Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus
Abstract
:1. Introduction
2. Exercise in Pregnancy and Gestational Diabetes
3. The Role of Exercise in Improving Glycemic Control and Overall Health in Insulin Resistance States
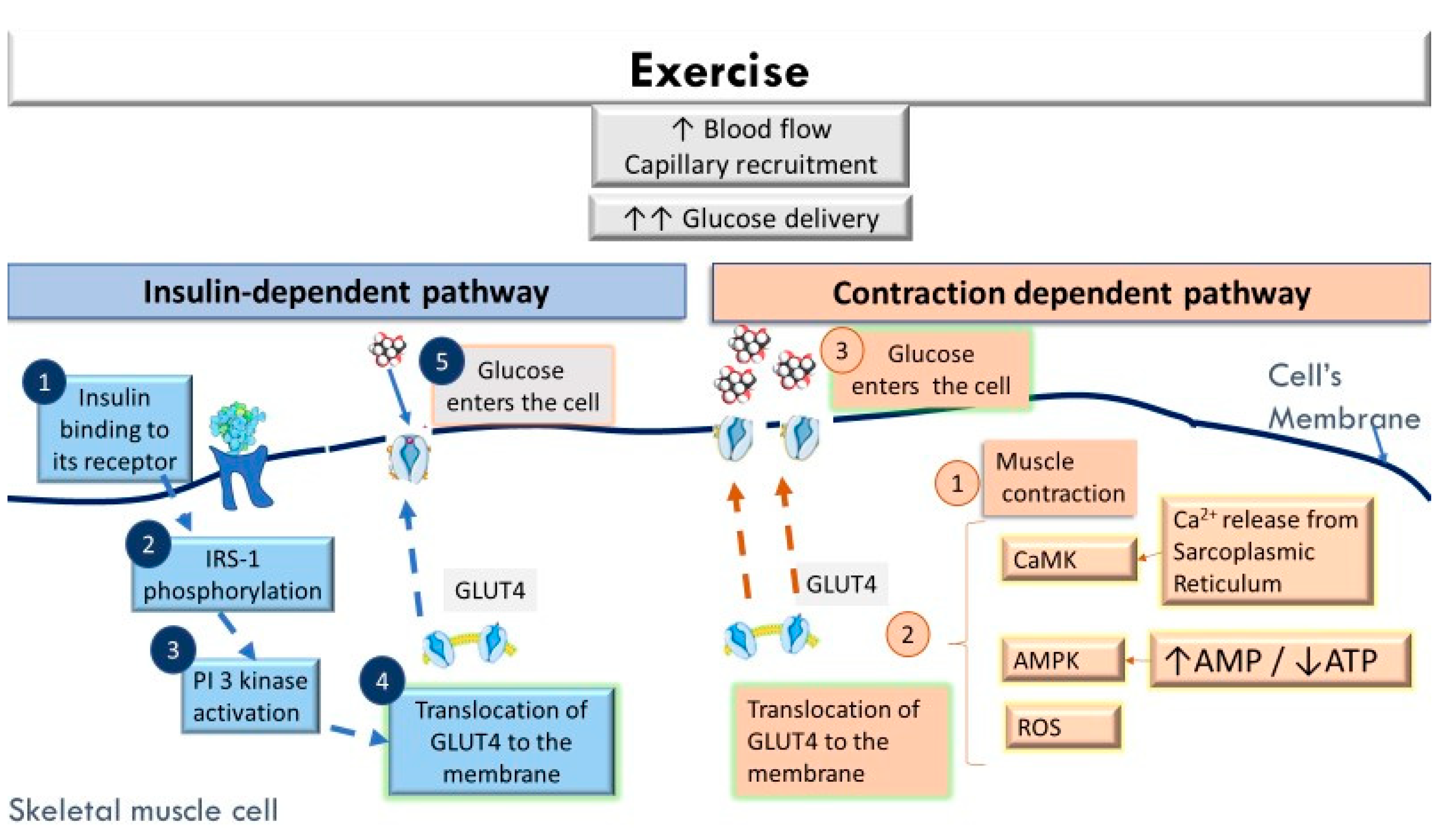
3.1. The Acute Effects of Exercise in Facilitating Greater Glucose Uptake by the Skeletal Muscles
3.2. The Long-Term Effects of Regular Exercise on Glycemic Control and Overall Health in Insulin Resistance States
4. Exercise Prescription in Gestational Diabetes Mellitus
4.1. Acute Cardiovascular and Metabolic Adaptations to Exercise in Pregnant Women with Uncomplicated Pregnancies
4.1.1. Physiological Alterations in Pregnancy
4.1.2. Physiology of Exercise in Uncomplicated Pregnancies
4.2. Metabolic, Neural, and Vascular Alterations that Can Affect the Responses to Exercise in Gestational Diabetes
4.3. Exercise Prescription Characteristics in Gestational Diabetes Mellitus
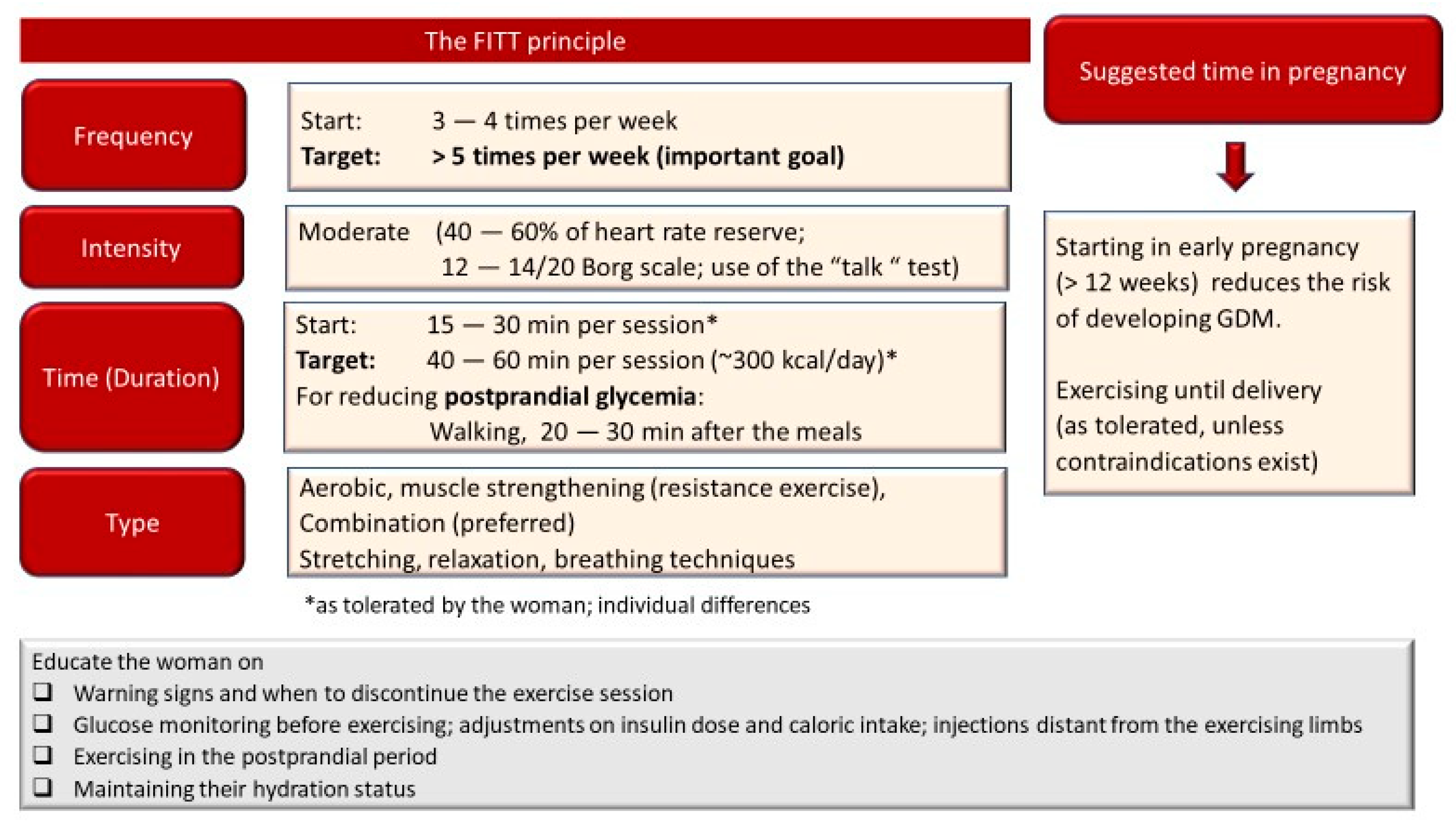
4.3.1. Important Characteristics of the Exercise Program
4.3.2. Avoiding Exercise-Induced Hypoglycemia in Insulin Treated Women
4.3.3. Which Type of Exercise Can Result in the Most Favorable Glycemic and Health Benefits?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Metzger, B.E.; Gabbe, S.G.; Persson, B.; Buchanan, T.A.; Catalano, P.A.; Damm, P.; Dyer, A.R.; de Leiva, A.; Hod, M.; Kitzmiler, J.L.; et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [Green Version]
- American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetes—2021. Diabetes Care 2021, 44, S15–S33. [Google Scholar] [CrossRef]
- Dias, J.; Echeverria, S.; Mayer, V.; Janevic, T. Diabetes risk and control in multi-ethnic US immigrant populations. Curr. Diab. Rep. 2020, 20, 73. [Google Scholar] [CrossRef] [PubMed]
- Yue, D.K.; Molyneaux, L.M.; Ross, G.P.; Constantino, M.I.; Child, A.G.; Turtle, J.R. Why does ethnicity affect prevalence of gestational diabetes? The underwater volcano theory. Diabet. Med. 1996, 13, 748–752. [Google Scholar] [CrossRef]
- International Federation Diabetes IDF Diabetes Atlas, 9th ed. Available online: https://www.diabetesatlas.org/en/resources/ (accessed on 25 March 2021).
