Abstract
Cancer is one of the major causes of death worldwide. Chemotherapeutic drugs have become a popular choice as anticancer agents. Despite the therapeutic benefits of chemotherapeutic drugs, patients often experience side effects and drug resistance. Biopolymers could be used to overcome some of the limitations of chemotherapeutic drugs, as well as be used either as anticancer agents or drug delivery vehicles. Chitosan is a biocompatible polymer derived from chitin. Chitosan, chitosan derivatives, or chitosan nanoparticles have shown their promise as an anticancer agent. Additionally, functionally modified chitosan can be used to deliver nucleic acids, chemotherapeutic drugs, and anticancer agents. More importantly, chitosan-based drug delivery systems improved the efficacy, potency, cytotoxicity, or biocompatibility of these anticancer agents. In this review, we will investigate the properties of chitosan and chemically tuned chitosan derivatives, and their application in cancer therapy.
1. Introduction
Cancer is one of the leading causes of death, and arises from abnormal proliferation of cells which later spreads to different parts of the human body [1]. In 2020, 19.3 million people were diagnosed with cancer and about 10 million cancer-related deaths occurred worldwide [2]. To reduce tumor burden and increase survival time, many cancer patients have to rely on chemotherapeutic drugs [3]. Though chemotherapeutic drugs improve the outcome of cancer therapy, some patients experience drug-induced side effects [4]. More importantly, the requirement of high-dose, poor bioavailability, low selectivity index, development of drug resistance, and non-specific interactions are some of the major drawbacks of chemotherapeutic drugs [5,6]. Therefore, the development of a suitable drug delivery system is urgently needed to reduce the therapeutic dose or frequency, and thereby minimize the toxic effects of the anticancer drugs.
Biopolymers are largely used as carriers in the delivery of active pharmaceutical ingredients. They play a key role in the development of a range of delivery systems, including hydrogels, micelles, tablets, capsules, and particulate systems (nanoparticles, beads, and micro) [7]. As a carrier, polymers have to be non-toxic, biodegradable, and biocompatible, the two latter properties help remove the carrier after administration of the drug [8,9]. Cellulose, chitosan, alginate, gellan gum, pectin, gum arabica, guar gum, starch, gelatin, chondroitin sulfate, and hyaluronic acid are some of the biopolymers used in drug delivery systems [10]. Among these, chitosan is one of the most widely used polymers in the pharmaceutical industry [11].
Chitosan is a biocompatible and biodegradable polymer derived from chitin [12]. Chitosan shows a range of biochemical activities such as antiproliferative and antimicrobial activity, immune activation, lowering cholesterol level, eliciting production of phytoalexin, antihypertensive action(s), and neuroprotective, wound-healing, and antiulcer activities [13,14]. Due to its biocompatibility, chitosan is often used as a delivery vehicle for nucleic acids, chemotherapeutic drugs, or anticancer compounds [15,16]. Furthermore, chemically tuned or chemically modified chitosan derivatives can be used as antiproliferative agents, either alone or in combination with other anticancer agents [15]. For example, in many cancer cells, alteration of glycosylation occurs, which can be targeted using chitosan-based glycoconjugates where carbohydrate incorporated into the pharmacophore offers higher penetration of anticancer agents [15,17].
In this review, we have investigated the anticancer activities of chitosan or chitosan derivatives, their application in the delivery of gene, chemotherapeutic drugs, or anticancer compounds, and their efficacy as cancer therapeutic agents. Additionally, we scrutinized the promise of chitosan or chitosan derivatives in cancer immunotherapy.
2. Properties of Chitosan and Its Derivatives
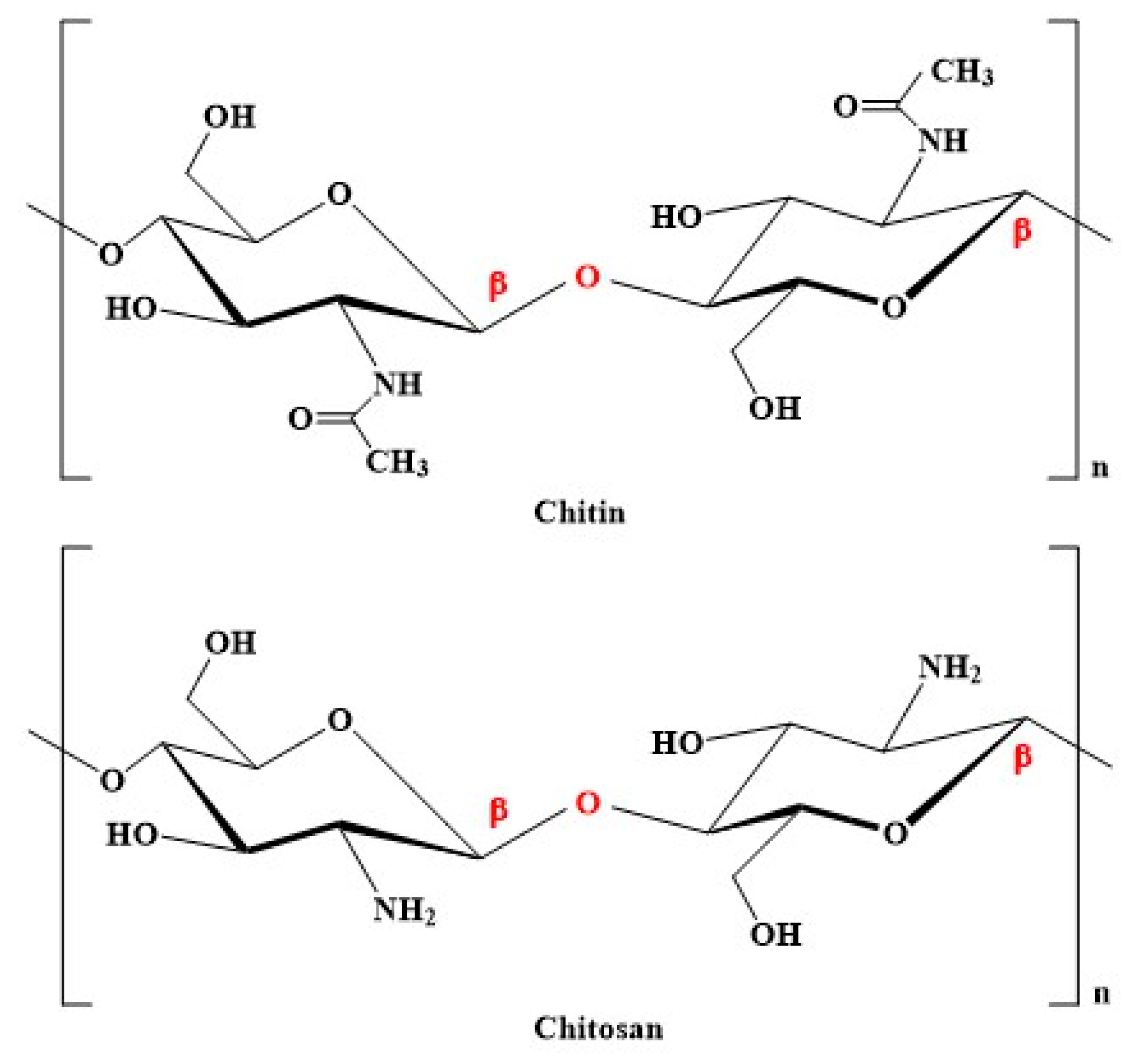
Chitosan is a polycationic linear polysaccharide comprising N-acetyl-glucosamine and D-glucosamine monosaccharides connected via β-(1→4) glycosidic linkages (Figure 1) [12]. It is a polysaccharide extracted from chitin. Under strict alkaline conditions, this chitin is partially deacetylated into chitosan [18]. Insects, crustaceans, squid, algae, and fungi are the primary source of chitosan [12]. Chitosan can be grouped into three categories based on molecular weight: (1) low molecular weight chitosan, (2) medium molecular weight chitosan, and (3) high molecular weight chitosan. High molecular weight chitosan contains a long chain and more hydroxyl groups [19].
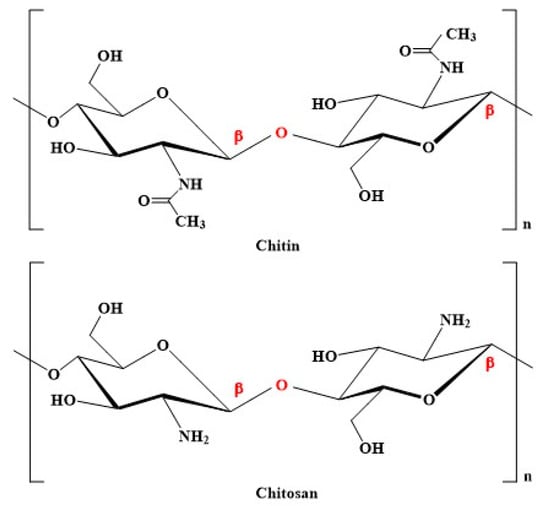
Figure 1.
Structure of chitin and chitosan. Chitosan can be prepared upon deacetylation of chitin. The degree of deacetylation can be represented as the molar fraction of GlcN (×) and GlcNAc (100-×) units. When the degree of chitin deacetylation is more than 50%, it is called chitosan. N-acetylated glucosamine: GlcNAc; 2-amino-2-d-glucose: GlcN.
The degree of deacetylation (DD) is the ratio of the relative number of D-glucosamine units to the cumulative number of N-acetyl-D-glucosamine and D-glucosamine units [20]. When the DD of chitin is more than 50%, it is called chitosan. Generally, the DD of chitosan is about 60–98% [20,21]. While the degree of substitution (DS) represents the percentage of newly added groups into the chitosan, the DS is linked with the pH, solubility, and biological activities of chitosan derivatives [22].
Chitosan is a weak base, insoluble in water and organic solvent. However, chitosan is soluble in an aqueous solvent under acidic conditions [23]. Poor dissolution of chitosan due to the highly crystalline hydrogen linkages, and reduced solubility in water or physiological condition restrict the use of chitosan in drug delivery [24,25]. Chitosan is often chemically modified to overcome its limitations as a drug delivery vehicle. O-substitution (e.g., carboxymethylation) and N-substitution of chitosan (e.g., reductive amination) can be used to create new covalent linkages and such modification may improve the property of chitosan [26,27]. Furthermore, hydrogel formation forms covalent linkages with different crosslinkers to improve the drug distribution property of chitosan [25,28].
3. Anticancer Activities of Chitosan and Its Derivatives
Chitosan or chitosan derivatives including chitosan-thymine conjugate (CTC), carboxymethyl chitosan (CMC), sulfated chitosan (SCS), sulfated benzaldehyde chitosan (SBCS), and polypyrrole-chitosan (PPC) displayed promising anticancer activity against many cancer cells [13,15]. The anticancer activities of chitosan derivatives can be further modified by combination with nanocomposite or other chemical agents. For example, conjugation of 3-amino-2-phenyl-4(3H)-quinazolinone on PPC-silver chloride nanocomposite enhanced bioavailability of chitosan to tumor cells. This formulation sequestered molecules from noncancerous cells and exhibited sustained release of chitosan to cancer cells [29,30].
Chitosan or its derivatives selectively penetrate tumor cells and exhibit antiproliferative activities via antiangiogenic, immunoenhancing, antioxidant defense, apoptosis, and enzymatic regulation [15]. Low molecular weight water-soluble chitosans (21 and 46 kDa) exhibited antiproliferative activity towards sarcoma 180-tumor bearing mice, while high molecular weight water-soluble chitosan of 130 kDa had no antitumor activity. The 21 and 46 kDa chitosans activated macrophages via cytokine (e.g., IFN-γ, IL-12, and IL-18) production from the intestinal intraepithelial lymphocytes [31]. Low molecular weight chitosan arrests cancer cells at the G1/S phase and induces apoptosis [32]. Furthermore, chitosan regulates cell cycle-related genes (e.g., Cdc25A, p21/Cip, and p27/Kip), upregulates transcriptional growth factor β (TGF-β), and induces apoptosis by controlling nuclear factor k-light-chain enhancer of activated B cells (NF-kB) mediated signaling pathways [32,33]. CTC exhibited antiproliferative activity towards HepG2 liver carcinoma cells, while it had no cytotoxic activity towards NIH3T3 normal mouse fibroblast cells, indicating selective targeting of cancer cells [34]. Similarly, PPC displayed anticancer activity towards Ehrlich ascites carcinoma (EAC) cells and MCF-7 breast cancer cells [35]. Furthermore, CTC and PPC showed antimicrobial activity, therefore could be used to prevent secondary infections in cancer patients [34,35,36]. While CMC increased the immune response by upregulating interferon γ (IFN-γ) and tumor necrosis factor α (TNF-α) level and reduced angiogenesis by decreasing vascular endothelial growth factor (VEGF) expression [37], it could be used as a transporter of chemotherapeutic drugs (e.g., doxorubicin, and 5-fluorouracil) or anticancer agents (e.g., curcumin) [5]. Likewise, SCS and SBCS exhibited anticancer activity towards MCF-7 cells by triggering apoptosis and blocking FGF-2-triggered activation of extracellular signal-regulated kinases [38]. Among these two derivatives (SCS and SBCS), SBCS was slightly more potent than SCS in terms of IC50 value (Table 1). Additionally, the apoptotic activity of SCS and SBCS was significantly greater than chitosan. They significantly increased apoptosis (more than 46%) in MCF-7 cells compared to chitosan at 40 μM [38]. Moreover, chitosan oligosaccharide (COS) exhibits anticancer activity towards SW480 (colorectal cancer cells), Hep3B (hepatocellular carcinoma cells), and HeLa (human cervical cancer cells) regardless of their positive or negative charge. Though quaternized amino chitooligosaccharides (QCOS) and sulfated chitooligosaccharides (SCOS) were equally potent against HeLa cells, negatively charged SCOS was more potent than positively charged QCOS towards SW480 cells in terms of IC50 value. DNA fragmentation and fluorescence microscopic studies showed that these charged COS derivatives induced necrosis rather than apoptosis [39]. Similar to COS, hexamers of COS (chitohexose, chitopentaose, chitobiose, and chitotriose) form displays antiproliferative activity. Among the five hexamer derivatives of COS, chitohexose was more potent towards A549 lung cancer cells. Chitohexose downregulated cell cycle linked cyclin D1 and pro-survival protein bcl-xl mRNA expression, thereby induced apoptosis [40].

Table 1.
Anticancer activity of chitosan and chitosan derivatives.
4. Ligand Decorated Chitosan for Targeted Drug Delivery
Ligand-targeted drug delivery helps to elicit distinct therapeutic effects, maximize therapeutic response, and minimize the toxicity of drugs [42,43]. Ligands attach to clustered receptors which are overexpressed on the cancer cell surface. Chitosan-based ligand conjugated nanoformulations can deliver drugs reliably to the target site, which eliminates unwanted effects of non-targeted drug delivery [43]. For example, Chen et al. [44] combined trimethyl chitosan with ligand CSKSSDYQC to treat breast cancer via oral administration. The incorporation of ligand CSKSSDYQC increased bioavailability due to its selective affinity towards intestinal goblet cells and enhanced intestinal uptake of the chitosan-based drug candidate [44].
