Metabolic Associated Fatty Liver Disease as a Risk Factor for the Development of Central Nervous System Disorders
Abstract
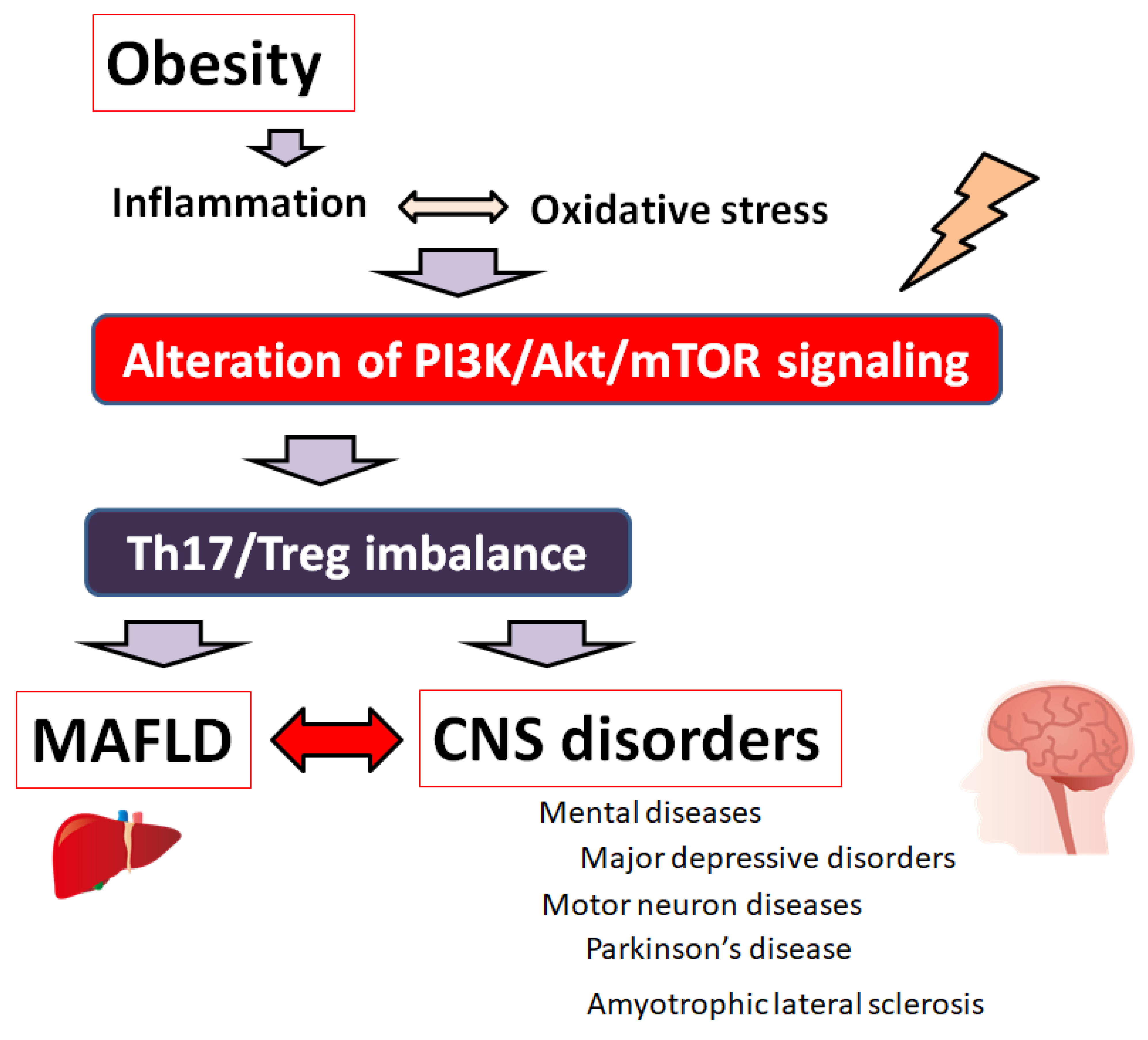
1. Introduction
2. Th17/Treg Balance Involved in MAFLD and/or Psychiatric Disorders
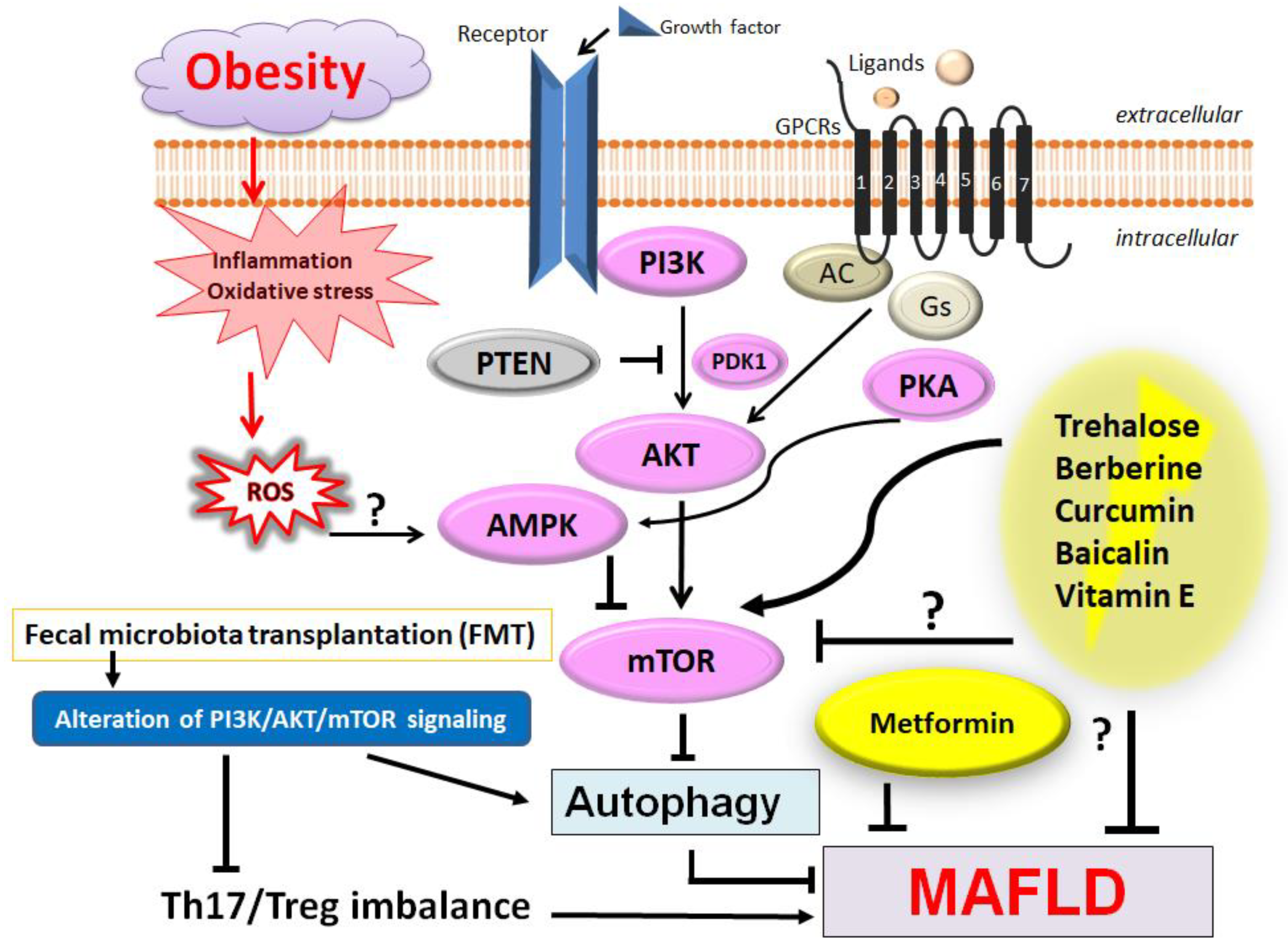
3. PI3K/AKT/mTOR Signaling Pathway Involved in the Regulation of Th17/Treg Balance of Various Diseases
4. Therapeutic Strategies for Patients with MAFLD and/or Various CNS Disorders
5. Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALS | amyotrophic lateral sclerosis |
| AMP | Adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| BMI | body mass index |
| CNS | central nervous system |
| FMT | fecal microbiota transplantation |
| NAFLD | non-alcoholic fatty liver disease |
| MAFLD | metabolic-associated fatty liver disease |
| NASH | non-alcoholic steatohepatitis |
| mTOR | mammalian/mechanistic target of rapamycin |
| PI3K | phosphoinositide-3 kinase |
| PKA | protein kinase A |
| PTEN | phosphatase and tensin homologue deleted on chromosome 10 |
| QOL | quality of life |
| ROS | reactive oxygen species |
| UC | ulcerative colitis |
References
- Cotter, T.G.; Rinella, M. Nonalcoholic Fatty Liver Disease 2020: The State of the Disease. Gastroenterology 2020, 158, 1851–1864. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A. Renaming NAFLD to MAFLD: Could the LDE System Assist in This Transition? J. Clin. Med. 2021, 10, 492. [Google Scholar] [CrossRef] [PubMed]
- Eslam, M.; Sanyal, A.J.; George, J.; on behalf of the International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology 2020, 158, 1999–2014.e1. [Google Scholar] [CrossRef] [PubMed]
- Labenz, C.; Huber, Y.; Michel, M.; Nagel, M.; Galle, P.R.; Kostev, K.; Schattenberg, J.M. Nonalcoholic Fatty Liver Disease Increases the Risk of Anxiety and Depression. Hepatol. Commun. 2020, 4, 1293–1301. [Google Scholar] [CrossRef]
- Adibhatla, R.M.; Hatcher, J.F. Altered lipid metabolism in brain injury and disorders. Subcell Biochem. 2008, 49, 241–268. [Google Scholar]
- Johnson, B.; Kokkinis, A.; Gai, N.; Shamim, E.A.; Blackstone, C.; Fischbeck, K.H.; Grunseich, C. Nonalcoholic Fatty Liver Disease in Patients with Inherited and Sporadic Motor Neuron Degeneration. Genes 2022, 13, 936. [Google Scholar] [CrossRef]
- Guber, R.D.; Takyar, V.; Kokkinis, A.; Fox, D.A.; Alao, H.; Kats, I.; Bakar, D.; Remaley, A.T.; Hewitt, S.M.; Kleiner, D.E.; et al. Nonalcoholic fatty liver disease in spinal and bulbar muscular atrophy. Neurology 2017, 89, 2481–2490. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Laukkanen, J.A. Gamma glutamyltransferase and risk of future dementia in middle-aged to older Finnish men: A new prospective cohort study. Alzheimers Dement 2016, 12, 931–941. [Google Scholar] [CrossRef]
- Joo, J.H.; Kim, H.J.; Park, E.C.; Jang, S.I. Association between sitting time and non-alcoholic fatty live disease in South Korean population: A cross-sectional study. Lipids Health Dis. 2020, 19, 212. [Google Scholar] [CrossRef]
- Deguise, M.O.; Baranello, G.; Mastella, C.; Beauvais, A.; Michaud, J.; Leone, A.; De Amicis, R.; Battezzati, A.; Dunham, C.; Selby, K.; et al. Abnormal fatty acid metabolism is a core component of spinal muscular atrophy. Ann. Clin. Transl. Neurol. 2019, 6, 1519–1532. [Google Scholar] [CrossRef]
- Fabbrini, E.; Sullivan, S.; Klein, S. Obesity and nonalcoholic fatty liver disease: Biochemical, metabolic, and clinical implications. Hepatology 2010, 51, 679–689. [Google Scholar] [CrossRef]
