1. Introduction
The enzyme urease has a variety of industrial applications, including biocementation of soil and pet stain removal. However, commercial sources of the enzyme are either highly purified and expensive laboratory and food grade powder or unprocessed meal from urease-rich agricultural products. This hinders the potential for industrial applications that require kilogram-scale supplies of the free enzyme. In particular, the adoption of enzyme induced carbonate precipitation (EICP), an emerging technique for biocementation of soil and repair of cracks in concrete, is hindered by the high cost of commercially available urease [
1]. A low-cost source of free urease enzyme would greatly facilitate the adoption of this potentially sustainable technique for various infrastructure construction and environmental protection applications that require binding together granular soil particles.
EICP binds granular soil particles together through the precipitation of calcium carbonate. The precipitate is formed from a solution containing free calcium ions, urea, and urease enzyme, with the urease acting as a catalyst for the hydrolysis of the urea (i.e., for ureolysis) [
2,
3]. As noted by Khodadadi et al. [
4], one of the primary barriers to EICP being applied in practice is the cost of the free urease enzyme. To combat this cost, Khodadadi et al. [
4] showed that urease extracted from jack beans using a simple, inexpensive technique can effectively catalyze ureolysis. However, the storage and shelf life of the crude extract needs to be investigated to address the commercial viability of the simple extraction process for mass production of urease.
Generally, enzymes are not stable in an aqueous solution: their activity is reduced or lost over time. Urease activity is defined as the micromoles of ammonia released per minute by 1 mL (if liquid extract) or 1 g (if powder in solution) of urease enzyme (i.e., U/mL or U/g). Total units, U, is therefore a measure of the urease content of a urease solution. Danial et al. [
5] stored commercial lab grade purified urease after hydration in an aqueous solution at room temperature for 60 days. These investigators observed an 85% loss of activity in the solution after just 7 days and 100% loss of activity after 60 days. El-Hefnawy et al. [
6] purified urease from germinating Pisum Sativum L. seeds and investigated the effects of storage in aqueous solution at 4 °C for up to 60 days on urease activity. They observed the activity of the aqueous solution decreased with time even when stored at 4 °C. They reported that activity decreased to 80% of the initial value on the tenth day and retained only about 14% of the initial activity after 60 days when stored at this temperature [
5,
7]. Sehgal et al. [
8] first soaked jack beans for 20 to 24 h in water and then the beans which were swollen were husked and frozen at −20 °C. Upon thawing the seeds after 4 to 6 months of storage and extracting the enzyme, they observed a loss of approximately 3% to 5% in urease activity for the soaked and frozen seeds. Das et al. [
9] found that when purified urease was stored in 0.1 M Tris–acetate buffer with pH of 6.8 at −20 °C, the activity half-life was about 50 days and the half-life decreased to 30 days when the urease was kept at 4 °C. They also observed that by adding of 5 mM Dithiothreitol (DTT) the half-life at −20 °C increased to 3 months [
10].
Powderization of an aqueous medium by freeze drying (lyophilization) is often used to facilitate transportation and storage of biological materials. In particular, lyophilization is a common method for preparing protein samples for long-term storage. However, the freezing and drying processes can cause protein denaturation [
11,
12]. Preservation of protein structure and function during lyophilization may require stabilization against both freezing and drying with appropriate additives. Dextran and sucrose have both been used for this purpose, though they protect proteins during these stresses by distinctly different mechanisms [
12,
13]. Schneider et al. [
14] reported that purified commercial urease in the form of powder retained only 20% of its activity upon re-hydration immediately after lyophilization and less than 1% following rehydration after 6 weeks of storage at 33 °C. However, urease stabilized with the addition of dextran prior to lyophilization retained 100% of its activity following rehydration after 3 months storage.
