Treated with an Intravenous Insulin Infusion for Acute Prednisolone-Induced Hyperglycemia. Diabetes Technol. Ther. 2024, 26, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Finn, E.; Schlichting, L.; Grau, L.; Douglas, I.S.; Pereira, R.I. Real-World Accuracy of CGM in Inpatient Critical and Noncritical Care Settings at a Safety-Net Hospital. Diabetes Care 2023, 46, 1825–1830. [Google Scholar] [CrossRef]
- Friman, O.; Lind, M.; Thobaben, R.; Zetterqvist, P.; Perner, A.; Rooijackers, O.; Oldner, A.; Mårtensson, J. Accuracy of Glucose Trends by Subcutaneous Continuous Monitoring vs Intermittent Arterial Measurements in Critically Ill Patients. J. Diabetes Sci. Technol. 2025, 19322968251358830. [Google Scholar] [CrossRef]
- Friman, O.; Soltani, N.; Lind, M.; Zetterqvist, P.; Balintescu, A.; Perner, A.; Oldner, A.; Rooyackers, O.; Mårtensson, J. Performance of Subcutaneous Continuous Glucose Monitoring in Adult Critically Ill Patients Receiving Vasopressor Therapy. Diabetes Technol. Ther. 2024, 26, 763–772. [Google Scholar] [CrossRef]
- Ge, S.; Zhang, H.; Wang, J.; Li, H.; Su, X.; Ding, D.; Ma, J. Accuracy of a Novel Real-Time Continuous Glucose Monitoring System: A Prospective Self-Controlled Study in Thirty Hospitalized Patients with Type 2 Diabetes. Front. Endocrinol. 2024, 15, 1374496. [Google Scholar] [CrossRef]
- Giovannetti, E.R.; Lee, R.O.; Thomas, R.L.; Wolinsky, T.; Talbot, A.V.; Ali, R.S.; Cavaiola, T.S.; Kulasa, K.; Boeder, S.C. Continuous Glucose Monitoring-Guided Insulin Infusion in Critically Ill Patients Promotes Safety, Improves Time Efficiency, and Enhances Provider Satisfaction. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2025, 31, 1143–1149. [Google Scholar] [CrossRef]
- Gu, J.; Zhao, Z.; Li, H.; Li, B.; Chen, S.; Cao, Y.; Li, N.; Lu, X. Accuracy of Freestyle Libre Continuous Glucose Monitoring System in Critically Ill Patients after Cardiac Surgery. J. Cardiothorac. Surg. 2025, 20, 75. [Google Scholar] [CrossRef]
- Insler, S.R.; Wakefield, B.; Debs, A.; Brake, K.; Nwosu, I.; Isaacs, D.; Bena, J.; Lansang, M.C. Continuous Glucose Monitoring Using the Dexcom G6 in Cardiac Surgery During the Postoperative Period. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2024, 30, 610–615. [Google Scholar] [CrossRef]
- Janssen, H.; Dias, P.; Ahuja, S.; Alharbi, S.; Hiller, L.; Khan, K.; Iyer, K.; Sundar, A.; Abousharkh, I.; Thomas, C.; et al. Accuracy of Continuous Glucose Monitoring during Noncardiac Surgery: A Prospective, Blinded Observational Multicentre Cohort Study. Br. J. Anaesth. 2025, 135, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Krutkyte, G.; Rolfes, E.-D.; Herzig, D.; Guensch, D.P.; Schweizer, T.; Wuethrich, P.Y.; Beldi, G.; Vogt, A.P.; Bally, L. Performance of the DEXCOM G7 CGM System during and after Major Surgery. Diabetes Obes. Metab. 2025, 27, 4586–4589. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, J.; Chun, X.; Gao, Y.; Yao, R.; Liang, Y.; Zhu, L.; He, Y.; Huang, W. Performance of Continuous Glucose Monitoring in Patients with Acute Respiratory Failure: A Prospective, Single-Center Observational Study. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2024, 30, 795–801. [Google Scholar] [CrossRef]
- Moon, S.-J.; Kim, M.-S.; Kim, Y.T.; Lee, H.-E.; Lee, Y.-W.; Lee, S.-J.; Chung, E.-S.; Park, C.-Y. Use of an Insulin Titration Protocol Based on Continuous Glucose Monitoring in Postoperative Cardiac Surgery Patients with Type 2 Diabetes and Prediabetes: A Randomized Controlled Trial. Cardiovasc. Diabetol. 2025, 24, 210. [Google Scholar] [CrossRef]
- Narasaki, Y.; Kalantar-Zadeh, K.; Daza, A.C.; You, A.S.; Novoa, A.; Peralta, R.A.; Siu, M.K.M.; Nguyen, D.V.; Rhee, C.M. Accuracy of Continuous Glucose Monitoring in Hemodialysis Patients with Diabetes. Diabetes Care 2024, 47, 1922–1929. [Google Scholar] [CrossRef]
- Olsen, M.T.; Jensen, S.H.; Rasmussen, L.M.; Klarskov, C.K.; Lindegaard, B.; Andersen, J.A.; Gottlieb, H.; Lunding, S.; Hansen, K.B.; Pedersen-Bjergaard, U.; et al. Most Hospitalised Patients with Type 2 Diabetes Benefit from Continuous Glucose Monitoring Compared to Point-of-Care Glucose Testing in a Non-Intensive Care Unit Setting: A Heterogeneity of Treatment Effect Analysis. Diabetes Obes. Metab. 2025, 27, 2857–2863. [Google Scholar] [CrossRef]
- Price, C.E.; Fanelli, J.E.; Aloi, J.A.; Anzola, S.C.; Vishneski, S.R.; Saha, A.K.; Woody, C.C.; Segal, S. Feasibility of Intraoperative Continuous Glucose Monitoring: An Observational Study in General Surgery Patients. J. Clin. Anesth. 2023, 87, 111090. [Google Scholar] [CrossRef] [PubMed]