Epidermal growth factor receptor (EGFR) is overexpressed in A549 cells [45]. Nascimento et al. [46] constructed EGFR-targeted chitosan-based carrier by conjugating EGFR, chitosan, and polyethylene glycol (PEG) linker loaded with small interfering RNA and Mad2 gene. Such chitosan-based carrier was selectively internalized by A549 cells and silenced Mad2 gene expression which caused premature anaphase leading to the death of A549 cells [46]. In addition, overexpression of estrogen receptor (ER) is observed in breast cancer cells [47]. Therefore, estrone ligand-modified glycol chitosan nanoparticles were prepared by Yin et al. [48] for targeted drug delivery in breast cancer cells. Qindeel et al. [49] reported that the folate–chitosan complex was rapidly uptaken into lysosome and mitochondria of MCF-7 cells through folate receptor. Moreover, Jin et al. [43] synthesized folic acid (FA), chitosan, and ursolic acid conjugated nanocarrier, which was rapidly internalized by MCF-7 cells through a folate receptor-regulated endocytic pathway. Similarly, a chitosan-derived nanocomplex, CN@CFTC, was fabricated by conjugating chitosan, FA, and fluorescein isothiocyanate (CFTC) which was electrostatically assembled to cellulose nanocrystals. The resulting CN@CFTC contains targeting ligand FA that facilitates the internalization of CN@CFTC into tumor cells expressing FR-receptor (i.e., MDA-MB-231 human breast cancer cells) and the elevation of the chitosan derivative (CFTC) concentration increases the fluorescent intensity five-fold compared to control [50]. Moreover, cluster determinant 44 (CD44) is overexpressed in many tumor cells, and CD44 overexpressed cells can be targeted using chitosan-based complexes containing hyaluronic acid (HA) and chondroitin sulfate (CS). It should be noted that HA and CS show affinity towards CD44 [51].
5. Chitosan-Mediated Gene Therapy
Gene therapy primarily focuses to introduce exogenous genetic elements into the targeted cell which paves the way to control genomic expression [52]. However, the negatively charged gene is not capable of penetrating the cell membrane through passive diffusion [53]. Moreover, injecting DNA directly into cells causes rapid degradation by nuclease [54]. The outcome of gene therapy is linked with several parameters, including the targeting of selective cells, protecting nucleotides in the extracellular milieu from degradation, and delivering adequate amounts of nucleic acid to generate a therapeutic effect [55]. Therefore, both viral and non-viral vectors are employed to achieve effective gene transfer to specific cells [56]. While developing vectors for gene therapy, the biocompatibility, immunogenicity, and efficacy of the drug delivery system should be considered [57]. Although high transfection efficiency could be achieved via a viral vector, a non-viral vector is preferred due to producing minimum toxicity and immunogenicity [58]. Among the non-viral vectors, chitosan-derived polymers having cationic properties have emerged as promising vectors in gene delivery systems [57].
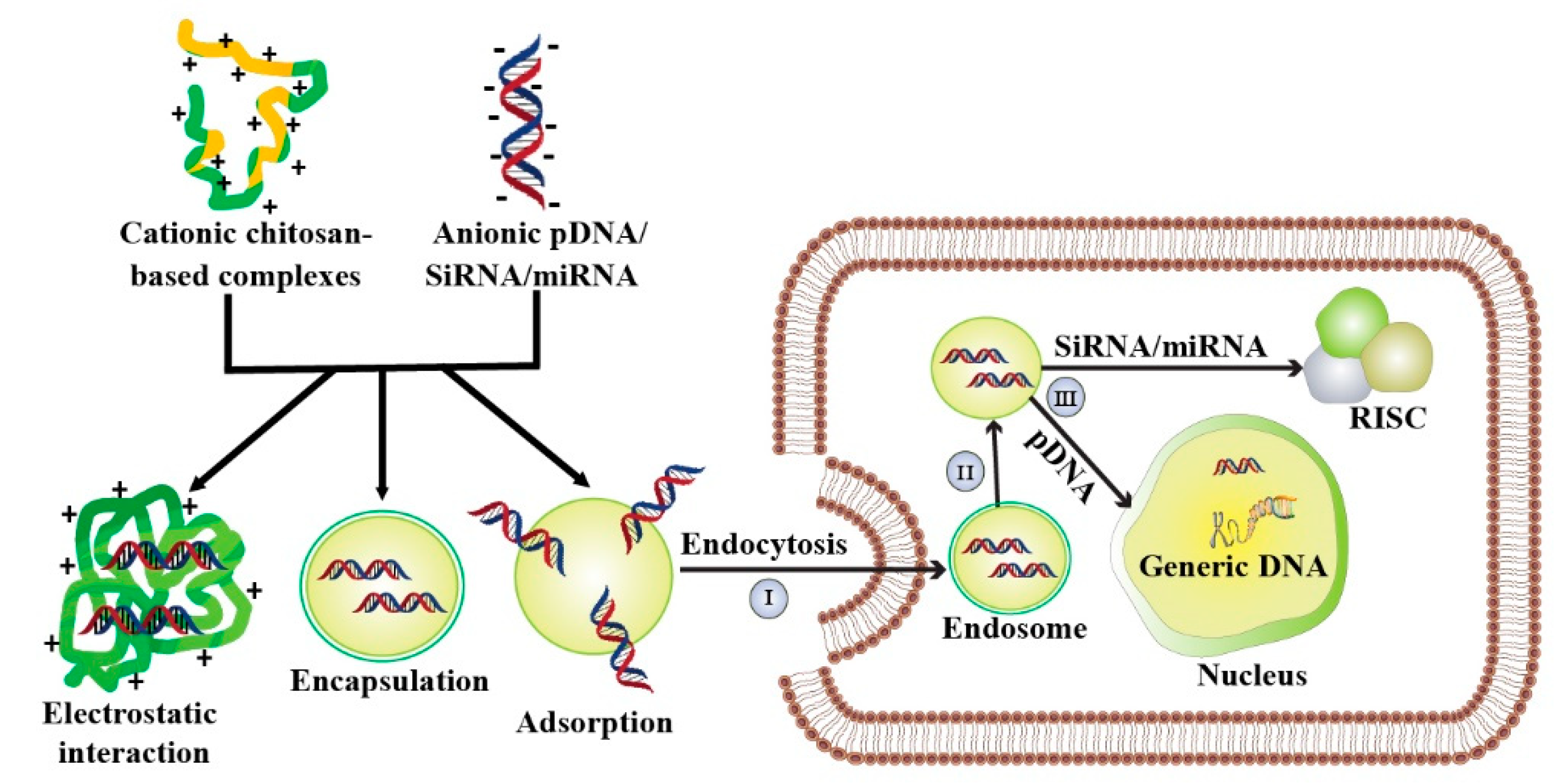
Three main strategies involved in the preparation of chitosan-based nucleic acids carriers are polyplex formation by simple complexation, encapsulation via ionic gelation method, and adsorption onto the performed systems (Figure 2). Fabrication of polyelectrolyte complexation or polyplexes is the simplest approach and can be achieved by mixing both chitosan and nucleic acids, followed by incubation [59]. Chitosan backbone contains protonated amino group which stimulate cationic chitosan to interact electrostatically with anionic nucleic acids to form polyplex. The resulting polyplex protects nucleic acids from enzymatic degradation [60]. An alternative approach of chitosan-nucleic acids formulation is ionic gelation where nucleic acids are encapsulated within the chitosan matrix. In this method, nucleic acids and chitosan interact through ionic crosslinker agents besides electrostatic interaction [61]. Such ionic crosslinker agents (i.e., tripolyphosphate (TPP), sodium sulfate, thiamine pyrophosphate, poly-γ-glutamic acid, dextran sulfate, and hyaluronic acid) are negatively charged and bind with chitosan [62,63]. The as-prepared chitosan-based nanostructures exhibited extended stability and entrapped nucleic acids for a longer time [64]. Another fabrication method of chitosan-based nucleic acids carrier is adsorption of nucleic acids onto the surface of chitosan modified poly(lactide-co-glycolide) (PLGA). Such formulation can load nucleic acids, protect them from nuclease degradation, and mediate the controlled release of nucleic acids [65].
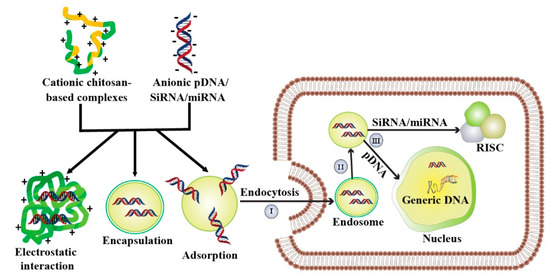
Figure 2.
Fabrication strategy and transfection mechanism of chitosan-based nucleic acids carrier. Cationic chitosan interacts with the anionic nucleic acids (i.e., pDNA, siRNA, and miRNA) to formulate complexes for gene delivery. Three main fabrication processes of chitosan-based complexes are (1) Polyplex formation, (2) Encapsulation via ionic gelation method, and (3) Adsorption. The chitosan-based vectors are taken up by endocytosis (I), followed by an escape from endosome (II). After releasing from endosome (III), siRNA and miRNA bind to the RNA-induced silencing complex (RISC) to trigger the RNA interfering (RNAi) pathway. The pDNA is translocated to the nucleus and subsequently performs desired anticancer activity.
Chitosan was first used in 1995 for plasmid transfection as a non-viral gene delivery agent [66]. Chitosan backbone contains primary amino group that undergoes protonation at marginally acidic pH and becomes positively charged which electrostatically interacts with nucleic acid [57]. Chitosan exerts fine bioavailability, low toxicity, and immunogenicity in the in vitro and in vivo approaches. Additionally, chitosan contains functional groups which can be chemically modified to facilitate endo-lysosomal escape and condensation of genetic material [67,68]. However, the disadvantages arise as the chitosan-based carriers are only soluble in acidic conditions. To improve the solubility of chitosan, its amino and hydroxyl groups are targeted for chemical modification [69,70]. Low molecular weight water-soluble O-carboxymethyl chitosan containing free amine group has already shown promise as gene carriers [71]. As O-carboxymethyl chitosan is anionic in nature, Nam et al. [71] conjugated it with cationic poly(ethyleneimine) to interact with negatively charged siRNA (Table 2). Another drawback of chitosan is its poor transfection potential [70]. Chemical modifications such as PEGylation [72], galactosylation [73], block copolymerization [74], histidine modification [75], and methylation [76] are often employed to increase the transfection efficiency of chitosan.
Liang et al. [77] developed novel cationic polymeric liposomes (CPLs), TQCMC-DOPE, by combining polymeric surfactants of quaternized (carboxymethyl)chitosan with different carbon chains (dodecyl, tetradecyl, hexadecyl, and octadecyl) (TQCMC) and L-α-phosphatidylethanolamine (DOPE). The resulting structure of TQCMC-DOPE CPLs was structurally similar to lipid bilayer, and the nanocomplex showed high thermal stability similar to cationic liposomes (Lipofectamine 2000). Interestingly, TQCMC-DOPE CPLs were two-fold less cytotoxic than Lipofectamine 2000 towards mouse fibroblast (L929), human ovarian carcinoma (HO-8910), and HepG2 cells. Different factors (i.e., NP size, NP components, surface charge, and microstructure) that affect gene transfection efficiency were analyzed, and TQCMC-DOPE CPLs, having apposite size (184.4 nm) and positive ζ potentials (27.5 mV), were the best to use against different cancer cell lines. In vitro study confirms that TQCMC-DOPE-pDNA CPLs exhibited 20–50% higher transfection efficiency than control against human embryonic kidney (293T), human gastric cancer (SGC-7901), prostate adenocarcinoma (PC-3), HO-8910, human primary glioblastoma (U87), and human hepatocellular carcinoma (SMMC-7721) cells. In vivo study in BALB/c mice revealed that TQCMC CPLs delivered reporter gene efficiently into U87 and SMMC-7721 tumors and caused less cell cytotoxicity, a reduction in cytokine production, and low hepatic tissue injury [77]. Covalent conjugation of folic acid (FA) to chitosan helps in targeted DNA delivery specifically to folate receptor overexpressing cancer cells [43]. Lee et al. [78] introduced FA to the primary amine group of chitosan to prepare folate-chitosan complex and found that FA was responsible for increasing gene transfer potentiality of chitosan on folate-receptor overexpressing MCF-7 cells. FA-conjugated chitosan augmented improved gene transfer than unmodified chitosan in folate receptor overexpressing cancer cells [78]. Similarly, Wang et al. [72] synthesized another type of multifunctional vector. The core enzyme complex (PHD) consisted of poly(amidoamine) (PAMAM), high mobility group box 1 (HMGB1), and pDNA. The PHD was then coated with FA modified polyethylene glycol tethered carboxylated chitosan (FA-PEG-CC). Conjugation of FA-PEG-CC and PHD results in a pH-sensitive FPCPHD complex which exhibited excellent in vitro and in vivo gene delivery efficacy. FPCPHDs showed resistance to digestion by DNase I and heparin replacement. Additionally, the encapsulation efficiency of FPCPHD increases (10–95%) with the increase in N/P ratio (0.5–30). Moreover, FPCPHDs exhibited efficient transfection in folate receptor (FR) positive cancer cells and displayed low toxicity against HepG2 and KB (epidermoid carcinoma) cells [72]. In the same way, chitosan-stabilized and gold-coated PLGA with FA nanoparticles (FA-Au-C-PLGA NPs) were prepared which exhibited excellent in vitro transgene activity, low cytotoxicity, fine cellular uptake in FR positive MCF-7 cells, and protection of pDNA from nuclease degradation [79]. In another study, a pH-sensitive nanocarrier was fabricated by combining chitosan and FA, and later conjugated with pDNA via the ionic gelation method. Encapsulation efficiency of the pDNA increased proportionally with the increase in the molecular weight of chitosan [53]. Furthermore, N, N-diethyl N-methyl chitosan (DEMC) was developed by Safari et al. [80] to deliver genes in human pancreatic cancer cells (AsPC-1). They used pDNA-EGFP to evaluate the gene delivery efficacy. DEMC condensed pDNA and effectively delivered pDNA into AsPC-1 cells. The transfection efficiency was enhanced by 16.7-fold with the increase in the charge ratio (5–40) [80]. Moreover, Wang et al. [81] cultured TC-2 cells in 3D chitosan-alginate (CA) porous scaffolds and obtained high pDNA delivery in vitro. The outstanding in vitro transfection efficiency predicts the in vivo gene delivering potentiality of CA [81].

Table 2.