Scope of This Study
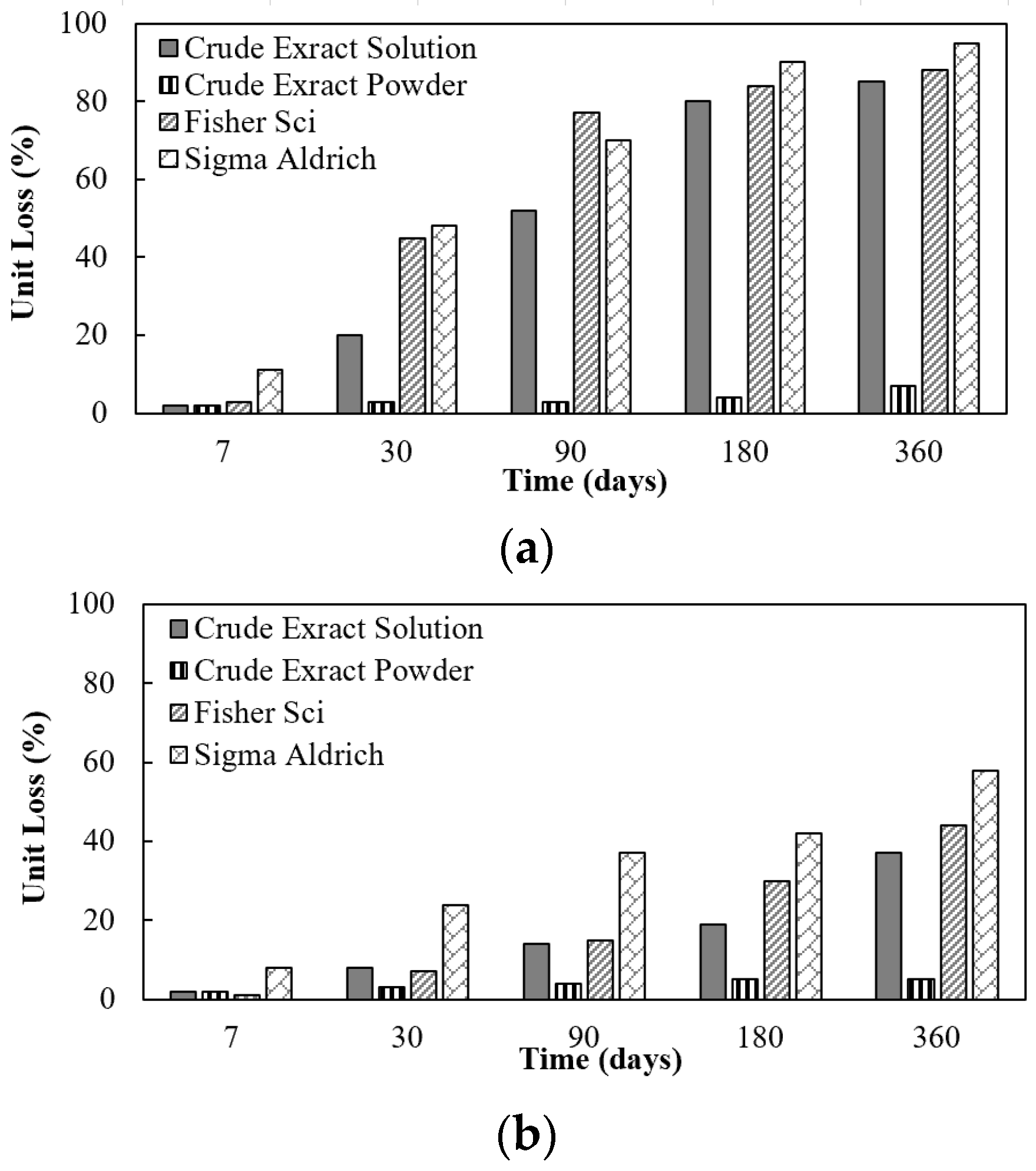
As part of an effort to lower the cost of the urease enzyme used in EICP for soil improvement, the stability of three crude urease extracts from jack bean, a urease-rich agricultural product, was evaluated and compared to the stability of commercially available urease powder. The effect of dehusking the jack beans and of filtration on the urease activity of the crude extract after storage as an aqueous solution for a period of up to one year was also evaluated. Three crude extract solutions were prepared: (a) a solution prepared using dehusked beans followed by filtration; (b) an unfiltered solution prepared using dehusked beans; and (c) an unfiltered solution prepared using whole crushed jack beans (without removal of the husks prior to crushing the beans). The effect of storage of the crude extract as an aqueous liquid, of the crude extract upon rehydration following storage in the form of powder (i.e., after lyophilization), and of two commercial urease powders when dissolved in an aqueous solution on urease activity was also evaluated. The effect of storage of the extract in aqueous solution and after rehydration of commercial powdered urease was evaluated both at room temperature (RT) and at 4 °C. In addition, the effect of treatment of the crude extract with dextran and with dextran and sucrose before lyophilization on its stability was investigated.
