Exploring the Limits of Reactivity of N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) with Disubstituted Bicycloalkadienes in the Homo-Diels–Alder Reaction
Abstract
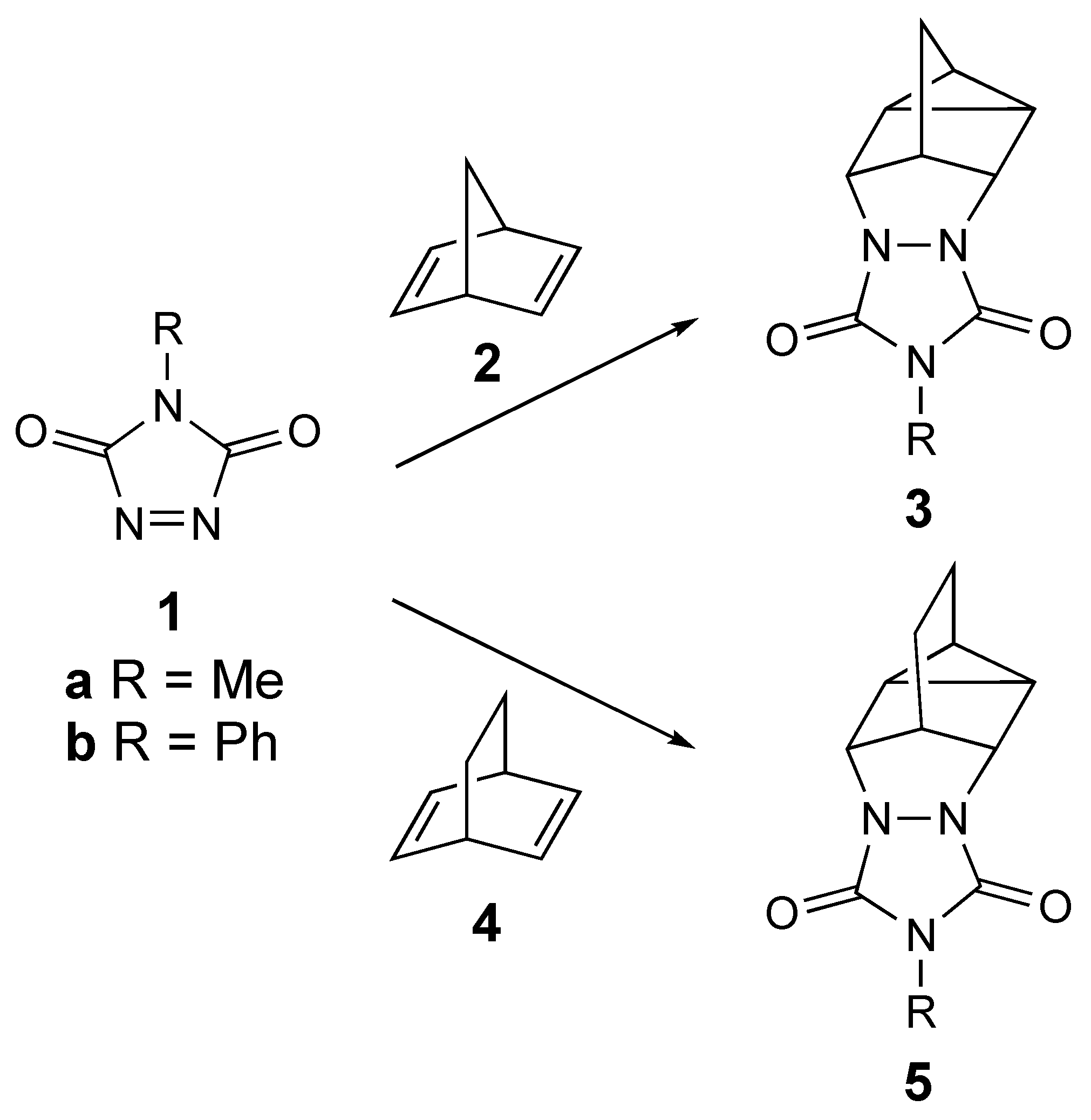
1. Introduction
2. Materials and Methods
2.1. General Methods
2.2. Experimental Procedures
2.2.1. Reaction of MeTAD with 9a
2.2.2. Attempted Reaction of MeTAD with 9b
2.2.3. Reaction of MeTAD with 10a
2.2.4. Attempted Reaction of MeTAD with 10b
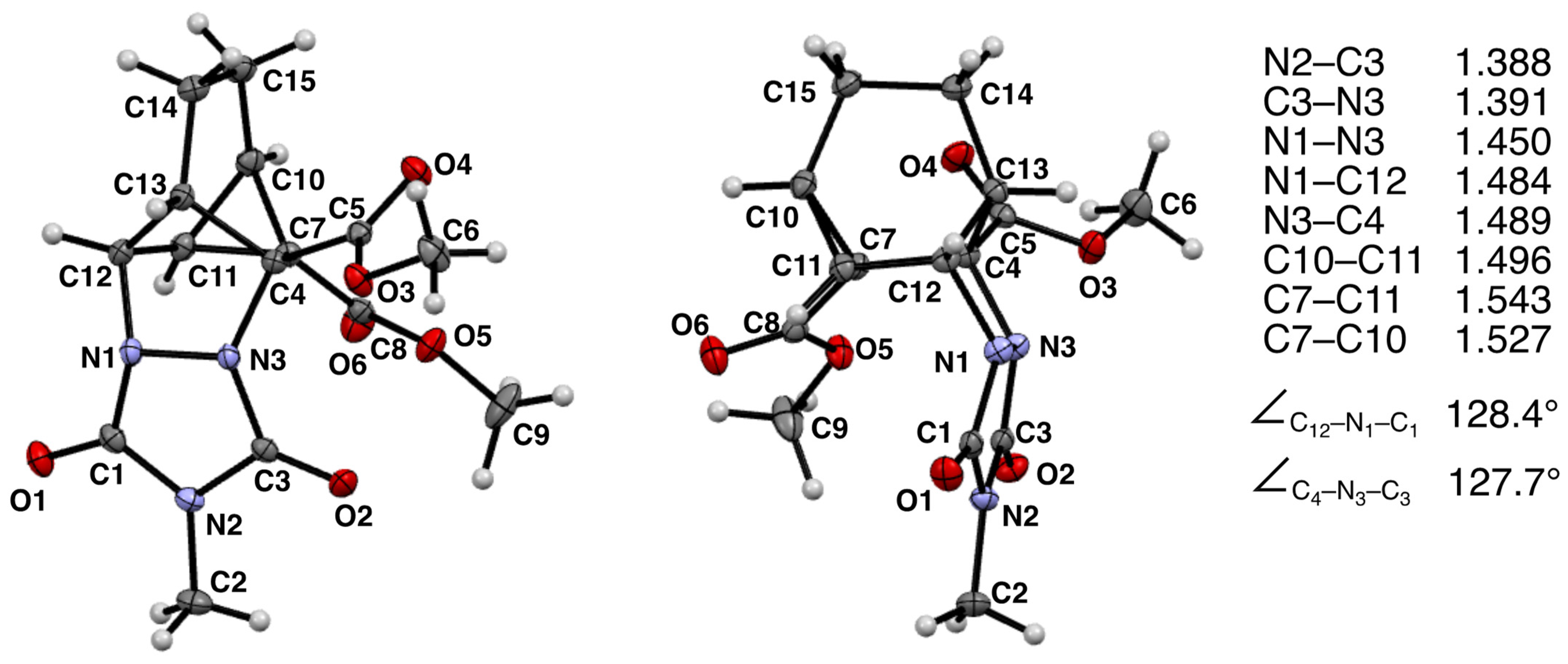
2.3. X-ray Crystallographic Analysis
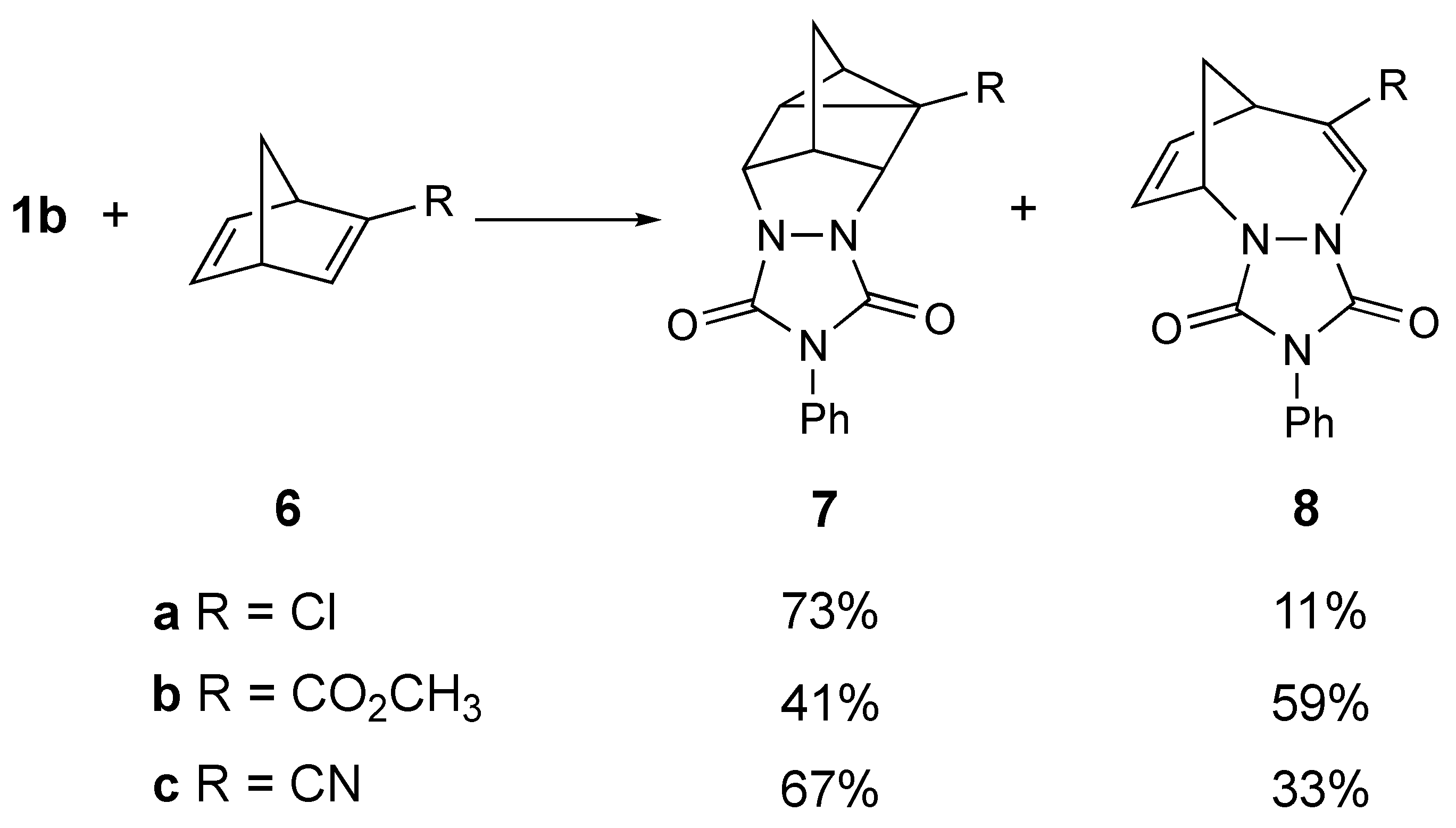
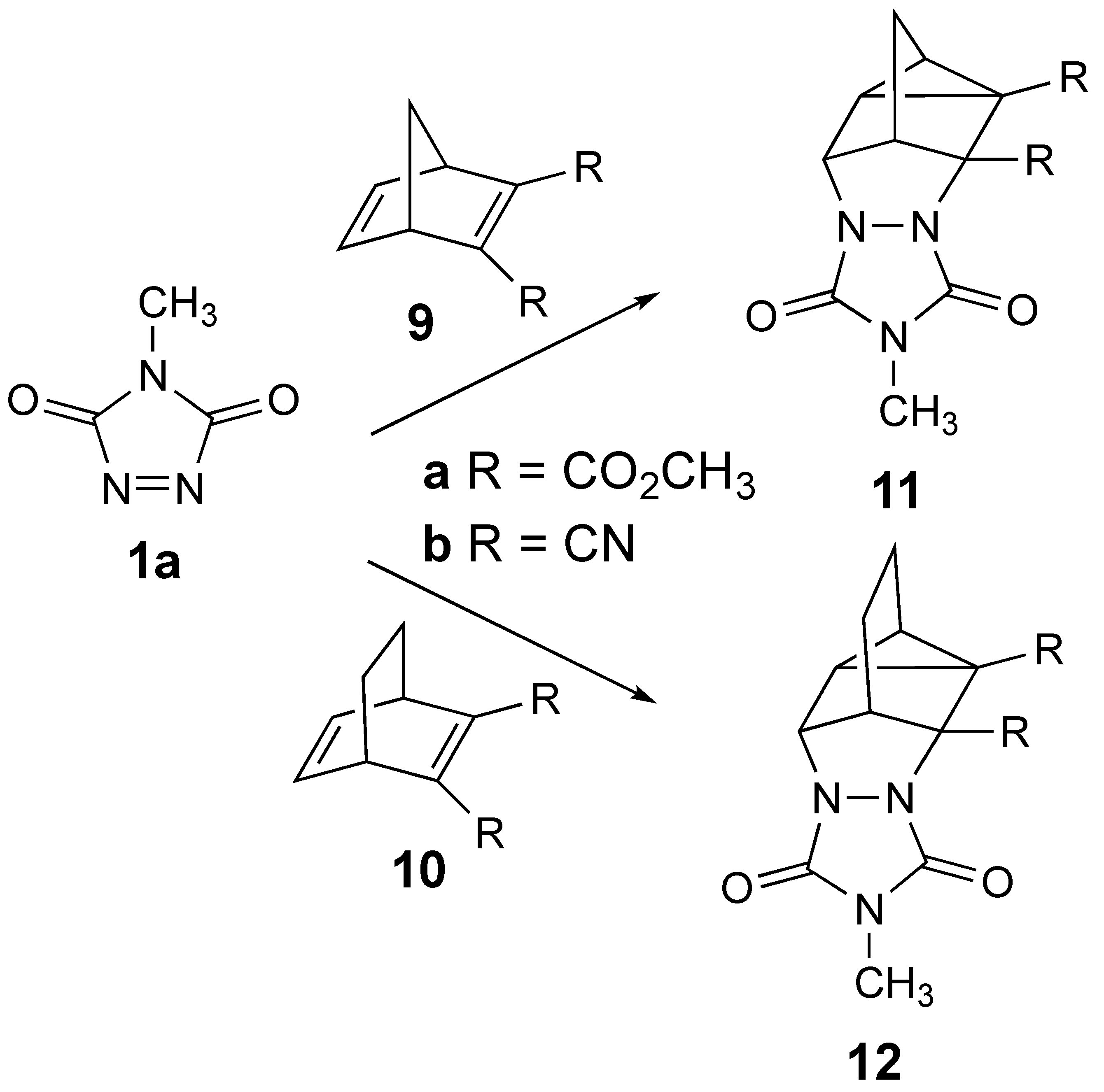
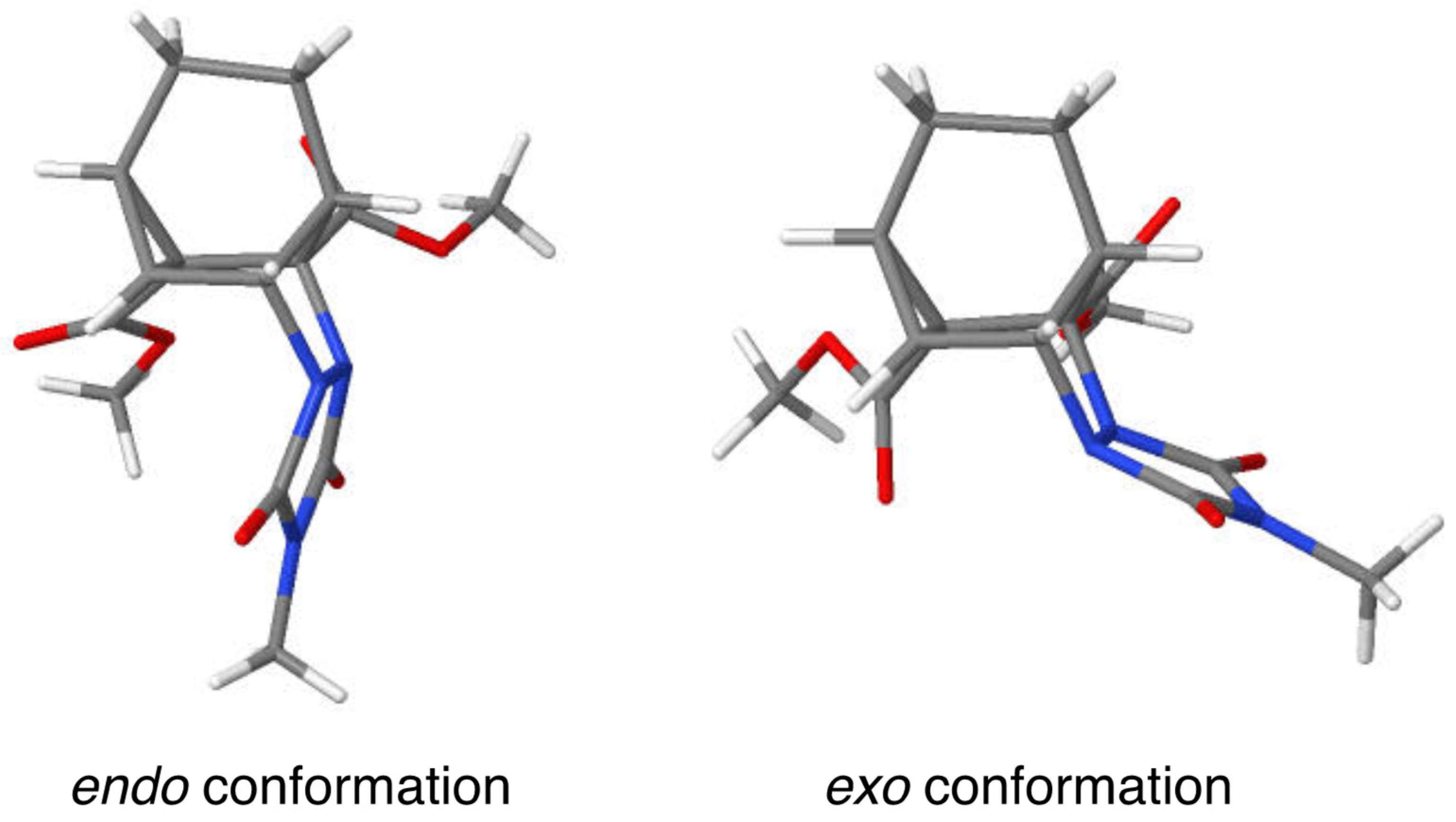
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cookson, R.C.; Gilani, S.S.H.; Stevens, I.D.R. Diels-Alder Reactions of 4-Phenyl-1,2,4-triazoline-3,5-dione. J. Chem. Soc. C. 1967, 1905–1909. [Google Scholar] [CrossRef]
- Allred, E.L.; Johnson, A.L. The Influence of Geometry on Cyclopropyl Participation in the Thermolysis of Azo Compounds. J. Am. Chem. Soc. 1971, 93, 1300–1301. [Google Scholar] [CrossRef]