Physicochemical properties and transfection report of some chitosan-based complexes.
Table 2.
Physicochemical properties and transfection report of some chitosan-based complexes.
| Chitosan Modified Complexes | Nucleic Acids | CytotoxiCity Assay | CytotoxiCity Status | Average Particle Size (nm) | Zeta Potential (mV) | InvestiGated Cell Lines | Transfection Investigated | Ref. |
|---|---|---|---|---|---|---|---|---|
| HPOCP | siRNA, pDNA, | MTT | No cytotoxicity | 100–~ 300 | − | HCT119 | in vitro | [71] |
| FPCPHDs | pDNA | MTT | <10% (HepG2), and 20% (KB) less cytotoxic than control | 51–305 | −0.9–+27.3 | HepG2, KB | in vitro and in vivo | [72] |
| TQCMC- DOPE | pDNA | MTT | 5–40% (L929), 0–70% (HO8910), and 0–60% (HepG2) more cytotoxic than Lipofectamine 2000 | 184.4 | 27.5 | 293T, SGC-7901, PC-3, HO-8910, HepG2, U87, SMMC-7721 | in vitro and in vivo | [77] |
| FA-Au-C-PLGA | pDNA | MTT | 0–10% less cytotoxic than control | 199.4 | 35.7 | HepG2, HEK293, MCF-7 | in vitro | [79] |
| DEMC | pDNA | MTT | 45–70% less cytotoxic than control | 114.24–570.4 | 6.14–16.45 | AsPC-1 | in vitro | [80] |
| C–miRNA | miRNA | MTT | Almost no cytotoxicity | ~80–190 | −20–+20 | MCF-7 | in vitro | [82] |
Abbreviations: O-carboxymethyl chitosan + poly(ethyleneimine) + HER-2/neu: HPOCP; Tetradecyl-quaternized (carboxymethy)chitosan: TQCMC; Chitosan+miRNA-145: C–miRNA; Chitosan + gold + poly(lactide-co-glycolide) + folic acid: FA-Au-C-PLGA; N,N-diethyl N-methyl chitosan: DEMC; and poly(amidoamine)+high mobility group box 1 + Pdna + polymer polyethylene glycol + folic acid: FPCPHD.
Nam et al. constructed a novel vector HPOCP by combining water-soluble O-carboxymethyl chitosan, branched-low molecular weight poly(ethyleneimine) (bPEI) and targeting ligand (epitope type, HER-2/neu). Low cytotoxicity, site-specific delivery, and excellent transfection efficiency made HPOCP copolymer an ideal candidate for gene therapy. HPOCP polyplexes conjugated with the green fluorescent protein encoded plasmid DNA (pDNA-EGFP) displayed high gene transfection efficiency in HCT119 (human colorectal cancer cells). Moreover, HPOCP efficiently bound with siRNA and siRNA/HPOCP 10% polyplexes did not exhibit cytotoxicity in HEK 293 (human embryonic kidney cells) [71]. However, other studies reported that chitosan-siRNA complexes showed 20–40% cytotoxicity in H1299 cells [83], whereas it was 30–40% in NIH3T3 cells [84]. The ability of chitosan-based formulations to deliver siRNA inspired Santos-Carballal et al. to fabricate chitosan–miRNA-145 (C–miRNA) and investigate in vitro transfection efficiency in MCF-7 cells. They reported that the ideal complex can be achieved by using chitosan with 40 kDa molecular weight and 12% degree of acetylation; a (+/−) charge ratio of 1.5. C–miRNA was able to deliver miRNA successfully in MCF-7 cells, and no cytotoxicity was reported at 0.05–0.5 nmol concentration. Furthermore, as miRNA-145 downregulates junction adhesion molecule A (JAM-A) mRNA, it was used to assess the transfection efficiency of miRNA-145. The level of JAM-A mRNA expression was reduced nearly 1.33 to 1.66-fold than control at a dose concentration ranging 0.05–0.5 nmol, which proved the transfection of miRNA by C–miRNA [82].
6. Photodynamic Therapy of Chitosan-Based Complexes
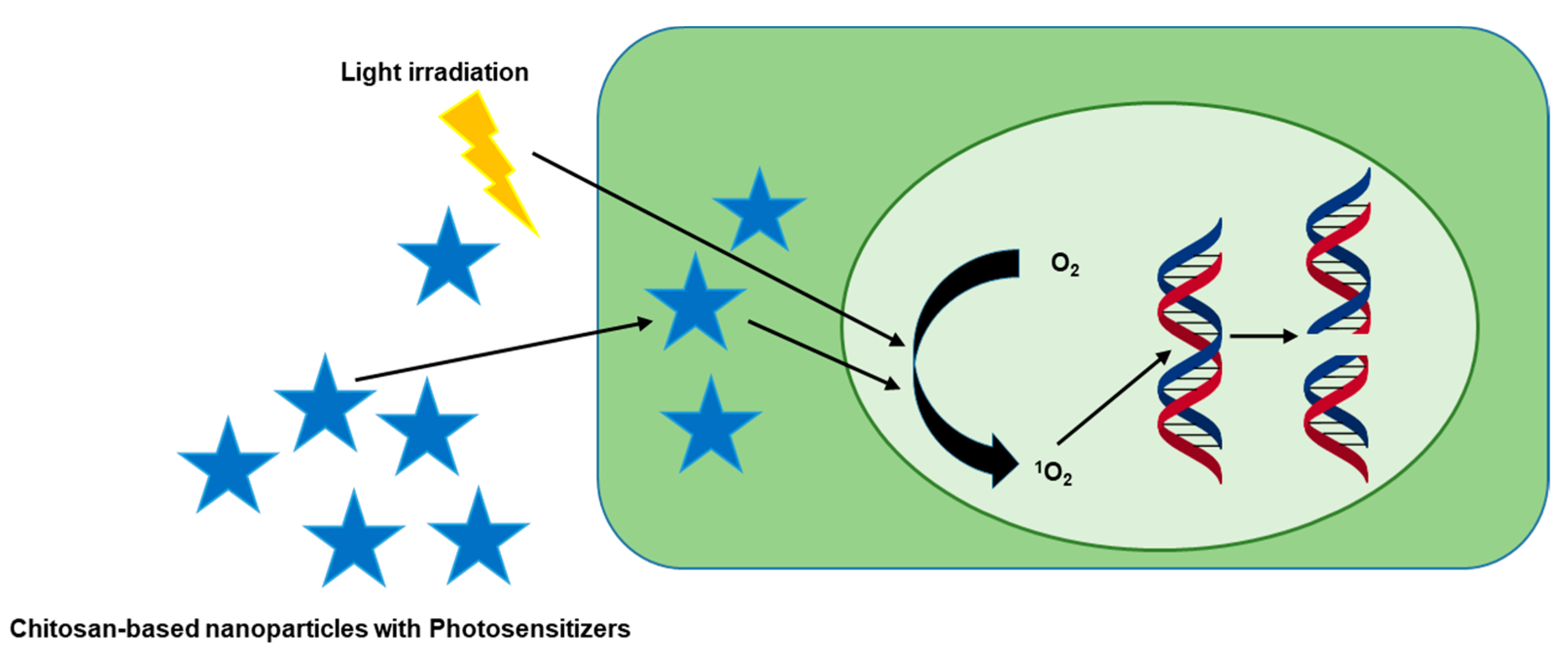
Photodynamic therapy (PDT) is a non-invasive approach to destroy tumor cells through the production of ROS by irradiation with light [85,86]. Lights with a specific wavelength stimulate photosensitizers (PS) to induce photochemical reactions which convert oxygen (O2) to singlet oxygen (1O2) and subsequently destroy tumors [87]. PS localizes into the tumors and generates in situ 1O2 with appropriate irradiation which leads to killing the tumors [85]. Most PS have poor water solubility and prolonged cutaneous photosensitivity which interrupts their clinical application. Therefore, chitosan-based nanoformulation could be an efficient approach in PS delivery and improving PDT outcomes (Figure 3) [88].
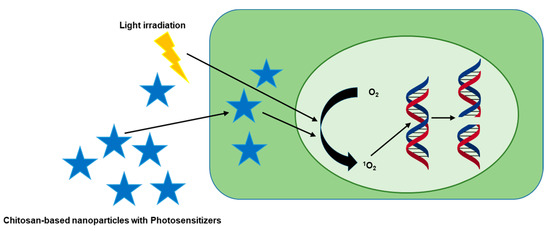
Figure 3.
Photodynamic therapy mediated by chitosan-based complexes. Some chitosan-based complexes contain photosensitizers that localize into tumor cells. Upon irradiation at a specific wavelength, the photosensitizers convert O2 to 1O2 which destroys cancer cells via photodynamic therapy.
Zhao et al. [89] constructed a chitosan-based glycolipid nanoparticle by conjugating chitosan, octadecanoic acid, and gadopentetic acid which self-assembled into micelle to load a PS, chlorin e6. The obtained nanostructure, COG@Ce6, could realize MRI-guided PDT in both 4T1 breast cancer cells and 4T1 tumor-bearing mice. Results obtained from singlet oxygen sensor green assay indicated that COG@Ce6 with laser irradiation at 660 nm elevated in vitro ROS generation of 4T1 cells. COG@Ce6 also showed in vivo anticancer activity through MRI-guided PDT in the 4T1 tumor-bearing mice model. COG@Ce6 with laser irradiation reduced tumor volume and tumor weight by nearly four- and five-times, respectively, compared to control. Moreover, COG@Ce6 with laser irradiation reduced tumor volume and weight 2.67- and 3-times, respectively, than COG@Ce6 without laser irradiation [89]. In another study, chitosan-based nanoparticle, CPCNPs, was developed by conjugating tetraphenylchlorin as PS and was used to load cytostatic drugs mertansine (MRT) or cabazitaxel (CBZ). Upon irradiation at 420 nm, PS mediated PDT effect on breast cancer cell lines (i.e., MDA-MB231 and MDA-MB-468 cells). Both empty and drug-loaded CPCNPs killed 40–50% more breast cancer cells than MRT and CBZ. Moreover, PS partially masked the toxicity of the loaded drugs and increased the in vivo suitability of the drugs [90]. Similarly, a polymeric nano-drug was fabricated by Jia et al. [91] by combining glycol chitosan, polyethylene glycol, and a PS, protoporphyrin IX (CGP). The resulting CGP was self-assembled in aquatic medium and the fluorescence of protoporphyrin IX moieties was quenched due to strong π–π stacking. However, upon encountering the plasma membranes of A549 cells, the CGP was disassembled and the membrane affinity of protoporphyrin IX led CGPs to attach to the plasma membrane. Upon irradiation at 635 nm, the protoporphyrin IX generated 1O2, which disrupted membrane integrity and induced nanoparticles to influx into A549 cells which led to cellular death. Moreover, in vivo experimentation with U14 squamous cell carcinoma tumor-bearing mice showed that irradiation by laser (635 nm) for 20 min stimulated CGP to eliminate the tumor through PDT and there was no sign of tumor relapse up to 22 days [91].
7. Delivery of Chemotherapeutic Drugs
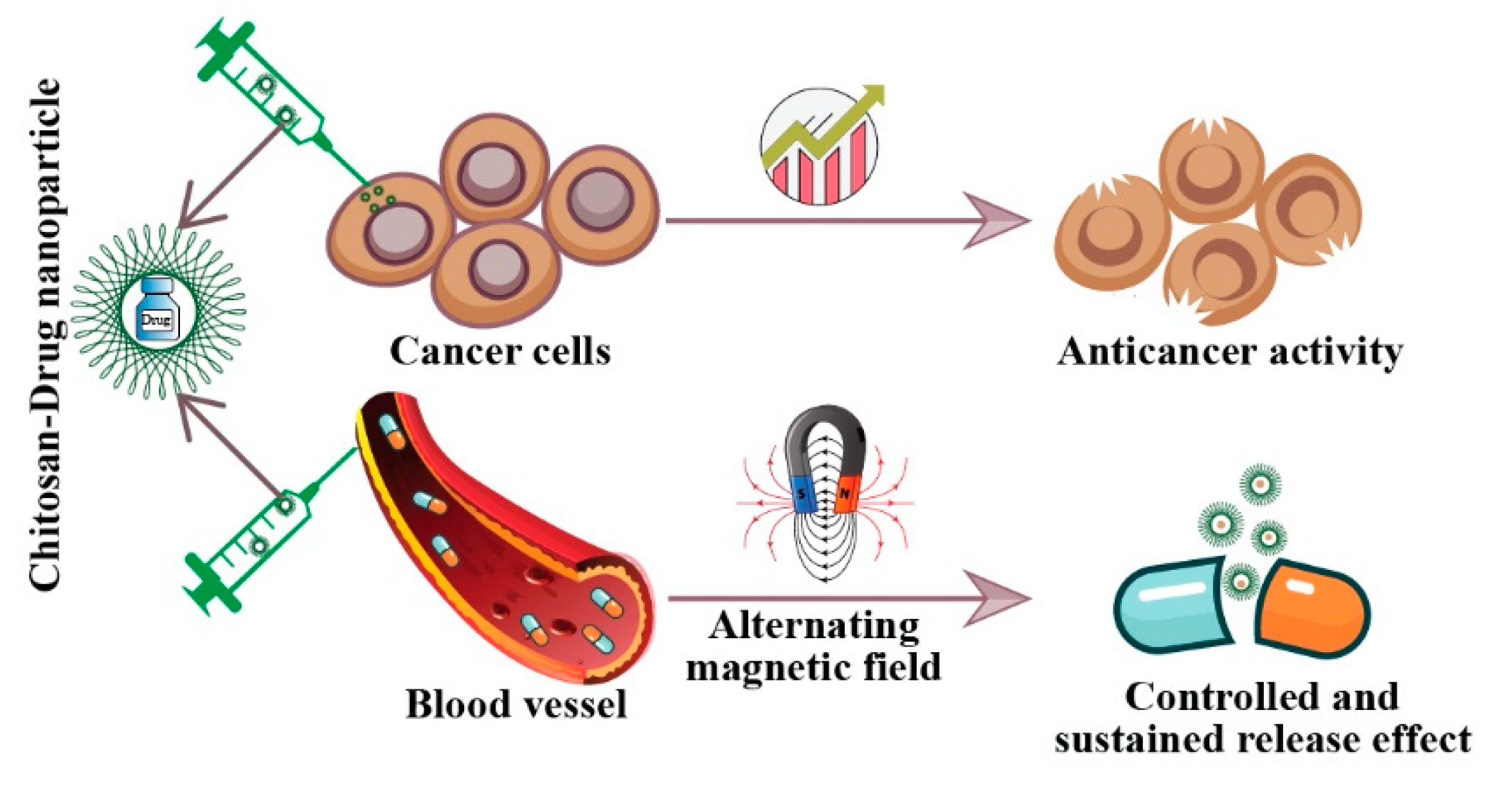
Lipophilicity is one of the major parameters that determine the cellular uptake of cytotoxic drugs [92], and is also linked with drug pharmacokinetic profile and potency [93]. There must be a balance between lipophilicity and hydrophobicity to be an ideal chemotherapeutic agent [92,94]. Chitosan improved the delivery of hydrophobic and hydrophilic conventional chemotherapeutic drugs (Figure 4) [94].