2. Experimental Program
2.1. Materials
Jack beans (Canavalia Gladiata, Sheffield’s Seed) were obtained from a commercial vendor. Commercial lab grade urease enzymes from Sigma Aldrich (U1500, Type III, powder, 42,700 U/g activity, Sigma Aldrich, St. Louis, MO) and Fisher Scientific (U2125, powder, no activity reported on the label, Fisher Scientific, Hampton, NH, USA) were also used in this study.
2.2. Urease Extraction and Solution Preparation
Jack beans with and without husk were soaked in tap water, rather than in a buffer as used in traditional extraction, and stored at 4 °C for 24 h. Dehusking was performed manually, a laborious and time consuming procedure. The ratio of jack beans to tap water was 1:4 (w/v). For example, to obtain 20 g of dehusked jack beans, approximately 23 g of jack bean were manually dehusked (husk comprises approximately 14% of the mass of jack beans) and the 20 g of dehusked jack beans were then soaked in 80 mL tap water. The soaked jack beans were then pulverized in a kitchen blender for 2 min and filtered through a muslin cloth to remove any coarse solids from the solution. The beans tested without dehusking, referred to herein as whole crushed beans, were simply soaked, without dehusking, and then pulverized and filtered as described above. After that, the remaining solution (i.e., the supernatant) was filtered through a thick layer of glass wool to reduce the amount of fats present. The solution obtained through this process is referred to herein as “crude extract.” In one case the crude extract from dehusked beans was filtered through a 0.25 nm filter. This extract is referred to as the filtered crude extract. Note that that the ultracentrifugation step typically used for extraction was not used in the crude extraction procedure.
Thirty (30) ml of crude extract made from dehusked jack beans and 30 mL made from whole crushed beans with measured activities of 286 and 141 U/mL, respectively, were stored at RT and at 4 °C prior to additional activity measurement after different storage times. Both unfiltered and filtered solution made from the dehusked beans were tested (they had approximately the same activity) and stored. The solution made from the whole crushed beans was not filtered.
Aqueous solutions prepared from commercially available powdered urease purchased from Fisher Scientific and from Sigma Aldrich and from lyophilized crude extract were also stored and tested in the same manner as the liquid crude extract. A solution containing 355 enzyme units was prepared by dissolving 0.15 g of Fisher Scientific enzyme with a measured activity of 2500 U/g in 10 mL of deionized (DI) water. A solution containing 770 enzyme units was prepared by dissolving 0.05 g of high activity Sigma Aldrich enzyme with a measured activity of 15,000 U/g in 10 mL of DI water. A solution containing 560 enzyme units (U) was prepared by dissolving 0.15 g of the lyophilized crude extract in 10 mL DI water.
2.3. Urease Activity Measurement
Enzyme activity of the crude extract was assayed in a 300 mM urea solution (pH = 7). An aliquot (4.7 mL) of DI water and 5 mL of 300 mM urea were mixed at room temperature in a vial. The ureolysis reaction was then started by adding 0.3 mL of crude urease solution to the DI water-urea solution and the vial was immediately capped and shaken gently at time intervals of 3, 5, and 10 min, 5 mL of 15% trichloroacetic acid was immediately added to the reaction solution (i.e., to the water, urea, and urease solution) and then the vials were opened to stop the reaction. After stopping the reaction, the solution was diluted 100-fold using DI water in a volumetric flask. Then 2 mL of this solution was mixed in a cuvette containing 100 µL of Nessler’s reagent. After 2 min, the cuvette was placed in a spectrophotometer to measure the optical density of the solution at a wavelength of 412 nm (i.e., OD412) to determine the concentration of ammonium.
2.4. Lyophilization
Each 20 g of dehusked jack beans when mixed with 80 mL of tap water yielded 32 mL of jack bean crude extract after the extraction process described above. Fifty (50) ml test tubes containing the crude extract were then placed in a −80 °C freezer. After freezing, the samples in the test tubes were capped with parafilm (leaving small holes on the top of each tube) and then placed onto the racks of a Labconco Freezone 6 lyophilizer and processed at a vacuum pressure of 50 mT (millitorr) at −50 °C. The lyophilizer uses a condenser that is kept colder than the samples and a vacuum to drive the sublimation process, extracting water vapor from the test tube and depositing it upon the coil. The samples were retained in the lyophilizer until the extract turned to powder, indicating that the residual moisture has been removed. The mass of the powder from lyophilizing 32 mL of crude extract was approximately 3.91 g. After the lyophilization, the powdered extract was stored at RT and 4 °C until it was rehydrated.