- Rivas-Montenegro, A.; Añez-Ramos, R.; Galdón-Sanz Pastor, A.; González-Albarrán, O. Continuous Glucose Monitoring in Hospitalized Patients with Type 2 Diabetes: A Step Forward in Inpatient Glycemic Control. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2025, 31, 564–570. [Google Scholar] [CrossRef]
- Sakjirapapong, C.; Sirinvaravong, S.; Preechasuk, L.; Thongtang, N. Prospective Study on Self-Calibrating Continuous Glucose Monitoring Practicality and Accuracy in Noncritically Ill COVID-19 Hospitalized Patients. J. Diabetes Res. 2025, 2025, 7538573. [Google Scholar] [CrossRef] [PubMed]
- Ullal, J.; Spanbauer, C.; Baran, J.D.; Bais, R.; Chiang, H.-H.; Chao, J.H.; Khakpour, D.; Panfil, P.; Pasquel, F.J.; Gligorijevic, N.; et al. Accuracy of Dexcom G6 Pro Continuous Glucose Monitor in the Intensive Care Setting: TIGHT ICU G6 Study. Diabetes Technol. Ther. 2025, online ahead of print. [Google Scholar] [CrossRef]
- Voglová Hagerf, B.; Protus, M.; Nemetova, L.; Mraz, M.; Kieslichova, E.; Uchytilova, E.; Indrova, V.; Lelito, J.; Girman, P.; Haluzík, M.; et al. Accuracy and Feasibility of Real-Time Continuous Glucose Monitoring in Critically Ill Patients After Abdominal Surgery and Solid Organ Transplantation. Diabetes Care 2024, 47, 956–963. [Google Scholar] [CrossRef]
- Wang, R.; Kyi, M.; Krishnamoorthi, B.; Tjahyadi, J.; Connell, A.; Chiang, C.; Renouf, D.; Barmanray, R.; Fourlanos, S. Accuracy of Continuous Glucose Monitoring in Adults with Type 1 Diabetes Admitted to Hospital: A Real-World Multicenter Observational Study. Diabetes Technol. Ther. 2025, 27, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Zelnick, L.R.; Trikudanathan, S.; Hall, Y.N.; Ayers, E.; Anderson, L.; Ashford, N.; Jones, E.; Hoofnagle, A.N.; de Boer, I.H.; Hirsch, I.B. Accuracy of Dexcom G6 Pro and G7 Continuous Glucose Monitors in Patients Treated with Maintenance Dialysis. Diabetes Technol. Ther. 2025, online ahead of print. [Google Scholar] [CrossRef]
- Zhang, R.; Wu, Y.; Xv, R.; Wang, W.; Zhang, L.; Wang, A.; Li, M.; Jiang, W.; Jin, G.; Hu, X. Clinical Application of Real-Time Continuous Glucose Monitoring System during Postoperative Enteral Nutrition Therapy in Esophageal Cancer Patients. Nutr. Clin. Pract. Off. Publ. Am. Soc. Parenter. Enter. Nutr. 2024, 39, 837–849. [Google Scholar] [CrossRef]
- Freckmann, G.; Pleus, S.; Eichenlaub, M.; Eriksson Boija, E.; Fokkert, M.; Hinzmann, R.; Jendle, J.; Klonoff, D.C.; Makris, K.; Nichols, J.H.; et al. Recommendations on the Collection of Comparator Measurement Data in the Performance Evaluation of Continuous Glucose Monitoring Systems. J. Diabetes Sci. Technol. 2025, 19, 1072–1081. [Google Scholar] [CrossRef]
- Freckmann, G.; Pleus, S.; Grady, M.; Setford, S.; Levy, B. Measures of Accuracy for Continuous Glucose Monitoring and Blood Glucose Monitoring Devices. J. Diabetes Sci. Technol. 2019, 13, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Spanakis, E.K.; Urrutia, A.; Galindo, R.J.; Vellanki, P.; Migdal, A.L.; Davis, G.; Fayfman, M.; Idrees, T.; Pasquel, F.J.; Coronado, W.Z.; et al. Continuous Glucose Monitoring-Guided Insulin Administration in Hospitalized Patients with Diabetes: A Randomized Clinical Trial. Diabetes Care 2022, 45, 2369–2375. [Google Scholar] [CrossRef]
- Hirsch, I.B.; Draznin, B.; Buse, J.B.; Raghinaru, D.; Spanbauer, C.; Umpierrez, G.E.; Ullal, J.; Jones, M.S.; Low Wang, C.C.; Spanakis, E.K.; et al. Results from a Randomized Trial of Intensive Glucose Management Using CGM Versus Usual Care in Hospitalized Adults with Type 2 Diabetes: The TIGHT Study. Diabetes Care 2025, 48, 118–124. [Google Scholar] [CrossRef]
- Agarwal, S.; Demidowich, A.P.; Soliman, D.; Umpierrez, G.E.; Galindo, R.J. Wearable Diabetes Technology for Hospitalized People with Diabetes and End-Stage Kidney Disease, Peripartum State, and Steroid Use. J. Diabetes Sci. Technol. 2025, 19322968251364276. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Kyi, M.; Krishnamoorthi, B.; Connell, A.; Chiang, C.; Renouf, D.; Barmanray, R.; Dwyer, K.; Fourlanos, S. Accuracy of Continuous Glucose Monitoring in People with Type 1 Diabetes Receiving Hemodialysis in Hospital. J. Diabetes Sci. Technol. 2025, 19, 859–861. [Google Scholar] [CrossRef] [PubMed]
- Molitch, M.E. Hyperglycaemia Management in Non-Critical Care Settings: Guidelines in Focus. Lancet Diabetes Endocrinol. 2022, 10, 614–616. [Google Scholar] [CrossRef]
- Honarmand, K.; Sirimaturos, M.; Hirshberg, E.L.; Bircher, N.G.; Agus, M.S.D.; Carpenter, D.L.; Downs, C.R.; Farrington, E.A.; Freire, A.X.; Grow, A.; et al. Society of Critical Care Medicine Guidelines on Glycemic Control for Critically Ill Children and Adults 2024. Crit. Care Med. 2024, 52, e161–e181. [Google Scholar] [CrossRef]
