Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation
Abstract
:1. Introduction
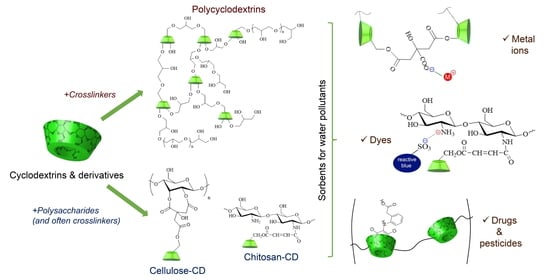
2. Cyclodextrin-Based Polymers in Pollutant Removal
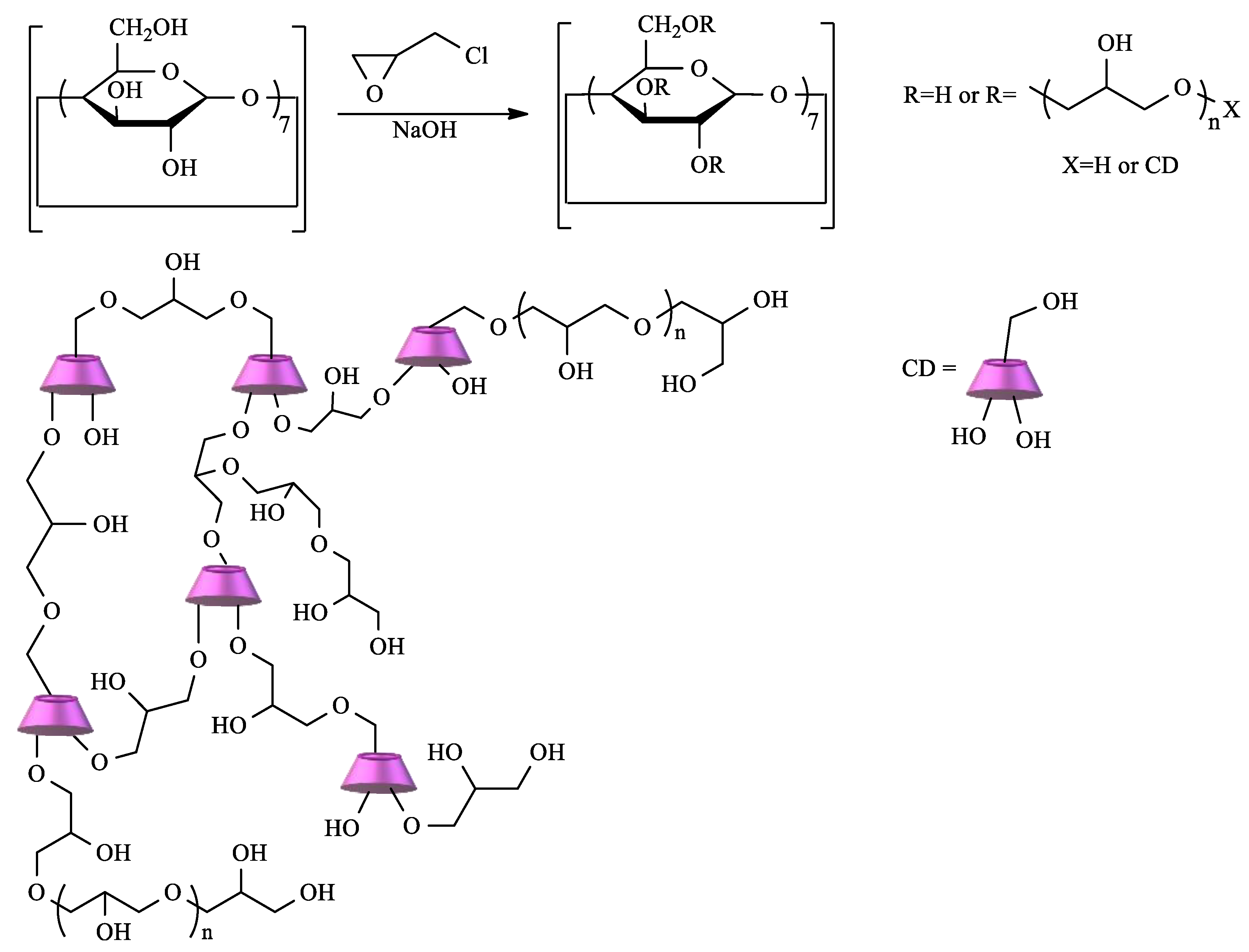
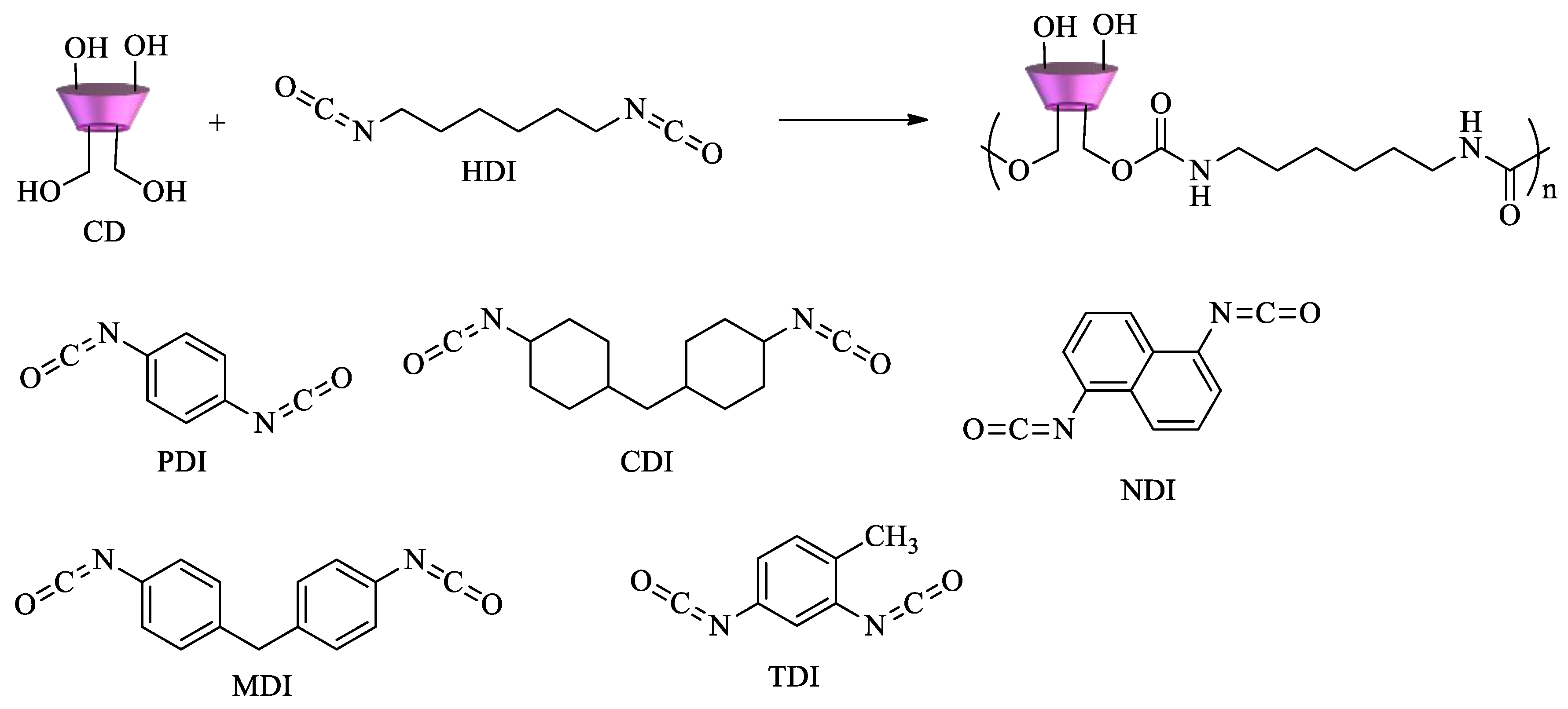
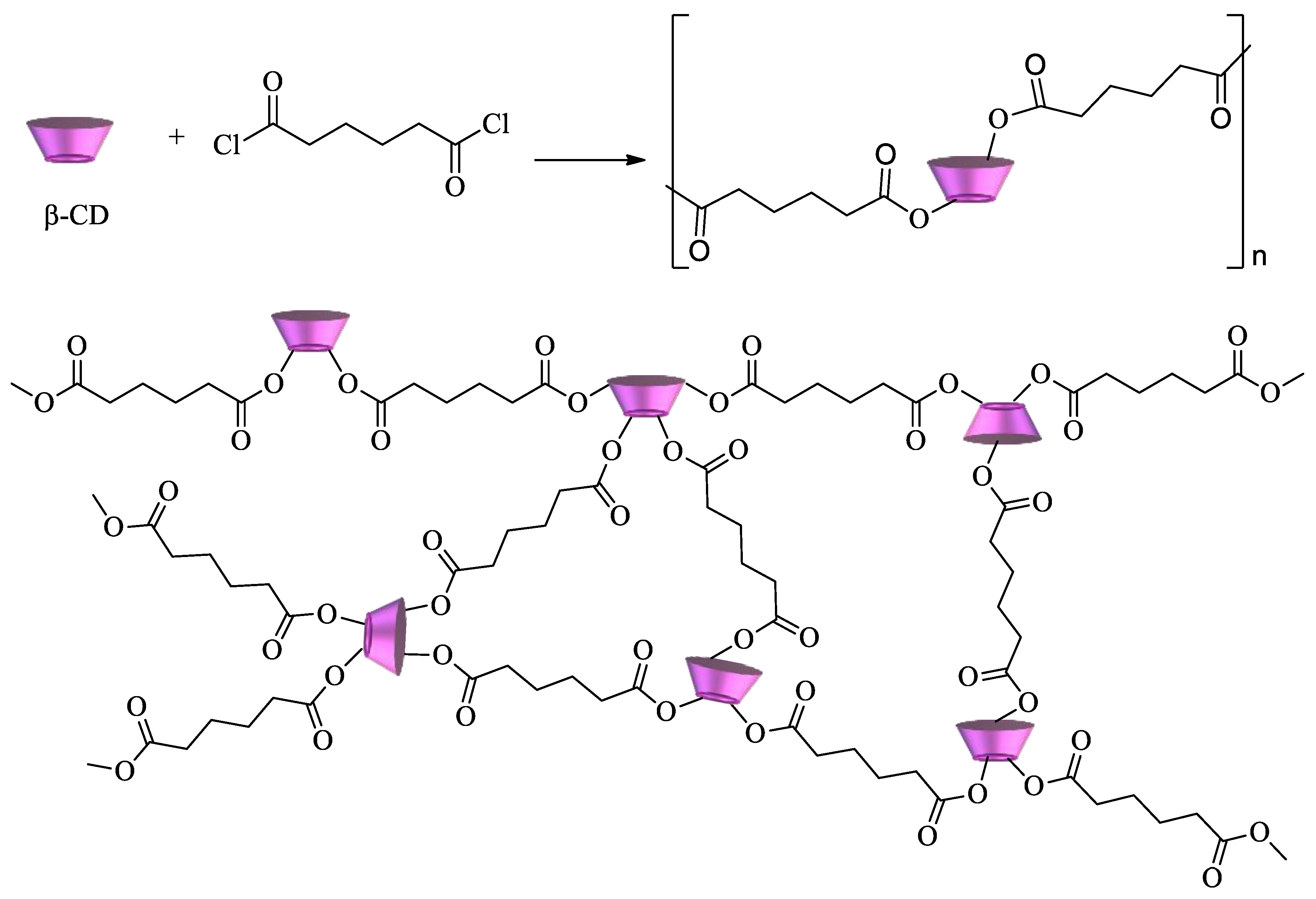
2.1. Synthesis and Properties of CD-Based Materials
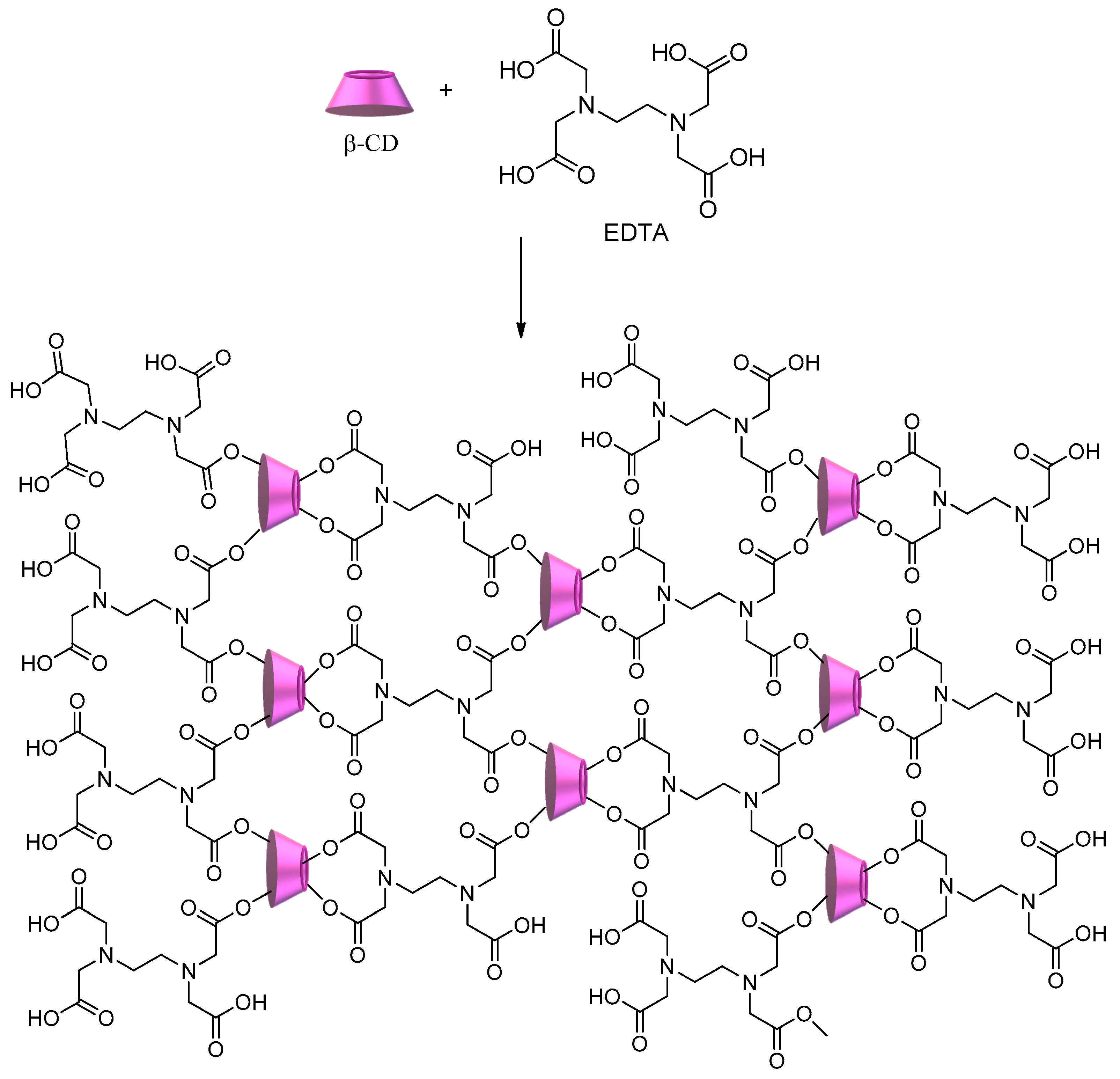
2.1.1. Polycyclodextrins
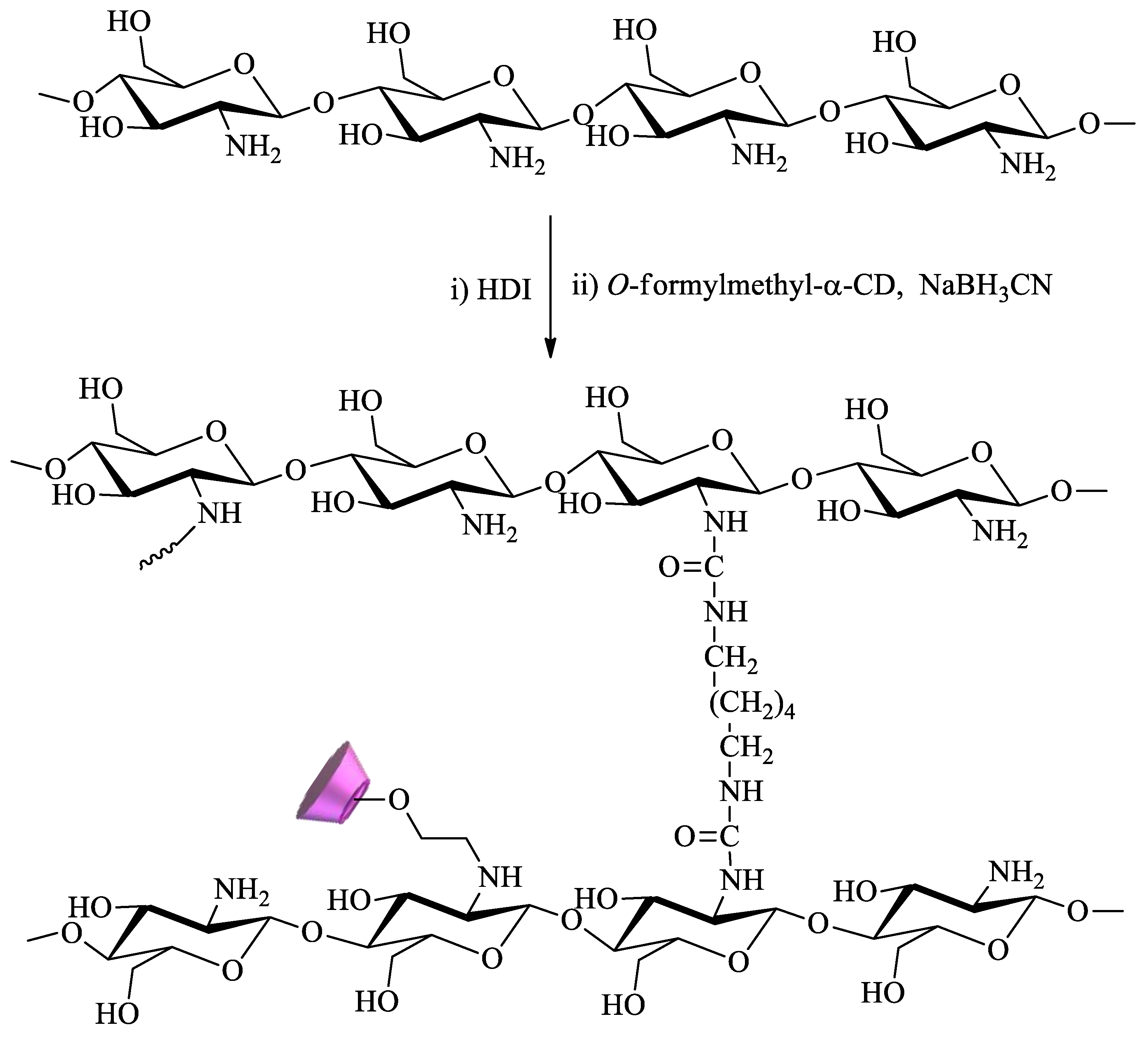
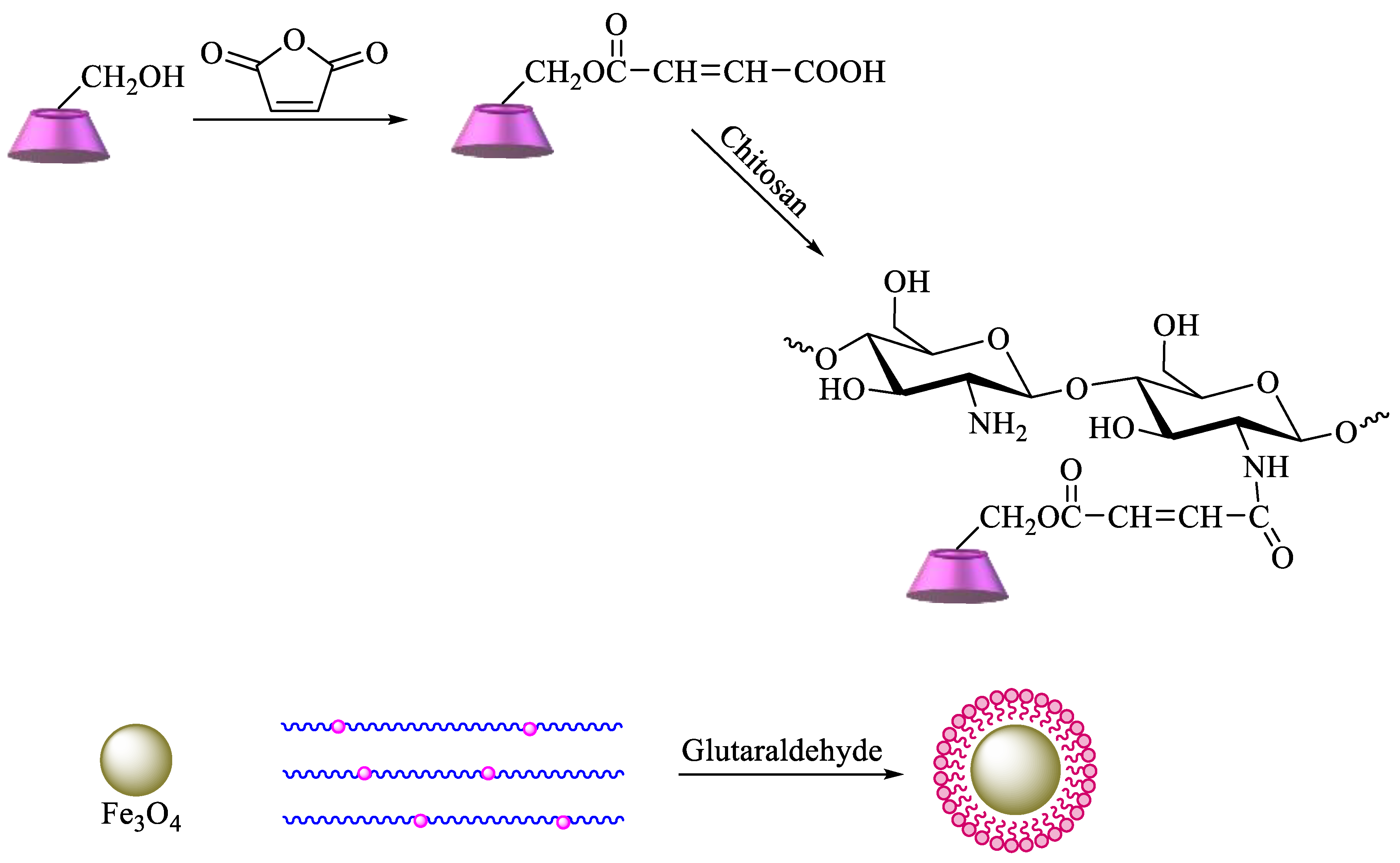
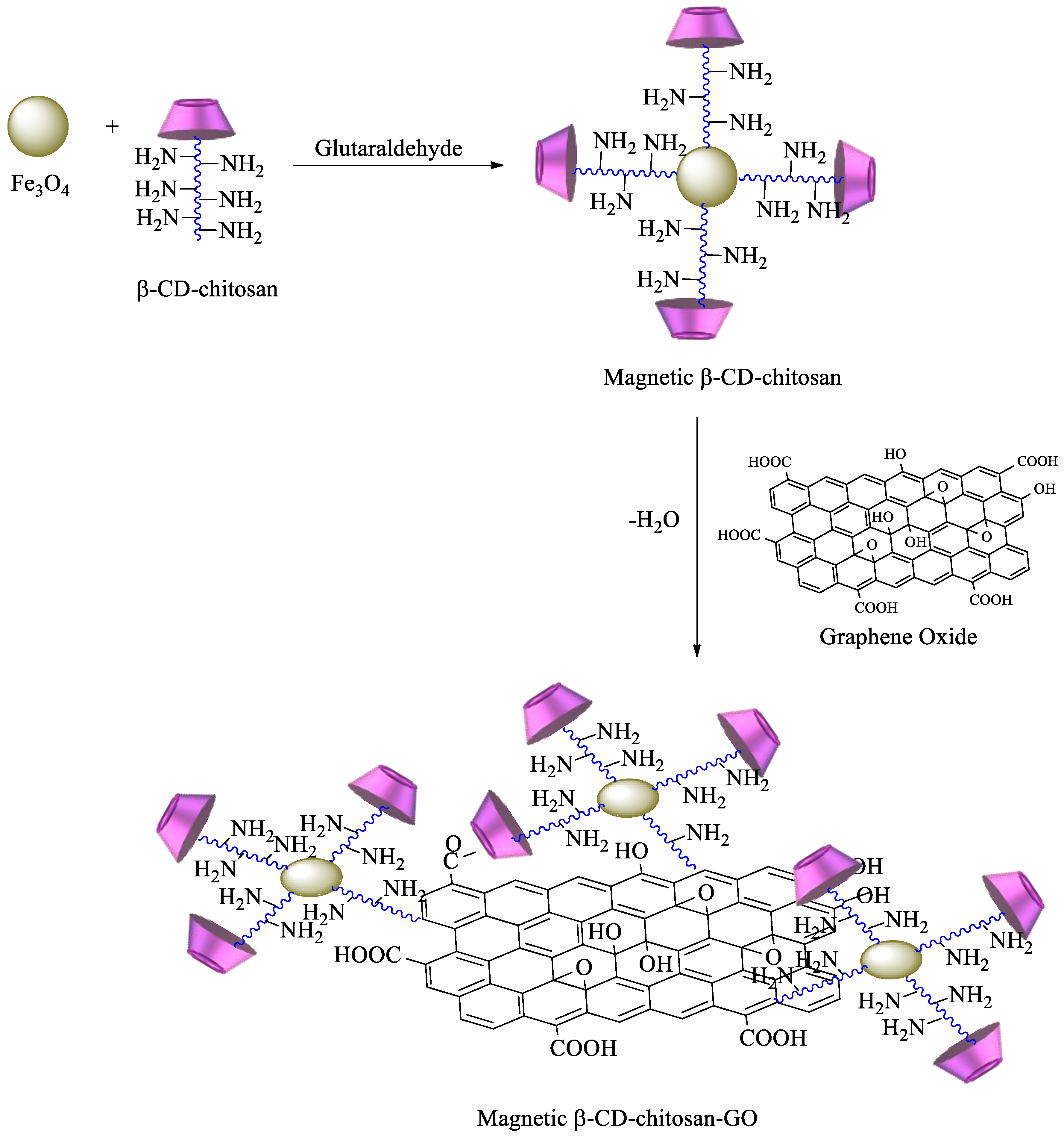
2.1.2. Polysaccharides Having CDs as Pendant Groups
