A Three-Stage Process of CO-Selective Methanation Based on Its Reaction Characteristics: Achieving a High Gas Hourly Space Velocity
Abstract
1. Introduction
2. Experimental
2.1. Catalyst
2.2. Apparatus
2.3. CO-SMET Performance Experiments
3. Results and Discussion
3.1. Effect of Space Velocity on the Reaction Characteristics of CO-SMET
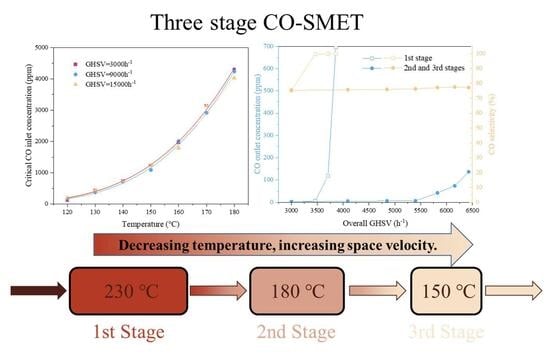
3.2. Effect of Space Velocity on Three-Stage CO-SMET Performance
3.3. Effect of Elevating Temperatures Under High GHSVs on the Three-Stage CO-SMET Performance
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lian, J.; Zhang, Y.; Ma, C.; Yang, Y.; Chaima, E. A review on recent sizing methodologies of hybrid renewable energy systems. Energy Convers. Manag. 2019, 199, 112027. [Google Scholar] [CrossRef]
- Shaker, H.; Zareipour, H.; Wood, D. Impacts of large-scale wind and solar power integration on California׳s net electrical load. Renew. Sustain. Energy Rev. 2016, 58, 761–774. [Google Scholar] [CrossRef]
- Li, Q.; Ren, B.; Tang, W.; Wang, D.; Wang, C.; Lv, Z. Analyzing the inertia of power grid systems comprising diverse conventional and renewable energy sources. Energy Rep. 2022, 8, 15095–15105. [Google Scholar] [CrossRef]
- Albadi, M.H.; El-Saadany, E.F. Overview of wind power intermittency impacts on power systems. Electr. Power Syst. Res. 2010, 80, 627–632. [Google Scholar] [CrossRef]
- Ren, G.; Liu, J.; Wan, J.; Guo, Y.; Yu, D. Overview of wind power intermittency: Impacts, measurements, and mitigation solutions. Appl. Energy 2017, 204, 47–65. [Google Scholar] [CrossRef]
- Elalfy, D.A.; Gouda, E.; Kotb, M.F.; Bureš, V.; Sedhom, B.E. Comprehensive review of energy storage systems technologies, objectives, challenges, and future trends. Energy Strategy Rev. 2024, 54, 101482. [Google Scholar] [CrossRef]
- Khan, M.K.; Raza, M.; Shahbaz, M.; Farooq, U.; Akram, M.U. Akram, Recent advancement in energy storage technologies and their applications. J. Energy Storage 2024, 92, 112112. [Google Scholar] [CrossRef]
- Kamran, M.; Turzyński, M. Exploring hydrogen energy systems: A comprehensive review of technologies, applications, prevailing trends, and associated challenges. J. Energy Storage 2024, 96, 112601. [Google Scholar] [CrossRef]
- Ge, L.; Zhang, B.; Huang, W.; Li, Y.; Hou, L.; Xiao, J.; Mao, Z.; Li, X. A review of hydrogen generation, storage, and applications in power system. J. Energy Storage 2024, 75, 109307. [Google Scholar] [CrossRef]
- Sikiru, S.; Oladosu, T.L.; Amosa, T.I.; Olutoki, J.O.; Ansari, M.N.M.; Abioye, K.J.; Rehman, Z.U.; Soleimani, H. Hydrogen-powered horizons: Transformative technologies in clean energy generation, distribution, and storage for sustainable innovation. Int. J. Hydrogen Energy 2024, 56, 1152–1182. [Google Scholar] [CrossRef]
- Badawi, M.N.; Agrawal, N.; Luqman, M.; Ramesh, S.; Ramesh, K.; Khan, M.; Adil, S.F. Developments and challenges in batteries, and hydrogen as a future fuel, and storage and carrier devices. Int. J. Hydrogen Energy 2025, 105, 1242–1260. [Google Scholar] [CrossRef]
- Padmanabhan, N.T.; Clarizia, L.; Ganguly, P. Advancing hydrogen storage: Critical insights to potentials, challenges, and pathways to sustainability. Curr. Opin. Chem. Eng. 2025, 48, 101135. [Google Scholar] [CrossRef]