- Grunberger, G.; Sherr, J.; Allende, M.; Blevins, T.; Bode, B.; Handelsman, Y.; Hellman, R.; Lajara, R.; Roberts, V.L.; Rodbard, D.; et al. American Association of Clinical Endocrinology Clinical Practice Guideline: The Use of Advanced Technology in the Management of Persons with Diabetes Mellitus. Endocr. Pract. 2021, 27, 505–537. [Google Scholar] [CrossRef] [PubMed]
- Avari, P.; Lumb, A.; Flanagan, D.; Rayman, G.; Misra, S.; Dhatariya, K.; Choudhary, P. Continuous Glucose Monitoring Within Hospital: A Scoping Review and Summary of Guidelines from the Joint British Diabetes Societies for Inpatient Care. J. Diabetes Sci. Technol. 2023, 17, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Galindo, R.J.; Umpierrez, G.E.; Rushakoff, R.J.; Basu, A.; Lohnes, S.; Nichols, J.H.; Spanakis, E.K.; Espinoza, J.; Palermo, N.E.; Awadjie, D.G.; et al. Continuous Glucose Monitors and Automated Insulin Dosing Systems in the Hospital Consensus Guideline. J. Diabetes Sci. Technol. 2020, 14, 1035–1064. [Google Scholar] [CrossRef]
- Avari, P.; Lumb, A.; Flanagan, D.; Rayman, G.; Misra, S.; Choudhary, P.; Dhatariya, K. Insulin Pumps and Hybrid Close Loop Systems Within Hospital: A Scoping Review and Practical Guidance from the Joint British Diabetes Societies for Inpatient Care. J. Diabetes Sci. Technol. 2023, 17, 625–634. [Google Scholar] [CrossRef]
- Tian, T.; Aaron, R.E.; Yeung, A.M.; Huang, J.; Drincic, A.; Seley, J.J.; Wallia, A.; Gilbert, G.; Spanakis, E.K.; Masharani, U.; et al. Use of Continuous Glucose Monitors in the Hospital: The Diabetes Technology Society Hospital Meeting Report 2023. J. Diabetes Sci. Technol. 2023, 17, 1392–1418. [Google Scholar] [CrossRef]
- Shaw, J.L.V.; Bannuru, R.R.; Beach, L.; ElSayed, N.A.; Freckmann, G.; Füzéry, A.K.; Fung, A.W.S.; Gilbert, J.; Huang, Y.; Korpi-Steiner, N.; et al. Consensus Considerations and Good Practice Points for Use of Continuous Glucose Monitoring Systems in Hospital Settings. Diabetes Care 2024, 47, 2062–2075. [Google Scholar] [CrossRef]
- Gómez, A.M.; Umpierrez, G.E.; Muñoz, O.M.; Herrera, F.; Rubio, C.; Aschner, P.; Buendia, R. Continuous Glucose Monitoring Versus Capillary Point-of-Care Testing for Inpatient Glycemic Control in Type 2 Diabetes Patients Hospitalized in the General Ward and Treated with a Basal Bolus Insulin Regimen. J. Diabetes Sci. Technol. 2015, 10, 325–329. [Google Scholar] [CrossRef]
- Olsen, M.T.; Klarskov, C.K.; Jensen, S.H.; Rasmussen, L.M.; Lindegaard, B.; Andersen, J.A.; Gottlieb, H.; Lunding, S.; Pedersen-Bjergaard, U.; Hansen, K.B.; et al. In-Hospital Diabetes Management by a Diabetes Team and Insulin Titration Algorithms Based on Continuous Glucose Monitoring or Point-of-Care Glucose Testing in Patients with Type 2 Diabetes (DIATEC): A Randomized Controlled Trial. Diabetes Care 2025, 48, 569–578. [Google Scholar] [CrossRef]
- Thabit, H.; Rubio, J.; Karuppan, M.; Mubita, W.; Lim, J.; Thomas, T.; Fonseca, I.; Fullwood, C.; Leelarathna, L.; Schofield, J. Use of Real-Time Continuous Glucose Monitoring in Non-Critical Care Insulin-Treated Inpatients under Non-Diabetes Speciality Teams in Hospital: A Pilot Randomized Controlled Study. Diabetes Obes. Metab. 2024, 26, 5483–5487. [Google Scholar] [CrossRef]
- Cavalcante Lima Chagas, G.; Teixeira, L.; Clemente, M.R.C.; Cavalcante Lima Chagas, R.; Santinelli Pestana, D.V.; Rodrigues Silva Sombra, L.; Lima, B.B.; Galindo, R.J.; Abreu, M. Use of Continuous Glucose Monitoring and Point-of-Care Glucose Testing in Hospitalized Patients with Diabetes Mellitus in Non-Intensive Care Unit Settings: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Diabetes Res. Clin. Pract. 2025, 220, 111986. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Liu, Y.; Chen, Y.; Deng, W.; Ran, X.; Chen, L.; Zhu, D.; Yang, J.; Shin, J.; Lee, S.W.; et al. Multicentre Randomized Controlled Trial with Sensor-Augmented Pump vs Multiple Daily Injections in Hospitalized Patients with Type 2 Diabetes in China: Time to Reach Target Glucose. Diabetes Metab. 2017, 43, 359–363. [Google Scholar] [CrossRef]
- Davis, G.M.; Hropot, T.; Pasquel, F.J. Diabetes Technology in the Hospital. Diabetes Technol. Ther. 2025, 27, S183–S188. [Google Scholar] [CrossRef]
- Gómez Medina, A.M.; Henao-Carrillo, D.C.; Yepes, C.; Silva, J.; Gómez González, J.A.; Cortes, D.; Robledo, S.; Mejía, G.; Rondon, M. Glycemic Control Metrics in a Cohort of Hospitalized Patients with Type 1 Diabetes Using Hybrid Closed-Loop and Advanced Hybrid Closed-Loop Systems. Diabetes Res. Clin. Pract. 2023, 204, 110897. [Google Scholar] [CrossRef]