3. Removal of Pollutants from Water and Wastewaters
3.1. Metal Ions
3.2. Dyes
3.2.1. CD Polymers
3.2.2. Chitosan-Based Sorbents
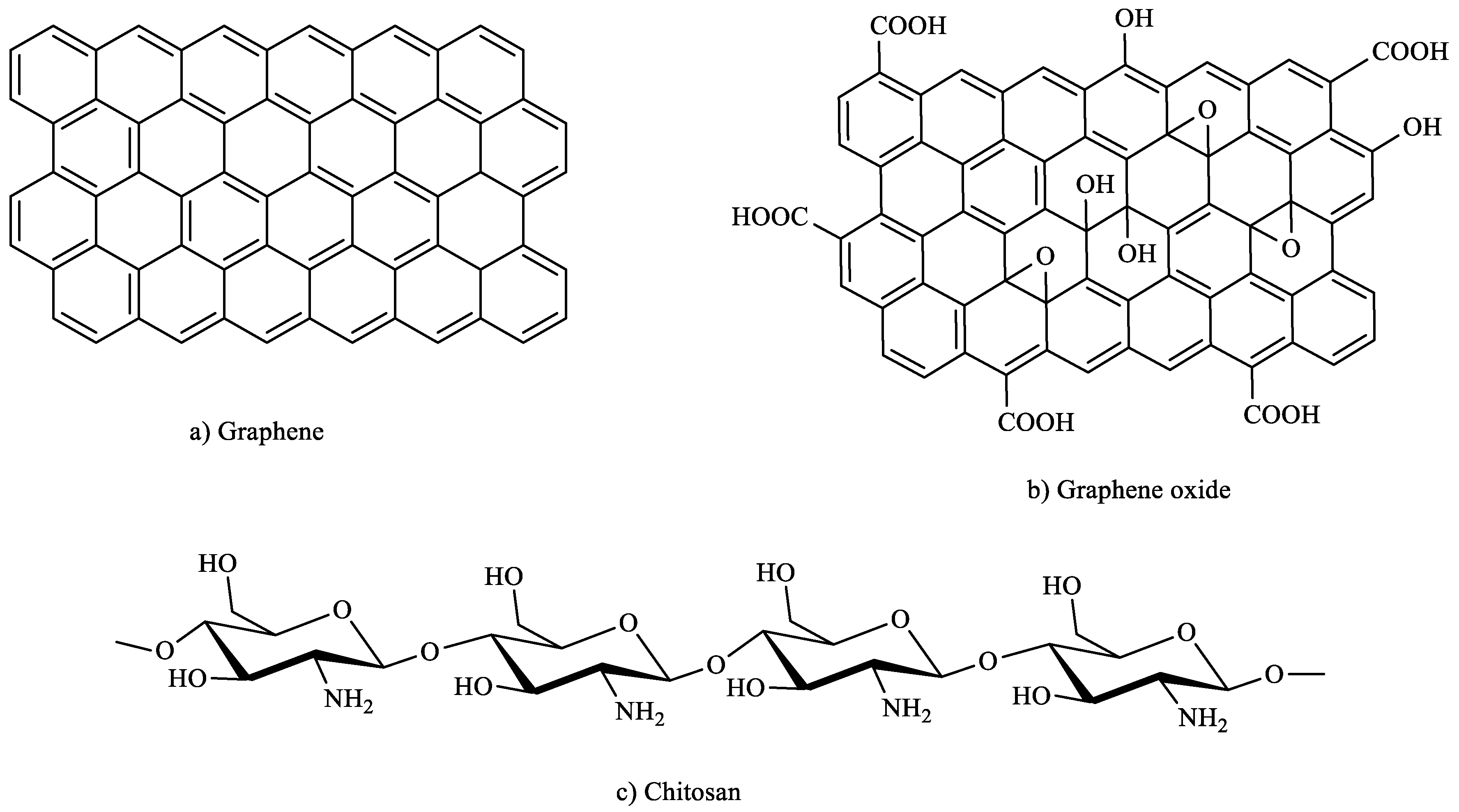
3.2.3. Cellulose-Based Sorbents
3.3. Pesticides
3.4. Pharmaceutical Compounds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BPA | bisphenol A |
| CBZ | carbamazepine |
| CD | cyclodextrin |
| CMC | carboxymethylcellulose |
| EDTA | ethylenediamine tetraacetic acid |
| EPI | epichlorohydrin |
| GO | graphene oxide |
| HDI | 1,6-hexamethylene diisocyanate |
| HP-β-CD | hydroxypropyl-β-cyclodextrin |
| MB | methylene blue |
| MDI | 4,4′-diphenylmethane diisocyanate |
| NDI | 1,5-naphtalene diisocyanate |
| PCMX | chloroxylenol |
| PDI | 1,4-phenylene diisocyanate |
| PS | pine sawdust |
| TDI | 2,4-toluene diisocyanate |
References
- European Commission. Commission Outlines Road to Zero Pollution Action Plan; European Commission: Brussels, Belgium, 2020. [Google Scholar]
- Wakeham, H. Zero Pollutions Conference 2020. Available online: https://wwtonline.co.uk/news/collaborative-approach-needed-to-achieve-zero-pollution (accessed on 2 November 2020).