- Xia, Y.; Ren, M.; Yang, J.; Cai, C.; Cheng, W.; Zhou, X.; Lu, D.; Ji, F. Gut microbiome and microbial metabolites in NAFLD and after bariatric surgery: Correlation and causality. Front. Microbiol. 2022, 13, 1003755. [Google Scholar] [CrossRef]
- Funuyet-Salas, J.; Pérez-San-Gregorio, M.Á.; Martín-Rodríguez, A.; Romero-Gómez, M. Vitality, mental health and role-physical mediate the influence of coping on depressive symptoms and self-efficacy in patients with non-alcoholic fatty liver disease: A cross-sectional study. J. Psychosom. Res. 2022, 162, 111045. [Google Scholar] [CrossRef] [PubMed]
- Manning, P.J.; Sutherland, W.H.; McGrath, M.M.; de Jong, S.A.; Walker, R.J.; Williams, M.J. Postprandial cytokine concentrations and meal composition in obese and lean women. Obesity 2008, 16, 2046–2052. [Google Scholar] [CrossRef] [PubMed]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef] [PubMed]
- Ter Horst, K.W.; Serlie, M.J. Fructose Consumption, Lipogenesis, and Non-Alcoholic Fatty Liver Disease. Nutrients 2017, 9, 981. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, H.; Yao, Y.; Zhang, X.; Guan, Y.; Zheng, F. CD4+ T cell activation and inflammation in NASH-related fibrosis. Front. Immunol. 2022, 13, 967410. [Google Scholar] [CrossRef]
- He, B.; Wu, L.; Xie, W.; Shao, Y.; Jiang, J.; Zhao, Z.; Yan, M.; Chen, Z.; Cui, D. The imbalance of Th17/Treg cells is involved in the progression of nonalcoholic fatty liver disease in mice. BMC Immunol. 2017, 18, 33. [Google Scholar] [CrossRef]
- Rau, M.; Schilling, A.K.; Meertens, J.; Hering, I.; Weiss, J.; Jurowich, C.; Kudlich, T.; Hermanns, H.M.; Bantel, H.; Beyersdorf, N.; et al. Progression from Nonalcoholic Fatty Liver to Nonalcoholic Steatohepatitis Is Marked by a Higher Frequency of Th17 Cells in the Liver and an Increased Th17/Resting Regulatory T Cell Ratio in Peripheral Blood and in the Liver. J. Immunol. 2016, 196, 97–105. [Google Scholar] [CrossRef]
- Saigusa, R.; Winkels, H.; Ley, K. T cell subsets and functions in atherosclerosis. Nat. Rev. Cardiol. 2020, 17, 387–401. [Google Scholar] [CrossRef]
- Fabre, T.; Kared, H.; Friedman, S.L.; Shoukry, N.H. IL-17A enhances the expression of profibrotic genes through upregulation of the TGF-β receptor on hepatic stellate cells in a JNK-dependent manner. J. Immunol. 2014, 193, 3925–3933. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, H.; Wang, Y.; Brown, Z.J.; Xia, Y.; Huang, Z.; Shen, C.; Hu, Z.; Beane, J.; Ansa-Addo, E.A.; et al. Regulatory T-cell and neutrophil extracellular trap interaction contributes to carcinogenesis in non-alcoholic steatohepatitis. J. Hepatol. 2021, 75, 1271–1283. [Google Scholar] [CrossRef] [PubMed]
- Van Herck, M.A.; Weyler, J.; Kwanten, W.J.; Dirinck, E.L.; De Winter, B.Y.; Francque, S.M.; Vonghia, L. The Differential Roles of T Cells in Non-alcoholic Fatty Liver Disease and Obesity. Front. Immunol. 2019, 10, 82. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Lv, J.; Jiang, H.; Zheng, K.; Chen, Y. Inflammatory Cytokines, Adipocytokines, and Th17/Treg Balance in Patients with Nonalcoholic Fatty Liver Disease following Administration of Dahuang Zhechong Pills. Genet. Res. 2022, 2022, 8560831. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, T.; Chen, P.; Ouyang, J.; Xu, G.; Zeng, Z.; Sun, Y. Emerging tendency towards autoimmune process in major depressive patients: A novel insight from Th17 cells. Psychiatry Res. 2011, 188, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, A.; Ahmad, S.F.; Al-Harbi, N.O.; Fardan, A.S.; El-Sherbeeny, A.M.; Ibrahim, K.E.; Attia, S.M. IL-17A causes depression-like symptoms via NFκB and p38MAPK signaling pathways in mice: Implications for psoriasis associated depression. Cytokine 2017, 97, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Mon, M.A.; Gomez-Lahoz, A.M.; Orozco, A.; Lahera, G.; Sosa-Reina, M.D.; Diaz, D.; Albillos, A.; Quintero, J.; Molero, P.; Monserrat, J.; et al. Blunted Expansion of Regulatory T Lymphocytes Is Associated With Increased Bacterial Translocation in Patients With Major Depressive Disorder. Front. Psychiatry 2021, 11, 591962. [Google Scholar] [CrossRef]
- Fu, J.; Huang, Y.; Bao, T.; Liu, C.; Liu, X.; Chen, X. The role of Th17 cells/IL-17A in AD, PD, ALS and the strategic therapy targeting on IL-17A. J. Neuroinflamm. 2022, 19, 98. [Google Scholar] [CrossRef]
- Shi, Y.; Wei, B.; Li, L.; Wang, B.; Sun, M. Th17 cells and inflammation in neurological disorders: Possible mechanisms of action. Front. Immunol. 2022, 13, 932152. [Google Scholar] [CrossRef]
- Jin, M.; Akgün, K.; Ziemssen, T.; Kipp, M.; Günther, R.; Hermann, A. Interleukin-17 and Th17 Lymphocytes Directly Impair Motoneuron Survival of Wildtype and FUS-ALS Mutant Human iPSCs. Int. J. Mol. Sci. 2021, 22, 8042. [Google Scholar] [CrossRef]
- Rolla, S.; Alchera, E.; Imarisio, C.; Bardina, V.; Valente, G.; Cappello, P.; Mombello, C.; Follenzi, A.; Novelli, F.; Carini, R. The balance between IL-17 and IL-22 produced by liver-infiltrating T-helper cells critically controls NASH development in mice. Clin. Sci. 2016, 130, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Zeng, P.; Jiang, Z.; Huang, Z.; Huang, Y.; Xu, H.; Chen, C.; Ma, W. PI3K/AKT/mTOR Signaling Pathway Is Downregulated by Runzaoling (RZL) in Sjögren’s Syndrome. Mediat. Inflamm. 2022, 2022, 7236118. [Google Scholar] [CrossRef]
- He, H.; Chen, Q.; Fan, H.; Leng, X.Y.; Zhu, F.; Gao, F.; Zhou, Q.; Dong, Y.; Yang, J. Extracellular vesicles produced by bone marrow mesenchymal stem cells overexpressing programmed death-ligand 1 ameliorate dextran sodium sulfate-induced ulcerative colitis in rats by regulating Th17/Treg cell balance through PTEN/PI3K/AKT/mTOR axis. J. Gastroenterol. Hepatol. 2022, 37, 2243–2254. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Luo, J.; Liu, Y.; Yu, X.B.; Liu, X.Y.; Tao, Q.W. Network Pharmacology Analysis and Experimental Validation to Investigate the Mechanism of Total Flavonoids of Rhizoma Drynariae in Treating Rheumatoid Arthritis. Drug Des. Devel. Ther. 2022, 16, 1743–1766. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Tang, Q.; Miao, X.; Wu, Y.; Qian, J.; Zhao, W.; Wang, L.; Li, L.; Zhang, D. ErbB3 binding protein 1 (EBP1) participates in the regulation of intestinal inflammation via mediating Akt signaling pathway. Mol. Immunol. 2015, 67, 540–551. [Google Scholar] [CrossRef]
- Cao, Q.; Li, Y.; Li, Y.; Li, L. miR-151-5p alleviates corneal allograft rejection by activating PI3K/AKT signaling pathway and balancing Th17/Treg after corneal transplantation via targeting IL-2Rɑ. Ann. Transl. Med. 2021, 9, 1410. [Google Scholar] [CrossRef]
- Zhang, W.; Zhou, Q.; Wei, Y.; Da, M.; Zhang, C.; Zhong, J.; Liu, J.; Shen, J. The exosome-mediated PI3k/Akt/mTOR signaling pathway in cervical cancer. Int. J. Clin. Exp. Pathol. 2019, 12, 2474–2484. [Google Scholar]
