Parallel Synthesis of Aurones Using a Homogeneous Scavenger
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bate-Smith, E.C.; Geissman, T.A. Benzalcoumaranones. Nature 1951, 167, 688. [Google Scholar] [CrossRef]
- Mazziotti, I.; Petrarolo, G.; La Motta, C. Aurones: A Golden Resource for Active Compounds. Molecules 2022, 27, 2. [Google Scholar] [CrossRef] [PubMed]
- Sui, G.; Li, T.; Zhang, B.; Wang, R.; Hao, H.; Zhou, W. Recent advances on synthesis and biological qactivities of aurones. Bioorg. Med. Chem. 2021, 29, 115895. [Google Scholar] [CrossRef]
- Sutton, C.L.; Taylor, Z.E.; Farone, M.B.; Handy, S.T. Antifungal activity of substituted aurones. Bioorg. Med. Chem. Lett. 2017, 27, 901. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Nelson, D.E.; Taylor, Z.E.; Hayes, J.B.; Cunningham, K.D.; Arivett, B.A.; Ghosh, R.; Wolf, L.C.; Taylor, K.M.; Farone, M.B.; et al. Suppression of LPS-induced NF-κB activity in macrophages by the synthetic aurone, (Z)-2-((5-(hydroxymethyl) furan-2-yl) methylene) benzofuran-3(2H)-one. Int. Immunopharm. 2017, 43, 116. [Google Scholar] [CrossRef]
- Schmitt, J.; Handy, S.T. A golden opportunity: Benzofuranone modifications of aurones and their influence on optical properties, toxicity, and potential as dyes. Beilstein J. Org. Chem. 2019, 15, 1781–1785. [Google Scholar] [CrossRef] [PubMed]
- Algahtani, F.M.; Arivett, B.A.; Taylor, Z.E.; Handy, S.T.; Farone, A.L.; Farone, M.B. Chemogenomic profiling to understand the antifungal action of a bioactive aurone compound. PLoS ONE 2019, 14, e0226068. [Google Scholar]
- Alqahtani, F.M.; Handy, S.T.; Sutton, C.L.; Farone, M.B. Combining Genome-Wide Gene Expression Analysis (RNA-seq) and a Gene Editing Platform (CRISPR-Cas9) to Uncover the Selectively Pro-oxidant Activity of Aurone Compounds Against Candida albicans. Front. Microbiol. 2021, 12, 708267. [Google Scholar] [CrossRef]
- Iwashina, T. The structure and distribution of the flavonoids in plants. J. Plant Res. 2000, 113, 287. [Google Scholar] [CrossRef]
- Agrawal, N.N.; Soni, P.A. A New Process for the Synthesis of Aurones by Using Mercury (II) Acetate in Pyridine and Cupric Bromide in Dimethyl Sulfoxide. Indian J. Chem. 2006, 45, 1301–1303. [Google Scholar] [CrossRef]
- Thanigaimalai, P.; Yang, H.M. Structural requirement of chalcones for the inhibitory activity of interleukin-5. Bioorg. Med. Chem. 2010, 18, 4441–4445. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.; Bolshan, Y. Metal-Free Synthesis of Ynones from Acyl Chlorides and Potassium Alkynyltrifluoroborate Salts. Tetrahedron Lett. 2015, 56, 4392–4396. [Google Scholar] [CrossRef]
- Harkat, H.; Blanc, A.; Weibel, J.-M.; Pale, P. Versatile and expeditious synthesis of aurones via Au I-catalyzed cyclization. J. Org. Chem. 2008, 73, 1620–1623. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.M.; Liu, M.W.; Yue, Y.Y.; Zhang, N.F.; Zhang, Y.I.; Zhuo, K.I. Construction of the flavones and aurones through regioselective carbonylative annulation of 2-bromophenols and terminal alkynes. Tetrahedron Lett. 2013, 54, 1802–1807. [Google Scholar] [CrossRef]
- Qi, X.X.; Li, R.; Wu, X.F. Selective Palladium-catalyzed carbonylative synthesis of aurones with formic acid as the CO source. RSC Adv. 2016, 6, 2810–62813. [Google Scholar] [CrossRef]