- Corsi, I.; Fiorati, A.; Grassi, G.; Bartolozzi, I.; Daddi, T.; Melone, L.; Punta, C. Environmentally sustainable and ecosafe polysaccharide-based materials for water nano-treatment: An eco-design study. Materials 2018, 11, 1228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morin-Crini, N.; Winterton, P.; Fourmentin, S.; Wilson, L.D.; Fenyvesi, É.; Crini, G. Water-insoluble β-cyclodextrin–epichlorohydrin polymers for removal of pollutants from aqueous solutions by sorption processes using batch studies: A review of inclusion mechanisms. Prog. Polym. Sci. 2018, 78, 1–23. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Fourmentin, M.; Fourmentin, S.; Torri, G.; Crini, G. Synthesis of silica materials containing cyclodextrin and their applications in wastewater treatment. Environ. Chem. Lett. 2019, 17, 683–696. [Google Scholar] [CrossRef]
- Alaba, P.A.; Oladoja, N.A.; Sani, Y.M.; Ayodele, O.B.; Mohammed, I.Y.; Olupinla, S.F.; Daud, W.M.W. Insight into wastewater decontamination using polymeric adsorbents. J. Environ. Chem. Eng. 2018, 6, 1651–1672. [Google Scholar] [CrossRef]
- Marican, A.; Durán-Lara, E.F. A review on pesticide removal through different processes. Environ. Sci. Pollut. Res. 2018, 25, 2051–2064. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cui, F.; Pan, Y.; Hou, L.; Zhang, B.; Li, Y.; Zhu, L. Hierarchically micro-mesoporous β-cyclodextrin polymers used for ultrafast removal of micropollutants from water. Carbohydr. Polym. 2019, 213, 352–360. [Google Scholar] [CrossRef]
- Yang, C.; Huang, H.; Ji, T.; Zhang, K.; Yuan, L.; Zhou, C.; Tang, K.; Yi, J.; Chen, X. A cost-effective crosslinked β-cyclodextrin polymer for the rapid and efficient removal of micropollutants from wastewater. Polym. Int. 2019, 68, 805–811. [Google Scholar] [CrossRef]
- Zhang, S.; He, Y.; Wu, L.; Wan, J.; Ye, M.; Long, T.; Yan, Z.; Jiang, X.; Lin, Y.; Lu, X. Remediation of Organochlorine Pesticide-Contaminated Soils by Surfactant-Enhanced Washing Combined with Activated Carbon Selective Adsorption. Pedosphere 2019, 29, 400–408. [Google Scholar] [CrossRef]
- Guerra, F.D.; Attia, M.F.; Whitehead, D.C.; Alexis, F. Nanotechnology for Environmental Remediation: Materials and Applications. Molecules 2018, 23, 1760. [Google Scholar] [CrossRef] [Green Version]
- Salazar, S.; Guerra, D.; Yutronic, N.; Jara, P. Removal of aromatic chlorinated pesticides from aqueous solution using β-cyclodextrin polymers decorated with Fe3O4 nanoparticles. Polymers 2018, 10, 1038. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Liu, M.; Jia, J.; Wu, D.; Gao, T.; Wang, X.; Yu, J.; Li, F. β-Cyclodextrin-based hollow nanoparticles with excellent adsorption performance towards organic and inorganic pollutants. Nanoscale 2019, 11, 18653–18661. [Google Scholar] [CrossRef] [PubMed]
- Cova, T.F.G.G.; Murtinho, D.; Pais, A.A.C.C.; Valente, A.J.M. Cyclodextrin-based Materials for Removing Micropollutants From Wastewater. Curr. Org. Chem. 2018, 22, 2150–2181. [Google Scholar] [CrossRef]
- Landy, D.; Mallard, I.; Ponchel, A.; Monflier, E.; Fourmentin, S. Remediation technologies using cyclodextrins: An overview. Environ. Chem. Lett. 2012, 10, 225–237. [Google Scholar] [CrossRef]
- Murcia-Salvador, A.; Pellicer, J.A.; Fortea, M.I.; Gómez-López, V.M.; Rodríguez-López, M.I.; Núñez-Delicado, E.; Gabaldón, J.A. Adsorption of Direct Blue 78 Using Chitosan and Cyclodextrins as Adsorbents. Polymers 2019, 11, 1003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pellicer, J.; Rodríguez-López, M.; Fortea, M.; Lucas-Abellán, C.; Mercader-Ros, M.; López-Miranda, S.; Gómez-López, V.; Semeraro, P.; Cosma, P.; Fini, P.; et al. Adsorption Properties of β- and Hydroxypropyl-β-Cyclodextrins Cross-Linked with Epichlorohydrin in Aqueous Solution. A Sustainable Recycling Strategy in Textile Dyeing Process. Polymers 2019, 11, 252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morin-Crini, N.; Crini, G. Environmental applications of water-insoluble β-cyclodextrin–epichlorohydrin polymers. Prog. Polym. Sci. 2013, 38, 344–368. [Google Scholar] [CrossRef]
- Crini, G.; Fourmentin, S.; Fenyvesi, É.; Torri, G.; Fourmentin, M.; Morin-Crini, N. Fundamentals and applications of cyclodextrins. In Cyclodextrin Fundamentals, Reactivity and Analysis; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–55. [Google Scholar]
- Crini, G. Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment. Prog. Polym. Sci. 2005, 30, 38–70. [Google Scholar] [CrossRef]
- Cova, T.F.; Murtinho, D.; Pais, A.A.C.C.; Valente, A.J.M. Combining Cellulose and Cyclodextrins: Fascinating Designs for Materials and Pharmaceutics. Front. Chem. 2018, 6. [Google Scholar] [CrossRef]
- Cova, T.F.; Milne, B.F.; Pais, A.A.C.C. Host flexibility and space filling in supramolecular complexation of cyclodextrins: A free-energy-oriented approach. Carbohydr. Polym. 2019, 205, 42–54. [Google Scholar] [CrossRef]
- Cova, T.F.G.G.; Milne, B.F.; Nunes, S.C.C.; Pais, A.A.C.C. Drastic Stabilization of Junction Nodes in Supramolecular Structures Based on Host–Guest Complexes. Macromolecules 2018, 51, 2732–2741. [Google Scholar] [CrossRef]
- Kono, H.; Onishi, K.; Nakamura, T. Characterization and bisphenol A adsorption capacity of beta-cyclodextrin-carboxymethylcellulose-based hydrogels. Carbohydr. Polym. 2013, 98, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, P.; Hu, F.; Zhao, Y.; Zhu, L. A crosslinked beta-cyclodextrin polymer used for rapid removal of a broad-spectrum of organic micropollutants from water. Carbohydr. Polym. 2017, 177, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liu, B.; Xu, J.; Pan, K.; Hou, H.; Hu, J.; Yang, J. Cross-linked chitosan/beta-cyclodextrin composite for selective removal of methyl orange: Adsorption performance and mechanism. Carbohydr. Polym. 2018, 182, 106–114. [Google Scholar] [CrossRef]
- Wycisk, A.; Döring, A.; Schneider, M.; Schönhoff, M.; Kuckling, D. Synthesis of β-cyclodextrin-based star block copolymers with thermo-responsive behavior. Polymers 2015, 7, 921–938. [Google Scholar] [CrossRef] [Green Version]
- Bhattarai, B.; Muruganandham, M.; Suri, R.P.S. Development of high efficiency silica coated beta-cyclodextrin polymeric adsorbent for the removal of emerging contaminants of concern from water. J. Hazard. Mater. 2014, 273, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Morales-Sanfrutos, J.; Javier Lopez-Jaramillo, F.; Elremaily, M.A.A.; Hernandez-Mateo, F.; Santoyo-Gonzalez, F. Divinyl Sulfone Cross-Linked Cyclodextrin-Based Polymeric Materials: Synthesis and Applications as Sorbents and Encapsulating Agents. Molecules 2015, 20, 3565–3581. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Wang, Y.; Zhou, Y.; Han, P.; Lu, X. A Facile Removal of Phenol in Wastewater Using Crosslinked beta-Cyclodextrin Particles with Ultrasonic Treatment. Clean-Soil Air Water 2014, 42, 51–55. [Google Scholar] [CrossRef]
- Alsbaiee, A.; Smith, B.J.; Xiao, L.; Ling, Y.; Helbling, D.E.; Dichtel, W.R. Rapid removal of organic micropollutants from water by a porous beta-cyclodextrin polymer. Nature 2016, 529, 190-U146. [Google Scholar] [CrossRef]
- Kayaci, F.; Aytac, Z.; Uyar, T. Surface modification of electrospun polyester nanofibers with cyclodextrin polymer for the removal of phenanthrene from aqueous solution. J. Hazard. Mater. 2013, 261, 286–294. [Google Scholar] [CrossRef]
- Li, C.; Klemes, M.J.; Dichtel, W.R.; Helbling, D.E. Tetrafluorotereplithalomtrile-crosslinked beta-cyclodexfrin polymers for efficient extraction and recovery of organic micropollutants from water. J. Chromatogr. A 2018, 1541, 52–56. [Google Scholar] [CrossRef]
- Zhao, F.; Repo, E.; Yin, D.; Meng, Y.; Jafari, S.; Sillanpää, M. EDTA-Cross-Linked β-Cyclodextrin: An Environmentally Friendly Bifunctional Adsorbent for Simultaneous Adsorption of Metals and Cationic Dyes. Environ. Sci. Technol. 2015, 49, 10570–10580. [Google Scholar] [CrossRef] [PubMed]