- Zhang, W.; Wen, C.; Zhang, X.; Chen, L.; Zhang, Q.; Ma, L. A Review of Green Methanol Production: Technologies, Economic Evaluation, and Carbon Emission Analysis. Energy Fuel 2025, 39, 18733–18750. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Q.; Mei, D.; Wang, Y. Development of highly efficient methanol steam reforming system for hydrogen production and supply for a low temperature proton exchange membrane fuel cell. Int. J. Hydrogen Energy 2020, 45, 25317–25327. [Google Scholar] [CrossRef]
- Thirumalesh, B.S.; Asapu, R. State of the Art of Methanol Reforming for Hydrogen Generation. Chembioeng Rev. 2024, 11, 543–554. [Google Scholar] [CrossRef]
- Harkou, E.; Wang, H.; Manos, G.; Constantinou, A.; Tang, J. Advances in catalyst and reactor design for methanol steam reforming and PEMFC applications. Chem. Sci. 2025, 16, 3810–3831. [Google Scholar] [CrossRef] [PubMed]
- Achomo, M.A.; Kumar, A.; Peela, N.R.; Muthukumar, P. Hydrogen production from steam reforming of methanol: A comprehensive review on thermodynamics, catalysts, reactors, and kinetic studies. Int. J. Hydrogen Energy 2024, 58, 1640–1672. [Google Scholar] [CrossRef]
- Pei, P.; Xu, Y.; Wang, M.; Ren, P. Effects of carbon monoxide on proton exchange membrane fuel cells and elimination techniques. Int. J. Hydrogen Energy 2024, 69, 1287–1304. [Google Scholar] [CrossRef]
- Sahebdelfar, S.; Ravanchi, M.T. Carbon monoxide clean-up of the reformate gas for PEM fuel cell applications: A conceptual review. Int. J. Hydrogen Energy 2023, 48, 24709–24729. [Google Scholar] [CrossRef]
- Snytnikov, P.V.; Zyryanova, M.M.; Sobyanin, V.A. CO-Cleanup of Hydrogen-Rich Stream for LT PEM FC Feeding: Catalysts and Their Performance in Selective CO Methanation. Top. Catal. 2016, 59, 1394–1412. [Google Scholar] [CrossRef]
- Liu, K.; Wang, A.Q.; Zhang, T. Recent Advances in Preferential Oxidation of CO Reaction over Platinum Group Metal Catalysts. ACS Catal 2012, 2, 1165–1178. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Ercolino, G.; Specchia, S.; Specchia, V. Final step for CO syngas clean-up: Comparison between CO-PROX and CO-SMET processes. Int. J. Hydrogen Energy 2014, 39, 18109–18119. [Google Scholar] [CrossRef]
- Wang, Y.C.; Wu, Q.; Mei, D.; Wang, Y. A methanol fuel processing system with methanol steam reforming and CO selective methanation modules for PEMFC application. Int. J. Energy Res. 2021, 45, 6163–6173. [Google Scholar] [CrossRef]
- Du, Z.M.; Liu, C.M.; Zhai, J.X.; Guo, X.Y.; Xiong, Y.L.; Su, W.; He, G.L. A Review of Hydrogen Purification Technologies for Fuel Cell Vehicles. Catalysts 2021, 11, 393. [Google Scholar] [CrossRef]
- Cisneros, S.; Chen, S.; Diemant, T.; Bansmann, J.; Abdel-Mageed, A.M.; Goepel, M.; Olesen, S.E.; Welter, E.S.; Parlinska-Wojtan, M.; Gläser, R.; et al. Effects of SiO2-doping on high-surface-area Ru/TiO2 catalysts for the selective CO methanation. Appl. Catal. B Environ. 2021, 282, 119483. [Google Scholar] [CrossRef]
- Shi, Z.; Feng, J.; Dong, X. Ru–Ni/GA-MMO composites as highly active catalysts for CO selective methanation in H2-rich gases. Int. J. Hydrogen Energy 2023, 48, 24640–24651. [Google Scholar] [CrossRef]