- Yin, N.; Wang, Y.; Lu, X.; Liu, R.; Zhang, L.; Zhao, W.; Yuan, W.; Luo, Q.; Wu, H.; Luan, X.; et al. hPMSC transplantation restoring ovarian function in premature ovarian failure mice is associated with change of Th17/Tc17 and Th17/Treg cell ratios through the PI3K/Akt signal pathway. Stem Cell Res. Ther. 2018, 9, 37. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, J.; Zhou, G. The mTOR-glycolytic pathway promotes T-cell immunobiology in oral lichen planus. Immunobiology 2020, 225, 151933. [Google Scholar] [CrossRef]
- Zhang, Y.; Jing, Y.; Qiao, J.; Luan, B.; Wang, X.; Wang, L.; Song, Z. Activation of the mTOR signaling pathway is required for asthma onset. Sci. Rep. 2017, 7, 4532. [Google Scholar] [CrossRef]
- Wan, C.F.; Zheng, L.L.; Liu, Y.; Yu, X. Houttuynia cordata Thunb reverses oxaliplatin-induced neuropathic pain in rat by regulating Th17/Treg balance. Am. J. Transl. Res. 2016, 8, 1609–1614. [Google Scholar] [PubMed]
- Gao, L.; Dong, Y.; Lin, R.; Meng, Y.; Wu, F.; Jia, L. The imbalance of Treg/Th17 cells induced by perinatal bisphenol A exposure is associated with activation of the PI3K/Akt/mTOR signaling pathway in male offspring mice. Food Chem. Toxicol. 2020, 137, 111177. [Google Scholar] [CrossRef] [PubMed]
- Feng, F.; Du, J.; Meng, Y.; Guo, F.; Feng, C. Louqin Zhisou Decoction Inhibits Mucus Hypersecretion for Acute Exacerbation of Chronic Obstructive Pulmonary Disease Rats by Suppressing EGFR-PI3K-AKT Signaling Pathway and Restoring Th17/Treg Balance. Evid. Based Complement. Altern. Med. 2019, 2019, 6471815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Dai, Q.; Jin, X.; Liang, D.; Li, X.; Lu, H.; Liu, Y.; Ding, J.; Gao, Q.; Wen, Y. Phosphoinositide 3-kinase/protein kinase B inhibition restores regulatory T cell’s function in pulmonary sarcoidosis. J. Cell Physiol. 2019, 234, 19911–19920. [Google Scholar] [CrossRef]
- Ma, B.; Athari, S.S.; Mehrabi Nasab, E.; Zhao, L. PI3K/AKT/mTOR and TLR4/MyD88/NF-κB Signaling Inhibitors Attenuate Pathological Mechanisms of Allergic Asthma. Inflammation 2021, 44, 1895–1907. [Google Scholar] [CrossRef]
- Feng, X.; Jiang, S.; Leung, W.; Wang, L.; Gober, H.J.; Chen, L.; Zhang, Y.; Wang, L. BuShen HuoXue Decoction Promotes Decidual Stromal Cell Proliferation via the PI3K/AKT Pathway in Unexplained Recurrent Spontaneous Abortion. Evid. Based Complement. Altern. Med. 2020, 2020, 6868470. [Google Scholar] [CrossRef]
- Shen, P.; Jiao, Y.; Miao, L.; Chen, J.H.; Momtazi-Borojeni, A.A. Immunomodulatory effects of berberine on the inflamed joint reveal new therapeutic targets for rheumatoid arthritis management. J. Cell Mol. Med. 2020, 24, 12234–12245. [Google Scholar] [CrossRef]
- Sun, X.; Zheng, X.; Zhang, X.; Zhang, Y.; Luo, G. Exosomal microRNA-23b-3p from bone marrow mesenchymal stem cells maintains T helper/Treg balance by downregulating the PI3k/Akt/NF-κB signaling pathway in intracranial aneurysm. Brain Res. Bull. 2020, 165, 305–315. [Google Scholar] [CrossRef]
- Van Herck, M.A.; Vonghia, L.; Kwanten, W.J.; Julé, Y.; Vanwolleghem, T.; Ebo, D.G.; Michielsen, P.P.; De Man, J.G.; Gama, L.; De Winter, B.Y.; et al. Diet Reversal and Immune Modulation Show Key Role for Liver and Adipose Tissue T Cells in Murine Nonalcoholic Steatohepatitis. Cell Mol. Gastroenterol. Hepatol. 2020, 10, 467–490. [Google Scholar] [CrossRef]