- Sikder, M.T.; Rahman, M.M.; Jakariya, M.; Hosokawa, T.; Kurasaki, M.; Saito, T. Remediation of water pollution with native cyclodextrins and modified cyclodextrins: A comparative overview and perspectives. Chem. Eng. J. 2019, 355, 920–941. [Google Scholar] [CrossRef]
- Skold, M.E.; Thyne, G.D.; Drexler, J.W.; McCray, J.E. Solubility enhancement of seven metal contaminants using carboxymethyl-β-cyclodextrin (CMCD). J. Contam. Hydrol. 2009, 107, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Badruddoza, A.Z.M.; Hazel, G.S.S.; Hidajat, K.; Uddin, M.S. Synthesis of carboxymethyl-β-cyclodextrin conjugated magnetic nano-adsorbent for removal of methylene blue. Colloids Surf. A Physicochem. Eng. Asp. 2010, 367, 85–95. [Google Scholar] [CrossRef]
- Olteanu, A.A.; Arama, C.-C.; Bleotu, C.; Lupuleasa, D.; Monciu, C.M. Investigation of Cyclodextrin based Nanosponges Complexes with angiotensin I converting enzyme inhibitors (enalapril, captopril, cilazapril). Farmacia 2015, 63, 492–503. [Google Scholar]
- Okoli, C.P.; Adewuyi, G.O.; Zhang, Q.; Diagboya, P.N.; Guo, Q. Mechanism of dialkyl phthalates removal from aqueous solution using gamma-cyclodextrin and starch based polyurethane polymer adsorbents. Carbohydr. Polym. 2014, 114, 440–449. [Google Scholar] [CrossRef] [Green Version]
- Nojavan, S.; Yazdanpanah, M. Micro-solid phase extraction of benzene, toluene, ethylbenzene and xylenes from aqueous solutions using water-insoluble beta-cyclodextrin polymer as sorbent. J. Chromatogr. A 2017, 1525, 51–59. [Google Scholar] [CrossRef]
- Kopperi, M.; Riekkola, M.-L. Non-targeted evaluation of selectivity of water-compatible class selective adsorbents for the analysis of steroids in wastewater. Anal. Chim. Acta 2016, 920, 47–53. [Google Scholar] [CrossRef]
- Danquah, M.K.; Aruei, R.C.; Wilson, L.D. Phenolic Pollutant Uptake Properties of Molecular Templated Polymers Containing beta-Cyclodextrin. J. Phys. Chem. B 2018, 122, 4748–4757. [Google Scholar] [CrossRef]
- Sikder, M.T.; Islam, M.S.; Kikuchi, T.; Suzuki, J.; Saito, T.; Kurasaki, M. Removal of Copper Ions from Water Using Epichlorohydrin Cross-Linked beta-Cyclodextrin Polymer: Characterization, Isotherms and Kinetics. Water Environ. Res. 2014, 86, 296–304. [Google Scholar] [CrossRef]
- Liu, H.; Cai, X.; Wang, Y.; Chen, J. Adsorption mechanism-based screening of cyclodextrin polymers for adsorption and separation of pesticides from water. Water Res. 2011, 45, 3499–3511. [Google Scholar] [CrossRef] [PubMed]
- Sevillano, X.; Isasi, J.R.; Penas, F.J. Feasibility study of degradation of phenol in a fluidized bed bioreactor with a cyclodextrin polymer as biofilm carrier. Biodegradation 2008, 19, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Hu, Y.; Li, Y.; Zhou, Y.; Niu, D.; Lei, Z.; Zhang, Z. Citric acid-crosslinked β-cyclodextrin for simultaneous removal of bisphenol A, methylene blue and copper: The roles of cavity and surface functional groups. J. Taiwan Inst. Chem. Eng. 2018, 82, 189–197. [Google Scholar] [CrossRef]
- Tu, Y.; Xu, G.; Jiang, L.; Hu, X.; Xie, X.; Li, A. Amphiphilic hyper-crosslinked porous cyclodextrin polymer with high specific surface area for rapid removal of organic micropollutants. Chem Eng. J. 2020, 382, 123015. [Google Scholar] [CrossRef]
- Trotta, F.; Tumiatti, W. Cross-Linked Cyclodextrins for Decontamination of Liquid, Gaseous or Solid Matrices, Is Obtainable by Reacting Cyclodextrin with Carbonyl Compound. US2005154198-A1, 1 July 2005. [Google Scholar]
- Nagy, Z.M.; Molnár, M.; Fekete-Kertész, I.; Molnár-Perl, I.; Fenyvesi, E.; Gruiz, K. Removal of emerging micropollutants from water using cyclodextrin. Sci. Total Environ. 2014, 485–486, 711–719. [Google Scholar] [CrossRef]
- Mak, Y.W.; Leung, W.W.-F. Crosslinking of genipin and autoclaving in chitosan-based nanofibrous scaffolds: Structural and physiochemical properties. J. Mater. Sci. 2019, 54, 10941–10962. [Google Scholar] [CrossRef]
- Bezerra, F.M.; Lis, M.J.; Firmino, H.B.; Silva, J.G.D.; Valle, R.C.S.C.; Valle, J.A.B.; Scacchetti, F.A.P.; Tessaro, A.L. The role of β-cyclodextrin in the textile industry-review. Molecules 2020, 25, 3624. [Google Scholar] [CrossRef]
- Qin, X.; Bai, L.; Tan, Y.; Li, L.; Song, F.; Wang, Y. β-Cyclodextrin-crosslinked polymeric adsorbent for simultaneous removal and stepwise recovery of organic dyes and heavy metal ions: Fabrication, performance and mechanisms. Chem. Eng. J. 2019, 372, 1007–1018. [Google Scholar] [CrossRef]
- Folch-Cano, C.; Yazdani-Pedram, M.; Olea-Azar, C. Inclusion and functionalization of polymers with cyclodextrins: Current applications and future prospects. Molecules 2014, 19, 14066–14079. [Google Scholar] [CrossRef] [Green Version]
- Krause, R.W.; Mamba, B.B.; Bambo, F.M.; Malefetse, T.J. Cyclodextrin polymers: Synthesis and application in water treatment. In Cyclodextrins: Chemistry and Physic; Transworld Research Network: Trivandrum, India, 2010; pp. 1–25. [Google Scholar]
- Mocanu, G.; Vizitiu, D.; Carpov, A. Cyclodextrin polymers. J. Bioact. Compat. Polym. 2001, 16, 315–342. [Google Scholar] [CrossRef]
- Renard, E.; Sebille, B.; Barnathan, G.; Deratani, A. Polycondensation of cyclodextrins with epichlorohydrin. Influence of reaction conditions on the polymer structure. Macromol. Symp. 1997, 122, 229–234. [Google Scholar] [CrossRef]
- Renard, E.; Deratani, A.; Volet, G.; Sebille, B. Preparation and characterization of water soluble high molecular weight β-cyclodextrin-epichlorohydrin polymers. Eur. Polym. J. 1997, 33, 49–57. [Google Scholar] [CrossRef]
- Gidwani, B.; Vyas, A. Synthesis, characterization and application of Epichlorohydrin-β-cyclodextrin polymer. Colloids Surf. B: Biointerfaces 2014, 114, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Exposito Saintemarie, A.; Rocchi, S.; Fourmentin, M.; Jeanvoine, A.; Millon, L.; Morin-Crini, N. Simultaneous removal of five triazole fungicides from synthetic solutions on activated carbons and cyclodextrin-based adsorbents. Heliyon 2017, 3, e00380. [Google Scholar] [CrossRef]
- Chen, Q.; Wen, Y.; Cang, Y.; Li, L.; Guo, X.; Zhang, R. Selective removal of phenol by spherical particles of α-, β- and γ-cyclodextrin polymers: Kinetics and isothermal equilibrium. Front. Chem. Sci. Eng. 2013, 7, 162–169. [Google Scholar] [CrossRef]
- Pratt, D.Y.; Wilson, L.D.; Kozinski, J.A.; Mohart, A.M. Preparation and sorption studies of β-cyclodextrin/epichlorohydrin copolymers. J. Appl. Polym. Sci. 2010, 116, 2982–2989. [Google Scholar] [CrossRef]
- Orprecio, R.; Evans, C.H. Polymer-immobilized cyclodextrin trapping of model organic pollutants in flowing water streams. J. Appl. Polym. Sci. 2003, 90, 2103–2110. [Google Scholar] [CrossRef]
- Zhang, K.-D.; Tsai, F.-C.; Ma, N.; Xia, Y.; Liu, H.-L.; Guo, X.; Yu, X.-Y.; Jiang, T.; Chiang, T.-C.; Chang, C.C.-J. Removal of azo dye from aqueous solution by host-guest interaction with β-cyclodextrin. Desalination Water Treat. 2017, 86. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Hu, Y.; Xu, G.; Li, M.; Zhu, Y.; Jiang, L.; Tu, Y.; Zhu, X.; Xie, X.; Li, A. Green synthesis of a magnetic β-cyclodextrin polymer for rapid removal of organic micro-pollutants and heavy metals from dyeing wastewater. Environ. Res. 2020, 180, 108796. [Google Scholar] [CrossRef]
- Zhang, X.M.; Peng, C.S.; Xu, G.C. Synthesis of modified β-cyclodextrin polymers and characterization of their fuchsin adsorption. J. Incl. Phenom. Macrocycl. Chem. 2012, 72, 165–171. [Google Scholar] [CrossRef]
- Zhao, D.; Zhao, L.; Zhu, C.-S.; Wang, J.; Lv, X.-H. A novel β-cyclodextrin polymer modified by sulfonate groups. J. Incl. Phenom. Macrocycl. Chem. 2012, 73, 93–98. [Google Scholar] [CrossRef]