- Li, Z.; Ma, J.; Dong, X. RuNi/TiZr-MMO Catalysts Derived from Zr-Modified NiTi-LDH for CO-Selective Methanation. Molecules 2024, 29, 3309. [Google Scholar] [CrossRef]
- Bobadilla, L.F.; Muñoz-Murillo, A.; Gándara-Loe, J.; Pérez, A.; Laguna, O.H.; Martínez, T.L.M.; Penkova, A.; Centeno, M.A.; Odriozola, J.A. Effect of noble metal addition over active Ru/TiO2 catalyst for CO selective methanation from H2 rich- streams. Int. J. Hydrogen Energy 2023, 48, 25065–25074. [Google Scholar] [CrossRef]
- Li, X.; Han, Y.; Huang, Y.; Lin, J.; Pan, X.; Zhao, Z.; Zhou, Y.; Wang, H.; Yang, X.; Wang, A.; et al. Hydrogenated TiO2 supported Ru for selective methanation of CO in practical conditions. Appl. Catal. B Environ. 2021, 298, 120597. [Google Scholar] [CrossRef]
- Ping, D.; Dong, X.; Zang, Y.; Feng, X. Highly efficient Ru/TiO2-NiAl mixed oxide catalysts for CO selective methanation in hydrogen-rich gas. Int. J. Energy Res. 2017, 41, 2308–2317. [Google Scholar] [CrossRef]
- Djinović, P.; Galletti, C.; Specchia, S.; Specchia, V. CO Methanation Over Ru-Al2O3 Catalysts: Effects of Chloride Doping on Reaction Activity and Selectivity. Top. Catal. 2011, 54, 1042–1053. [Google Scholar] [CrossRef]
- Konishcheva, M.V.; Potemkin, D.I.; Snytnikov, P.V.; Stonkus, O.A.; Belyaev, V.D.; Sobyanin, V.A. The insights into chlorine doping effect on performance of ceria supported nickel catalysts for selective CO methanation. Appl. Catal. B-Environ. 2018, 221, 413–421. [Google Scholar] [CrossRef]
- Miyao, T.; Shen, W.H.; Chen, A.H.; Higashiyama, K.; Watanabe, M. Mechanistic study of the effect of chlorine on selective CO methanation over Ni alumina-based catalysts. Appl. Catal. A Gen. 2014, 486, 187–192. [Google Scholar] [CrossRef]
- Shimoda, N.; Shoji, D.; Tani, K.; Fujiwara, M.; Urasaki, K.; Kikuchi, R.; Satokawa, S. Role of trace chlorine in Ni/TiO2 catalyst for CO selective methanation in reforrnate gas. Appl. Catal. B-Environ. 2015, 174, 486–495. [Google Scholar] [CrossRef]
- Truszkiewicz, E.; Zegadło, K.; Wojda, D.; Mierzwa, B.; Kępiński, L. The Effect of the Ruthenium Crystallite Size on the Activity of Ru/Carbon Systems in CO Methanation. Top. Catal. 2017, 60, 1299–1305. [Google Scholar] [CrossRef]
- Panagiotopoulou, P.; Kondarides, D.I.; Verykios, X.E. Selective methanation of CO over supported noble metal catalysts: Effects of the nature of the metallic phase on catalytic performance. Appl. Catal. A-Gen. 2008, 344, 45–54. [Google Scholar] [CrossRef]
- Panagiotopoulou, P.; Kondarides, D.I.; Verykios, X.E. Selective methanation of CO over supported Ru catalysts. Appl. Catal. B-Environ. 2009, 88, 470–478. [Google Scholar] [CrossRef]
- Konishcheva, M.V.; Potemkin, D.I.; Badmaev, S.D.; Snytnikov, P.V.; Paukshtis, E.A.; Sobyanin, V.A.; Parmon, V.N. On the Mechanism of Co and CO2 Methanation over Ni/CeO2 Catalysts. Top. Catal. 2016, 59, 1424–1430. [Google Scholar] [CrossRef]
- Miao, B.; Ma, S.S.K.; Wang, X.; Su, H.B.; Chan, S.H. Catalysis Mechanisms of CO2 and CO Methanation. Catal. Sci. Technol. 2016, 6, 4048–4058. [Google Scholar] [CrossRef]
- Zhang, S.T.; Yan, H.; Wei, M.; Evans, D.G.; Duan, X. Hydrogenation Mechanism of Carbon Dioxide and Carbon Monoxide on Ru(0001) Surface: A Density Functional Theory Study. RSC Adv. 2014, 4, 30241–30249. [Google Scholar] [CrossRef]