Figure 4.
Co-delivery of chemotherapeutic drugs using chitosan-based nanocarriers. Chitosan-based drug delivery systems improve the TI of chemotherapeutic drugs through a controlled and sustained release effect. Chitosan-drug nanocarriers also reduce the therapeutic concentration of approved chemotherapeutic drugs, thereby minimize the toxic effects on healthy cells. This diagram reflects potential outcomes, and does not indicate any experimental findings. Therapeutic index: TI.
7.1. Delivery of Hydrophilic Anticancer Drugs
Doxorubicin (DOX) is a water-soluble chemotherapeutic drug used in the treatment of different cancers such as lung, thyroid, breast, and hematological malignancies [95,96]. Yousefpour et al. [97] conjugated DOX with chitosan via succinic anhydride spacer, and then self-assembled to form chitosan-DOX nanoparticles. Chitosan-DOX nanoparticles were then decorated with trastuzumab which targets the Herceptin receptor. Trastuzumab-loaded chitosan-DOX (Tra-chitosan-DOX) nanoparticles showed higher cytotoxic activity towards SKOV3 ovarian cancer cells (Her2 positive) compared to free DOX. Fluorescence microscopy study confirmed that Tra-chitosan-DOX selectively was taken up by SKOV3 compared to control MCF-7 cells (Her2 negative). It should be noted that the conjugation of chitosan with DOX reduced the cytotoxic activity of DOX against SKOV3 cells [97]. In another strategy, DOX loaded onto the surface of chitosan mesoporous magnetic nanoparticles (DOX-CMN) displayed a controlled and sustained release effect of the drug. Application of alternating current magnetic field increased the release of DOX from DOX-CMN and thereby augmented the cytotoxic activity of DOX towards MCF-7 cells [98]. Similarly, encapsulation of DOX into pluronic F127 polymer-chitosan micelle enhanced anticancer activity of DOX compared to free DOX [94].
7.2. Delivery of Hydrophobic Anticancer Drugs
Paclitaxel (PTX) is a broad-spectrum hydrophobic anticancer drug that is used in the treatment of liver, lung, breast, ovarian, and other cancers [99,100]. However, PTX is poorly soluble in water (less than 0.1 μg/mL), which profoundly affects its bioavailability [100]. To increase the solubility, PTX is traditionally formulated with Cremophor EL, a conventional taxol carrier [99]. However, some complications associated with Cremophor EL demand the development of alternative carrier systems such as liposomes, emulsions, microspheres, and polymeric nanoparticles. Water-soluble chitosan derivatives, i.e., glycol chitosan, are considered as a novel carrier of PTX owing to their solubility and in vivo biocompatibility [99]. Kim et al. [99] developed hydrophobically modified glycol chitosan (HGC) nanoparticles as a PTX carrier to make the sustained release of PTX. PTX was loaded on HGC (PTX-HGC) nanoparticles via the dialysis method. Due to the sustained delivery, PTX-HGC nanoparticles showed less cytotoxicity than Cremophor EL formulated PTX. Treatment of MCF-7 cells using PTX-HGC nanoparticles (10 μg/mL) enhanced 60% cell viability compared to Cremophor EL formulated PTX at the same concentration. In vivo study in the C57BL/6 mice model showed that PTX-HGC nanoparticles had similar anticancer activity compared to Cremophor EL-based PTX at 20 mg/kg dose. At higher concentration, PTX (50 mg/kg) in HGC nanoformulation completely regressed tumors in four mice (n = 6) [99]. In another study, chitosan and glyceryl monooleate (CS-GMN) nanoparticles were prepared through multiple emulsion (o/w/o) solvent evaporation methods). PTX was then loaded into CS-GMN nanoparticles to accelerate the therapeutic effects of PTX. CS-GMN-PTX displayed sustained release effect and enhanced PTX (4-times) in MDA-MB-231 than PTX alone. Furthermore, the IC50 value of the nano-PTX formulation was also reduced by 1000 folds than the conventional PTX solution. Moreover, nano-PTX minimized the PTX associated side effects; therefore, this drug delivery system could enhance the chemotherapeutic effects of PTX [101].
Cisplatin (CIS) is an anticancer drug used in the treatment of ovarian, head, neck, bladder, lung, and testicular cancer [102]. CIS is poorly soluble in water and exhibits several side effects, such as chronic neurotoxicity, acute nephrotoxicity, nausea, and vomiting [102,103]. Kim et al. [103] loaded CIS on HGC nanoparticles (CIS-HGC) through the dialysis method to facilitate sustained release of CIS. Due to the slow and sustained drug delivery, CIS-HGC (10 μg/mL) showed ~10% less cytotoxicity in SCC7 squamous carcinoma cells and A549 cells compared to free CIS following 72 h treatment. Moreover, in vivo studies on SCC7 tumor-bearing male C3H/HeN mice (changes in tumor volume and body weight, survival rates, and, immunohistological findings) also confirmed that CIS-HGC possessed higher antitumor activity and lower cytotoxicity compared to free CIS [103]. On the other hand, CIS conjugated with chitosan encapsulated solid-lipid nanoparticles (CIS-CSN) exhibited higher cytotoxic potential against HeLa with an IC50 value of 0.6125 μg/mL compared to free CIS (IC50 1.602 μg/mL). While the cell viability remained > 95% after CSN treatment indicating the biocompatibility of CSN towards the HeLa cell, the possible reason behind the higher cytotoxic activity of CIS-CSN could be higher internalization of CIS-CSN into the HeLa cells or sustained release of CIS. Moreover, CIS-CSN also induced ~28% more apoptosis in HeLa cells compared to CIS following 24 h incubation [104].
5-Fluorouracil (5-FU) is an anticancer drug used in the treatment of nasopharyngeal, breast, lung and colorectal cancer [105,106]. Similar to CIS, 5-FU shows less solubility in water [106], and patients can experience life-threatening complications such as myelosuppression [107]. Conjugation of biopolymer enhanced selectivity of 5-FU towards cancer cells [97]. Rajon et al. [108] grafted 5-FU into hyaluronidase (HL) encapsulated chitosan-copolymer nanoparticles and prepared three different nanopolymer composites, namely chitosan-hyaluronidase-5-fluorouracil (CHL-5-FU), CHL-5-FU-polyethylene glycol (CHL-5-FU-PEG), and CHL-5-FU-PEG-gelatin (CHL-5-FU-PEG-GE), via ionic gelation technique. HL-mediated encapsulation of these polymers facilitates site-specific targeting of the nanoparticles towards COLO-205 and HT-29 colon cancer cell lines, and thereby enhances the bioavailability of 5-FU. MTT assay results showed that 5-FU loaded nanopolymer composites were less potent towards COLO-205 and HT-29 cells compared to 5-FU following 3–12 h treatment. It should be noted that CHL-5-FU-PEG-GE was less cytotoxic compared to CHL-5-FU-PEG and CHL-5-FU [108]. In another study, Cavalli et al. [109] encapsulated 5-FU with chitosan nanospheres (5FCN) via combined coacervation and emulsion droplet coalescence method for the efficient delivery of 5-FU into HT-29 and PC-3 cancer cell lines. In vitro studies demonstrated that 5FCN not only declined tumor cell growth in a concentration and time-dependent manner, but also prohibited both tumor cell adhesion to human umbilical vein endothelial cells (HUVEC) following 48 h treatment [109].
Docetaxel (DTL) is another lipophilic drug used to treat a wide range of solid tumors including breast, prostate, ovarian, and non-small cell lung cancer [110]. Hydrophobicity and DTL-associated adverse effects (anaphylaxis, myelosuppression, etc.) limit the clinical usage of DTL [110,111,112]. To enhance the therapeutic potential of DTL, sodium tripolyphosphate cross-linked DTL-loaded chitosan nanoparticles (DTLC) were developed by using the ionotropic gelation method. MTT assay showed that 72 h incubation of MDA-MB-231 cells with DTLC (5.0 μg/mL) increased cytotoxicity by ~25% compared to free DTL at the same concentration. This study indicates that ionically cross-linked DTL grafted chitosan nanoparticles could be used to deliver chemotherapeutic drugs [110].
8. Chitosan-Based Surfactant in Cancer Treatment
Surface-active compounds have special structural features that allow them to absorb at an oil–water or air–water interface and solid surface [113]. There are two types of surfactants: (1) ionic surfactant and (2) non-ionic surfactant. Though both types of surfactants are used to dissolve poorly soluble drugs, non-ionic surfactants are more effective compared to ionic surfactants [114].
Surfactant-based drug delivery system enhanced anticancer activity and minimized the toxicity of chemotherapeutic drugs via selective targeting of cancer cells, pH-responsive drug release, and sustained release effect [114,115,116,117]. Alshraim et al. [115] synthesized chitosan-coated flexible liposome (FL) to improve the delivery of chemotherapeutic drug docetaxel (DTX). This nanoformulation was further modified using sodium deoxycholate and dicetyl phosphate as non-ionic surfactants. The surfactant-loaded FL displayed a sustained release effect and higher cytotoxic activity towards HT-29 cells compared to the control drug onkotaxel. In vitro studies showed that FL showed ~27% hemolytic activity due to its high positive surface charge, therefore preclinical studies using this FL should be performed to investigate its safe dose [115]. In another study, Scheeren et al. [116] incorporated pH-sensitive lysine-based amphiphile surfactant into the surface of PEGylated and poloxamer-modified chitosan nanoparticles. DOX was then entrapped into this nanoformulation to trigger the pH-responsive release of DOX. In an acidic environment (pH 6.6), 100% of DOX was released after 6 h, while only 71% of the drug was released at pH 7.4. Photodegradation studies confirmed that the nanoformulation enhanced the stability of DOX under UVA radiation. While MTT assay results showed that anticancer activity of DOX towards HeLa cells remained unaffected after nanoformulation, it indicated the promise of this pH-sensitive drug delivery system in targeting cancer cells and the cellular microenvironment [116].
9. Chitosan-Based Emulsion in Cancer Therapy
An emulsion is a colloid of two immiscible liquids with one of them, the dispersed phase, producing small droplets in the other, namely the continuous phase. The phenomenon of droplets formation during emulsification is employed to produce polymeric nanocarriers [118]. Chitosan-based carriers are usually fabricated as water-in-oil emulsions, where a small aqueous phase is dispersed into a large oil phase [119,120]. The emulsifying capacity of chitosan may be due to its amphipathic nature. Due to the presence of both hydrophilic (D-glucosamine) and hydrophobic (N-acetylated) sites, chitosan may localize itself in both the oil and water phase, therefore acting as an emulsifier [121,122].
Gemcitabine (GEM) is a difluoro analog of deoxycytidine that inhibits ribonucleotide reductase and impedes DNA synthesis. Non-specific cytotoxicity is one of the major limitations of this chemotherapeutic agent. Utilizing the mucoadhesive properties of chitosan, Trickler et al. [123] formulated GEM with chitosan/glyceryl monooleate (GMO) nanostructures by multiple emulsion solvent evaporation methods to assess the cytotoxicity and intracellular accumulation of GEM on Mia PaCa-2 or BxPC-3 human pancreatic cell lines. HPLC studies showed that, compared with free gemcitabine, GEM-chitosan/GMO nanoparticles doubled the level of intracellular accumulation of GEM. Furthermore, GEM-chitosan/GMO nanoparticles were more potent than free GEM in terms of IC50 values [123]. Similar to GEM, the application of camptothecin (CPT) as an anticancer agent faces numerous hindrances, mainly due to its high lipophilicity, poor aqueous solubility, and side effects. To overcome these obstacles, Natesan et al. [124] developed chitosan conjugated nanoemulsions carrying CPT and evaluated its activity in preclinical breast cancer animal models. CPT was dissolved in the oil phase consisting of benzyl alcohol, TG, and D-α-Tocopheryl polyethylene glycol 1000 succinate to produce NEs before mixing with chitosan as an aqueous phase to produce CPT-CHI-NEs. Coformulation with chitosan prolonged the half-life of the NEs by reducing their clearance by the RES and liver accumulation. The drug release pattern of CHI-CPT-NEs showed an initial burst release, followed by a sustained release for a prolonged period which ensured acute drug action(s) and a long therapeutic window. Furthermore, in CHI-CPT-NEs treated BALB/c mice bearing T1-breast tumor xenograft showed passive accumulation of 2495.22 ± 174.66 ng/gm of CPT which was higher than the non-stabilized NEs (1677.58 ± 134.21 ng/gm) that might be attributed to the slightly acidic pKa of chitosan leading to rapid solubilization of the NEs in the tumor microenvironment leading to a quicker release of CPT [124]. In another study, Rosch et al. [125] encapsulated DOX via a water-in-oil emulsification process involving an oil phase mixture of cyclohexane and dodecylamine with alginate/chitosan in the water phase to produce sub-100 nm polymeric nanoparticles. At physiological pH, the nanoparticles showed a controlled release of DOX, whereas, at pH 5.5, a burst release of DOX was observed. This phenomenon can maximize the therapeutic efficacy by achieving a higher drug concentration at the tumor vicinity. Cellular uptake of DOX was comparable in both free and encapsulated forms which is necessary to ensure the efficacy and cytotoxic effects on the tumor cells. Free DOX and alginate-chitosan encapsulated DOX were equally potent towards 4T1 cells in terms of IC50 values [125].
10. Chitosan-Based Hydrogel in Cancer Treatment
Chitosan-based hydrogel unveils a new area in cancer therapy. Hydrogel-based formulations enhanced the antitumor activity of well-known anticancer drugs, improving bioavailability and biocompatibility, eliciting an immune response, and, more importantly, the antibacterial activity of chitosan could offer an extra advantage in the prevention of secondary infections in cancer patients [126,127,128,129,130,131].
The PD-1/PDL1 pathway inhibitors have emerged as a significant addition to the anticancer armament capable of resisting immune evasion by tumor cells [132]. Although monoclonal antibodies are primarily employed to inhibit PD-1/PDL1 interaction, numerous small molecule and peptide inhibitors have been shown to exhibit similar activity [133,134]. Trimethyl chitosan has been used to formulate a proteolysis resistant oral PDL-1 inhibitor, Oral PD-L1 Binding Peptide 1 (OPBP-1), which could selectively bind PD-L1 and block its interaction with PD-1. OPBP-1 showed favorable oral bioavailability and durable half-life in rats when loaded with trimethyl chitosan hydrogel, and significantly inhibited tumor proliferation in murine colorectal CT26 and melanoma B16-OVA models [126].