- Davis, G.M.; Hughes, M.S.; Brown, S.A.; Sibayan, J.; Perez-Guzman, M.C.; Stumpf, M.; Thompson, Z.; Basina, M.; Patel, R.M.; Hester, J.; et al. Automated Insulin Delivery with Remote Real-Time Continuous Glucose Monitoring for Hospitalized Patients with Diabetes: A Multicenter, Single-Arm, Feasibility Trial. Diabetes Technol. Ther. 2023, 25, 677–688. [Google Scholar] [CrossRef]
- Ueda, K.; Mizumoto, H.; Shibata, H.; Miyauchi, Y.; Sato, M.; Hata, D. Continuous Glucose Monitoring for Suspected Dumping Syndrome in Infants after Nissen Fundoplication. Pediatr. Int. Off. J. Jpn. Pediatr. Soc. 2013, 55, 782–785. [Google Scholar] [CrossRef] [PubMed]
- O’Kelly, R.; Quigley, E.; Byrne, K.; Kareem, M.A.; Mhaoinigh, N.N.; Healy, P.; Fanning, M.; Reynolds, J.V.; Donohoe, C.L.; Doyle, S.L. Influence of Dietary Intake and Eating Patterns on Reactive Hypoglycemic Events in Patients Postesophagectomy: A Prospective Observational Study Using Continuous Glucose Monitoring. Nutr. Clin. Pract. Off. Publ. Am. Soc. Parenter. Enter. Nutr. 2025. online ahead of print. [Google Scholar] [CrossRef]
- Chesser, H.; Abdulhussein, F.; Huang, A.; Lee, J.Y.; Gitelman, S.E. Continuous Glucose Monitoring to Diagnose Hypoglycemia Due to Late Dumping Syndrome in Children After Gastric Surgeries. J. Endocr. Soc. 2021, 5, bvaa197. [Google Scholar] [CrossRef]
- Kubota, T.; Shoda, K.; Ushigome, E.; Kosuga, T.; Konishi, H.; Shiozaki, A.; Kudo, M.; Arita, T.; Murayama, Y.; Morimura, R.; et al. Utility of Continuous Glucose Monitoring Following Gastrectomy. Gastric Cancer Off. J. Int. Gastric Cancer Assoc. Jpn. Gastric Cancer Assoc. 2020, 23, 699–706. [Google Scholar] [CrossRef]
- Price, B.; Bertrand, M.; Estrade, A.; Brinas, P.; Tuyeras, G.; Guillaume, E.; Bordes, S.; Montastier, E.; Hanaire, H.; Ritz, P. Validity of Continuous Glucose Monitoring for the Diagnosis of Dumping Syndrome After Metabolic Surgery, in Comparison to the Oral Glucose Tolerance Test. Obes. Surg. 2024, 34, 4189–4195. [Google Scholar] [CrossRef]
- Li, L. Continuous Glucose Monitoring in Metabolic Surgery: Evaluating Diagnosis of Dumping Syndrome and Glycemic Variability. Obes. Surg. 2024, 34, 4583–4585. [Google Scholar] [CrossRef]
- Volčanšek, Š.; Rahne Perc, U.; Lunder, M.; Pongrac Barlovič, D. No Indices of Increased Type 2 Diabetes Risk in Individuals with Reactive Postprandial Hypoglycemia. Metabolites 2022, 12, 1232. [Google Scholar] [CrossRef]
- Lim, H.A.; Kim, M.; Kim, N.J.; Huh, J.; Jeong, J.-O.; Hwang, W.; Choi, H. The Performance of Continuous Glucose Monitoring During the Intraoperative Period: A Scoping Review. J. Clin. Med. 2024, 13, 6169. [Google Scholar] [CrossRef] [PubMed]
- Kalmovich, B.; Bar-Dayan, Y.; Boaz, M.; Wainstein, J. Continuous Glucose Monitoring in Patients Undergoing Cardiac Surgery. Diabetes Technol. Ther. 2012, 14, 232–238. [Google Scholar] [CrossRef]
- Schierenbeck, F.; Franco-Cereceda, A.; Liska, J. Accuracy of 2 Different Continuous Glucose Monitoring Systems in Patients Undergoing Cardiac Surgery. J. Diabetes Sci. Technol. 2017, 11, 108–116. [Google Scholar] [CrossRef]
- Song, I.-K.; Lee, J.-H.; Kang, J.-E.; Park, Y.-H.; Kim, H.-S.; Kim, J.-T. Continuous Glucose Monitoring System in the Operating Room and Intensive Care Unit: Any Difference According to Measurement Sites? J. Clin. Monit. Comput. 2017, 31, 187–194. [Google Scholar] [CrossRef]
- Herzig, D.; Vettoretti, M.; Guensch, D.P.; Melmer, A.; Schürch, D.; Roos, J.; Goerg, A.M.C.; Krutkyte, G.; Cecchini, L.; Facchinetti, A.; et al. Performance of the Dexcom G6 Continuous Glucose Monitoring System During Cardiac Surgery Using Hypothermic Extracorporeal Circulation. Diabetes Care 2023, 46, 864–867. [Google Scholar] [CrossRef]
- Agarwal, S.; Mathew, J.; Davis, G.M.; Shephardson, A.; Levine, A.; Louard, R.; Urrutia, A.; Perez-Guzman, C.; Umpierrez, G.E.; Peng, L.; et al. Continuous Glucose Monitoring in the Intensive Care Unit During the COVID-19 Pandemic. Diabetes Care 2021, 44, 847–849. [Google Scholar] [CrossRef]
- Krinsley, J.S.; Chase, J.G.; Gunst, J.; Martensson, J.; Schultz, M.J.; Taccone, F.S.; Wernerman, J.; Bohe, J.; De Block, C.; Desaive, T.; et al. Continuous Glucose Monitoring in the ICU: Clinical Considerations and Consensus. Crit. Care Lond. Engl. 2017, 21, 197. [Google Scholar] [CrossRef]
- Chee, F.; Fernando, T.; van Heerden, P.V. Closed-Loop Control of Blood Glucose Levels in Critically Ill Patients. Anaesth. Intensive Care 2002, 30, 295–307. [Google Scholar] [CrossRef]