- Burrage, M.E.; Cookson, R.C.; Gupte, S.S.; Stevens, I.D.R. Substituent and Solvent Effects on the Diels-Alder Reactions of Triazolinediones. J. Chem. Soc. Perkin Trans. 2 1975, 1325–1334. [Google Scholar] [CrossRef]
- Erden, I.; de Meijere, A. Convenient New Synthesis of Snoutene Utilizing a Dipolar Cycloaddition of 4-Phenyl-1,2,4-triazolin-3,5-dione. Tetrahedron Lett. 1980, 21, 1837–1840. [Google Scholar] [CrossRef]
- Gassman, P.G.; Hoye, R.C. Anomalous Cycloaddition Reactions of Distorted Cyclohexa-1,4-dienes. Cycloaddition of N-phenyltriazolinedione to (i,o)-Bicyclo[n.2.2]alkadienes. J. Am. Chem. Soc. 1981, 103, 2496–2498. [Google Scholar] [CrossRef]
- Adam, W.; De Lucchi, O. Reaction of Bicyclo[3.2.1]octadiene with 4-Methyl-1,2,4-triazoline-3,5-dione: Competitive Dipolar and Homo-Cycloaddition. Tetrahedron Lett. 1981, 22, 3501–3504. [Google Scholar] [CrossRef]
- Adam, W.; De Lucchi, O.; Peters, K.; Peters, E.-M.; von Schnering, H.G. Reaction of Bicyclo[3.2.1]octadiene with 1,2,4-triazoline-3,5-diones: Competitive Dipolar and HomoCycloaddition. J. Am. Chem. Soc. 1982, 104, 161–166. [Google Scholar] [CrossRef]