11. Immunotherapy Using Chitosan
In recent years, cancer vaccines or other active immunotherapies are being considered as adjuvant and neoadjuvant therapies in treating cancer. These treatment strategies have been found to reinforce the efficacy of the primary antitumor treatments by inducing robust and long-lasting tumor-specific immune responses to impede tumor growth and metastasis [135]. As mentioned earlier, chitosan received attention as a means of targeted gene delivery due to its biocompatibility, biodegradability, and low toxicity [60]. Maiyo et al. [136] developed chitosan-coated selenium nanoparticles (SeNPs) with a folate targeting moiety to deliver systemic mRNA to cancer cells. Functionalized SeNPs could be used in tumor vaccination or cancer immunotherapy to achieve a higher immune response. The SeNPs efficiently delivered the luciferase mRNA into cells in vitro and induced its expression, while the FA-targeting moiety increased the uptake of SeNPs in folate receptor-positive cells. The SeNPs protected the mRNA from RNase degradation and this nanoformulation exhibited low cytotoxicity towards HEK293, MCF-7, and KB nasopharyngeal cancer cells [136].
Chitosan-based pro-inflammatory cytokine therapy has shown immense potential against solid tumors in preclinical studies [137]. To circumvent the systemic toxicity associated with administration of IL-12 alone, Vo et al. [138] co-formulated IL-12 with chitosan (CS/IL-12) and investigated its activity in barring tumor growth and metastasis in spontaneously metastatic 4T1 mammary adenocarcinoma bearing mice. They demonstrated that intratumoral or intravesical administration of simple mixtures of CS/IL-12 significantly enhanced local IL-12 retention and mediated complete tumor regression. Tumor-specific, protective immunity was confirmed following neoadjuvant CS/IL-12 immunotherapy, as mice receiving the mixture were shown to exhibit resistance to rechallenge with 4T1 tumor as opposed to manifesting susceptibility to primary challenge with RENCA cells in cured mice. Neoadjuvant CS/IL-12 therapy also significantly reduced the instances of metastases, characterized by the reduced number of nodules in the lung and having a higher survival percentage compared to the cohort of mice receiving either chitosan or IL-12 alone (Figure 5) [138]. Furthermore, CS/IL-12 co-formulation displayed higher anticancer and antimetastatic activity compared to free IL-12. This might be due to the immunostimulatory capacity of chitosan, which induces the secretion of type I Interferon (IFN) via activation of mitochondrial DNA-mediated cGAS-STING (Cyclic GMP-AMP synthase-Stimulator of Interferon Genes) pathway [139]. CS/IL-12 also led to the expression of pro-inflammatory, cytotoxic, and antitumor TH1 cytokines (IL-12p70, IFN-γ, and tumor-necrosis factor-α) by resident T cells and macrophage in the bladder [140]. IFN-γ induction increased CD3+, CD4+, and CD8+ T-cell infiltration, and reduced the number of myeloid-derived suppressor cells and immunosuppressive regulatory T cells in the tumor vicinity [141].
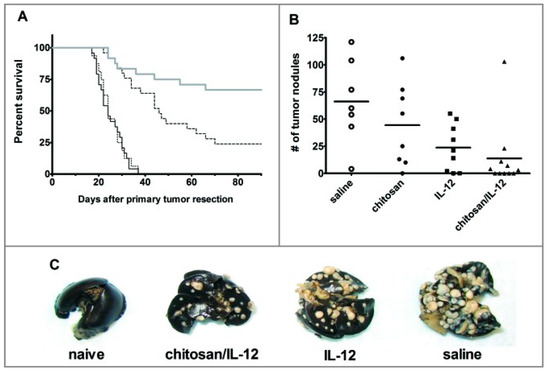
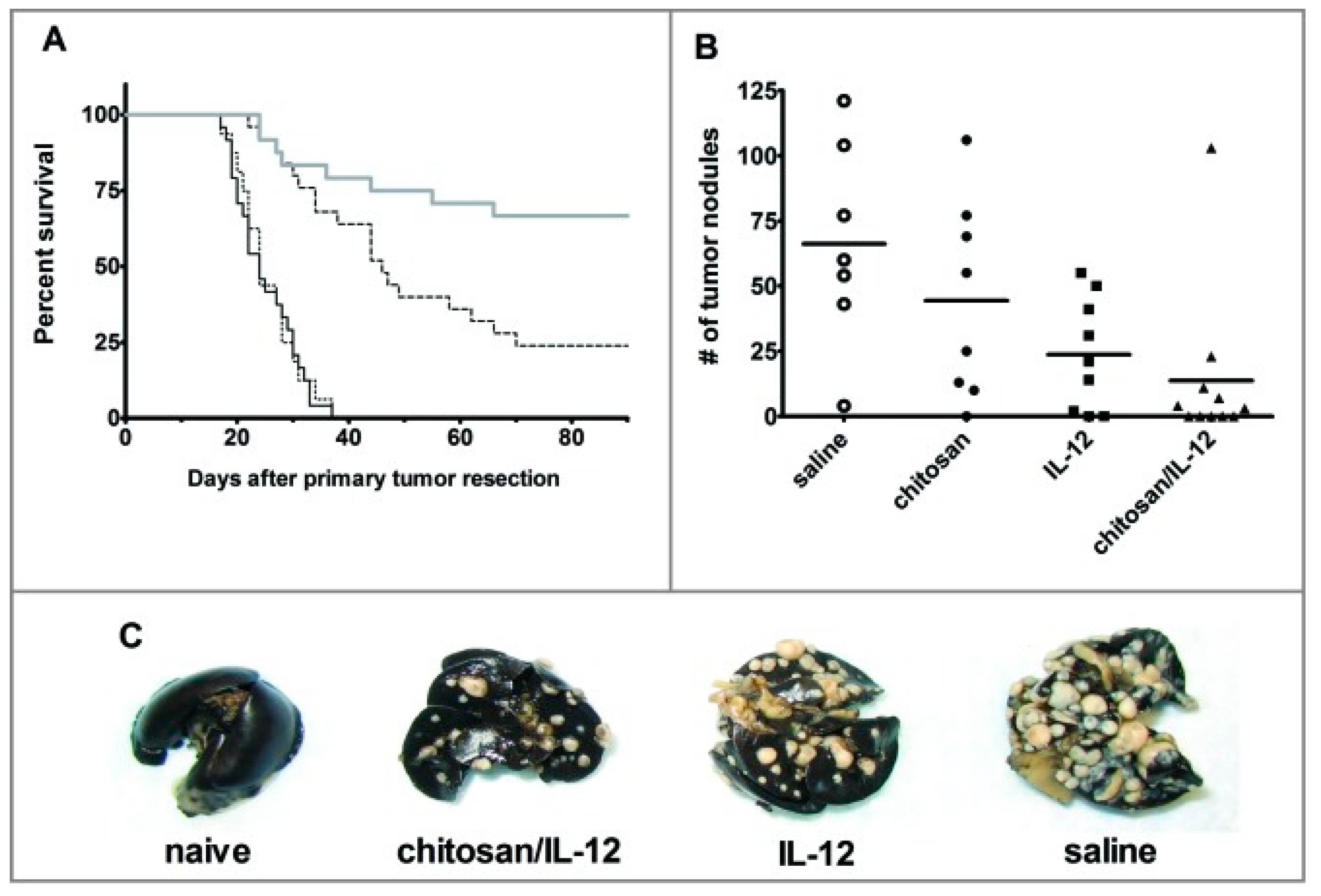
Figure 5.
Neoadjuvant chitosan/IL-12 treatment enhances survival and limits distant site metastasis of breast cancer. (A) BALB/c mice implanted with 4T1 mammary adenocarcinoma were intratumorally administered chitosan/IL-12 (2 μg) (thick gray line), chitosan alone (dotted line), IL-12 alone (dashed line), or saline (solid black) line on the 6th, 9th, and 12th post-implantation days. On the 15th day, implanted primary tumors were resected and mice were followed onwards for symptoms of lung metastasis. Mice manifesting signs of severe distress were euthanized to assess the presence of metastatic lesions. Surviving mice were followed for at least 90 days post-resection of the primary tumor. (B) In a different study, mice were euthanized after five weeks of primary tumor resection and the number of metastatic nodules in the lungs of mice from four different treatment arms was enumerated using a stereomicroscope following infusion of the removed lungs with India ink. (C) The lungs images of control mice and mice treated with chitosan/IL-12, free IL-12, or saline at five weeks post-resection. This figure is adapted from Vo et al. (2015) [138], non-commercial uses of this study do not require any further copyright permission from Taylor & Francis Group, under the license (Available online: http://creativecommons.org/licenses/by-nc/3.0/ (accessed on 6 August 2021)).
In the treatment of bladder cancer, chitosan can be of benefit to intravesical drug delivery due to its positive charge facilitating adhesion to negatively charged mucosal surfaces, while the viscosity of chitosan solution reduces the removal of the therapeutic agent during voiding of the bladder. Chitosan may also open the gap junctions between epithelial cells and enhance drug absorption in the bladder [142]. In preclinical bladder cancer models, intravesical CS/IL-12-based immunotherapy was applied in the treatment of orthotopic MB49 or MBT-2 bladder cancer [140,141]. Intravesical treatment with CS/IL-12 induced adaptive tumor-specific immunity, as evident in recovered mice. The recovered mice group rejected 100% of intravesical tumor rechallenges and about 50–100% of distant subcutaneous rechallenges [141]. Intravesical administration of CS/IL-12 for four weeks (once a week) led to the 80–100% elimination of MB49 mouse bladder tumors in C57BL/6J mice or MBT-2 tumor in the C3H/HeJ mice model, leading to long term survival (more than 60 days), while standard BCG treated groups died within 60 days of post-tumor implantation [140]. CS/IL-12-based neoadjuvant immunotherapy receiving mice exhibited elevated delayed-type hypersensitivity responses associated with positive prognosis of breast tumor [138]. Smith et al. [143] unveiled the systemic and local immunological mechanism of actions of intravesical CS/IL-12 immunotherapy. Depletion studies elucidated the role of CD8+ cells in orchestrating initial tumor rejection, whereas the CD4+ cells were found to mediate memory response by conferring durable protection against tumor rechallenge. Successive administration of CS/IL-12 led to dynamic shifts in the immune cell kinetics within secondary lymphoid organs and tumor region characterized by preliminary infiltration of granulocytes and macrophages, and then an increase in CD4+ and CD8 + effector-memory cells [143].
Dendritic cell (DC)-based vaccines have shown remarkable potential against cancer and have been validated in numerous clinical studies [144]. Chitosan has been previously reported to prolong the sequestration of antigen in the administration site, allowing more efficient antigen presentation [10]. In addition, the immunostimulatory properties of chitosan make it a suitable adjuvant to be used in cancer vaccines [139]. Han et al. [145] encapsulated ovalbumin (OVA) and polyinosinic-polycytidylic acid with chitosan nanoparticles (CH (OVA+poly I:C)-NPs) to deliver tumor-specific antigens to DCs. Flow cytometry and confocal microscopy confirmed intracellular uptake of the nanoparticles by DCs. This led to the activation of the DCs characterized by higher expression of surface maturation markers and subsequent generation of antigen-specific cytotoxic CD8+ T cells following DC-mediated antigen presentation. The therapeutic efficacy and tumor specificity conferred by antigen-carrying nanoparticles were demonstrated in EG.7-OVA tumor carrying mice where CH (OVA+poly I:C)-NPs-injected mice showed significantly reduced tumor growth compared to the control group, soluble OVA, or CH-NPs-injected mice [145]. In contrast, no therapeutic effect was shown in the OVA-negative TC-1 tumor model following CH (OVA+poly I:C)-NPs injection. The DC-maturation effect of the NPs was shown to be dependent on the molecular weight of chitosan, with polymers having a molecular weight greater than 310 kDa producing the greatest effect [146].
Chitosan-based therapeutic agents have been demonstrated to enhance the efficacy of conventional anti-cancer treatment strategies [13]. For instance, chitosan nanoparticles have been shown to enhance the radiosensitivity of breast tumors in mice models [147]. Castro et al. [147] demonstrated that combining chitosan/poly γ-glutamic acid nanoparticles (Ch/γ-PGA NPs) with radiotherapy induces antitumor immunity in the 4T1 orthotopic breast tumor mouse model. Combination therapy potentiated CD4+ and CD8+ cell-induced dendritic cell maturation and macrophages activation, respectively, impaired 4T1 tumor progression, systemic immunosuppression, and lung metastases. Notably, animals from the combination therapy exhibited less and smaller lung metastatic foci and lower levels of the systemic pro-tumor cytokines IL-3, IL-4, and IL-10, and of the CCL4 chemokine, in comparison to non-treated animals [147].
Chitosan-based theranostic nanoparticles improve the efficiency of anticancer agents [148,149]. In thermotherapy and photothermal therapy, specific enrichment and excitation of a therapeutic agent in the tumor vicinity lead to the conversion of electromagnetic radiation into heat energy resulting in hyperthermic cell death [150,151,152]. Kumar et al. [148] fabricated IR 820 encapsulated polycaprolactone glycol chitosan: poloxamer blend nanoparticles (PP-IR NPs) for imaging and effective photo-immunotherapy in human estrogen-positive MCF-7 cells. An elevated level of ROS was observed in PP-IR NP treated cells upon laser irradiation. The combined effect of hyperthermia and stimulation of TNF-α response might be linked with the ROS production. The elevated ROS caused DNA fragmentation, apoptotic bodies formation, and ultimately cell death [148]. In another study, Tang et al. [149] developed chitosan microcapsules by encapsulating ion liquids with Fe3O4 nanoparticles. The microcapsules showed high biocompatibility, and were used for magnetic resonance imaging-guided microwave irradiation leading to the thermal killing of H22 mouse hepatocellular carcinoma in mice [149].
12. Conclusions and Future Perspective
Chitosan is a natural, biocompatible, and biodegradable polymer. Chitosan displayed promise in its application to cancer therapy, including gene therapy, chemotherapeutic drug delivery, and immunotherapy. One of the major pitfalls of chitosan as a drug delivery vehicle is its poor solubility in water. Therefore, chemically modified chitosan could be used to improve the solubility of chitosan. Furthermore, chemically modified chitosan or its derivatives in different formulations (e.g., nanoformulation, emulsion, surfactant, and hydrogel) improves the efficacy of anticancer agents, biocompatibility, selectivity, and, more importantly, reduces the therapeutic dose. As the molecular weight of chitosan plays a role in the potency of the chitosan-derivatives, this factor should be taken into account while preparing different formulations. Moreover, chitosan-based biomaterials induce both humoral and cellular immune responses and, therefore, chitosan could be used in the development of therapeutic cancer vaccines. Finally, before the clinical trial of chitosan-based anticancer agents, an extensive preclinical study should be performed to examine their safety, efficacy, and short and long-time side effects.