- Holzinger, U.; Warszawska, J.; Kitzberger, R.; Wewalka, M.; Miehsler, W.; Herkner, H.; Madl, C. Real-Time Continuous Glucose Monitoring in Critically Ill Patients: A Prospective Randomized Trial. Diabetes Care 2010, 33, 467–472. [Google Scholar] [CrossRef]
- Beardsall, K.; Thomson, L.; Guy, C.; Iglesias-Platas, I.; van Weissenbruch, M.M.; Bond, S.; Allison, A.; Kim, S.; Petrou, S.; Pantaleo, B.; et al. Real-Time Continuous Glucose Monitoring in Preterm Infants (REACT): An International, Open-Label, Randomised Controlled Trial. Lancet Child Adolesc. Health 2021, 5, 265–273. [Google Scholar] [CrossRef]
- Faulds, E.R.; Hester, J.C.; Badakhshi, Y.; Miller, J.D.; Basil, R.C.; Chandra, S.; Chang, A.S.; Garcia, M.; Jones, L.; Pintor, K.A.; et al. Continuous Glucose Monitoring in the Intensive Care Unit: A Multicenter, Retrospective Hospital-Based Analysis. J. Diabetes Sci. Technol. 2025, 19322968251343108. [Google Scholar] [CrossRef]
- Nielsen, C.G.; Grigonyte-Daraskeviciene, M.; Olsen, M.T.; Møller, M.H.; Nørgaard, K.; Perner, A.; Mårtensson, J.; Pedersen-Bjergaard, U.; Kristensen, P.L.; Bestle, M.H. Accuracy of Continuous Glucose Monitoring Systems in Intensive Care Unit Patients: A Scoping Review. Intensive Care Med. 2024, 50, 2005–2018. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Yuan, Z.; Zhang, Z.; Zhou, Q.; Zou, Y.; Wang, W. Effectiveness of Continuous Glucose Monitoring on Short-Term, In-Hospital Mortality Among Frail and Critically Ill Patients With COVID-19: Randomized Controlled Trial. J. Med. Internet Res. 2025, 27, e67012. [Google Scholar] [CrossRef]
- Danne, T.; Garg, S.; Peters, A.L.; Buse, J.B.; Mathieu, C.; Pettus, J.H.; Alexander, C.M.; Battelino, T.; Ampudia-Blasco, F.J.; Bode, B.W.; et al. International Consensus on Risk Management of Diabetic Ketoacidosis in Patients with Type 1 Diabetes Treated with Sodium-Glucose Cotransporter (SGLT) Inhibitors. Diabetes Care 2019, 42, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Kyi, M.; Krishnamoorthi, B.; Connell, A.; Chiang, C.; Renouf, D.; Barmanray, R.; Fourlanos, S. Continuous Glucose Monitoring during Intravenous Insulin Infusion Treatment: Assessing Accuracy to Enable Future Clinical Utility. Diabet. Med. J. Br. Diabet. Assoc. 2025, 42, e70076. [Google Scholar] [CrossRef] [PubMed]
- Battelino, T.; Danne, T.; Bergenstal, R.M.; Amiel, S.A.; Beck, R.; Biester, T.; Bosi, E.; Buckingham, B.A.; Cefalu, W.T.; Close, K.L.; et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations from the International Consensus on Time in Range. Diabetes Care 2019, 42, 1593–1603. [Google Scholar] [CrossRef]
- Olsen, M.T.; Liarakos, A.L.; Wilmot, E.G.; Dhatariya, K.; Thabit, H.; Sánchez-García, D.; Nørgaard, K.; Pedersen-Bjergaard, U.; Hansen, K.B.; Vangoitsenhoven, R.; et al. Implementation Strategies for Inpatient Continuous Glucose Monitoring-Based Diabetes Management: A Systematic Review. J. Clin. Endocrinol. Metab. 2025, 110, e2411–e2419. [Google Scholar] [CrossRef]
- Olsen, M.T.; Pedersen-Bjergaard, U.; Jensen, S.H.; Rasmussen, L.M.; Klarskov, C.K.; Lindegaard, B.; Andersen, J.A.; Gottlieb, H.; Lunding, S.; Hansen, K.B.; et al. Evaluation of a Continuous Glucose Monitoring-Based Insulin Titration Protocol for Inpatients with Type 2 Diabetes in Nonintensive Care Unit Settings. J. Diabetes Sci. Technol. 2025, 19322968251331628. [Google Scholar] [CrossRef]
- Davasgaium, A.; Robbins, T.; Leca, B.; Epure, A.; Sankar, S.; Randeva, H. Comparing Glucose Monitoring Methods: Efficiency Insights in a Simulated Hospital Setting. Front. Clin. Diabetes Healthc. 2025, 6, 1517161. [Google Scholar] [CrossRef]
- Buschur, E.O.; Faulds, E.; Dungan, K. CGM in the Hospital: Is It Ready for Prime Time? Curr. Diab. Rep. 2022, 22, 451–460. [Google Scholar] [CrossRef]
- Faulds, E.R.; Dungan, K.M.; McNett, M.; Jones, L.; Poindexter, N.; Exline, M.; Pattison, J.; Pasquel, F.J. Nursing Perspectives on the Use of Continuous Glucose Monitoring in the Intensive Care Unit. J. Diabetes Sci. Technol. 2023, 17, 649–655. [Google Scholar] [CrossRef]
- Olsen, M.T.; Vikner, M.E.; Jensen, S.H.; Rasmussen, L.M.; Klarskov, C.K.; Lindegaard, B.; Andersen, J.A.; Gottlieb, H.; Lunding, S.; Nørgaard, K.; et al. Satisfaction with Continuous Glucose Monitoring and Diabetes Care among Hospitalised Patients with Type 2 Diabetes Managed by Inpatient Diabetes Teams. Diabet. Med. J. Br. Diabet. Assoc. 2025, 42, e70114. [Google Scholar] [CrossRef] [PubMed]
- Bergenstal, R.M. Roadmap to the Effective Use of Continuous Glucose Monitoring: Innovation, Investigation, and Implementation. Diabetes Spectr. Publ. Am. Diabetes Assoc. 2023, 36, 327–336. [Google Scholar] [CrossRef]
