Can High-Entropy Interlayers Develop Intermetallic-Free Welded Joints of Dissimilar Metals?
Abstract
1. Background and Purpose
1.1. High-Entropy Alloys
1.2. Intermetallic Compounds in Dissimiar Metals Welded Joints
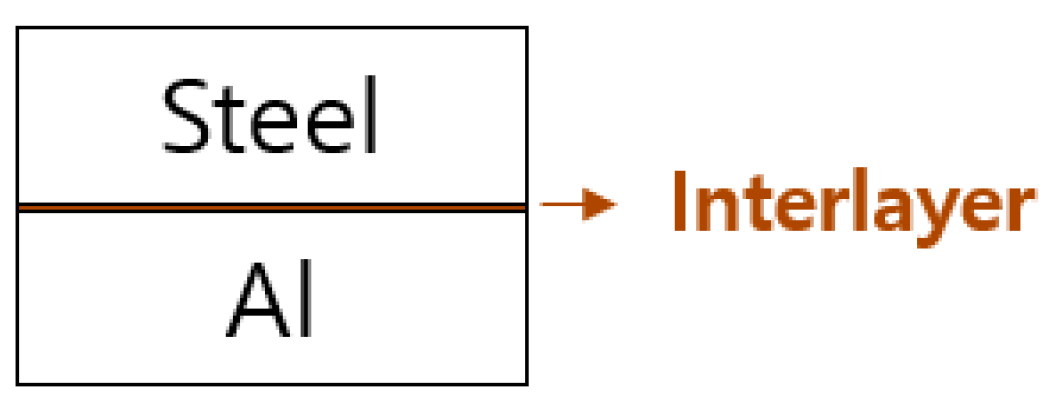
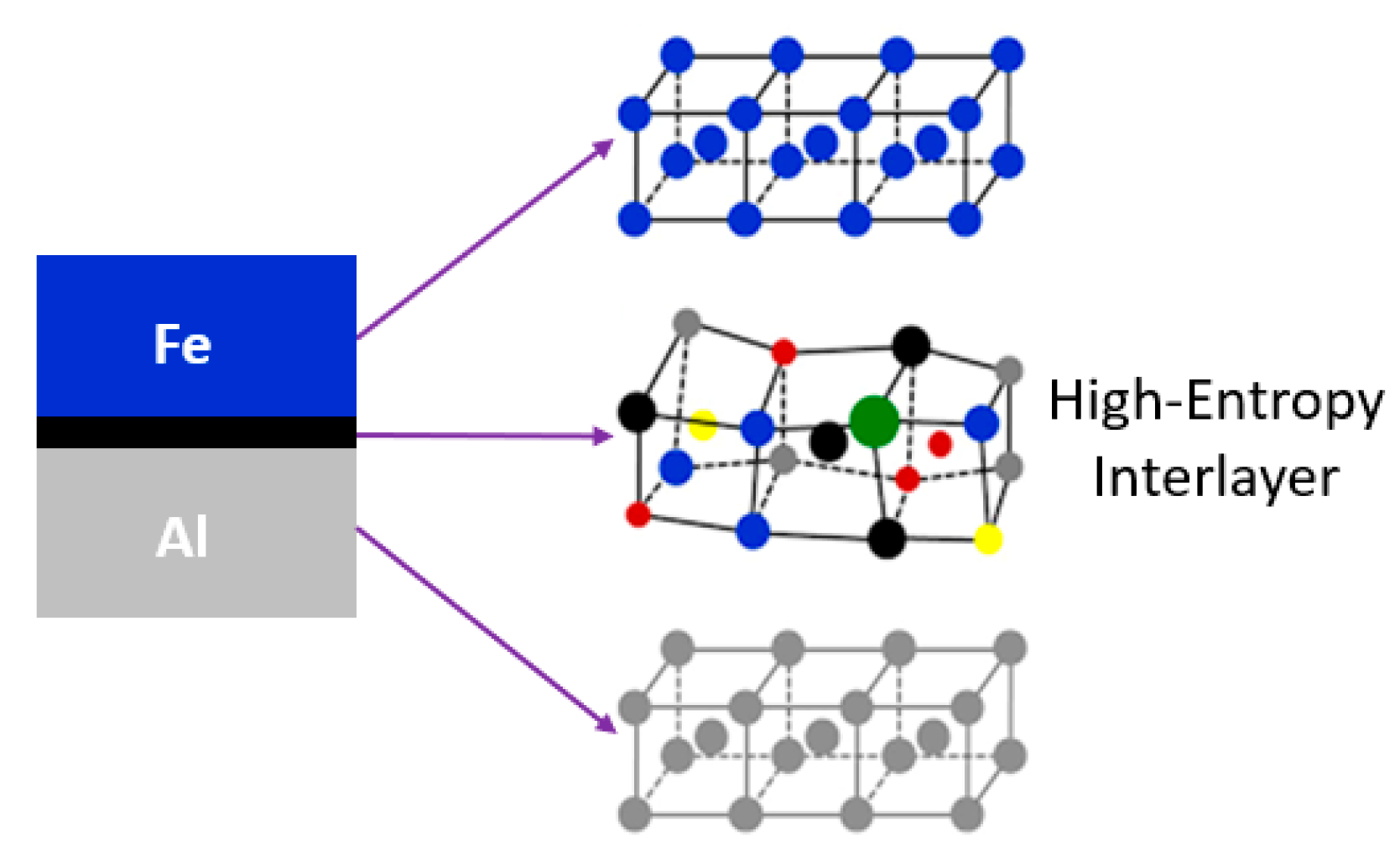
2. High-Entropy Interlayer (HEI)—An Unorthodox Approach
3. Conclusions
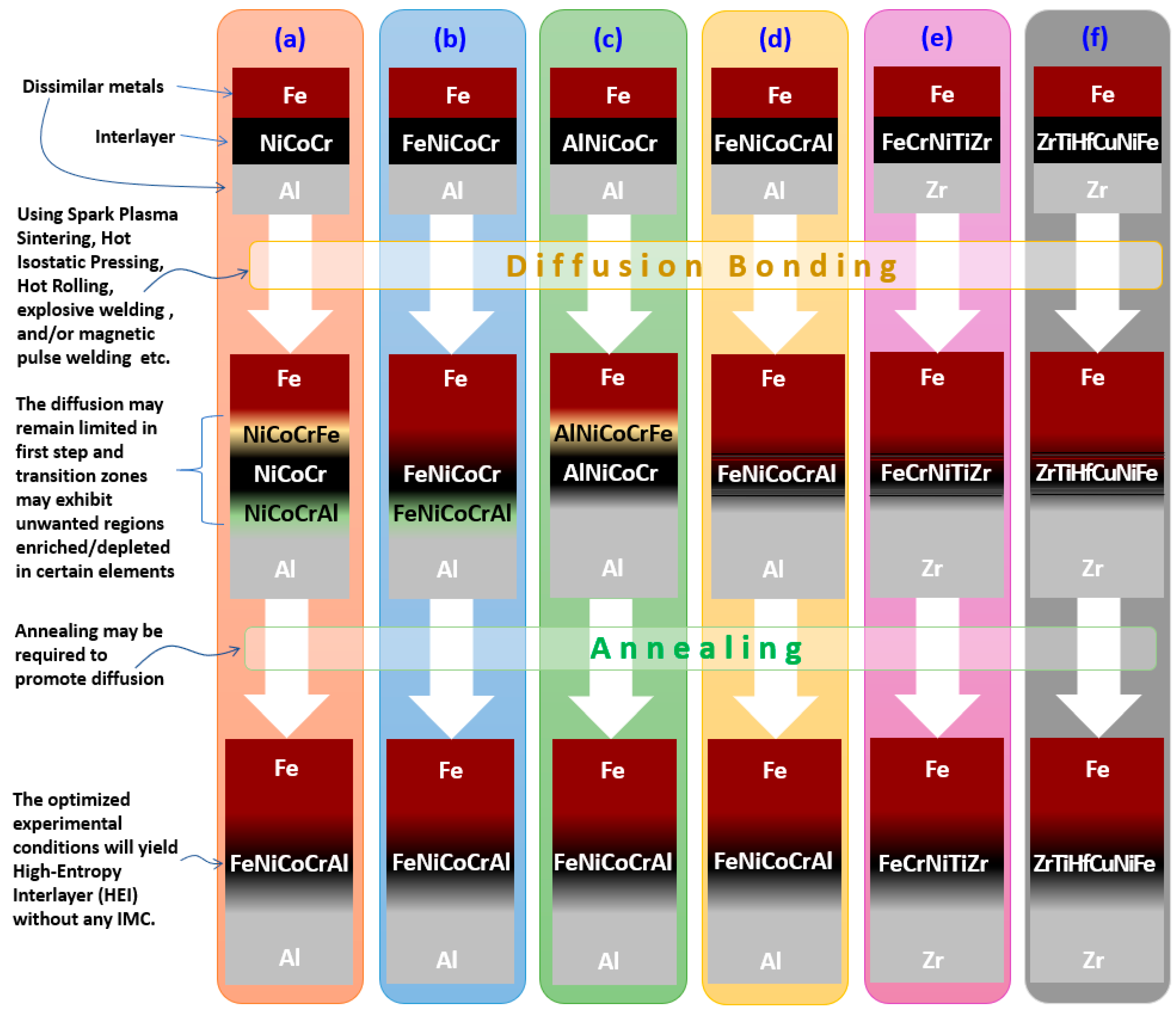
4. Experimental Validation
5. Impact of HEI Concept
Author Contributions
Funding
Conflicts of Interest
References
- Anand, G.; Goodall, R.; Freeman, C.L. Role of configurational entropy in body-centred cubic or face-centred cubic phase formation in high entropy alloys. Scr. Mater. 2016, 124, 90–94. [Google Scholar] [CrossRef]
- Egami, T.; Guo, W.; Rack, P.D.; Nagase, T. Irradiation resistance of multicomponent alloys. Metall. Mater. Trans. A 2014, 45, 180–183. [Google Scholar] [CrossRef]
- Nagase, T.; Rack, P.D.; Noh, J.H.; Egami, T. In-situ TEM observation of structural changes in nano-crystalline CoCrCuFeNi multicomponent high-entropy alloy (HEA) under fast electron irradiation by high voltage electron microscopy (HVEM). Intermetallics 2015, 59, 32–42. [Google Scholar] [CrossRef]
- Hao, X.; Dong, H.; Xia, Y.; Li, P. Microstructure and mechanical properties of laser welded TC4 titanium alloy/304 stainless steel joint with (CoCrFeNi)100-xCux high-entropy alloy interlayer. J. Alloys Compd. 2019, 803, 649–657. [Google Scholar] [CrossRef]
- Tsai, M.H.; Yeh, J.W.; Gan, J.Y. Diffusion barrier properties of AlMoNbSiTaTiVZr high-entropy alloy layer between copper and silicon. Thin Solid Film. 2008, 516, 5527–5530. [Google Scholar] [CrossRef]