Author Contributions
Conceptualization, M.S.S.; writing—original draft preparation, M.S.S., K.M.M., M.S., M.S.N., S.K.H., M.S.H. and M.F.U.; writing—review and editing, M.S.S. and M.A.H.; and project administration, M.S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This review article did not receive any external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in this article.
Acknowledgments
We are grateful to Md. Asiful Islam from Department of Haematology, School of Medical Sciences, Universiti Sains Malaysia, Kubang Kerian 16150, Malaysia and Mozammel Haque Bhuiyan from Center for Bioengineering and Nanomedicine, Faculty of Dentistry, University of Otago, 362 Leith St, North Dunedin, Dunedin 9016, New Zealand for checking the similarity content of this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ward, R.A.; Fawell, S.; Floc’h, N.; Flemington, V.; McKerrecher, D.; Smith, P.D. Challenges and Opportunities in Cancer Drug Resistance. Chem. Rev. 2021, 121, 3297–3351. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Falzone, L.; Salomone, S.; Libra, M. Evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front. Pharm. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [Green Version]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal. Transduct Target. 2018, 3, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayhan, M.; Hossen, M.; Niloy, M.S.; Bhuiyan, M.H.; Paul, S.; Shakil, M. Biopolymer and Biomaterial Conjugated Iron Oxide Nanomaterials as Prostate Cancer Theranostic Agents: A Comprehensive Review. Symmetry 2021, 13, 974. [Google Scholar] [CrossRef]
- Parhi, R. Drug delivery applications of chitin and chitosan: A review. Environ. Chem. Lett. 2020, 18, 577–594. [Google Scholar] [CrossRef]
- Dass, C.R.; Choong, P.F. The use of chitosan formulations in cancer therapy. J. Microencapsul. 2008, 25, 275–279. [Google Scholar] [CrossRef]
- Prabaharan, M. Chitosan derivatives as promising materials for controlled drug delivery. J. Biomater. Appl. 2008, 23, 5–36. [Google Scholar] [CrossRef]
- Hasnain, M.S.; Ahmed, S.A.; Alkahtani, S.; Milivojevic, M.; Kandar, C.C.; Dhara, A.K.; Nayak, A.K. Biopolymers for Drug Delivery. In Advanced Biopolymeric Systems for Drug Delivery; Nayak, A.K., Hasnain, M.S., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–29. [Google Scholar]
- Ahmed, T.A.; Aljaeid, B.M. Preparation, characterization, and potential application of chitosan, chitosan derivatives, and chitosan metal nanoparticles in pharmaceutical drug delivery. Drug Des. Dev. Ther. 2016, 10, 483. [Google Scholar] [CrossRef] [Green Version]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and Chitosan: Production and Application of Versatile Biomedical Nanomaterials. Int. J. Adv. Res. 2016, 4, 411–427. [Google Scholar]
- Prateeksha; Sharma, V.K.; Liu, X.; Oyarzún, D.A.; Abdel-Azeem, A.M.; Atanasov, A.G.; Hesham, A.E.; Barik, S.K.; Gupta, V.K.; Singh, B.N. Microbial polysaccharides: An emerging family of natural biomaterials for cancer therapy and diagnostics. In Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2021. [Google Scholar]
- Shakil, M.S.; Hasan, M.A.; Uddin, M.F.; Islam, A.; Nahar, A.; Das, H.; Khan, M.N.I.; Dey, B.P.; Rokeya, B.; Hoque, S.M. In Vivo Toxicity Studies of Chitosan-Coated Cobalt Ferrite Nanocomplex for Its Application as MRI Contrast Dye. ACS Appl. Bio Mater. 2020, 3, 7952–7964. [Google Scholar] [CrossRef]
- Adhikari, H.S.; Yadav, P.N. Anticancer Activity of Chitosan, Chitosan Derivatives, and Their Mechanism of Action. Int. J. Biomater. 2018, 2018, 2952085. [Google Scholar] [CrossRef] [Green Version]
- Park, B.K.; Kim, M.M. Applications of chitin and its derivatives in biological medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franconetti, A.; López, Ó.; Fernandez-Bolanos, J.G. Carbohydrates: Potential Sweet Tools Against Cancer. Curr. Med. Chem. 2020, 27, 1206–1242. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, M.B.; Struszczyk-Swita, K.; Li, X.; Szczęsna-Antczak, M.; Daroch, M. Enzymatic Modifications of Chitin, Chitosan, and Chitooligosaccharides. Front. Bioeng. Biotechnol. 2019, 7, 243. [Google Scholar] [CrossRef] [Green Version]
- Ang, L.F.; Por, L.Y.; Yam, M.F. Study on different molecular weights of chitosan as an immobilization matrix for a glucose biosensor. PLoS ONE 2013, 8, e70597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Machado, D.I.; López-Cervantes, J.; Correa-Murrieta, M.A.; Sánchez-Duarte, R.G.; Cruz-Flores, P.; de la Mora-López, G.S. Chitosan. In Nonvitamin and Nonmineral Nutritional Supplements; Seyed Mohammad Nabavi, A.S.S., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 485–493. [Google Scholar]
- Chen, J.K.; Shen, C.R.; Liu, C.L. N-acetylglucosamine: Production and applications. Mar. Drugs 2010, 8, 2493–2516. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.C.; Chou, C.C.; Li, C.F. Antibacterial activity of N-alkylated disaccharide chitosan derivatives. Int. J. Food Microbiol 2005, 97, 237–245. [Google Scholar] [CrossRef]
- Qin, C.; Li, H.; Xiao, Q.; Liu, Y.; Zhu, J.; Du, Y. Water-solubility of chitosan and its antimicrobial activity. Carbohydr. Polym. 2006, 63, 367–374. [Google Scholar] [CrossRef]
- Li, J.; Cai, C.; Li, J.; Li, J.; Li, J.; Sun, T.; Wang, L.; Wu, H.; Yu, G. Chitosan-Based Nanomaterials for Drug Delivery. Molecules 2018, 23, 2661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Meng, Q.; Li, Q.; Liu, J.; Zhou, M.; Jin, Z.; Zhao, K. Chitosan Derivatives and Their Application in Biomedicine. Int. J. Mol. Sci. 2020, 21, 487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jing, Z.W.; Ma, Z.W.; Li, C.; Jia, Y.Y.; Luo, M.; Ma, X.X.; Zhou, S.Y.; Zhang, B.L. Chitosan cross-linked with poly(ethylene glycol)dialdehyde via reductive amination as effective controlled release carriers for oral protein drug delivery. Bioorg Med. Chem. Lett. 2017, 27, 1003–1006. [Google Scholar] [CrossRef]
- Bukzem, A.L.; Signini, R.; Dos Santos, D.M.; Lião, L.M.; Ascheri, D.P. Optimization of carboxymethyl chitosan synthesis using response surface methodology and desirability function. Int. J. Biol. Macromol 2016, 85, 615–624. [Google Scholar] [CrossRef]
- Giri, T.K.; Thakur, A.; Alexander, A.; Badwaik, H.; Tripathi, D.K. Modified chitosan hydrogels as drug delivery and tissue engineering systems: Present status and applications. Acta Pharm. Sin. B 2012, 2, 439–449. [Google Scholar] [CrossRef] [Green Version]
- Salahuddin, N.; Elbarbary, A.A.; Alkabes, H.A. Quinazolinone Derivatives Loaded Polypyrrole/Chitosan Core–Shell Nanoparticles with Different Morphologies: Antibacterial and Anticancer Activities. Nano 2017, 12, 1750002. [Google Scholar] [CrossRef]
- Salahuddin, N.; Elbarbary, A.A.; Alkabes, H.A. Antibacterial and antitumor activities of 3-amino-phenyl-4 (3H)-quinazolinone/polypyrrole chitosan core shell nanoparticles. Polym. Bull. 2017, 74, 1775–1790. [Google Scholar] [CrossRef]
- Maeda, Y.; Kimura, Y. Antitumor effects of various low-molecular-weight chitosans are due to increased natural killer activity of intestinal intraepithelial lymphocytes in sarcoma 180-bearing mice. J. Nutr. 2004, 134, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Wimardhani, Y.S.; Suniarti, D.F.; Freisleben, H.J.; Wanandi, S.I.; Siregar, N.C.; Ikeda, M.A. Chitosan exerts anticancer activity through induction of apoptosis and cell cycle arrest in oral cancer cells. J. Oral Sci. 2014, 56, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Nawaz, A.; Wong, T.W. Chitosan as Anticancer Compound and Nanoparticulate Matrix for Cancer Therapeutics. Encycl. Mar. Biotechnol. 2020, 3, 1737–1752. [Google Scholar]
- Kumar, S.; Koh, J.; Kim, H.; Gupta, M.K.; Dutta, P.K. A new chitosan-thymine conjugate: Synthesis, characterization and biological activity. Int. J. Biol. Macromol 2012, 50, 493–502. [Google Scholar] [CrossRef]
- Salahuddin, N.; Elbarbary, A.A.; Salem, M.L.; Elksass, S. Antimicrobial and antitumor activities of 1,2,4-triazoles/polypyrrole chitosan core shell nanoparticles. J. Phys. Org. Chem. 2017, 30, e3702. [Google Scholar] [CrossRef]
- Vedham, V.; Divi, R.L.; Starks, V.L.; Verma, M. Multiple infections and cancer: Implications in epidemiology. Technol. Cancer Res. Treat. 2014, 13, 177–194. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Z.; Han, B.; Li, H.; Yang, Y.; Liu, W. Carboxymethyl chitosan represses tumor angiogenesis in vitro and in vivo. Carbohydr. Polym. 2015, 129, 1–8. [Google Scholar] [CrossRef]
- Jiang, M.; Ouyang, H.; Ruan, P.; Zhao, H.; Pi, Z.; Huang, S.; Yi, P.; Crepin, M. Chitosan derivatives inhibit cell proliferation and induce apoptosis in breast cancer cells. Anticancer Res. 2011, 31, 1321–1328. [Google Scholar]
- Huang, R.; Mendis, E.; Rajapakse, N.; Kim, S.K. Strong electronic charge as an important factor for anticancer activity of chitooligosaccharides (COS). Life Sci. 2006, 78, 2399–2408. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Chen, X.; Tian, J.; Li, L.; Zhao, M.; Jiao, Y.; Zhou, C. Effect of chitooligosaccharides on cyclin D1, bcl-xl and bcl-2 mRNA expression in A549 cells using quantitative PCR. Chin. Sci. Bull. 2011, 56, 1629. [Google Scholar] [CrossRef] [Green Version]
- Cui, Z.; Ni, N.C.; Wu, J.; Du, G.Q.; He, S.; Yau, T.M.; Weisel, R.D.; Sung, H.W.; Li, R.K. Polypyrrole-chitosan conductive biomaterial synchronizes cardiomyocyte contraction and improves myocardial electrical impulse propagation. Theranostics 2018, 8, 2752–2764. [Google Scholar] [CrossRef] [PubMed]
- Srinivasarao, M.; Low, P.S. Ligand-Targeted Drug Delivery. Chem Rev. 2017, 117, 12133–12164. [Google Scholar] [CrossRef]
- Nandgude, T.; Pagar, R. Plausible role of chitosan in drug and gene delivery against resistant breast cancer cells. Carbohydr. Res. 2021, 506, 108357. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Svirskis, D.; Lu, W.; Ying, M.; Huang, Y.; Wen, J. N-trimethyl chitosan nanoparticles and CSKSSDYQC peptide: N-trimethyl chitosan conjugates enhance the oral bioavailability of gemcitabine to treat breast cancer. J. Control. Release 2018, 277, 142–153. [Google Scholar] [CrossRef]
- Zhou, P.; Hu, J.; Wang, X.; Wang, J.; Zhang, Y.; Wang, C. Epidermal growth factor receptor expression affects proliferation and apoptosis in non-small cell lung cancer cells via the extracellular signal-regulated kinase/microRNA 200a signaling pathway. Oncol. Lett. 2018, 15, 5201–5207. [Google Scholar] [CrossRef] [Green Version]
- Nascimento, A.V.; Singh, A.; Bousbaa, H.; Ferreira, D.; Sarmento, B.; Amiji, M.M. Mad2 Checkpoint Gene Silencing Using Epidermal Growth Factor Receptor-Targeted Chitosan Nanoparticles in Non-Small Cell Lung Cancer Model. Mol. Pharm. 2014, 11, 3515–3527. [Google Scholar] [CrossRef] [Green Version]
- Tanimoto, K.; Eguchi, H.; Yoshida, T.; Hajiro-Nakanishi, K.; Hayashi, S. Regulation of estrogen receptor alpha gene mediated by promoter B responsible for its enhanced expressionin human breast cancer. Nucleic Acids Res. 1999, 27, 903–909. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Tang, C.; Yin, C. Estrone-modified pH-sensitive glycol chitosan nanoparticles for drug delivery in breast cancer. Acta Biomater. 2018, 73, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Qindeel, M.; Ahmed, N.; Khan, G.M.; Rehman, A.U. Ligand decorated chitosan as an advanced nanocarrier for targeted delivery: A critical review. Nanomedicine 2019, 14, 1623–1642. [Google Scholar] [CrossRef]
- Pinto, R.J.B.; Lameirinhas, N.S.; Guedes, G.; Rodrigues da Silva, G.H.; Oskoei, P.; Spirk, S.; Oliveira, H.; Duarte, I.F.; Vilela, C.; Freire, C.S.R. Cellulose Nanocrystals/Chitosan-Based Nanosystems: Synthesis, Characterization, and Cellular Uptake on Breast Cancer Cells. Nanomaterials 2021, 11, 2057. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Sun, J.; Zhang, W.; Zhao, Y.; Zhang, S.; Zhang, S. Drug delivery systems based on CD44-targeted glycosaminoglycans for cancer therapy. Carbohydr. Polym. 2021, 251, 117103. [Google Scholar] [CrossRef]
- Mali, S. Delivery systems for gene therapy. Indian J. Hum. Genet. 2013, 19, 3–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammadzadeh, R.; Shahim, P.; Akbari, A.J.B. Formulation of a pH-sensitive cancer cell-targeted gene delivery system based on folate–chitosan conjugated nanoparticles. Biotechnol. Appl. Biochem. 2021, 68, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Jiang, H.-L.; Jere, D.; Park, I.-K.; Cho, M.-H.; Nah, J.-W.; Choi, Y.-J.; Akaike, T.; Cho, C.-S. Chemical modification of chitosan as a gene carrier in vitro and in vivo. Prog. Polym. Sci. 2007, 32, 726–753. [Google Scholar] [CrossRef]