- Kayal, S.; Mukherjee, S. Catalytic enantioselective cascade Michael/cyclization reaction of 3-isothiocyanato oxindoles with exocyclic α,β-unsaturated ketones en route to 3,2′-pyrrolidinyl bispirooxindoles. Org. Biomol. Chem. 2016, 14, 10175–10179. [Google Scholar] [CrossRef]
- Lee, Y.H.; Shin, M.C.; Yun, Y.D.; Shin, S.Y.; Kim, J.M.; Seo, J.M.; Kim, N.-J.; Ryu, J.H.; Lee, Y.S. Synthesis of aminoalkyl-substituted aurone derivatives as acetylcholinesterase inhibitors. Bioorg. Med. Chem. 2015, 23, 231–240. [Google Scholar] [CrossRef]
- Varma, R.S.; Varma, M. Alumina-mediated condensation. A simple synthesis of aurones. Tetrahedron Lett. 1992, 33, 5937. [Google Scholar] [CrossRef]
- Villemin, D.; Martin, B.; Bar, N. Application of Microwave in Organic Synthesis. Dry Synthesis of 2-Arylmethylene-3(2)-naphthofuranones. Molecules 1998, 3, 88. [Google Scholar] [CrossRef] [Green Version]
- Venkateswarlu, S.; Murty, G.N.; Saryanarayana, M. “On water” synthesis of aurones: First synthesis of 4,5,3′,4′,5′-pentamethoxy-6-hydroxyaurone from Smilax riparia. ARKIVOC 2017, 4, 303. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, I.; Handy, S.T. Synthesis of aurones under neutral conditions using a deep eutectic solvent. Tetrahedron 2013, 69, 9200. [Google Scholar] [CrossRef]
- Yellol, G.S.; Sun, C.-M. Green Techniques for Organic Synthesis and Medicinal Chemistry; Zhang, W., Cue, B.W., Jr., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2012; pp. 393–442. [Google Scholar]
- Lu, J.; Toy, P.H. Organic polymer supports for synthesis and for reagent and catalyst immobilization. Chem. Rev. 2009, 109, 815–836. [Google Scholar] [CrossRef] [PubMed]
- Moses, J.E.; Moorhouse, A.D. The growing applications of click chemistry. Chem. Soc. Rev. 2007, 36, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Le Lamer, A.-C.; Gouault, N.; David, M.; Boustie, J.; Uriac, P. Method for the parallel synthesis of alpha-methylene-gamma-lactones from a fluorous acrylate. J. Comb. Chem. 2006, 8, 643–645. [Google Scholar] [CrossRef]
- Ley, S.V.; Baxendale, I.R.; Bream, R.N.; Jackson, P.S.; Leach, A.G.; Longbottom, D.A.; Nesi, M.; Scott, J.S.; Storer, R.I.; Taylor, S.J. Multi-step organic synthesis using solid-supported reagents and scavengers: A new paradigm in chemical library generation. J. Chem. Soc. Perkin Trans. 1 2000, 23, 3815. [Google Scholar] [CrossRef]
- Boucher, M.M.; Furigay, M.H.; Quach, P.K.; Brindle, C.S. Liquid–Liquid Extraction Protocol for the Removal of Aldehydes and Highly Reactive Ketones from Mixtures. Org. Process Res. Dev. 2017, 21, 1394–1403. [Google Scholar] [CrossRef]
- Kafle, A.; Bhatarai, S.; Handy, S.T. An Unusual Triazole Synthesis from Aurones. Synthesis 2020, 2337–2346. [Google Scholar]
- Taylor, K.M.; Taylor, Z.E.; Handy, S.T. Rapid synthesis of aurones under mild conditions using a combination of microwaves and deep eutectic solvents. Tetrahedron Lett. 2017, 58, 240–241. [Google Scholar] [CrossRef]
- Xu, H.; Ziao, H.; Hu, X.; Zuan, G.; Li, P.; Zhang, Z. Synthesis of Fully Substituted 5-(o-Hydroxybenzoyl)imidazoles via Iodine-Promoted Domino Reactions of Aurones with Amidines. J. Org. Chem. 2022, 87, 16204–16212. [Google Scholar] [CrossRef]
- Flynn, D.L.; Crich, J.Z.; Devraj, R.V.; Hockerman, S.L.; Parlow, J.J.; South, M.S.; Woodard, S. Chemical Library Purification Strategies Based on Principles of Complementary Molecular Reactivity and Molecular Recognition. J. Am. Chem. Soc. 1997, 119, 4874. [Google Scholar] [CrossRef]