- Singh, J.; Arora, K.S.; Shukla, D.K. Dissimilar MIG-CMT weld-brazing of aluminium to steel: A review. J. Alloys Compd. 2019, 783, 753–764. [Google Scholar] [CrossRef]
- Kobayashi, S.; Yakou, T. Control of intermetallic compound layers at interface between steel and aluminum by diffusion-treatment. Mater. Sci. Eng. A 2002, 338, 44–53. [Google Scholar] [CrossRef]
- Jimenez-Mena, N.; Jacques, P.J.; Ding, L.; Gauquelin, N.; Schryvers, D.; Idrissi, H.; Delannay, F.; Simar, A. Enhancement of toughness of Al-to-steel Friction Melt Bonded welds via metallic interlayers. Mater. Sci. Eng. A 2019, 740, 274–284. [Google Scholar] [CrossRef]
- Leshchinsky, E.; Sobiesiak, A.; Maev, R. Intermetallic Al-, Fe-, Co- and Ni-Based thermal barrier coatings prepared by cold spray for applications on low heat rejection diesel engines. J. Spray Technol. 2018, 27, 456–470. [Google Scholar] [CrossRef]
- Wu, Z.; Bei, H.; Otto, F.; Pharr, G.M.; George, E.P. Recovery, recrystallization, grain growth and phase stability of a family of FCC-structured multi-component equiatomic solid solution alloys. Intermetallics 2014, 46, 131–140. [Google Scholar] [CrossRef]
- Linden, Y.; Pinkas, M.; Munitz, A.; Meshi, L. Long-period antiphase domains and short-range order in a B2 matrix of the AlCoCrFeNi high-entropy alloy. Scr. Mater. 2017, 139, 49–52. [Google Scholar] [CrossRef]