- Adam, W.; Arias, L.A.; De Lucchi, O. Unusual Cycloaddition Behavior of 2-Substituted Norbornadienes with Triazolinediones. Tetrahedron Lett. 1982, 23, 399–402. [Google Scholar] [CrossRef]
- Adam, W.; de Lucchi, O.; Pasquato, L.; Will, B. Cycloaddition Behavior of 2-Substituted Norbornadienes towards 4-Phenyl-4H-1,2,4-triazole-3,5-dione (PTAD): Homo Diels-Alder Reactivity versus Insertion, Rearrangement, and [2+2] Cycloaddition. Chem. Ber. 1987, 120, 531–535. [Google Scholar] [CrossRef]
- Kiselev, V.D.; Shakirova, I.I.; Kornilov, D.A.; Kashaeva, H.A.; Potapova, L.N.; Konovalov, A.I. Homo-Diels-Alder Reaction of a Very Inactive Diene, Bicyclo[2,2,1]hepta-2,5-diene, with the Most Active Dienophile, 4-Phenyl-1,2,4-triazolin-3,5-dione. Solvent, Temperature, and High Pressure Influence on the Reaction Rate. J. Phys. Org. Chem. 2013, 26, 47–53. [Google Scholar] [CrossRef]
- Adam, W.; De Lucchi, O.; Erden, I. Bicyclic Azoalkanes via Urazoles Derived from Cycloaddition of N-Phenyl-1,2,4-triazoline-3,5-dione with Strained Bicycloalkenes. J. Am. Chem. Soc. 1980, 102, 4806–4809. [Google Scholar] [CrossRef]
- Adam, W.; Dorr, M.; Kron, J.; Rosenthal, R.J. On the Question of 1,4-Diradical Intermediates in the Di-p-methane Rearrangement of Benzobicyclo[3.2.1]octadienes: Azoalkanes as Mechanistic Probes. J. Am. Chem. Soc. 1987, 109, 7074–7081. [Google Scholar] [CrossRef]
- Zimmerman, H.W.; Boettcher, R.J.; Buehler, N.E.; Keck, G.E.; Steinmetz, M.G. Independent generation of cyclopropyldicarbinyl diradical species of the di-.pi.-methane rearrangement. Excited singlet, triplet, and ground-state hypersurfaces of barrelene photochemistry. Mechanistic and exploratory organic photochemistry. J. Am. Chem. Soc. 1976, 98, 7680–7689. [Google Scholar] [CrossRef]
- Li, Y.; Fang, D.-C. DFT Calculations on Kinetic Data for Some [4+2] Reactions in Solution. Phys. Chem. Chem. Phys. 2014, 16, 15224–15230. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-P.; Seeman, J.I.; Houk, K.N. Rolf Huisgen’s Classic Studies of Cyclic Triene Diels-Alder Reactions Elaborated by Modern Computational Analysis. Angew. Chem. Int. Ed. 2020, 59, 12506–12519. [Google Scholar] [CrossRef] [PubMed]
- Breton, G.W.; Turlington, M. Alternative Synthetic Routes to N-Methyl-1,2,4-Triazoline-3,5-Dione (MeTAD) and Other Triazolinedione Derivatives. Tetrahedron Lett. 2014, 55, 4661–4663. [Google Scholar] [CrossRef]
- Billiet, S.; De Bruycker, K.; Driessen, F.; Goossens, H.; Van Speybroeck, V.; Winne, J.M.; Du Prez, F.E. Triazolinediones Enable Ultrafast and Reversible Click Chemistry for the Design of Dynamic Polymer Systems. Nat. Chem. 2014, 6, 815–821. [Google Scholar] [CrossRef]
- Vialulin, R.A.; Arisco, T.M.; Kutateladze, A.G. Photoinduced Intramolecular Cyclopentanation vs. Photoprotolytic Oxametathesis in Polycyclic Alkenes Outfitted with Conformationally Constrained Aroylmethyl Chromophores. J. Org. Chem. 2013, 78, 2012–2025. [Google Scholar] [CrossRef]