- Mahato, R.I.; Smith, L.C.; Rolland, A. 4-Pharmaceutical Perspectives of Nonviral Gene Therapy. In Advances in Genetics; Hall, J.C., Dunlap, J.C., Friedmann, T., Giannelli, F., Eds.; Academic Press: Cambridge, MA, USA, 1999; Volume 41, pp. 95–156. [Google Scholar]
- Lundstrom, K.; Boulikas, T. Viral and non-viral vectors in gene therapy: Technology development and clinical trials. Technol. Cancer Res. Treat. 2003, 2, 471–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos-Carballal, B.; Fernández Fernández, E.; Goycoolea, F.M. Chitosan in Non-Viral Gene Delivery: Role of Structure, Characterization Methods, and Insights in Cancer and Rare Diseases Therapies. Polymers 2018, 10, 444. [Google Scholar] [CrossRef] [Green Version]
- Ramamoorth, M.; Narvekar, A. Non viral vectors in gene therapy-an overview. J. Clin. Diagn. Res. JCDR 2015, 9, Ge01–Ge06. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, N.; Mourya, V.K. Chitosan and anionic polymers—Complex formation and applications. In Polysaccharide: Development, Properties and Applications; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 333–377. [Google Scholar]
- Cao, Y.; Tan, Y.F.; Wong, Y.S.; Liew, M.W.J.; Venkatraman, S. Recent Advances in Chitosan-Based Carriers for Gene Delivery. Mar. Drugs 2019, 17, 381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babu, A.; Ramesh, R. Multifaceted Applications of Chitosan in Cancer Drug Delivery and Therapy. Mar. Drugs 2017, 15, 96. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Mysore, K.; Flannery, E.; Michel, K.; Severson, D.W.; Zhu, K.Y.; Duman-Scheel, M. Chitosan/interfering RNA nanoparticle mediated gene silencing in disease vector mosquito larvae. J. Vis. Exp. JoVE 2015, 97, 52523. [Google Scholar] [CrossRef] [Green Version]
- Ragelle, H.; Riva, R.; Vandermeulen, G.; Naeye, B.; Pourcelle, V.; Le Duff, C.S.; D’Haese, C.; Nysten, B.; Braeckmans, K.; De Smedt, S.C.; et al. Chitosan nanoparticles for siRNA delivery: Optimizing formulation to increase stability and efficiency. J. Control. Release 2014, 176, 54–63. [Google Scholar] [CrossRef]
- Mohammed, M.A.; Syeda, J.T.M.; Wasan, K.M.; Wasan, E.K. An Overview of Chitosan Nanoparticles and Its Application in Non-Parenteral Drug Delivery. Pharmaceutics 2017, 9, 53. [Google Scholar] [CrossRef] [Green Version]
- Guo, C.; Gemeinhart, R.A. Understanding the adsorption mechanism of chitosan onto poly(lactide-co-glycolide) particles. Eur. J. Pharm. Biopharm. Off. J. Arb. Fur Pharm. Verfahr. E.V 2008, 70, 597–604. [Google Scholar] [CrossRef] [Green Version]
- Mumper, R.; Wang, J.; Claspell, J.M.; Rolland, A. Novel polymeric condensing carriers for gene delivery. Proc. Natl. Symp. Control. Rel. Bioact. Mater. 1995, 22, 178–179. [Google Scholar]
- Carreño-Gómez, B.; Duncan, R. Evaluation of the biological properties of soluble chitosan and chitosan microspheres. Int. J. Pharm. 1997, 148, 231–240. [Google Scholar] [CrossRef]
- Richardson, S.W.; Kolbe, H.J.; Duncan, R. Potential of low molecular mass chitosan as a DNA delivery system: Biocompatibility, body distribution and ability to complex and protect DNA. Int. J. Pharm. 1999, 178, 231–243. [Google Scholar] [CrossRef]
- Herdiana, Y.; Wathoni, N.; Shamsuddin, S.; Joni, I.M.; Muchtaridi, M. Chitosan-Based Nanoparticles of Targeted Drug Delivery System in Breast Cancer Treatment. Polymers 2021, 13, 1717. [Google Scholar] [CrossRef]
- Zhang, E.; Xing, R.; Liu, S.; Qin, Y.; Li, K.; Li, P. Advances in chitosan-based nanoparticles for oncotherapy. Carbohydr. Polym. 2019, 222, 115004. [Google Scholar] [CrossRef]
- Nam, J.P.; Nah, J.W. Target gene delivery from targeting ligand conjugated chitosan-PEI copolymer for cancer therapy. Carbohydr. Polym. 2016, 135, 153–161. [Google Scholar] [CrossRef]
- Wang, M.; Hu, H.; Sun, Y.; Qiu, L.; Zhang, J.; Guan, G.; Zhao, X.; Qiao, M.; Cheng, L.; Cheng, L.; et al. A pH-sensitive gene delivery system based on folic acid-PEG-chitosan—PAMAM-plasmid DNA complexes for cancer cell targeting. Biomaterials 2013, 34, 10120–10132. [Google Scholar] [CrossRef]
- Song, B.; Zhang, W.; Peng, R.; Huang, J.; Nie, T.; Li, Y.; Jiang, Q.; Gao, R. Synthesis and cell activity of novel galactosylated chitosan as a gene carrier. Colloids Surf. B Biointerfaces 2009, 70, 181–186. [Google Scholar] [CrossRef]
- Germershaus, O.; Mao, S.; Sitterberg, J.; Bakowsky, U.; Kissel, T. Gene delivery using chitosan, trimethyl chitosan or polyethylenglycol-graft-trimethyl chitosan block copolymers: Establishment of structure-activity relationships in vitro. J. Control. Release 2008, 125, 145–154. [Google Scholar] [CrossRef]
- Chang, K.-L.; Higuchi, Y.; Kawakami, S.; Yamashita, F.; Hashida, M. Efficient Gene Transfection by Histidine-Modified Chitosan through Enhancement of Endosomal Escape. Bioconjugate Chem. 2010, 21, 1087–1095. [Google Scholar] [CrossRef]
- Kean, T.; Roth, S.; Thanou, M. Trimethylated chitosans as non-viral gene delivery vectors: Cytotoxicity and transfection efficiency. J. Control. Release 2005, 103, 643–653. [Google Scholar] [CrossRef]
- Liang, X.; Li, X.; Chang, J.; Duan, Y.; Li, Z. Properties and evaluation of quaternized chitosan/lipid cation polymeric liposomes for cancer-targeted gene delivery. Langmuir ACS J. Surf. Colloids 2013, 29, 8683–8693. [Google Scholar] [CrossRef]
- Lee, D.; Lockey, R.; Mohapatra, S. Folate receptor-mediated cancer cell specific gene delivery using folic acid-conjugated oligochitosans. J. Nanosci. Nanotechnol. 2006, 6, 2860–2866. [Google Scholar] [CrossRef]
- Akinyelu, J.; Singh, M. Chitosan Stabilized Gold-Folate-Poly(lactide-co-glycolide) Nanoplexes Facilitate Efficient Gene Delivery in Hepatic and Breast Cancer Cells. J. Nanosci. Nanotechnol. 2018, 18, 4478–4486. [Google Scholar] [CrossRef]
- Safari, S.; Dorkoosh, F.A.; Soleimani, M.; Zarrintan, M.H.; Akbari, H.; Larijani, B.; Tehrani, M.R. N-Diethylmethyl chitosan for gene delivery to pancreatic cancer cells and the relation between charge ratio and biologic properties of polyplexes via interpolations polynomial. Int. J. Pharm. 2011, 420, 350–357. [Google Scholar] [CrossRef]
- Wang, K.; Kievit, F.M.; Florczyk, S.J.; Stephen, Z.R.; Zhang, M. 3D Porous Chitosan–Alginate Scaffolds as an In Vitro Model for Evaluating Nanoparticle-Mediated Tumor Targeting and Gene Delivery to Prostate Cancer. Biomacromolecules 2015, 16, 3362–3372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos-Carballal, B.; Aaldering, L.J.; Ritzefeld, M.; Pereira, S.; Sewald, N.; Moerschbacher, B.M.; Götte, M.; Goycoolea, F.M. Physicochemical and biological characterization of chitosan-microRNA nanocomplexes for gene delivery to MCF-7 breast cancer cells. Sci. Rep. 2015, 5, 13567. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Howard, K.A.; Dong, M.; Andersen, M.; Rahbek, U.L.; Johnsen, M.G.; Hansen, O.C.; Besenbacher, F.; Kjems, J. The influence of polymeric properties on chitosan/siRNA nanoparticle formulation and gene silencing. Biomaterials 2007, 28, 1280–1288. [Google Scholar] [CrossRef] [PubMed]
- Howard, K.A.; Rahbek, U.L.; Liu, X.; Damgaard, C.K.; Glud, S.Z.; Andersen, M.Ø.; Hovgaard, M.B.; Schmitz, A.; Nyengaard, J.R.; Besenbacher, F.; et al. RNA Interference in Vitro and in Vivo Using a Novel Chitosan/siRNA Nanoparticle System. Mol. Ther. 2006, 14, 476–484. [Google Scholar] [CrossRef]
- Chen, J.; Fan, T.; Xie, Z.; Zeng, Q.; Xue, P.; Zheng, T.; Chen, Y.; Luo, X.; Zhang, H. Advances in nanomaterials for photodynamic therapy applications: Status and challenges. Biomaterials 2020, 237, 119827. [Google Scholar] [CrossRef]
- Kuo, W.S.; Chang, Y.T.; Cho, K.C.; Chiu, K.C.; Lien, C.H.; Yeh, C.S.; Chen, S.J. Gold nanomaterials conjugated with indocyanine green for dual-modality photodynamic and photothermal therapy. Biomaterials 2012, 33, 3270–3278. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, K.M.; Niloy, M.S.; Shakil, M.S.; Islam, M.A. Ruthenium Complexes: An Alternative to Platinum Drugs in Colorectal Cancer Treatment. Pharmaceutics 2021, 13, 1295. [Google Scholar] [CrossRef] [PubMed]
- Calixto, G.M.F.; de Annunzio, S.R.; Victorelli, F.D.; Frade, M.L.; Ferreira, P.S.; Chorilli, M.; Fontana, C.R. Chitosan-Based Drug Delivery Systems for Optimization of Photodynamic Therapy: A Review. AAPS PharmSciTech 2019, 20, 253. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Shen, R.; Bao, L.; Wang, C.; Yuan, H. Chitosan derived glycolipid nanoparticles for magnetic resonance imaging guided photodynamic therapy of cancer. Carbohydr. Polym. 2020, 245, 116509. [Google Scholar] [CrossRef]
- Pandya, A.D.; Øverbye, A.; Sahariah, P.; Gaware, V.S.; Høgset, H.; Masson, M.; Høgset, A.; Mælandsmo, G.M.; Skotland, T.; Sandvig, K.; et al. Drug-Loaded Photosensitizer-Chitosan Nanoparticles for Combinatorial Chemo- and Photodynamic-Therapy of Cancer. Biomacromolecules 2020, 21, 1489–1498. [Google Scholar] [CrossRef]
- Jia, H.R.; Jiang, Y.W.; Zhu, Y.X.; Li, Y.H.; Wang, H.Y.; Han, X.; Yu, Z.W.; Gu, N.; Liu, P.; Chen, Z.; et al. Plasma membrane activatable polymeric nanotheranostics with self-enhanced light-triggered photosensitizer cellular influx for photodynamic cancer therapy. J. Control. Release 2017, 255, 231–241. [Google Scholar] [CrossRef]
- Zhang, R.; Qin, X.; Kong, F.; Chen, P.; Pan, G. Improving cellular uptake of therapeutic entities through interaction with components of cell membrane. Drug Deliv. 2019, 26, 328–342. [Google Scholar] [CrossRef] [Green Version]
- Winiwarter, S.; Ridderström, M.; Ungell, A.-L.; Andersson, T.; Zamora, I. Use of molecular descriptors for absorption, distribution, metabolism, and excretion predictions. In Comprehensive Medicinal Chemistry II; Taylor, J.B., Ed.; Elsevier: Amsterdam, The Netherlands, 2007; Volume 5, pp. 531–554. [Google Scholar]
- Liu, Y.; Sun, M.; Wang, T.; Chen, X.; Wang, H. Chitosan-based self-assembled nanomaterials: Their application in drug delivery. View 2021, 2, 20200069. [Google Scholar] [CrossRef]
- Xu, J.; Zhao, Q.; Jin, Y.; Qiu, L. High loading of hydrophilic/hydrophobic doxorubicin into polyphosphazene polymersome for breast cancer therapy. Nanomedicine 2014, 10, 349–358. [Google Scholar] [CrossRef]
- Yun, U.J.; Lee, J.H.; Shim, J.; Yoon, K.; Goh, S.H.; Yi, E.H.; Ye, S.K.; Lee, J.S.; Lee, H.; Park, J.; et al. Anti-cancer effect of doxorubicin is mediated by downregulation of HMG-Co A reductase via inhibition of EGFR/Src pathway. Lab. Investig. 2019, 99, 1157–1172. [Google Scholar] [CrossRef]
- Yousefpour, P.; Atyabi, F.; Vasheghani-Farahani, E.; Movahedi, A.A.; Dinarvand, R. Targeted delivery of doxorubicin-utilizing chitosan nanoparticles surface-functionalized with anti-Her2 trastuzumab. Int. J. Nanomed. 2011, 6, 1977–1990. [Google Scholar]
- Zou, Y.; Liu, P.; Liu, C.H.; Zhi, X.T. Doxorubicin-loaded mesoporous magnetic nanoparticles to induce apoptosis in breast cancer cells. Biomed. Pharm. 2015, 69, 355–360. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, Y.S.; Kim, S.; Park, J.H.; Kim, K.; Choi, K.; Chung, H.; Jeong, S.Y.; Park, R.W.; Kim, I.S.; et al. Hydrophobically modified glycol chitosan nanoparticles as carriers for paclitaxel. J. Control. Release 2006, 111, 228–234. [Google Scholar] [CrossRef]
- Wu, C.; Gao, Y.; Liu, Y.; Xu, X. Pure paclitaxel nanoparticles: Preparation, characterization, and antitumor effect for human liver cancer SMMC-7721 cells. Int. J. Nanomed. 2018, 13, 6189–6198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trickler, W.J.; Nagvekar, A.A.; Dash, A.K. A novel nanoparticle formulation for sustained paclitaxel delivery. AAPS PharmSciTech 2008, 9, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Dasari, S.; Tchounwou, P.B. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur. J. Pharm. 2014, 740, 364–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.H.; Kim, Y.S.; Park, K.; Lee, S.; Nam, H.Y.; Min, K.H.; Jo, H.G.; Park, J.H.; Choi, K.; Jeong, S.Y.; et al. Antitumor efficacy of cisplatin-loaded glycol chitosan nanoparticles in tumor-bearing mice. J. Control. Release 2008, 127, 41–49. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wang, Y.; Meng, X. Chitosan Nanolayered Cisplatin-Loaded Lipid Nanoparticles for Enhanced Anticancer Efficacy in Cervical Cancer. Nanoscale Res. Lett. 2016, 11, 524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, N.; Yin, Y.; Xu, S.J.; Chen, W.S. 5-Fluorouracil: Mechanisms of resistance and reversal strategies. Molecules 2008, 13, 1551–1569. [Google Scholar] [CrossRef] [Green Version]