- Lim, K.R.; Lee, K.S.; Lee, J.S.; Kim, J.Y.; Chang, H.J.; Na, Y.S. Dual-phase high-entropy alloys for high-temperature structural applications. J. Alloys Compd. 2017, 728, 1235–1238. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, X.; Liaw, P.K. Alloy design and properties optimization of high-entropy alloys. Jom 2012, 64, 830–838. [Google Scholar] [CrossRef]
- Ji, W.; Fu, Z.; Wang, W.; Wang, H.; Zhang, J.; Wang, Y.; Zhang, F. Mechanical alloying synthesis and spark plasma sintering consolidation of CoCrFeNiAl high-entropy alloy. J. Alloys Compd. 2014, 589, 61–66. [Google Scholar] [CrossRef]
- Jumaev, E.; Hong, S.H.; Kim, J.T.; Park, H.J.; Kim, Y.S.; Mun, S.C.; Park, J.Y.; Song, G.; Lee, J.K.; Min, B.H.; et al. Chemical evolution-induced strengthening on AlCoCrNi dual-phase high-entropy alloy with high specific strength. J. Alloys Compd. 2019, 777, 828–834. [Google Scholar] [CrossRef]
- Pan, H.; Liu, B.; Guo, Y.; Liu, Y.; Quan, G. An investigation on diffusion bonding of Zircaloy-4 and 304L stainless steel with Ti and Ag multiple interlayers. Mater. Lett. 2019, 240, 185–188. [Google Scholar] [CrossRef]
- Eremin, E.N.; Yurov, V.M.; Laurinas, V.C.; Guchenko, S.A. Structure and properties of corrosion-resistant coatings of the Zr-Fe-Cr-Ni-Ti system. Nanosci. Nanotechnol. NANO SciTech. 2019, 2151. [Google Scholar] [CrossRef]
- Meng, Y.H.; Duan, F.H.; Pan, J.; Li, Y. Phase stability of B2-ordered ZrTiHfCuNiFe high entropy alloy. Intermetallics 2019, 111. [Google Scholar] [CrossRef]
- Ding, W.; Wang, X.; Liu, N.; Qin, L. Diffusion bonding of copper and 304 stainless steel with an interlayer of CoCrFeMnNi High-Entropy alloy. Acta Metall. Sin. 2020, 56, 1084–1090. [Google Scholar] [CrossRef]