2.5. Whole Bean Storage and Dextran and Sucrose Stabilization
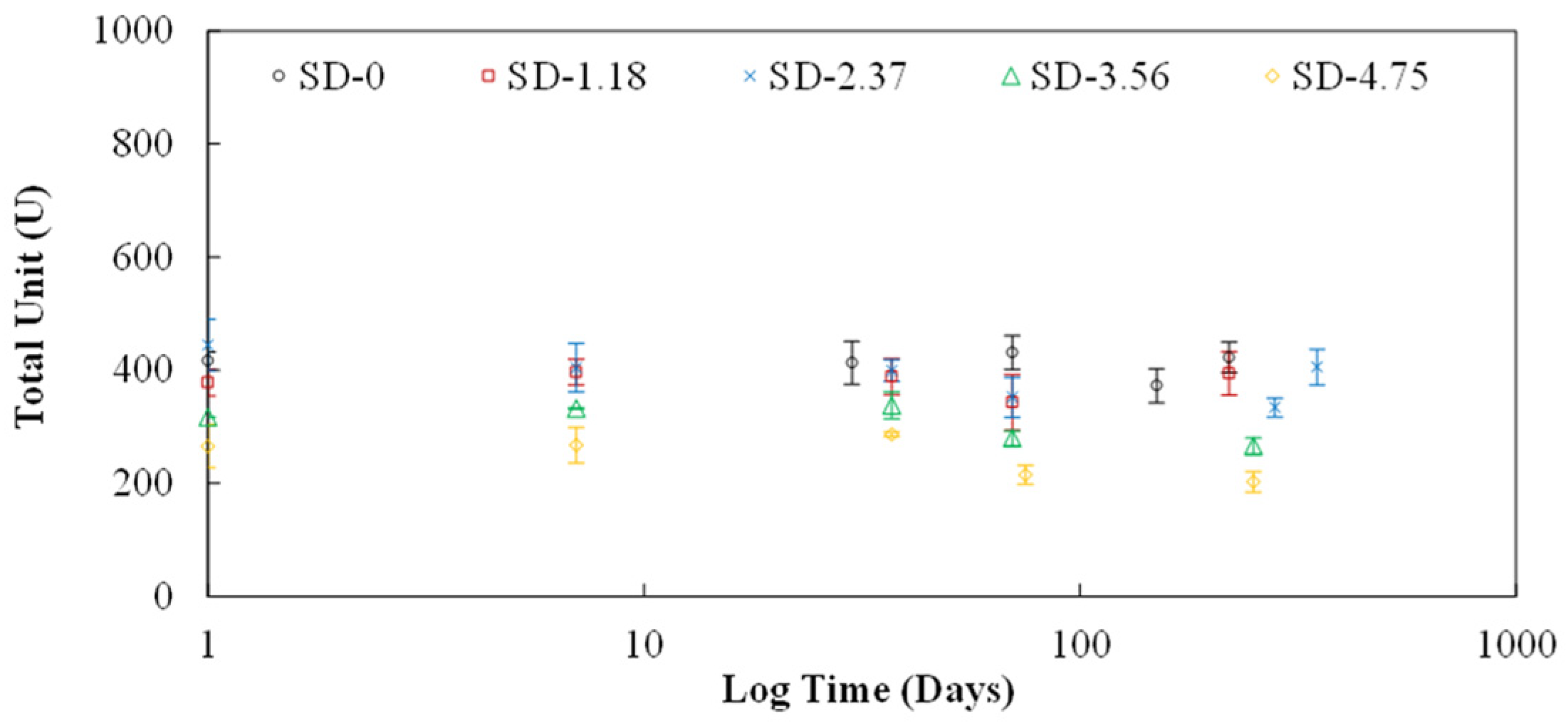
In addition to the effect of storage on the activity of the hydrated commercial enzymes, the liquid extract, and the lyophilized crude extract following rehydation, the effect of storage on unprocessed jack beans stored at RT and 4 °C was also examined. The jack beans were stored whole and then periodically sampled to create crude extract and make activity measurements in the same manner as described above (i.e., using the Nessler method). In addition, as part of the effort to enhance the stability of the crude extract, the effect of adding (a) dextran and (b) dextran with sucrose in the crude extract prior to lyophilization was evaluated. Various concentrations of dextran and dextran with sucrose were added to the crude extract solutions. The concentrations of dextran and sucrose used in this study were based upon previous work on enzyme stabilization using these substances [
15].