- Abildgaard, A.; Elfving, B.; Hokland, M.; Lund, S.; Wegener, G. Probiotic treatment protects against the pro-depressant-like effect of high-fat diet in Flinders Sensitive Line rats. Brain Behav. Immun. 2017, 65, 33–42. [Google Scholar] [CrossRef]
- Ferguson, D.; Finck, B.N. Emerging therapeutic approaches for the treatment of NAFLD and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2021, 17, 484–495. [Google Scholar] [CrossRef]
- Wu, Q.L.; Zeng, S.X.; Peng, J.Y.; Yuan, Y.; Zhu, Z.; Xie, Z.C.; Huang, Z.H.; Huang, J.S.; Lai, J.M.; Chen, J.A.; et al. Advances in metformin for the treatment of non-alcoholic fatty liver disease in children. Expert Rev. Gastroenterol. Hepatol. 2022, 16, 863–877. [Google Scholar] [CrossRef] [PubMed]
- Son, H.J.; Lee, J.; Lee, S.Y.; Kim, E.K.; Park, M.J.; Kim, K.W.; Park, S.H.; Cho, M.L. Metformin attenuates experimental autoimmune arthritis through reciprocal regulation of Th17/Treg balance and osteoclastogenesis. Mediat. Inflamm. 2014, 2014, 973986. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.J.; Kim, J.M.; Kim, I.K.; Ko, S.H.; Kim, J.S. Anti-inflammatory mechanism of metformin and its effects in intestinal inflammation and colitis-associated colon cancer. J. Gastroenterol. Hepatol. 2014, 29, 502–510. [Google Scholar] [CrossRef]
- Park, Y.H.; Jang, Y.J.; Choi, Y.; Lee, K.; Kim, H.J.; Cho, O.; Lee, H.R.; Heo, T.H. Combination of LMT-28 and Metformin Improves Beneficial Anti-Inflammatory Effect in Collagen-Induced Arthritis. Pharmacology 2021, 106, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Di Giacomo, M.; Zara, V. Antioxidant dietary approach in treatment of fatty liver: New insights and updates. World J. Gastroenterol. 2017, 23, 4146–4157. [Google Scholar] [CrossRef]
- Dinesh, P.; Rasool, M. Berberine mitigates IL-21/IL-21R mediated autophagic influx in fibroblast-like synoviocytes and regulates Th17/Treg imbalance in rheumatoid arthritis. Apoptosis 2019, 24, 644–661. [Google Scholar] [CrossRef]
- Gong, Y.; Lu, Q.; Liu, Y.; Xi, L.; Zhang, Z.; Liu, H.; Jin, J.; Yang, Y.; Zhu, X.; Xie, S.; et al. Dietary berberine alleviates high carbohydrate diet-induced intestinal damages and improves lipid metabolism in largemouth bass (Micropterus salmoides). Front. Nutr. 2022, 9, 1010859. [Google Scholar] [CrossRef]
- Li, Q.P.; Dou, Y.X.; Huang, Z.W.; Chen, H.B.; Li, Y.C.; Chen, J.N.; Liu, Y.H.; Huang, X.Q.; Zeng, H.F.; Yang, X.B.; et al. Therapeutic effect of oxyberberine on obese non-alcoholic fatty liver disease rats. Phytomedicine 2021, 85, 153550. [Google Scholar] [CrossRef]
- Xiao, Q.P.; Zhong, Y.B.; Kang, Z.P.; Huang, J.Q.; Fang, W.Y.; Wei, S.Y.; Long, J.; Li, S.S.; Zhao, H.M.; Liu, D.Y. Curcumin regulates the homeostasis of Th17/Treg and improves the composition of gut microbiota in type 2 diabetic mice with colitis. Phytother. Res. 2022, 36, 1708–1723. [Google Scholar] [CrossRef]
- Ngu, M.H.; Norhayati, M.N.; Rosnani, Z.; Zulkifli, M.M. Curcumin as adjuvant treatment in patients with non-alcoholic fatty liver (NAFLD) disease: A systematic review and meta-analysis. Complement. Ther. Med. 2022, 68, 102843. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.W.; Kong, Z.L.; Tsai, M.L.; Lo, C.Y.; Ho, C.T.; Lai, C.S. Tetrahydrocurcumin ameliorates free fatty acid-induced hepatic steatosis and improves insulin resistance in HepG2 cells. J. Food Drug Anal. 2018, 26, 1075–1085. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Liu, Y.; Wu, Y.; Chen, Y. Dihydrocurcumin ameliorates the lipid accumulation, oxidative stress and insulin resistance in oleic acid-induced L02 and HepG2 cells. Biomed. Pharmacother. 2018, 103, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Zhang, W.; Wu, Z.; Tian, X.; Xiang, J.; Li, L.; Li, Z.; Peng, X.; Wei, S.; Ma, X.; et al. Baicalin and the liver-gut system: Pharmacological bases explaining its therapeutic effects. Pharmacol. Res. 2021, 165, 105444. [Google Scholar] [CrossRef] [PubMed]
- Van Laar, A.; Grootaert, C.; Van Nieuwerburgh, F.; Deforce, D.; Desmet, T.; Beerens, K.; Van Camp, J. Metabolism and Health Effects of Rare Sugars in a CACO-2/HepG2 Coculture Model. Nutrients 2022, 14, 611. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.H.; Kwon, Y.; Huh, Y.E.; Choi, H.J. Trehalose ameliorates prodromal non-motor deficits and aberrant protein accumulation in a rotenone-induced mouse model of Parkinson’s disease. Arch. Pharm. Res. 2022, 45, 417–432. [Google Scholar] [CrossRef]
- Wieland, A.; Frank, D.N.; Harnke, B.; Bambha, K. Systematic review: Microbial dysbiosis and nonalcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2015, 42, 1051–1063. [Google Scholar] [CrossRef]
- Yao, C.; Lan, D.; Li, X.; Wang, Y.; Qi, S.; Liu, Y. Porphyromonas gingivalis is a risk factor for the development of nonalcoholic fatty liver disease via ferroptosis. Microbes Infect. 2022. Online ahead of print. [Google Scholar] [CrossRef]
- He, S.; Cui, S.; Song, W.; Jiang, Y.; Chen, H.; Liao, D.; Lu, X.; Li, J.; Chen, X.; Peng, L. Interleukin-17 Weakens the NAFLD/NASH Process by Facilitating Intestinal Barrier Restoration Depending on the Gut Microbiota. mBio 2022, 13, e0368821. [Google Scholar] [CrossRef]
- Feng, Q.; Liu, W.; Baker, S.S.; Li, H.; Chen, C.; Liu, Q.; Tang, S.; Guan, L.; Tsompana, M.; Kozielski, R.; et al. Multi-targeting therapeutic mechanisms of the Chinese herbal medicine QHD in the treatment of non-alcoholic fatty liver disease. Oncotarget 2017, 8, 27820–27838. [Google Scholar] [CrossRef]
- Riaz, F.; Wei, P.; Pan, F. Fine-tuning of regulatory T cells is indispensable for the metabolic steatosis-related hepatocellular carcinoma: A review. Front. Cell Dev. Biol. 2022, 10, 949603. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; DeBosch, B.J. Microbial and metabolic impacts of trehalose and trehalose analogues. Gut Microbes 2020, 11, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Yang, L.; Chu, H. The Gut Microbiota: A Novel Player in Autoimmune Hepatitis. Front. Cell Infect. Microbiol. 2022, 12, 947382. [Google Scholar] [CrossRef]
- Chen, P.; Hei, M.; Kong, L.; Liu, Y.; Yang, Y.; Mu, H.; Zhang, X.; Zhao, S.; Duan, J. One water-soluble polysaccharide from Ginkgo biloba leaves with antidepressant activities via modulation of the gut microbiome. Food Funct. 2019, 10, 8161–8171. [Google Scholar] [CrossRef] [PubMed]
- Estrada, J.A.; Contreras, I. Nutritional Modulation of Immune and Central Nervous System Homeostasis: The Role of Diet in Development of Neuroinflammation and Neurological Disease. Nutrients 2019, 11, 1076. [Google Scholar] [CrossRef] [PubMed]
- Mandrioli, J.; Amedei, A.; Cammarota, G.; Niccolai, E.; Zucchi, E.; D’Amico, R.; Ricci, F.; Quaranta, G.; Spanu, T.; Masucci, L. FETR-ALS Study Protocol: A Randomized Clinical Trial of Fecal Microbiota Transplantation in Amyotrophic Lateral Sclerosis. Front. Neurol. 2019, 10, 1021. [Google Scholar] [CrossRef] [PubMed]