- Kaydouh, M.-N.; El Hassan, N.; Osman, A.I.; Ahmed, H.; Alarifi, N.; Fakeeha, A.H.; Bin Jumah, A.; Al-Fatesh, A.S. Optimizing Co2 Methanation: Effect of Surface Basicity and Active Phase Reducibility on Ni-Based Catalysts. React. Chem. Eng. 2024, 9, 1933–1946. [Google Scholar] [CrossRef]
- Chen, A.; Miyao, T.; Higashiyama, K.; Yamashita, H.; Watanabe, M. High Catalytic Performance of Ruthenium-Doped Mesoporous Nickel-Aluminum Oxides for Selective Co Methanation. Angew. Chem.-Int. Ed. 2010, 49, 9895–9898. [Google Scholar]
- Xiong, J.; Dong, X.F.; Song, Y.B.; Dong, Y.C. A High Performance Ru-ZrO2/Carbon Nanotubes-Ni Foam Composite Catalyst for Selective Co Methanation. J. Power Sources 2013, 242, 132–136. [Google Scholar]
- Miyao, T.; Tanaka, J.; Shen, W.; Hayashi, K.; Higashiyama, K.; Watanabe, M. Catalytic Activity and Durability of a Mesoporous Silica-Coated Ni-Alumina-Based Catalyst for Selective Co Methanation. Catal. Today 2015, 251, 81–87. [Google Scholar]
- Yang, C.; Guo, F.; Luo, C.; Su, Q. A Novel Two-Step Ru/Al2O3 Catalyst Impregnation Method for CO Selective Methanation. Int. J. Hydrogen Energy 2024, 97, 845–855. [Google Scholar]
- Yang, C.; Luo, C.; Su, Q. Critical Co Concentration Driven Three-Stage Co Selective Methanation Design: Achieving Synergistic Enhancement of Co Removal Depth and Selectivity. Int. J. Hydrogen Energy 2025, 191, 152326. [Google Scholar] [CrossRef]
- Li, Y.; Luo, C.; Su, Q. Cold Start-up Study of Methanol Reformer Based on Chemical-Looping Combustion. Fuel 2022, 317, 122850. [Google Scholar] [CrossRef]
- Li, Y.; Luo, C.; Xu, J.; Su, Q. A Cold Start-up Method with Combining Chemical-Looping Combustion and Catalytic Combustion for a Methanol Reformer. Int. J. Hydrogen Energy 2024, 49, 668–679. [Google Scholar] [CrossRef]
- Ranjekar, A.M.; Yadav, G.D. Steam Reforming of Methanol for Hydrogen Production: A Critical Analysis of Catalysis, Processes, and Scope. Ind. Eng. Chem. Res. 2021, 60, 89–113. [Google Scholar] [CrossRef]
- Garcia, G.; Arriola, E.; Chen, W.-H.; De Luna, M.D. A Comprehensive Review of Hydrogen Production from Methanol Thermochemical Conversion for Sustainability. Energy 2021, 217, 119384. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yang, C.; Luo, C.; Su, Q. A Three-Stage Process of CO-Selective Methanation Based on Its Reaction Characteristics: Achieving a High Gas Hourly Space Velocity. Hydrogen 2026, 7, 39. https://doi.org/10.3390/hydrogen7010039
Yang C, Luo C, Su Q. A Three-Stage Process of CO-Selective Methanation Based on Its Reaction Characteristics: Achieving a High Gas Hourly Space Velocity. Hydrogen. 2026; 7(1):39. https://doi.org/10.3390/hydrogen7010039
Chicago/Turabian StyleYang, Changchang, Chunhuan Luo, and Qingquan Su. 2026. "A Three-Stage Process of CO-Selective Methanation Based on Its Reaction Characteristics: Achieving a High Gas Hourly Space Velocity" Hydrogen 7, no. 1: 39. https://doi.org/10.3390/hydrogen7010039
APA StyleYang, C., Luo, C., & Su, Q. (2026). A Three-Stage Process of CO-Selective Methanation Based on Its Reaction Characteristics: Achieving a High Gas Hourly Space Velocity. Hydrogen, 7(1), 39. https://doi.org/10.3390/hydrogen7010039