4. Conclusions
The storage capability of urease extracted from jack beans through a simple extraction procedure was investigated for three different extraction solutions, at two different temperatures, before and after lyophilization. The simple extraction process requires only a kitchen blender, muslin cloth, and glass wool, uses tap water rather than a buffer solution, and does not require centrifugation. The stability of the jack bean crude extract stored at room temperature and 4 °C in both solution and upon rehydration following lyophilization was studied by measuring the urease activity at storage times of up to one year (360 days). The key findings of this study can be summarized as follows:
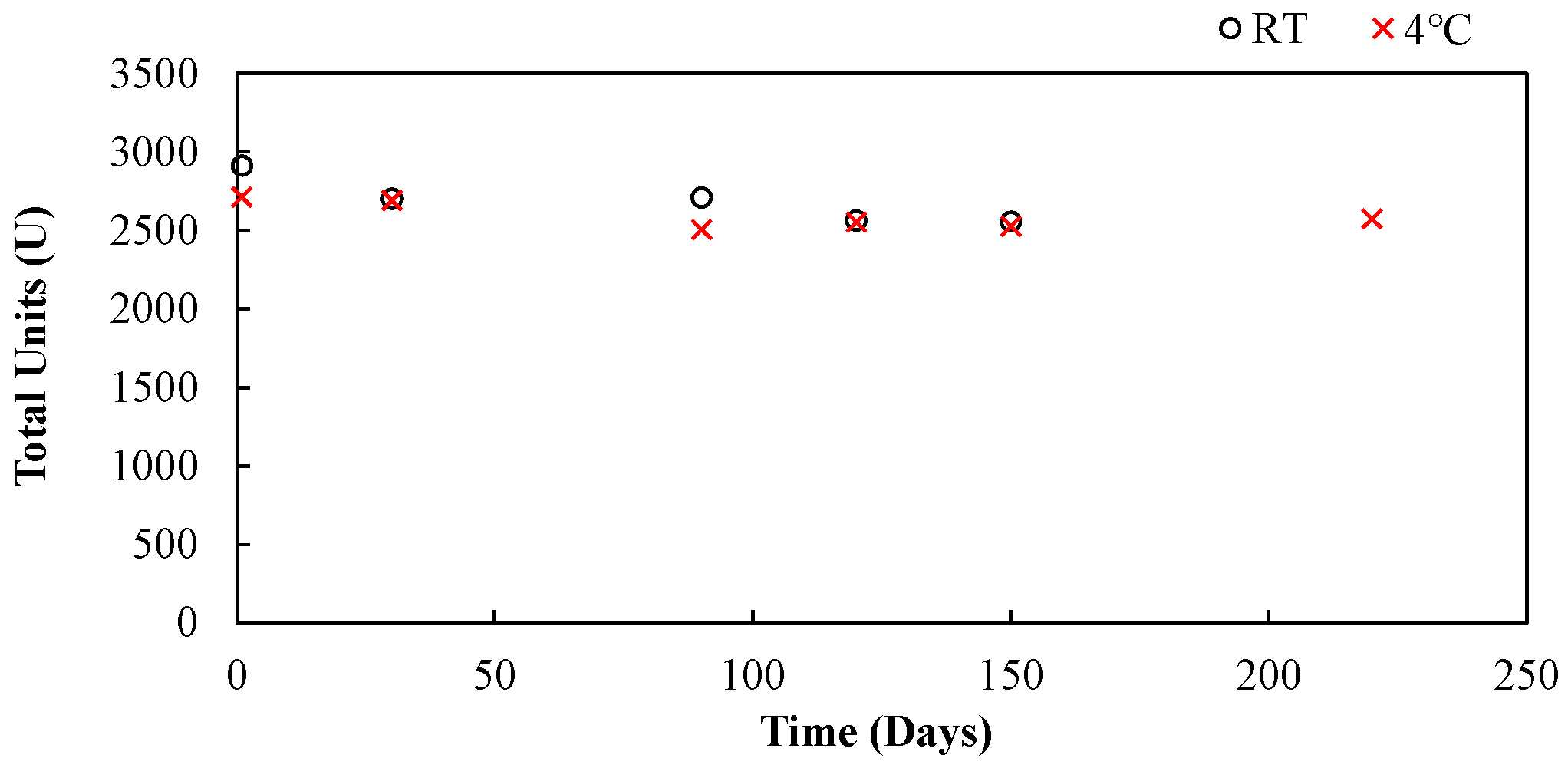
The effect of the storage on the whole jack beans prior to extraction when stored at room temperature and 4 °C was insignificant.