- Perumpail, B.J.; Khan, M.A.; Yoo, E.R.; Cholankeril, G.; Kim, D.; Ahmed, A. Clinical epidemiology and disease burden of nonalcoholic fatty liver disease. World J. Gastroenterol. 2017, 23, 8263–8276. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Yan, K.; Wang, Z.; Zhang, Q.; Gao, L.; Xu, T.; Sai, J.; Cheng, F.; Du, Y. The association between hypertension and nonalcoholic fatty liver disease (NAFLD): Literature evidence and systems biology analysis. Bioengineered 2021, 12, 2187–2202. [Google Scholar] [CrossRef]
- Tomeno, W.; Imajo, K.; Takayanagi, T.; Ebisawa, Y.; Seita, K.; Takimoto, T.; Honda, K.; Kobayashi, T.; Nogami, A.; Kato, T.; et al. Complications of Non-Alcoholic Fatty Liver Disease in Extrahepatic Organs. Diagnostics 2020, 10, 912. [Google Scholar] [CrossRef]
- Shao, Q.; Wu, Y.; Ji, J.; Xu, T.; Yu, Q.; Ma, C.; Liao, X.; Cheng, F.; Wang, X. Interaction Mechanisms Between Major Depressive Disorder and Non-alcoholic Fatty Liver Disease. Front. Psychiatry 2021, 12, 711835. [Google Scholar] [CrossRef]
- Kim, D.; Yoo, E.R.; Li, A.A.; Tighe, S.P.; Cholankeril, G.; Harrison, S.A.; Ahmed, A. Depression is associated with non-alcoholic fatty liver disease among adults in the United States. Aliment. Pharmacol. Ther. 2019, 50, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Yang, F.; Ma, B.; Jing, W.; Liu, J.; Guo, M.; Li, J.; Wang, Z.; Liu, M. Prevalence of nonalcoholic fatty liver disease in mental disorder inpatients in China: An observational study. Hepatol. Int. 2021, 15, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Filipović, B.; Marković, O.; Đurić, V.; Filipović, B. Cognitive Changes and Brain Volume Reduction in Patients with Nonalcoholic Fatty Liver Disease. Can. J. Gastroenterol. Hepatol. 2018, 2018, 9638797. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, M.; Siddique, K.; Baum, M.; Smith, S.A. Prenatal programming of hypertension induces sympathetic overactivity in response to physical stress. Hypertension 2013, 61, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Macavei, B.; Baban, A.; Dumitrascu, D.L. Psychological factors associated with NAFLD/NASH: A systematic review. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 5081–5097. [Google Scholar] [PubMed]
- Tomeno, W.; Kawashima, K.; Yoneda, M.; Saito, S.; Ogawa, Y.; Honda, Y.; Kessoku, T.; Imajo, K.; Mawatari, H.; Fujita, K.; et al. Non-alcoholic fatty liver disease comorbid with major depressive disorder: The pathological features and poor therapeutic efficacy. J. Gastroenterol. Hepatol. 2015, 30, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Kara, N.Z.; Toker, L.; Agam, G.; Anderson, G.W.; Belmaker, R.H.; Einat, H. Trehalose induced antidepressant-like effects and autophagy enhancement in mice. Psychopharmacology 2013, 229, 367–375. [Google Scholar] [CrossRef]
- Stachowicz, A.; Wiśniewska, A.; Kuś, K.; Kiepura, A.; Gębska, A.; Gajda, M.; Białas, M.; Totoń-Żurańska, J.; Stachyra, K.; Suski, M.; et al. The Influence of Trehalose on Atherosclerosis and Hepatic Steatosis in Apolipoprotein E Knockout Mice. Int. J. Mol. Sci. 2019, 20, 1552. [Google Scholar] [CrossRef]
- Ngo, S.T.; Steyn, F.J. The interplay between metabolic homeostasis and neurodegeneration: Insights into the neurometabolic nature of amyotrophic lateral sclerosis. Cell Regen. 2015, 4, 5. [Google Scholar] [CrossRef]