- Ono, N.; Hirao, A.; Takaisa, K.; Aramaki, S.; Okushima, T.; Hashimo, Y.; Akiyama, S. Manufacture of Heat-Stable Organic Pigments and Organic Electric Elements, Organic Pigment Precursors, and Phtalocyanie Analogs. JP2009215547, September 2009. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H.J. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Bourhis, L.J.; Dolomanov, O.V.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. The Anatomy of a Comprehensive Constrained, Restrained Refinement Program for the Modern Computing Environment—Olex2 Dissected. Acta Cryst. 2015, A71, 59–75. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT-Integrated Space-Group and Crystal-Structure Determination. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From Visualization to Analysis, Design and Prediction. J. Appl. Cryst. 2020, 53, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Carey, F.A.; Sundberg, R.J. Advanced Organic Chemistry, Part A: Structure and Mechanisms, 5th ed.; Springer: New York, NY, USA, 2007; pp. 335–344. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Breton, G.W.; Martin, K.L. Exploring the Limits of Reactivity of N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) with Disubstituted Bicycloalkadienes in the Homo-Diels–Alder Reaction. Organics 2023, 4, 41-48. https://doi.org/10.3390/org4010002
Breton GW, Martin KL. Exploring the Limits of Reactivity of N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) with Disubstituted Bicycloalkadienes in the Homo-Diels–Alder Reaction. Organics. 2023; 4(1):41-48. https://doi.org/10.3390/org4010002
Chicago/Turabian StyleBreton, Gary W., and Kenneth L. Martin. 2023. "Exploring the Limits of Reactivity of N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) with Disubstituted Bicycloalkadienes in the Homo-Diels–Alder Reaction" Organics 4, no. 1: 41-48. https://doi.org/10.3390/org4010002
APA StyleBreton, G. W., & Martin, K. L. (2023). Exploring the Limits of Reactivity of N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) with Disubstituted Bicycloalkadienes in the Homo-Diels–Alder Reaction. Organics, 4(1), 41-48. https://doi.org/10.3390/org4010002