The stability (storage capability) of the crude extract improved by storing it at 4 °C rather than room temperature.
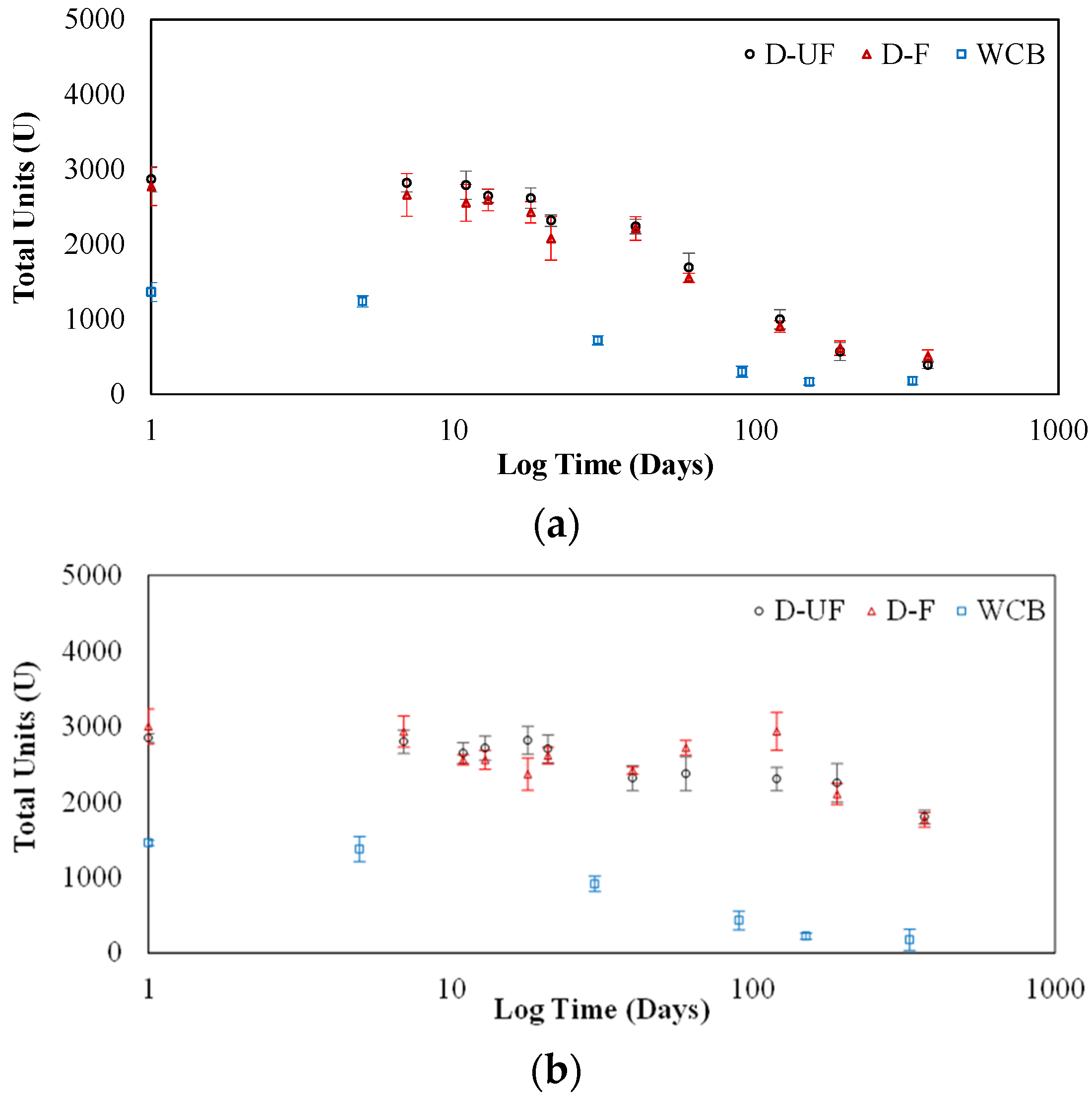
Filtration and dehusking steps are not required as part of the crude extraction process, further simplifying the process; however, the dehusking step was effective for maintaining the stability of the urease in the crude extract, particularly when stored at 4 °C.