- Nicholson, N.; Štotl, I. A Generic Framework for the Semantic Contextualization of Indicators. Front. Comput. Sci. 2024, 6, 1463989. [Google Scholar] [CrossRef]
- Witte, H.; Nakas, C.; Bally, L.; Leichtle, A.B. Machine Learning Prediction of Hypoglycemia and Hyperglycemia from Electronic Health Records: Algorithm Development and Validation. JMIR Form. Res. 2022, 6, e36176. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Castro-Revoredo, I.; Moazzami, B.; Nayberg, I.; Zabala, Z.; Galindo, R.J.; Vellanki, P.; Peng, L.; Klonoff, D.C. Randomized Study Comparing Continuous Glucose Monitoring and Capillary Glucose Testing in Patients with Type 2 Diabetes After Hospital Discharge. Endocr. Pract. Off. J. Am. Coll. Endocrinol. Am. Assoc. Clin. Endocrinol. 2025, 31, 286–291. [Google Scholar] [CrossRef]
- Vongsumran, N.; Buranapin, S.; Manosroi, W. Standardized Glycemic Management versus Conventional Glycemic Management and Postoperative Outcomes in Type 2 Diabetes Patients Undergoing Elective Surgery. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 2593–2601. [Google Scholar] [CrossRef] [PubMed]
- Vedantam, D.; Poman, D.S.; Motwani, L.; Asif, N.; Patel, A.; Anne, K.K. Stress-Induced Hyperglycemia: Consequences and Management. Cureus 2022, 14, e26714. [Google Scholar] [CrossRef]
- Ljungqvist, O.; Nygren, J.; Thorell, A.; Brodin, U.; Efendic, S. Preoperative Nutrition—Elective Surgery in the Fed or the Overnight Fasted State. Clin. Nutr. 2001, 20, 167–171. [Google Scholar] [CrossRef]
- Visser, M.M.; Vangoitsenhoven, R.; Gillard, P.; Mathieu, C. Review Article—Diabetes Technology in the Hospital: An Update. Curr. Diab. Rep. 2024, 24, 173–182. [Google Scholar] [CrossRef] [PubMed]

| Author (1.), Year | Study Design | Patient Characteristics | Clinical Setting | CGM System(s) Tested/ Reference Standard | Key Accuracy Metric | Key Clinical Outcome and Conclusions |
|---|---|---|---|---|---|---|
| Ang L et al., 2024 [57] | Prospective cohort | 59 postsurgical patients with hyperglycemia requiring insulin infusion | Cardiovascular ICU | Dexcom G6/ POC | MARD, Clarke error grid | MARD 13.2% Nurses reported CGMs being very or quite convenient, and it were favored over POC-BG testing |
| Avari P et al., 2025 [58] | Prospective cohort | 10 PwT1D and T2D | undergoing hemodialysis | Dexcom G7/laboratory/ POC | MARD, DTS error grid | MARD CGM vs. laboratory 10.4% |
| Aziz QUA et al., 2025 [59] | Prospective cohort | 22 patients with T2D after kidney transplant | Surgical ICU | NA | MARD, Clarke error grid | MARD 13.2% |
| Baker M et al., 2024 [60] | Prospective cohort | 30 hospitalized patients requiring POC, 80% with T2D | General wards | NA | MARD, surveillance error grid, Clarke error grid | MARD 12.5% |
| Bann et al., 2024 [54] | Prospective cohort | 28 adults (mixed diabetes and surgical) | Medical-surgical ICU | Dexcom G6/ laboratory | MARD, Clarke error grid | MARD 13.2% non-calibrated, MARD 9.6% calibrated—Calibration protocol improves accuracy |
| Chen AX et al., 2024 [61] | Prospective cohort | NA, hospitalized patients receiving insulin for prednisolone-associated hyperglycemia | Freestyle Libre Pro/ POC | MARD, Clarke error grid | ||
| Finn E et al., 2023 [62] | Retrospective analysis | 233 hospitalized adult patients | ICU, non-ICU | Dexcom G6/ POC/laboratory | MARD, Clarke error grid | POC-CGM MARD 17.1%, Laboratory-CGM MARD 12.2%, Real-world accuracy of in-patient CGM is acceptable for critically and non-critically ill patients |
| Friman et al., 2025 [63] | Prospective cohort | 40 ICU patients receiving insulin and organ-supportive therapies | ICU | Dexcom G6/ laboratory | Rate error grid | CGM demonstrated high overall trend accuracy relative to aBG. Trend accuracy was reduced at lower glucose ranges and during the initial 24 h of CGM use |
| Friman O et al., 2024 [64] | Prospective cohort | 40 ICU patients requiring mechanical ventilation, insulin infusion, and vasopressor therapy | ICU | Dexcom G6/ laboratory | MARD, Clarke error grid | MARD 12.7% |
| Ge S et al., 2024 [65] | Prospective cohort | 30 hospitalized PwT2D | General wards | Glunovo/FGM/POC | MARD, Clarke error grid | MARD 8.9% |
| Giovanetti et al., 2025 [66] | Retrospective analysis | 35 critically ill patients requiring insulin infusion | Surgical and medical ICUs | Dexcom G7/ POC | MARD, surveillance error grid, Parkes error grid | MARD 12.5%, Clinician time efficiency improved significantly; all surveyed nurses (n = 20) reported that CGM increased efficiency and improved safety, and preferred CGM with POC over POC testing alone |