- Alugoju, P.; Krishna; Swamy, V.K.D.; Anthikapalli, N.V.A.; Tencomnao, T. Health benefits of astaxanthin against age-related diseases of multiple organs: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2022. Online ahead of print. [Google Scholar] [CrossRef]
- Li, Y.; Guo, Y.; Wang, X.; Yu, X.; Duan, W.; Hong, K.; Wang, J.; Han, H.; Li, C. Trehalose decreases mutant SOD1 expression and alleviates motor deficiency in early but not end-stage amyotrophic lateral sclerosis in a SOD1-G93A mouse model. Neuroscience 2015, 298, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.P.; Hou, P.H.; Mao, F.C.; Chang, C.C.; Yang, W.C.; Wu, C.F.; Liao, H.J.; Lin, T.C.; Chou, L.S.; Hsiao, L.W.; et al. Risperidone Exacerbates Glucose Intolerance, Nonalcoholic Fatty Liver Disease, and Renal Impairment in Obese Mice. Int. J. Mol. Sci. 2021, 22, 409. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Xiong, Q.; Li, Y.; Yao, C.; Wu, R.; Wang, Q.; Luo, L.; Liu, H.; Feng, P. Traditional Chinese Medicine Regulates Th17/Treg Balance in Treating Inflammatory Bowel Disease. Evid. Based Complement. Altern. Med. 2022, 2022, 6275136. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Bonner, J.A.; Shi, L.Z. Metabolic checkpoints in neurodegenerative T helper 17 (TH17) and neuroregenerative regulatory T (Treg) cells as new therapeutic targets for multiple sclerosis. Neural Regen. Res. 2020, 15, 267–269. [Google Scholar] [PubMed]
- Li, Z.; Sun, Q.; Liu, Q.; Mu, X.; Wang, H.; Zhang, H.; Qin, F.; Wang, Q.; Nie, D.; Liu, A.; et al. Compound 511 ameliorates MRSA-induced lung injury by attenuating morphine-induced immunosuppression in mice via PI3K/AKT/mTOR pathway. Phytomedicine 2022, 108, 154475. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, Y.; Murakami, M.; Nakagawa, Y.; Tsuji, A.; Kitagishi, Y.; Matsuda, S. Diet induces hepatocyte protection in fatty liver disease via modulation of PTEN signaling. Biomed. Rep. 2020, 12, 295–302. [Google Scholar] [CrossRef]
- Hadjihambi, A.; Konstantinou, C.; Klohs, J.; Monsorno, K.; Le Guennec, A.; Donnelly, C.; Cox, I.J.; Kusumbe, A.; Hosford, P.S.; Soffientini, U.; et al. Partial MCT1 invalidation protects against diet-induced non-alcoholic fatty liver disease and the associated brain dysfunction. J. Hepatol. 2022, in press. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshikawa, S.; Taniguchi, K.; Sawamura, H.; Ikeda, Y.; Asai, T.; Tsuji, A.; Matsuda, S. Metabolic Associated Fatty Liver Disease as a Risk Factor for the Development of Central Nervous System Disorders. Livers 2023, 3, 21-32. https://doi.org/10.3390/livers3010002
Yoshikawa S, Taniguchi K, Sawamura H, Ikeda Y, Asai T, Tsuji A, Matsuda S. Metabolic Associated Fatty Liver Disease as a Risk Factor for the Development of Central Nervous System Disorders. Livers. 2023; 3(1):21-32. https://doi.org/10.3390/livers3010002
Chicago/Turabian StyleYoshikawa, Sayuri, Kurumi Taniguchi, Haruka Sawamura, Yuka Ikeda, Tomoko Asai, Ai Tsuji, and Satoru Matsuda. 2023. "Metabolic Associated Fatty Liver Disease as a Risk Factor for the Development of Central Nervous System Disorders" Livers 3, no. 1: 21-32. https://doi.org/10.3390/livers3010002
APA StyleYoshikawa, S., Taniguchi, K., Sawamura, H., Ikeda, Y., Asai, T., Tsuji, A., & Matsuda, S. (2023). Metabolic Associated Fatty Liver Disease as a Risk Factor for the Development of Central Nervous System Disorders. Livers, 3(1), 21-32. https://doi.org/10.3390/livers3010002