- De Oliveira, A.V.B.; Kartnaller, V.; Pedrosa, M.S.P.; Cajaiba, J.J. Isoniazid as an Aldehdye Scavenger: Analysis of Its Kinetics, Selectivity, and Practicality in Purifying Organic Reactions. Appl. Polym. Sci. 2015, 132, 1. [Google Scholar]
- Kassehin, U.C.; Gbaguidi, F.A.; Kapanda, C.N.; McCurdy, C.R.; Poupaert, J.H. Solvent effect and catalysis in the synthesis of thiosemicarbazone derivatives from ketones and 4’-phenylthiosemicarbazide. Afr. J. Pure App. Chem. 2014, 8, 110–115. [Google Scholar]

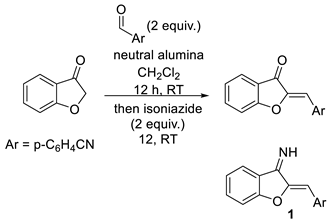 | |||||
|---|---|---|---|---|---|
| Entry | Scavenger a | Amount | % Yield | % Purity b | Cost c |
| 1 | p-Toluenesulfonyl hydroazide–polymer bound | 200 mg | 0 d | N/A | $18 |
| 2 | Sulfonylamide–polymer bound | 275 mg | 72 | >95 | $43 |
| 3 | Ethylenediamine-polymer bound | 100 mg | 72 | >95 | $12 |
| 4 | isoniazid | 55 mg | 70 | >95 | $0.22 |
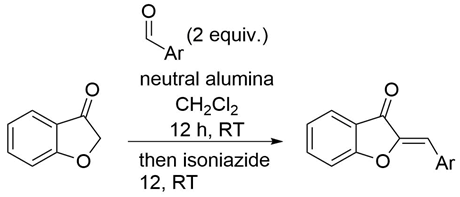 | |||
|---|---|---|---|
| Entry | Ar | % Yield | % Purity a |
| 1 | 4-Cyanophenyl | 70 | >95 |
| 2 | 4-Trifluoromethylphenyl | 44 | >95 |
| 3 | 4-Dimethylaminophenyl | 25 | >95 |
| 4 | 4-Methylphenyl | 57 | >95 |
| 5 | 4-Methoxyphenyl | 61 | >95 |
| 6 | 4-Methyl carboxyphenyl | 49 | 77 |
| 7 | 2-Bromophenyl | 64 | >95 |
| 8 | 3-Bromophenyl | 39 | >95 |
| 9 | 4-Bromophenyl | 53 | >95 |
| 10 | 2-Thiophenyl | 36 | >95 |
| 11 | 2-Furyl | 37 | >95 |
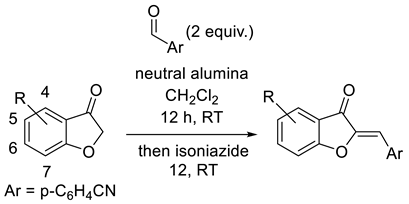 | |||
|---|---|---|---|
| Entry | Substituent | % Yield | % Purity a |
| 1 | 4-Chloro | 46 | >95 |
| 2 | 5-Chloro | 31 | >95 |
| 3 | 4-Fluoro | 55 | >95 |
| 4 | 6-Fluoro | 39 | >95 |
| 5 | 5-Bromo | 68 | >95 |
| Carbonyl Compound | Mean Rate Constant (103 s−1) | Relative Rate |
|---|---|---|
| Benzaldehyde | −0.410 ± 0.047 | 1.00 |
| 4-Nitrobenzaldehyde | −2.350 ± 0.500 | 5.73 |
| 4-Cyanobenzaldehyde | −1.399 ± 0.271 | 3.41 |
| 4-Bromobenzaldehyde | −0.584 ± 0.088 | 1.42 |
| 4-Methylbenzaldehyde | −0.141 ± 0.024 | 0.34 |
| 4-Methoxybenzaldehyde | −0.468 ± 0.054 | 1.14 |
| 3-Methoxybenzladehyde | −0.731 ± 0.053 | 1.78 |
| 2-Methoxybenzaldehyde | −2.583 ± 0.629 | 6.30 |
| Trans-cinnamaldehyde | −0.844 ± 0.084 | 2.06 |
| Dihydrocinnamaldehyde | −1.509 ± 0.620 | 3.68 |
| Thiophene-2-carboxyaldehyde | −0.320 ± 0.072 | 0.78 |
| Furan-2-carboxaldehyde | −1.433 ± 0.364 | 3.50 |
| 2-Octanone | −0.0436 ± 0.00548 | 0.11 |
| Cyclohexanone | −0.846 ± 0.0180 | 2.06 |
| Acetophenone | 0.00045 ± 0.0028 | 0.00 |
| Benzophenone | −0.0047 ± 0.0024 | 0.01 |
| Ethyl acetoacetate | −0.150 ± 0.0102 | 0.37 |
| Butyl acetate | −0.0013 ± 0.0025 | 0.00 |
| Methyl benzoate | −0.00024 ± 0.0014 | 0.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, Z.E.; Handy, S.T. Parallel Synthesis of Aurones Using a Homogeneous Scavenger. Organics 2023, 4, 51-58. https://doi.org/10.3390/org4010004
Taylor ZE, Handy ST. Parallel Synthesis of Aurones Using a Homogeneous Scavenger. Organics. 2023; 4(1):51-58. https://doi.org/10.3390/org4010004
Chicago/Turabian StyleTaylor, Zachary E., and Scott T. Handy. 2023. "Parallel Synthesis of Aurones Using a Homogeneous Scavenger" Organics 4, no. 1: 51-58. https://doi.org/10.3390/org4010004
APA StyleTaylor, Z. E., & Handy, S. T. (2023). Parallel Synthesis of Aurones Using a Homogeneous Scavenger. Organics, 4(1), 51-58. https://doi.org/10.3390/org4010004