| Gu J et al., 2025 [67] | Prospective cohort | 86 patients with hyperglycemia after cardiac surgery | Surgical ICU | Freestyle Libre/ laboratory | MARD, Clarke error grid | MARD 21.5% MARD aBG vs. vBG 8.4% |
| Insler SR et al., 2024 [68] | Prospective cohort | 29 patients after cardiac surgery | Surgical ICU | Dexcom G6 Pro/ laboratory/POC | MARD, Clarke error grid | MARD 21.6% |
| Janssen H et al., 2025 [69] | Prospective cohort | 118 surgical patients with or without diabetes | Perioperative, non-cardiac surgery | Dexcom G7/ laboratory | Overall mean difference (bias), MARD, surveillance error grid | MARD 12.0–18.3% |
| Krutkyte G et al., 2025 [70] | Retrospective analysis | 29 adult patients | During and after major surgery | Dexcom G7/ laboratory | MARD, Diabetes Technology Society error grid, Clarke error grid | MARD during surgery 12.5%, MARD during ECC 15.5%, MARD after surgery 9.0% CGM system exhibits adequate accuracy with no signal losses during surgery |
| Lee et al., 2024 [28] | Retrospective analysis | 135 PwD, 28.6% with an insulin pump | ICU, medical or surgical wards | Dexcom G6, FreeStyle Libre 2, Medtronic/POC | Clarke error grid | Implementation of a hospital-wide in-patient CGM policy supporting multiple CGM types with real-time accuracy monitoring and integration into the EHR |
| Liu Y et al., 2024 [71] | Prospective cohort | 40 ICU patients with acute respiratory failure | ICU | Freestyle Libre H/ Laboratory/POC | MARD, Clarke error grid | MARD CGM vs. aBG 13.8% MARD CGM vs. POC 14.7% |
| Moon et al., 2025 [72] | RCT | 54 cardiac surgery patients (60% PwD, 40% non-diabetic) | Cardiac surgery ICU | Dexcom G6/ POC | TIR | CGM with a specialized titration protocol demonstrated safe glycemic control with improvements in TIR |
| Narasaki Y et al., 2024 [73] | Prospective cohort | 31 PwD | on maintenance dialysis | Dexcom G6/ laboratory | MARD, Consensus error grid | MARD 20% Consensus error grids showed nearly all CGM values were clinically acceptable |
| O’Connor et al., 2024 [51] | Prospective cohort | 326 PwD | Non-ICU medical/surgical wards | Dexcom G6 Pro/ POC/laboratory | MARD, %20/20, Clarke error grid | MARD 19.2% Lower accuracy in severe anemia, renal dysfunction and edema. Once-daily morning calibration schedule improved accuracy (MARD 11.4%) |
| Olsen et al., 2025 [74] | RCT | 166 PwT2D | Non ICU | Dexcom G6/ POC | TIR | No heterogeneity of treatment effect was observed, suggesting that all patients benefited equally from CGM compared to POC glucose testing regarding glycemic outcomes |
| Price et al., 2023 [75] | Prospective cohort | 76 PwD undergoing major surgery | Perioperative, general surgery | Abbott Freestyle Libre 2.0 and/or Dexcom G6/ POC | Pearson correlation coefficient | CGM provided more glycemic data and glycemic trends. The required time of CGM warm-up was a barrier for intraoperative use, as well as unexplained sensor failure |
| Rivas-Montenegro et al., 2025 [76] | Pilot RCT | 37 PwT2D | Non-ICU medical/surgical wards | Abbott FreeStyle 2/3/ POC | TIR, MARD, DTS error grid | TIR was higher, and more asymptomatic hypoglycemia was detected in the CGM arm MARD 14.7% |
| Sakjirapapong C et al., 2025 [77] | Prospective cohort | 15 patients with COVID-19 receiving insulin | Non ICU | Medtronic Guardian Sensor 3/ POC | MARD, Clarke error grid | MARD 9.9% |
| Ullal J et al., 2025 [78] | Prospective multicenter cohort | 130 adult ICU PwD or stress hyperglycemia receiving insulin | ICU, non-ICU | Dexcom G6/ laboratory TWO sensors placed! | MARD | MARD 23% The accuracy of the Dexcom G6 Pro sensor in the ICU setting was worse than has previously been reported |
| Voglova Hagerf B et al., 2024 [79] | Prospective cohort | 61 patients after pancreas surgery or solid organ transplantation | Surgical ICU | Dexcom G6/ laboratory/POC | Overall mean difference (bias), MARD, surveillance error grid | MARD 9.4% |
| Wang et al., 2025 [80] | Multicenter retrospective observational | 146 PwT1D | ICU, non-ICU | Modern CGM devices (not specified)/ POC/laboratory | MARD, consensus error grid | POC-CGM MARD 12.3%, Laboratory-CGM MARD 14.3% Modern CGM devices could be safely and effectively used in hospitalized PwT1D |
| Zelnick et al., 2025 [81] | Prospective cohort | 12 PwD | on maintenance dialysis | Both Dexcom G6 pro and G7/ POC | MARD, DTS error grid | G6 Pro MARD 18.3%, G7 MARD 13.5% |
| Zhang R et al., 2024 [82] | Prospective cohort | NA, non-diabetic patients with esophageal cancer receiving postoperative EN | Surgical ICU | NA/ laboratory | MARD, Clarke error grid | MARD 13.5% |