- Mallard Favier, I.; Baudelet, D.; Fourmentin, S. VOC trapping by new crosslinked cyclodextrin polymers. J. Incl. Phenom. Macrocycl. Chem. 2011, 69, 433–437. [Google Scholar] [CrossRef]
- Pellicer, J.A.; Rodríguez-López, M.I.; Fortea, M.I.; Gabaldón Hernández, J.A.; Lucas-Abellán, C.; Mercader-Ros, M.T.; Serrano-Martínez, A.; Núñez-Delicado, E.; Cosma, P.; Fini, P.; et al. Removing of Direct Red 83:1 using α- and HP-α-CDs polymerized with epichlorohydrin: Kinetic and equilibrium studies. Dye. Pigment. 2018, 149, 736–746. [Google Scholar] [CrossRef]
- Wilson, L.D.; Mohamed, M.H.; Headley, J.V. Surface area and pore structure properties of urethane-based copolymers containing β-cyclodextrin. J. Colloid Interface Sci. 2011, 357, 215–222. [Google Scholar] [CrossRef]
- Mohamed, M.H.; Wilson, L.D.; Headley, J.V. Design and characterization of novel β-cyclodextrin based copolymer materials. Carbohydr. Res. 2011, 346, 219–229. [Google Scholar] [CrossRef]
- Ozmen, E.Y.; Yilmaz, M. Use of β-cyclodextrin and starch based polymers for sorption of Congo red from aqueous solutions. J. Hazard. Mater. 2007, 148, 303–310. [Google Scholar] [CrossRef]
- Ozmen, E.Y.; Sirit, A.; Yilmaz, M. A Calix [4] arene Oligomer and Two Beta-cyclodextrin Polymers: Synthesis and Sorption Studies of Azo Dyes. J. Macromol. Sci. Part A 2007, 44, 167–173. [Google Scholar] [CrossRef]
- Nkambule, T.I.; Krause, R.W.; Mamba, B.B.; Haarhoff, J. Removal of natural organic matter from water using ion-exchange resins and cyclodextrin polyurethanes. Phys. Chem. EarthParts A/B/C 2009, 34, 812–818. [Google Scholar] [CrossRef]
- Mamba, B.B.; Krause, R.W.; Malefetse, T.J.; Nxumalo, E.N. Monofunctionalized cyclodextrin polymers for the removal of organic pollutants from water. Environ. Chem. Lett. 2007, 5, 79–84. [Google Scholar] [CrossRef]
- Shabtai, I.A.; Mishael, Y.G. Polycyclodextrin–clay composites: Regenerable dual-site sorbents for bisphenol A removal from treated wastewater. ACS Appl. Mater. Interfaces 2018, 10, 27088–27097. [Google Scholar] [CrossRef]
- Leudjo Taka, A.; Pillay, K.; Yangkou Mbianda, X. Nanosponge cyclodextrin polyurethanes and their modification with nanomaterials for the removal of pollutants from waste water: A review. Carbohydr. Polym. 2017, 159, 94–107. [Google Scholar] [CrossRef]
- Zemel, H.; Koch, M.B. Preparation of Crosslinked Cyclodextrin Resins with Enhanced Porosity. US4958015A, 18 September 1990. [Google Scholar]
- Flores, J.; Jiménez, V.; Belmar, J.; Mansilla, H.D.; Alderete, J.B. Inclusion Complexation of Phenol Derivatives with a β-Cyclodextrin Based Polymer. J. Incl. Phenom. Macrocycl. Chem. 2005, 53, 63–68. [Google Scholar] [CrossRef]
- Weltrowski, M.; Morcellet, M.; Martel, B. Cyclodextrin Polymers and/or Cyclodextrin Derivatives with Complexing Properties and Ion-Exchange Properties and Method for the Production Thereof. US6660804B1, 9 December 2003. [Google Scholar]
- Martel, B.; Ruffin, D.; Weltrowski, M.; Lekchiri, Y.; Morcellet, M. Water-soluble polymers and gels from the polycondensation between cyclodextrins and poly(carboxylic acid)s: A study of the preparation parameters. J. Appl. Polym. Sci. 2005, 97, 433–442. [Google Scholar] [CrossRef]
- Girek, T.; Kozlowski, C.A.; Koziol, J.J.; Walkowiak, W.; Korus, I. Polymerisation of β-cyclodextrin with succinic anhydride. Synthesis, characterisation, and ion flotation of transition metals. Carbohydr. Polym. 2005, 59, 211–215. [Google Scholar] [CrossRef]
- Zhao, D.; Zhao, L.; Zhu, C.-S.; Huang, W.-Q.; Hu, J.-L. Water-insoluble β-cyclodextrin polymer crosslinked by citric acid: Synthesis and adsorption properties toward phenol and methylene blue. J. Incl. Phenom. Macrocycl. Chem. 2009, 63, 195–201. [Google Scholar] [CrossRef]
- Xiao, L.; Ling, Y.; Alsbaiee, A.; Li, C.; Helbling, D.E.; Dichtel, W.R. β-Cyclodextrin Polymer Network Sequesters Perfluorooctanoic Acid at Environmentally Relevant Concentrations. J. Am. Chem. Soc. 2017, 139, 7689–7692. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Zhou, M.; Jia, J.; Jia, Q. A water-insoluble viologen-based β-cyclodextrin polymer for selective adsorption toward anionic dyes. React. Funct. Polym. 2018, 126, 20–26. [Google Scholar] [CrossRef]
- Yang, J.S.; Yang, L. Preparation and application of cyclodextrin immobilized polysaccharides. J. Mater. Chem. B 2013, 1, 909–918. [Google Scholar] [CrossRef]
- Tojima, T.; Katsura, H.; Nishiki, M.; Nishi, N.; Tokura, S.; Sakairi, N. Chitosan beads with pendant α-cyclodextrin: Preparation and inclusion property to nitrophenolates. Carbohydr. Polym. 1999, 40, 17–22. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Chen, C.-C.; Chung, Y.-C. Removal of phthalate esters by α-cyclodextrin-linked chitosan bead. Bioresour. Technol. 2007, 98, 2578–2583. [Google Scholar] [CrossRef]
- Fan, L.; Zhang, Y.; Luo, C.; Lu, F.; Qiu, H.; Sun, M. Synthesis and characterization of magnetic β-cyclodextrin–chitosan nanoparticles as nano-adsorbents for removal of methyl blue. Int. J. Biol. Macromol. 2012, 50, 444–450. [Google Scholar] [CrossRef]
- Ali, I.; Basheer, A.A.; Mbianda, X.Y.; Burakov, A.; Galunin, E.; Burakova, I.; Mkrtchyan, E.; Tkachev, A.; Grachev, V. Graphene based adsorbents for remediation of noxious pollutants from wastewater. Environ. Int. 2019, 127, 160–180. [Google Scholar] [CrossRef]
- Fan, L.; Luo, C.; Sun, M.; Qiu, H.; Li, X. Synthesis of magnetic β-cyclodextrin–chitosan/graphene oxide as nanoadsorbent and its application in dye adsorption and removal. Colloids Surf. B: Biointerfaces 2013, 103, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Martel, B.; Devassine, M.; Crini, G.; Weltrowski, M.; Bourdonneau, M.; Morcellet, M. Preparation and sorption properties of a β-cyclodextrin-linked chitosan derivative. J. Polym. Sci. Part. A Polym. Chem. 2001, 39, 169–176. [Google Scholar] [CrossRef]
- Aoki, N.; Nishikawa, M.; Hattori, K. Synthesis of chitosan derivatives bearing cyclodextrin and adsorption of p-nonylphenol and bisphenol A. Carbohydr. Polym. 2003, 52, 219–223. [Google Scholar] [CrossRef]
- Xia, Y.; Wan, J. Preparation and adsorption of novel cellulosic fibers modified by β-cyclodextrin. Polym. Adv. Technol. 2008, 19, 270–275. [Google Scholar] [CrossRef]
- Sancey, B.; Trunfio, G.; Charles, J.; Badot, P.M.; Crini, G. Sorption onto crosslinked cyclodextrin polymers for industrial pollutants removal: An interesting environmental approach. J. Incl. Phenom. Macrocycl. Chem. 2011, 70, 315–320. [Google Scholar] [CrossRef]
- Duffus, J.H. “Heavy metals” a meaningless term? (IUPAC Technical Report). Pure Appl. Chem. 2002, 74, 793–807. [Google Scholar] [CrossRef] [Green Version]
- Bolan, N.; Choppala, G.; Kunhikrishnan, A.; Park, J.; Ravi, N. Microbial Transformation of Trace Elements in Soils in Relation to Bioavailability and Remediation. In Reviews of Environmental Contamination and Toxicology; Whitacre, M.D., Ed.; Springer: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Adriano, D.C.; Wenzel, W.W.; Vangronsveld, J.; Bolan, N.S. Role of assisted natural remediation in environmental cleanup. Geoderma 2004, 122, 121–142. [Google Scholar] [CrossRef]
- Adriano, D.C. Trace Elements in Terrestrial Environments: Biogeochemistry, Bioavailability, and Risks of Metals; Springer: New York, NY, USA, 2001. [Google Scholar] [CrossRef]
- Vareda, J.P.; Valente, A.J.M.; Durães, L. Assessment of heavy metal pollution from anthropogenic activities and remediation strategies: A review. J. Environ. Manag. 2019, 246, 101–118. [Google Scholar] [CrossRef]
- Vareda, J.P.; Valente, A.J.; Durães, L. Erratum to “Heavy metals in Iberian soils: Removal by current adsorbents/amendments and prospective for aerogels” [Adv. Colloid Interf. Sci. 237 (2016) 28–42]. Adv. Colloid Interface Sci. 2016, 237, 76–77. [Google Scholar] [CrossRef]