- Becker, N.; Gauthier, D.; Vidal, E.E. Fatigue properties of steel to aluminum transition joints produced by explosion welding. Int. J. Fatigue 2020, 139, 105736. [Google Scholar] [CrossRef]
- Bellmann, J.; Schettler, S.; Dittrich, S.; Lueg-Althoff, J.; Schulze, S.; Hahn, M.; Beyer, E.; Tekkaya, A.E. Experimental study on the magnetic pulse welding process of large aluminum tubes on steel rods. IOP Conf. Ser. Mater. Sci. Eng. 2019, 480, 012033. [Google Scholar] [CrossRef]
| Method | Structure | Reference |
|---|---|---|
| Arc-melting | BCC + B2 | [11] |
| Arc-melting | A2 + B2 | [12] |
| Arc-melting | BCC | [13] |
| Bridgman solidification | BCC | [13] |
| Mechanical alloying | BCC | [14] |
| Annealing of milled powder | BCC | [14] |
| Spark plasma sintering | BCC + FCC | [14] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waseem, O.A. Can High-Entropy Interlayers Develop Intermetallic-Free Welded Joints of Dissimilar Metals? Eng 2020, 1, 183-187. https://doi.org/10.3390/eng1020012
Waseem OA. Can High-Entropy Interlayers Develop Intermetallic-Free Welded Joints of Dissimilar Metals? Eng. 2020; 1(2):183-187. https://doi.org/10.3390/eng1020012
Chicago/Turabian StyleWaseem, Owais Ahmed. 2020. "Can High-Entropy Interlayers Develop Intermetallic-Free Welded Joints of Dissimilar Metals?" Eng 1, no. 2: 183-187. https://doi.org/10.3390/eng1020012
APA StyleWaseem, O. A. (2020). Can High-Entropy Interlayers Develop Intermetallic-Free Welded Joints of Dissimilar Metals? Eng, 1(2), 183-187. https://doi.org/10.3390/eng1020012