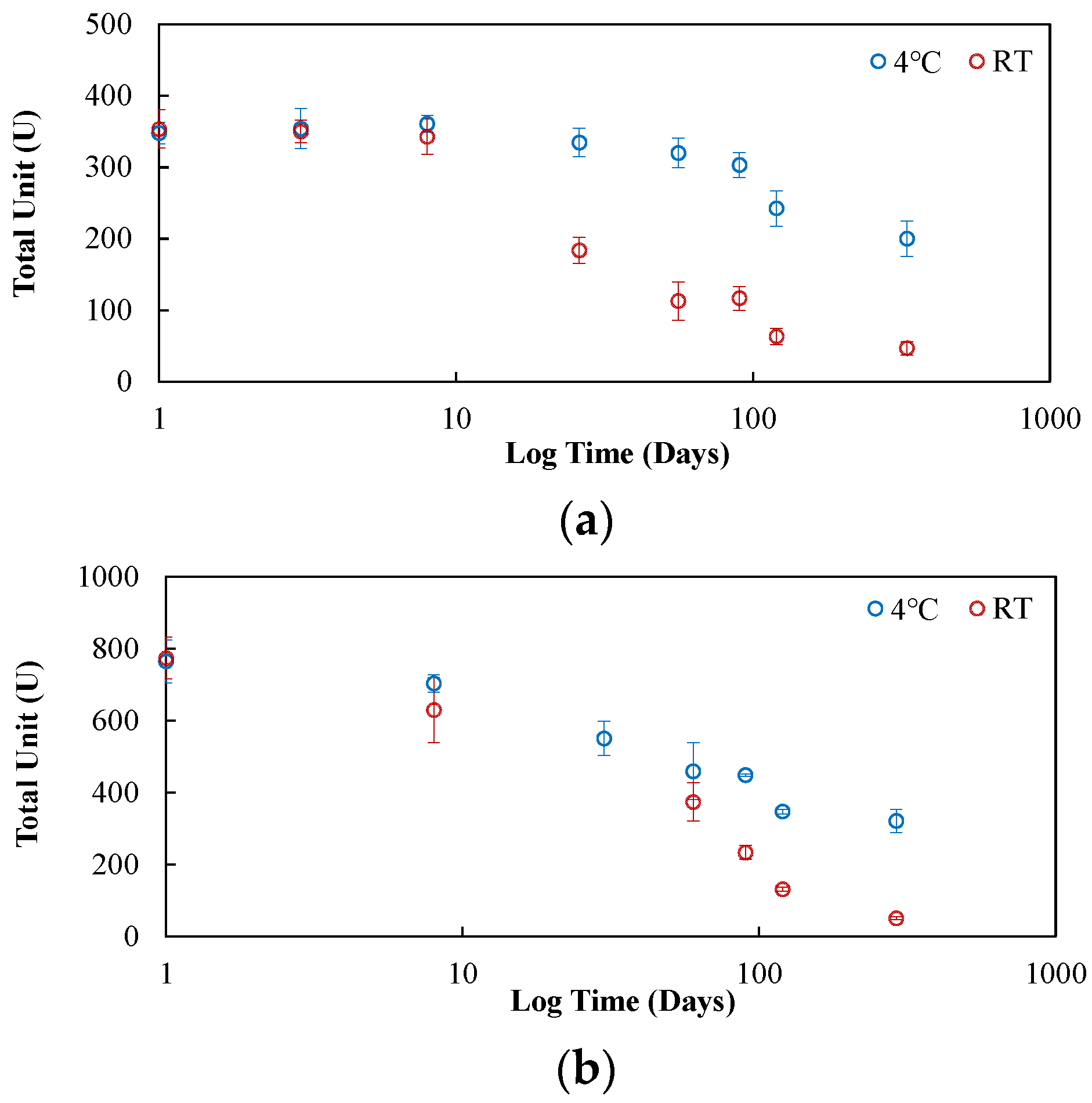
The jack bean crude extract both in solution and when lyophilized showed better stability (longer storage capability) compared to the commercialized enzymes in solution.
Lyophilization of the crude extract was found to be the most effective approach to prolong the storage capability of the enzyme. After storing the lyophilized crude extract for 365 days, the loss of urease activity following rehydration was less than 10% regardless of the storage temperature.