- Na, Y.; Lee, J.; Lee, S.H.; Kumar, P.; Kim, J.H.; Patel, R. Removal of heavy metals by polysaccharide: A review. Polym.-Plast. Technol. Mater. 2020, 59, 1770–1790. [Google Scholar] [CrossRef]
- Buvári, Á.; Barcza, L. β-cyclodextrin complexes of different type with inorganic compounds. Inorg. Chim. Acta 1979, 33, L179–L180. [Google Scholar] [CrossRef]
- Buvári, Á.; Barcza, L. Complex formation of inorganic salts withβ-cyclodextrin. J. Incl. Phenom. Mol. Recognit. Chem. 1989, 7, 379–389. [Google Scholar] [CrossRef]
- Ribeiro, A.; Lobo, V.; Valente, A.; Simões, S.; Sobral, A.; Ramos, M.; Burrows, H.D. Association between Ammonium Monovanadate and β-Cyclodextrin as seen by NMR and Transport Techniques. Polyhedron 2006, 25, 3581–3587. [Google Scholar] [CrossRef]
- Norkus, E.; Grincien, G.; Vaitkus, R. Interaction of lead(II) with β-cyclodextrin in alkaline solutions. Carbohydr. Res. 2002, 337, 1657–1661. [Google Scholar] [CrossRef]
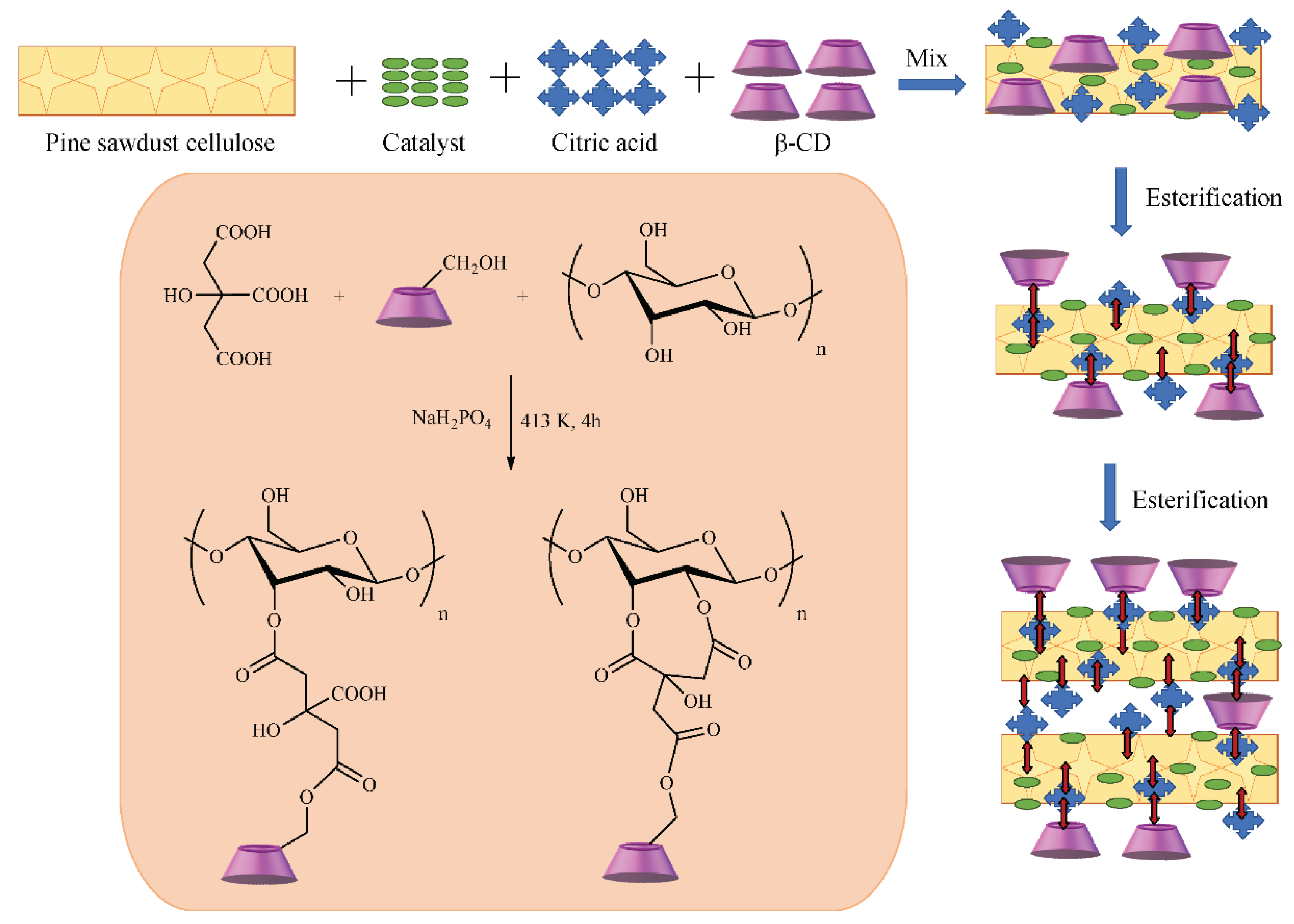
- Zhou, Y.; Gu, X.; Zhang, R.; Lu, J. Removal of Aniline from Aqueous Solution using Pine Sawdust Modified with Citric Acid and β-Cyclodextrin. Ind. Eng. Chem. Res. 2014, 53, 887–894. [Google Scholar] [CrossRef]
- Badruddoza, A.Z.M.; Shawon, Z.B.Z.; Tay, W.J.D.; Hidajat, K.; Uddin, M.S. Fe3O4/cyclodextrin polymer nanocomposites for selective heavy metals removal from industrial wastewater. Carbohydr. Polym. 2013, 91, 322–332. [Google Scholar] [CrossRef]
- Kyzas, G.; Bikiaris, D. Recent Modifications of Chitosan for Adsorption Applications: A Critical and Systematic Review. Mar. Drugs 2015, 13, 312–337. [Google Scholar] [CrossRef]
- Martins, A.F.; Bueno, P.V.A.; Almeida, E.A.M.S.; Rodrigues, F.H.A.; Rubira, A.F.; Muniz, E.C. Characterization of N-trimethyl chitosan/alginate complexes and curcumin release. Int. J. Biol. Macromol. 2013, 57, 174–184. [Google Scholar] [CrossRef] [Green Version]
- Malayoglu, U. Removal of heavy metals by biopolymer (chitosan)/nanoclay composites. Sep. Sci. Technol. 2018, 53, 2741–2749. [Google Scholar] [CrossRef]
- Zhang, L.; Zeng, Y.; Cheng, Z. Removal of heavy metal ions using chitosan and modified chitosan: A review. J. Mol. Liq. 2016, 214, 175–191. [Google Scholar] [CrossRef]
- Fan, L.; Li, M.; Lv, Z.; Sun, M.; Luo, C.; Lu, F.; Qiu, H. Fabrication of magnetic chitosan nanoparticles grafted with β-cyclodextrin as effective adsorbents toward hydroquinol. Colloids Surf. B Biointerfaces 2012, 95, 42–49. [Google Scholar] [CrossRef]
- Binello, A.; Cravotto, G.; Nano, G.M.; Spagliardi, P. Synthesis of chitosan–cyclodextrin adducts and evaluation of their bitter-masking properties. Flavour Fragr. J. 2004, 19, 394–400. [Google Scholar] [CrossRef]
- Elanchezhiyan, S.S.D.; Meenakshi, S. Facile Fabrication of Metal Ions-Incorporated Chitosan/β-Cyclodextrin Composites for Effective Removal of Oil from Oily Wastewater. ChemistrySelect 2017, 2, 11393–11401. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Graphene oxide and its application as an adsorbent for wastewater treatment. J. Chem. Technol. Biotechnol. 2014, 89, 196–205. [Google Scholar] [CrossRef]
- Zhang, N.; Qiu, H.; Si, Y.; Wang, W.; Gao, J. Fabrication of highly porous biodegradable monoliths strengthened by graphene oxide and their adsorption of metal ions. Carbon 2011, 49, 827–837. [Google Scholar] [CrossRef]
- Cruz, S.; Marques, P.; Valente, A. Supramolecular Graphene-Based Systems for Drug Delivery. In Handbook of Graphene; Ozkan, C., Ed.; Scrivener Publishing LLC: New York, NY, USA, 2019; pp. 443–480. [Google Scholar]
- Duru, İ.; Ege, D.; Kamali, A.R. Graphene oxides for removal of heavy and precious metals from wastewater. J. Mater. Sci. 2016, 51, 6097–6116. [Google Scholar] [CrossRef]
- Hu, X.-j.; Liu, Y.-g.; Wang, H.; Zeng, G.-m.; Hu, X.; Guo, Y.-m.; Li, T.-t.; Chen, A.-w.; Jiang, L.-h.; Guo, F.-y. Adsorption of copper by magnetic graphene oxide-supported β-cyclodextrin: Effects of pH, ionic strength, background electrolytes, and citric acid. Chem. Eng. Res. Des. 2015, 93, 675–683. [Google Scholar] [CrossRef]
- Cukierman, A.L.; Platero, E.; Fernandez, M.E.; Bonelli, P.R. Potentialities of Graphene-Based Nanomaterials for Wastewater Treatment. In Smart Materials for Waste Water Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; 2p. [Google Scholar]
- Li, L.; Fan, L.; Sun, M.; Qiu, H.; Li, X.; Duan, H.; Luo, C. Adsorbent for chromium removal based on graphene oxide functionalized with magnetic cyclodextrin–chitosan. Colloids Surf. B Biointerfaces 2013, 107, 76–83. [Google Scholar] [CrossRef]
- Weckhuysen, B.M.; Wachs, I.E.; Schoonheydt, R.A. Surface Chemistry and Spectroscopy of Chromium in Inorganic Oxides. Chem. Rev. 1996, 96, 3327–3350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Wei, S.; Gu, H.; Rapole, S.B.; Wang, Q.; Luo, Z.; Haldolaarachchige, N.; Young, D.P.; Guo, Z. One-Pot Synthesis of Magnetic Graphene Nanocomposites Decorated with Core@Double-shell Nanoparticles for Fast Chromium Removal. Environ. Sci. Technol. 2012, 46, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhou, Y.; Zhou, Y.; Lei, J.; Pu, S. Cyclodextrin modified filter paper for removal of cationic dyes/Cu ions from aqueous solutions. Water Sci. Technol. 2018, 78, 2553–2563. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.B.; Vakili, M.; Horri, B.A.; Poh, P.E.; Abdullah, A.Z.; Salamatinia, B. Adsorption of dyes by nanomaterials: Recent developments and adsorption mechanisms. Sep. Purif. Technol. 2015, 150, 229–242. [Google Scholar] [CrossRef]
- Forgacs, E.; Cserhati, T.; Oros, G. Removal of synthetic dyes from wastewaters: A review. Environ. Int. 2004, 30, 953–971. [Google Scholar] [CrossRef]
- Yaseen, D.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef] [Green Version]
- Sivaram, N.; Barik, D. Toxic Waste from Leather Industries. In Energy from Toxic Organic Waste for Heat and Power Generation; Elsevier: Amsterdam, The Netherlands, 2019; pp. 55–67. [Google Scholar]
- Yamjala, K.; Nainar, M.S.; Ramisetti, N.R. Methods for the analysis of azo dyes employed in food industry–a review. Food Chem. 2016, 192, 813–824. [Google Scholar] [CrossRef]
- Yu, Z.; Li, F.; Sun, L. Recent advances in dye-sensitized photoelectrochemical cells for solar hydrogen production based on molecular components. Energy Environ. Sci. 2015, 8, 760–775. [Google Scholar] [CrossRef]
- Wong, Y.; Szeto, Y.; Cheung, W.; McKay, G. Adsorption of acid dyes on chitosan—equilibrium isotherm analyses. Process. Biochem. 2004, 39, 695–704. [Google Scholar] [CrossRef]
- Mani, S.; Chowdhary, P.; Bharagava, R.N. Textile wastewater dyes: Toxicity profile and treatment approaches. In Emerging and Eco-Friendly Approaches for Waste Management; Springer: Berlin/Heidelberg, Germany, 2019; pp. 219–244. [Google Scholar]
- Mahmoud, H.R.; Ibrahim, S.M.; El-Molla, S.A. Textile dye removal from aqueous solutions using cheap MgO nanomaterials: Adsorption kinetics, isotherm studies and thermodynamics. Adv. Powder Technol. 2016, 27, 223–231. [Google Scholar] [CrossRef]
- Asghar, A.; Raman, A.A.A.; Daud, W.M.A.W. Advanced oxidation processes for in-situ production of hydrogen peroxide/hydroxyl radical for textile wastewater treatment: A review. J. Clean. Prod. 2015, 87, 826–838. [Google Scholar] [CrossRef] [Green Version]
- Babuponnusami, A.; Muthukumar, K. A review on Fenton and improvements to the Fenton process for wastewater treatment. J. Environ. Chem. Eng. 2014, 2, 557–572. [Google Scholar] [CrossRef]
- Martinez-Huitle, C.A.; Rodrigo, M.A.; Sires, I.; Scialdone, O. Single and coupled electrochemical processes and reactors for the abatement of organic water pollutants: A critical review. Chem. Rev. 2015, 115, 13362–13407. [Google Scholar] [CrossRef] [PubMed]