- Zorrilla-Veloz, R.I.; Stelzer, T.; López-Mejías, V. Measurement and Correlation of the Solubility of 5-Fluorouracil in Pure and Binary Solvents. J. Chem. Eng. Data 2018, 63, 3809–3817. [Google Scholar] [CrossRef]
- Raida, M.; Schwabe, W.; Häusler, P.; Van Kuilenburg, A.B.; Van Gennip, A.H.; Behnke, D.; Höffken, K. Prevalence of a common point mutation in the dihydropyrimidine dehydrogenase (DPD) gene within the 5’-splice donor site of intron 14 in patients with severe 5-fluorouracil (5-FU)- related toxicity compared with controls. Clin. Cancer Res. An. Off. J. Am. Assoc. Cancer Res. 2001, 7, 2832–2839. [Google Scholar]
- Rajan, M.; Raj, V.; Al-Arfaj, A.A.; Murugan, A.M. Hyaluronidase enzyme core-5-fluorouracil-loaded chitosan-PEG-gelatin polymer nanocomposites as targeted and controlled drug delivery vehicles. Int. J. Pharm. 2013, 453, 514–522. [Google Scholar] [CrossRef]
- Cavalli, R.; Leone, F.; Minelli, R.; Fantozzi, R.; Dianzani, C. New chitosan nanospheres for the delivery of 5-fluorouracil: Preparation, characterization and in vitro studies. Curr. Drug Deliv. 2014, 11, 270–278. [Google Scholar] [CrossRef] [Green Version]
- Jain, A.; Thakur, K.; Sharma, G.; Kush, P.; Jain, U.K. Fabrication, characterization and cytotoxicity studies of ionically cross-linked docetaxel loaded chitosan nanoparticles. Carbohydr. Polym. 2016, 137, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Ishida, S.; Masuguchi, K.; Kawashiri, T.; Tsuji, T.; Watanabe, H.; Akiyoshi, S.; Kubo, M.; Masuda, S.; Egashira, N. Effects of Diluent Volume and Administration Time on the Incidence of Anaphylaxis Following Docetaxel Therapy in Breast Cancer. Biol. Pharm. Bull. 2020, 43, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Zhou, C.; Liu, Y.; Su, K.; Jia, L.; Chen, L.; Li, M.; Ma, J.; Zhou, W.; Zhang, S.; et al. Genetic associations of docetaxel-based chemotherapy-induced myelosuppression in Chinese Han population. J. Clin. Pharm. 2020, 45, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Attwood, D. Surfactant Systems: Their Chemistry, Pharmacy and Biology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Kaur, P.; Garg, T.; Rath, G.; Murthy, R.S.; Goyal, A.K. Surfactant-based drug delivery systems for treating drug-resistant lung cancer. Drug Deliv. 2016, 23, 727–738. [Google Scholar] [CrossRef]
- Alshraim, M.O.; Sangi, S.; Harisa, G.I.; Alomrani, A.H.; Yusuf, O.; Badran, M.M. Chitosan-Coated Flexible Liposomes Magnify the Anticancer Activity and Bioavailability of Docetaxel: Impact on Composition. Molecules 2019, 24, 250. [Google Scholar] [CrossRef] [Green Version]
- Scheeren, L.E.; Nogueira, D.R.; Macedo, L.B.; Vinardell, M.P.; Mitjans, M.; Infante, M.R.; Rolim, C.M. PEGylated and poloxamer-modified chitosan nanoparticles incorporating a lysine-based surfactant for pH-triggered doxorubicin release. Colloids Surf. B Biointerfaces 2016, 138, 117–127. [Google Scholar] [CrossRef] [Green Version]
- Kamel, K.M.; Khalil, I.A.; Rateb, M.E.; Elgendy, H.; Elhawary, S. Chitosan-Coated Cinnamon/Oregano-Loaded Solid Lipid Nanoparticles to Augment 5-Fluorouracil Cytotoxicity for Colorectal Cancer: Extract Standardization, Nanoparticle Optimization, and Cytotoxicity Evaluation. J. Agric. Food Chem. 2017, 65, 7966–7981. [Google Scholar] [CrossRef]
- Tsirigotis-Maniecka, M.; Gancarz, R.; Wilk, K.A. Preparation and characterization of sodium alginate/chitosan microparticles containing esculin. Colloids Surf. A Physicochem. Eng. Asp. 2016, 510, 22–32. [Google Scholar] [CrossRef]
- Zhi, J.; Wang, Y.; Luo, G. Adsorption of diuretic furosemide onto chitosan nanoparticles prepared with a water-in-oil nanoemulsion system. React. Funct. Polym. 2005, 65, 249–257. [Google Scholar] [CrossRef]
- Paques, J.P.; van der Linden, E.; van Rijn, C.J.; Sagis, L.M. Alginate submicron beads prepared through w/o emulsification and gelation with CaCl2 nanoparticles. Food Hydrocoll. 2013, 31, 428–434. [Google Scholar] [CrossRef]
- Rodrıguez, M.; Albertengo, L.; Agulló, E. Emulsification capacity of chitosan. Carbohydr. Polym. 2002, 48, 271–276. [Google Scholar] [CrossRef]
- Schulz, P.; Rodriguez, M.; Del Blanco, L.; Pistonesi, M.; Agullo, E. Emulsification properties of chitosan. Colloid Polym. Sci. 1998, 276, 1159–1165. [Google Scholar] [CrossRef]
- Trickler, W.J.; Khurana, J.; Nagvekar, A.A.; Dash, A.K. Chitosan and glyceryl monooleate nanostructures containing gemcitabine: Potential delivery system for pancreatic cancer treatment. AAPS PharmSciTech 2010, 11, 392–401. [Google Scholar] [CrossRef] [Green Version]
- Natesan, S.; Sugumaran, A.; Ponnusamy, C.; Thiagarajan, V.; Palanichamy, R.; Kandasamy, R. Chitosan stabilized camptothecin nanoemulsions: Development, evaluation and biodistribution in preclinical breast cancer animal mode. Int. J. Biol. Macromol 2017, 104, 1846–1852. [Google Scholar] [CrossRef] [PubMed]
- Rosch, J.G.; Winter, H.; DuRoss, A.N.; Sahay, G.; Sun, C. Inverse-micelle synthesis of doxorubicin-loaded alginate/chitosan nanoparticles and in vitro assessment of breast cancer cytotoxicity. Colloid Interface Sci. Commun. 2019, 28, 69–74. [Google Scholar] [CrossRef]
- Li, W.; Zhu, X.; Zhou, X.; Wang, X.; Zhai, W.; Li, B.; Du, J.; Li, G.; Sui, X.; Wu, Y.; et al. An orally available PD-1/PD-L1 blocking peptide OPBP-1-loaded trimethyl chitosan hydrogel for cancer immunotherapy. J. Control. Release 2021, 334, 376–388. [Google Scholar] [CrossRef]
- Rezakhani, L.; Alizadeh, M.; Alizadeh, A. A three dimensional in vivo model of breast cancer using a thermosensitive chitosan-based hydrogel and 4 T1 cell line in Balb/c. J. Biomed. Mater. Res. A 2021, 109, 1275–1285. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, W.; Zhao, J.; Wu, C.; Ye, C.; Huang, M.; Wang, S. Preparation of injectable temperature-sensitive chitosan-based hydrogel for combined hyperthermia and chemotherapy of colon cancer. Carbohydr. Polym. 2019, 222, 115039. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Qian, J.; Zhang, Y.; Xu, W.; Xiao, J.; Suo, A. Growth of MCF-7 breast cancer cells and efficacy of anti-angiogenic agents in a hydroxyethyl chitosan/glycidyl methacrylate hydrogel. Cancer Cell Int. 2017, 17, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Zhang, P.; Shan, W.; Gao, J.; Liang, W. A novel chitosan-based thermosensitive hydrogel containing doxorubicin liposomes for topical cancer therapy. J. Biomater. Sci. Polym. Ed. 2013, 24, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- McCormick, M.; Friehling, E.; Kalpatthi, R.; Siripong, N.; Smith, K. Cost-effectiveness of levofloxacin prophylaxis against bacterial infection in pediatric patients with acute myeloid leukemia. Pediatr. Blood Cancer 2020, 67, e28469. [Google Scholar] [CrossRef]
- Topalian, S.L.; Taube, J.M.; Pardoll, D.M. Neoadjuvant checkpoint blockade for cancer immunotherapy. Science 2020, 367, 6547. [Google Scholar] [CrossRef]
- Wu, Q.; Jiang, L.; Li, S.C.; He, Q.J.; Yang, B.; Cao, J. Small molecule inhibitors targeting the PD-1/PD-L1 signaling pathway. Acta Pharm. Sin. 2021, 42, 1–9. [Google Scholar] [CrossRef]
- Pan, C.; Yang, H.; Lu, Y.; Hu, S.; Wu, Y.; He, Q.; Dong, X. Recent advance of peptide-based molecules and nonpeptidic small-molecules modulating PD-1/PD-L1 protein-protein interaction or targeting PD-L1 protein degradation. Eur J. Med. Chem. 2021, 213, 113170. [Google Scholar] [CrossRef]
- O’Donnell, J.S.; Hoefsmit, E.P.; Smyth, M.J.; Blank, C.U.; Teng, M.W.L. The Promise of Neoadjuvant Immunotherapy and Surgery for Cancer Treatment. Clin. Cancer Res. An. Off. J. Am. Assoc. Cancer Res. 2019, 25, 5743–5751. [Google Scholar] [CrossRef]
- Maiyo, F.; Singh, M. Folate-Targeted mRNA Delivery Using Chitosan-Functionalized Selenium Nanoparticles: Potential in Cancer Immunotherapy. Pharmaceuticals 2019, 12, 164. [Google Scholar] [CrossRef] [Green Version]
- Zaharoff, D.A.; Hance, K.W.; Rogers, C.J.; Schlom, J.; Greiner, J.W. Intratumoral immunotherapy of established solid tumors with chitosan/IL-12. J. Immunother. 2010, 33, 697–705. [Google Scholar] [CrossRef] [Green Version]
- Vo, J.L.; Yang, L.; Kurtz, S.L.; Smith, S.G.; Koppolu, B.P.; Ravindranathan, S.; Zaharoff, D.A. Neoadjuvant immunotherapy with chitosan and interleukin-12 to control breast cancer metastasis. Oncoimmunology 2014, 3, e968001. [Google Scholar] [CrossRef] [Green Version]
- Carroll, E.C.; Jin, L.; Mori, A.; Muñoz-Wolf, N.; Oleszycka, E.; Moran, H.B.T.; Mansouri, S.; McEntee, C.P.; Lambe, E.; Agger, E.M.; et al. The Vaccine Adjuvant Chitosan Promotes Cellular Immunity via DNA Sensor cGAS-STING-Dependent Induction of Type I Interferons. Immunity 2016, 44, 597–608. [Google Scholar] [CrossRef] [Green Version]
- Zaharoff, D.A.; Hoffman, B.S.; Hooper, H.B.; Benjamin, C.J., Jr.; Khurana, K.K.; Hance, K.W.; Rogers, C.J.; Pinto, P.A.; Schlom, J.; Greiner, J.W. Intravesical immunotherapy of superficial bladder cancer with chitosan/interleukin-12. Cancer Res. 2009, 69, 6192–6199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, S.G.; Koppolu, B.P.; Ravindranathan, S.; Kurtz, S.L.; Yang, L.; Katz, M.D.; Zaharoff, D.A. Intravesical chitosan/interleukin-12 immunotherapy induces tumor-specific systemic immunity against murine bladder cancer. Cancer Immunol. Immunother. 2015, 64, 689–696. [Google Scholar] [CrossRef] [Green Version]
- Dodane, V.; Amin Khan, M.; Merwin, J.R. Effect of chitosan on epithelial permeability and structure. Int. J. Pharm. 1999, 182, 21–32. [Google Scholar] [CrossRef]
- Smith, S.G.; Baltz, J.L.; Koppolu, B.P.; Ravindranathan, S.; Nguyen, K.; Zaharoff, D.A. Immunological mechanisms of intravesical chitosan/interleukin-12 immunotherapy against murine bladder cancer. Oncoimmunology 2017, 6, e1259050. [Google Scholar] [CrossRef]
- Santos, P.M.; Butterfield, L.H. Dendritic Cell-Based Cancer Vaccines. J. Immunol. 2018, 200, 443–449. [Google Scholar] [CrossRef]
- Han, H.D.; Byeon, Y.; Jang, J.H.; Jeon, H.N.; Kim, G.H.; Kim, M.G.; Pack, C.G.; Kang, T.H.; Jung, I.D.; Lim, Y.T.; et al. In vivo stepwise immunomodulation using chitosan nanoparticles as a platform nanotechnology for cancer immunotherapy. Sci. Rep. 2016, 6, 38348. [Google Scholar] [CrossRef] [Green Version]
- Choi, J.J.; Le, Q.V.; Kim, D.; Kim, Y.B.; Shim, G.; Oh, Y.K. High Molecular Weight Chitosan-Complexed RNA Nanoadjuvant for Effective Cancer Immunotherapy. Pharmaceutics 2019, 11, 680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro, F.; Pinto, M.L.; Pereira, C.L.; Serre, K.; Barbosa, M.A.; Vermaelen, K.; Gärtner, F.; Gonçalves, R.M.; De Wever, O.; Oliveira, M.J. Chitosan/γ-PGA nanoparticles-based immunotherapy as adjuvant to radiotherapy in breast cancer. Biomaterials 2020, 257, 120218. [Google Scholar] [CrossRef]
- Kumar, P.; Srivastava, R. IR 820 dye encapsulated in polycaprolactone glycol chitosan: Poloxamer blend nanoparticles for photo immunotherapy for breast cancer. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 57, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Du, Q.; Liu, T.; Tan, L.; Niu, M.; Gao, L.; Huang, Z.; Fu, C.; Ma, T.; Meng, X.; et al. In Vivo Magnetic Resonance Imaging and Microwave Thermotherapy of Cancer Using Novel Chitosan Microcapsules. Nanoscale Res. Lett. 2016, 11, 334. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Lovell, J.F.; Yoon, J.; Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674. [Google Scholar] [CrossRef] [PubMed]
- Bakht, M.K.; Sadeghi, M.; Pourbaghi-Masouleh, M.; Tenreiro, C. Scope of nanotechnology-based radiation therapy and thermotherapy methods in cancer treatment. Curr. Cancer Drug Targets 2012, 12, 998–1015. [Google Scholar] [CrossRef] [PubMed]
- Niloy, M.S.; Shakil, M.S.; Hossen, M.S.; Alam, M.; Rosengren, R.J. Promise of gold nanomaterials as a lung cancer theranostic agent: A systematic review. Int. Nano Lett. 2021, 11, 93–111. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).