| Guideline Body Latest Edition Guideline /Consensus Document | Primary Focus Area | Recommended Strength | Recommended Target Glucose Range | CGM/Insulin Pump Stance | CGM Limitations/ Contraindications/ Requirements |
|---|---|---|---|---|---|
| American Diabetes Association (ADA) (Standards of Care 2026) [8,9] | Clinical practice guideline for PwD | Conditional; evidence-based (Grading of Recommendations Assessment, Development and Evaluation [GRADE]) | 5.6–10.0 mmol/L = 100–180 mg/dL
7.8–10.0 mmol/L = 140–180 mg/dL
| Continuation of personal CGM/AID systems use with Hybrid testing protocols:
|
|
| Endocrine Society (ES) (2022 Update) Korytkowski et al., 2022 [6] | Clinical practice guideline for non-critical care adult in-patients | Conditional; evidence-based (Grading of Recommendations Assessment, Development and Evaluation [GRADE]) | 5.6–10.0 mmol/L = 100–180 mg/dL | Continuation of personal CGM/AID systems use
|
|
| Joint British Diabetes Societies (JBDS-IP) (Latest Guidance) Avari et al., 2023 [92,94] | Scoping review and guideline summary for CGM in the hospital | Conditional; consensus-based, moderate evidence | 6.0–10.0 mmol/L = 108–180 mg/dL
HIGH ALERT set at 15–18 mmol/L = 270–324 mg/dL LOW ALERT set at 4–5 mmol/L = 72–90 mg/dL | Continuation of personal CGM/AID systems use
| Discontinuation required:
|
| Joint British Diabetes Societies (JBDS-IP) (Latest Guidance) Avari et al., 2022, “Insulin Pumps and Hybrid Closed-Loop Systems” [11] | Scoping review and guidance for insulin pumps and hybrid closed-loop in the hospital | Conditional; consensus-based, moderate/low evidence | 6.0–10.0 mmol/L = 108–180 mg/dL
6.0–12.0 mmol/L = 108–216 mg/dL
15–18 mmol/L = 270–324 mg/dL LOW ALERT set at 4–5 mmol/L = 72–90 mg/dL LOOMING HYPOGLYCEMIA 4–6 mmol/L = 72–108 mg/dL | Hybrid testing protocols:
| Discontinuation required:
|
| Diabetes Technology Society (DTS) (Consensus Guidelines) Galindo et al., 2020 [93] | Consensus guideline for Continuous Glucose Monitoring (CGM) and automated insulin dosing in the hospital | Strong/mild consensus; evidence- and consensus-based | No mention of targets | CGM can be used as a primary monitoring tool, often in a hybrid protocol with POC checks. Recommend continuation of home CGM for patients not cognitively impaired and capable of self-management | Discontinuation required:
|
| Diabetes Technology Society (DTS) (Consensus Guidelines) Spanakis et al., 2023 [5] | Consensus statement on CGM metrics for in-patient trials | Strong/mild consensus; consensus-based | 5.6–10.0 mmol/L = 100 to 180 mg/dL
= 3.9–10 mmol/L = 70–180 mg/dL achieved in
| Individualized clinically acceptable target glucose ranges may vary
| |
| Diabetes Technology Society (DTS) Tian et al., 2023 [95] | Meeting report | Consensus-based | No mention Mentions separate metrics of glycemia;
| Meeting topics
| Cross-sector collaboration is critical to advance the state of EHR integration and interoperability: iCoDE-1 (Integration of Continuous Glucose Monitoring Data into the Electronic Health Record) focuses on moving diabetes technology data from various devices into the EHR |
| Multidisciplinary expert panel (International) Shaw et al., 2024 [96] | Good practice points for CGM in the hospital | Consensus-based The document reviews evidence on hospital CGM use | No mention of targets | Topics:
| Factors to consider for safe use of CGM systems in hospitals:
|
| Situation of Suspected CGM Inaccuracy | Need for Initial or Periodic POC Testing |
|---|---|
| Suspected Hypoglycemia |
|
| Unreliable CGM Readings |
|
| Calibration Required by the Device |
|
| During/Following Procedures |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Volčanšek, Š.; Janež, A.; Srpčič, M. The Evolving Role of Continuous Glucose Monitoring in Hospital Settings: Bridging the Analytical and Clinical Needs. Diabetology 2026, 7, 6. https://doi.org/10.3390/diabetology7010006
Volčanšek Š, Janež A, Srpčič M. The Evolving Role of Continuous Glucose Monitoring in Hospital Settings: Bridging the Analytical and Clinical Needs. Diabetology. 2026; 7(1):6. https://doi.org/10.3390/diabetology7010006
Chicago/Turabian StyleVolčanšek, Špela, Andrej Janež, and Matevž Srpčič. 2026. "The Evolving Role of Continuous Glucose Monitoring in Hospital Settings: Bridging the Analytical and Clinical Needs" Diabetology 7, no. 1: 6. https://doi.org/10.3390/diabetology7010006
APA StyleVolčanšek, Š., Janež, A., & Srpčič, M. (2026). The Evolving Role of Continuous Glucose Monitoring in Hospital Settings: Bridging the Analytical and Clinical Needs. Diabetology, 7(1), 6. https://doi.org/10.3390/diabetology7010006