- Mezohegyi, G.; van der Zee, F.P.; Font, J.; Fortuny, A.; Fabregat, A. Towards advanced aqueous dye removal processes: A short review on the versatile role of activated carbon. J. Environ. Manag. 2012, 102, 148–164. [Google Scholar] [CrossRef]
- Beslin, L. Textural Features-Indicators of Pollution. J. Environ. Anal. Toxicol. 2017, 7, 2161-0525.1000505. [Google Scholar] [CrossRef]
- Fei, Y.; Dexian, C.; Jie, M. Synthesis of Cyclodextrin-based Adsorbents and its Application for Organic Pollutant Removal from Water. Curr. Org. Chem. 2017, 21, 1976–1990. [Google Scholar] [CrossRef]
- Dsouza, R.N.; Pischel, U.; Nau, W.M. Fluorescent dyes and their supramolecular host/guest complexes with macrocycles in aqueous solution. Chem. Rev. 2011, 111, 7941–7980. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.; Li, B.; Wada, T.; Inoue, Y. Cooperative Multipoint Recognition of Organic Dyes by Bis (β-cyclodextrin) s with 2, 2′-Bipyridine-4, 4′-dicarboxy Tethers. Chem.–A Eur. J. 2001, 7, 2528–2535. [Google Scholar] [CrossRef]
- Hirai, H.; Toshima, N.; Uenoyama, S. Inclusion complex formation of cyclodextrin with large dye molecule. Polym. J. 1981, 13, 607. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Hu, Y.; Huang, W.; Cheng, G.; Cui, C.; Lu, J. A novel amphoteric β-cyclodextrin-based adsorbent for simultaneous removal of cationic/anionic dyes and bisphenol A. Chem. Eng. J. 2018, 341, 47–57. [Google Scholar] [CrossRef]
- Zhao, J.; Zou, Z.; Ren, R.; Sui, X.; Mao, Z.; Xu, H.; Zhong, Y.; Zhang, L.; Wang, B. Chitosan adsorbent reinforced with citric acid modified β-cyclodextrin for highly efficient removal of dyes from reactive dyeing effluents. Eur. Polym. J. 2018, 108, 212–218. [Google Scholar] [CrossRef]
- Wang, Q.Z.; Chen, X.G.; Liu, N.; Wang, S.X.; Liu, C.S.; Meng, X.H.; Liu, C.G. Protonation constants of chitosan with different molecular weight and degree of deacetylation. Carbohydr. Polym. 2006, 65, 194–201. [Google Scholar] [CrossRef]
- Yu, D.; Wu, L.L.; Wang, J.F.; Tao, Y.W.; Shen, Y.T.; Liang, B.Y.; Wang, H. Preparation of β-Cyclodextrin/Chitosan Membranes and its Application in the Wastewater Treatment of Acid Dyes. Adv. Mater. Res. 2013, 726′731, 2558–2562. [Google Scholar] [CrossRef]
- Cova, T.F.; Cruz, S.M.; Valente, A.J.; Abreu, P.E.; Marques, J.M.; Pais, A.A. Aggregation of Cyclodextrins: Fundamental Issues and Applications. In Cyclodextrin Fundamentals, Reactivity and Analysis; Springer Nature Switzerland AG Basel: Basel, Switzerland, 2018; 10.5772/intechopen.73532. [Google Scholar]
- Mourtzis, N.; Cordoyiannis, G.; Nounesis, G.; Yannakopoulou, K. Single and Double Threading of Congo Red into γ-Cyclodextrin. Solution Structures and Thermodynamic Parameters of 1:1 and 2:2 Adducts, as Obtained from NMR Spectroscopy and Microcalorimetry. Supramol. Chem. 2003, 15, 639–649. [Google Scholar] [CrossRef]
- Hamai, S.; Satou, H. Inclusion complexes of cyclodextrins with Methylene Blue and Acid Orange 7 in aqueous solutions. Bull. Chem. Soc. Jpn. 2000, 73, 2207–2214. [Google Scholar] [CrossRef]
- Hao, Z.; Yi, Z.; Bowen, C.; Yaxing, L.; Sheng, Z. Preparing γ-Cyclodextrin-Immobilized Starch and the Study of its Removal Properties to Dyestuff from Wastewater. Pol. J. Environ. Stud. 2019, 28, 1701–1711. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Huang, S.; Zhao, X.; Zhang, Y. Fabrication of three-dimensional porous β-cyclodextrin/chitosan functionalized graphene oxide hydrogel for methylene blue removal from aqueous solution. Colloids Surf. A: Physicochem. Eng. Asp. 2018, 539, 1–10. [Google Scholar] [CrossRef]
- Fennell Evans, D.; Wennerstrom, H.; Rajagopalan, R. The colloidal domain: Where physics, chemistry, biology, and technology meet. J. Colloid Interface Sci. 1995, 172, 541. [Google Scholar]
- Lindman, B.; Karlstrom, G.; Stigsson, L. On the mechanism of dissolution of cellulose. J. Mol. Liq. 2010, 156, 76–81. [Google Scholar] [CrossRef]
- O’Connell, D.W.; Birkinshaw, C.; O’Dwyer, T.F. Heavy metal adsorbents prepared from the modification of cellulose: A review. Bioresour. Technol. 2008, 99, 6709–6724. [Google Scholar] [CrossRef]
- Yue, X.; Huang, J.; Jiang, F.; Lin, H.; Chen, Y. Synthesis and characterization of cellulose-based adsorbent for removal of anionic and cationic dyes. J. Eng. Fibers Fabr. 2019, 14, 155892501982819. [Google Scholar] [CrossRef] [Green Version]
- Zhang, F.; Chen, Y.; Lin, H.; Lu, Y. Synthesis of an amino-terminated hyperbranched polymer and its application in reactive dyeing on cotton as a salt-free dyeing auxiliary. Coloration Technol. 2007, 123, 351–357. [Google Scholar] [CrossRef]
- An, F.; Feng, X.; Gao, B. Adsorption of aniline from aqueous solution using novel adsorbent PAM/SiO2. Chem. Eng. J. 2009, 151, 183–187. [Google Scholar] [CrossRef]
- Gross, K.C.; Seybold, P.G. Substituent effects on the physical properties and pKa of aniline. Int. J. Quantum Chem 2000, 80, 1107–1115. [Google Scholar] [CrossRef]
- Ghemati, D.; Aliouche, D. Study of the Sorption of Synthetic Dyes from Aqueous Solution onto Cellulosic Modified Polymer. J. Water Chem. Technol. 2014, 36, 265–272. [Google Scholar] [CrossRef]
- Gao, S.; Jiang, J.-Y.; Liu, Y.-Y.; Fu, Y.; Zhao, L.-X.; Li, C.-Y.; Ye, F. Enhanced Solubility, Stability, and Herbicidal Activity of the Herbicide Diuron by Complex Formation with β-Cyclodextrin. Polymers 2019, 11, 1396. [Google Scholar] [CrossRef] [Green Version]
- Junthip, J.; Jumrernsuk, N.; Klongklaw, P.; Promma, W.; Sonsupap, S. Removal of paraquat herbicide from water by textile coated with anionic cyclodextrin polymer. SN Appl. Sci. 2018, 1, 106. [Google Scholar] [CrossRef] [Green Version]
- Xiao, P.; Dudal, Y.; Corvini, P.F.X.; Shahgaldian, P. Polymeric cyclodextrin-based nanoparticles: Synthesis, characterization and sorption properties of three selected pharmaceutically active ingredients. Polym. Chem. 2011, 2, 120–125. [Google Scholar] [CrossRef]
- Moulahcene, L.; Kebiche-Senhadji, O.; Skiba, M.; Lahiani-Skiba, M.; Oughlis-Hammache, F.; Benamor, M. Cyclodextrin polymers for ibuprofen extraction in aqueous solution: Recovery, separation, and characterization. Desalination Water Treat. 2016, 57, 11392–11402. [Google Scholar] [CrossRef]
- Moulahcene, L.; Skiba, M.; Senhadji, O.; Milon, N.; Benamor, M.; Lahiani-Skiba, M. Inclusion and removal of pharmaceutical residues from aqueous solution using water-insoluble cyclodextrin polymers. Chem. Eng. Res. Des. 2015, 97, 145–158. [Google Scholar] [CrossRef]
- Xiao, P.; Weibel, N.; Dudal, Y.; Corvini, P.F.X.; Shahgaldian, P. A cyclodextrin-based polymer for sensing diclofenac in water. J. Hazard. Mater. 2015, 299, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Cheng, G.; Chen, K.; Lu, J.; Lei, J.; Pu, S. Adsorptive removal of bisphenol A, chloroxylenol, and carbamazepine from water using a novel β-cyclodextrin polymer. Ecotoxicol. Environ. Saf. 2019, 170, 278–285. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cova, T.F.; Murtinho, D.; Aguado, R.; Pais, A.A.C.C.; Valente, A.J.M. Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation. Polysaccharides 2021, 2, 16-38. https://doi.org/10.3390/polysaccharides2010002
Cova TF, Murtinho D, Aguado R, Pais AACC, Valente AJM. Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation. Polysaccharides. 2021; 2(1):16-38. https://doi.org/10.3390/polysaccharides2010002
Chicago/Turabian StyleCova, Tânia F., Dina Murtinho, Roberto Aguado, Alberto A. C. C. Pais, and Artur J. M. Valente. 2021. "Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation" Polysaccharides 2, no. 1: 16-38. https://doi.org/10.3390/polysaccharides2010002
APA StyleCova, T. F., Murtinho, D., Aguado, R., Pais, A. A. C. C., & Valente, A. J. M. (2021). Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation. Polysaccharides, 2(1), 16-38. https://doi.org/10.3390/polysaccharides2010002