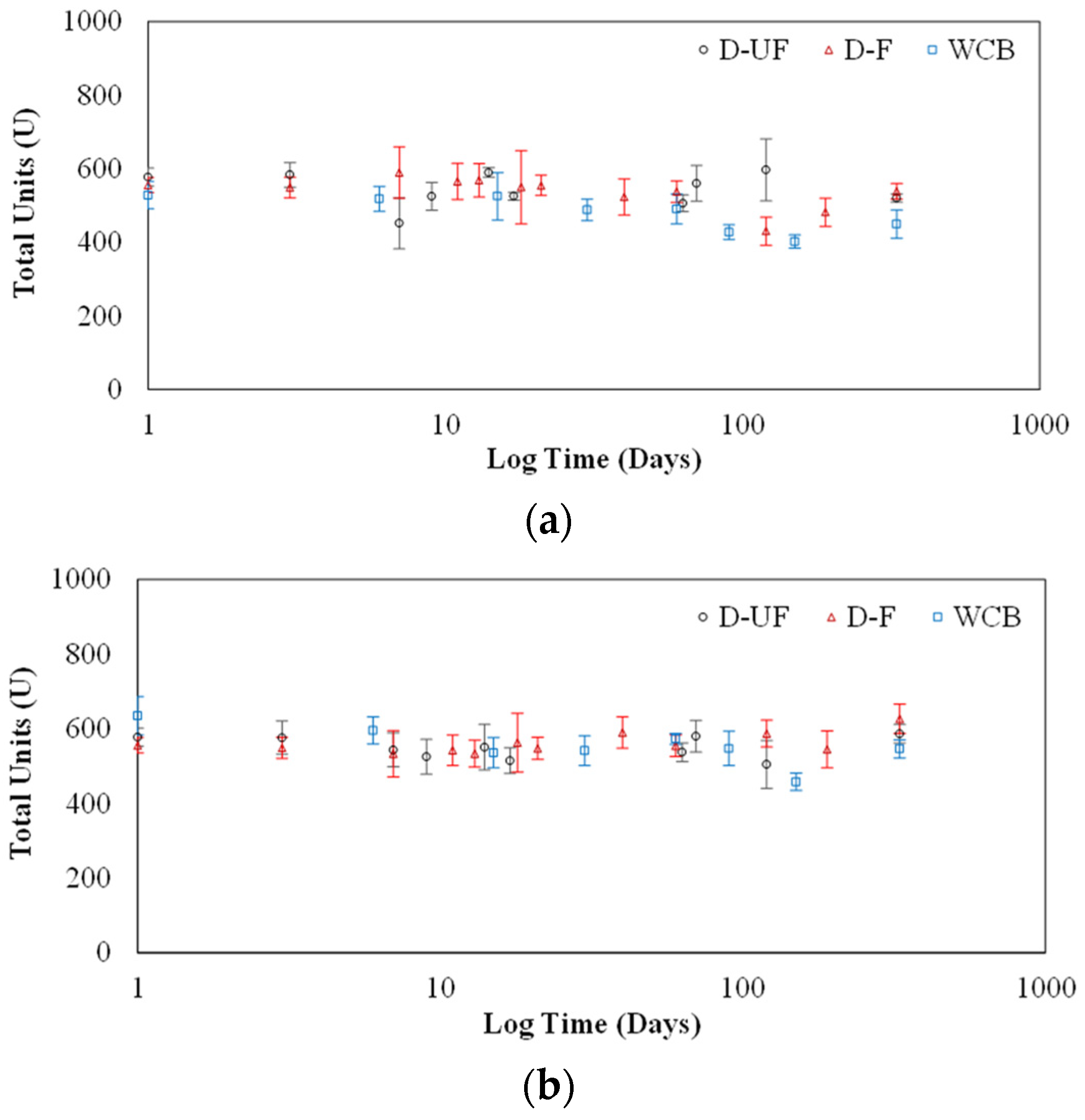
The addition of 2% dextran into lyophilized crude extract provided no significant benefit in maintaining stability of urease.
The use of hybrid dextran-sucrose at 2.37% enhanced the storage capability of the lyophilized crude extract, resulting in little to no loss of activity over 365 days of storage.
This demonstrates that the crude extract can be stored after production followed by lyophilization for periods in excess of one year without a significant loss of activity offers the potential for a significant reduction in the cost of applying enzyme induced carbonate precipitation (EICP) in engineering practice by mass production of the enzyme.