- Xu, Y.-H.; Shi, L.; Bao, Y.-P.; Chen, S.-J.; Shi, J.; Zhang, R.-L.; Lu, L. Association between sleep duration during pregnancy and gestational diabetes mellitus: A meta-analysis. Sleep Med. 2018, 52, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Reece, E.A.; Leguizamón, G.; Wiznitzer, A. Gestational diabetes: The need for a common ground. Lancet 2009, 373, 1789–1797. [Google Scholar] [CrossRef]
- Zhang, C.; Ning, Y. Effect of dietary and lifestyle factors on the risk of gestational diabetes: Review of epidemiologic evidence. Am. J. Clin. Nutr. 2011, 94, 1975S–1979S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poulakos, P.; Mintziori, G.; Tsirou, E.; Taousani, E.; Savvaki, D.; Harizopoulou, V.; Goulis, D.G. Comments on gestational diabetes mellitus: From pathophysiology to clinical practice. Hormones 2015, 14, 335–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saravanan, P. Gestational diabetes: Opportunities for improving maternal and child health. Lancet. Diabetes Endocrinol. 2020, 8, 793–800. [Google Scholar] [CrossRef]
- ACOG committee opinion No. 650: Physical activity and exercise during pregnancy and the postpartum period. Obstet. Gynecol. 2015, 126, e135-42. [CrossRef]
- Physical activity and exercise during pregnancy and the postpartum period: ACOG committee opinion, number 804. Obstet. Gynecol. 2020, 135, e178–e188. [CrossRef] [PubMed] [Green Version]
- Lifestyle management: Standards of medical care in diabetes-2019. Diabetes Care 2019, 42, S46–S60. [CrossRef] [Green Version]
- Peters, T.M.; Brazeau, A.-S. Exercise in pregnant women with diabetes. Curr. Diab. Rep. 2019, 19, 80. [Google Scholar] [CrossRef]
- Dipietro, L.; Evenson, K.R.; Bloodgood, B.; Sprow, K.; Troiano, R.P.; Piercy, K.L.; Vaux-Bjerke, A.; Powell, K.E. Benefits of physical activity during pregnancy and postpartum: An umbrella review. Med. Sci. Sports Exerc. 2019, 51, 1292–1302. [Google Scholar] [CrossRef]
- Taylor, N. Critically Appraised Papers: An aerobic and resistance exercise program can improve glycaemic control in women with gestational diabetes mellitus [synopsis]. J. Physiother. 2018, 64, 124. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Pelaez, M.; Cordero, Y.; Perales, M.; Lopez, C.; Coteron, J.; Mottola, M.F. Exercise during pregnancy protects against hypertension and macrosomia: Randomized clinical trial. Am. J. Obstet. Gynecol. 2016, 214, 649. e1–649. e8. [Google Scholar] [CrossRef] [PubMed]
- Laredo-Aguilera, J.A.; Gallardo-Bravo, M.; Rabanales-Sotos, J.A.; Cobo-Cuenca, A.I.; Carmona-Torres, J.M. Physical activity programs during pregnancy are effective for the control of gestational diabetes mellitus. Int. J. Environ. Res. Public Health 2020, 17, 6151. [Google Scholar] [CrossRef]
- Davenport, M.H.; Mottola, M.F.; McManus, R.; Gratton, R. A walking intervention improves capillary glucose control in women with gestational diabetes mellitus: A pilot study. Appl. Physiol. Nutr. Metab. = Physiol. Appl. Nutr. Metab. 2008, 33, 511–517. [Google Scholar] [CrossRef]
- Qiu, S.-H.; Sun, Z.-L.; Cai, X.; Liu, L.; Yang, B. Improving patients’ adherence to physical activity in diabetes mellitus: A review. Diabetes Metab. J. 2012, 36, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Richter, E.A. Is GLUT4 translocation the answer to exercise-stimulated muscle glucose uptake? Am. J. Physiol. Endocrinol. Metab. 2021, 320, E240–E243. [Google Scholar] [CrossRef]
- Kennedy, J.W.; Hirshman, M.F.; Gervino, E.V.; Ocel, J.V.; Forse, R.A.; Hoenig, S.J.; Aronson, D.; Goodyear, L.J.; Horton, E.S. Acute exercise induces GLUT4 translocation in skeletal muscle of normal human subjects and subjects with type 2 diabetes. Diabetes 1999, 48, 1192–1197. [Google Scholar] [CrossRef]
- Kraniou, G.; Cameron-Smith, D.; Hargreaves, M. Effect of short-term training on GLUT-4 mRNA and protein expression in human skeletal muscle. Exp. Physiol. 2004, 89, 559–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraniou, G.; Cameron-Smith, D.; Hargreaves, M. Acute exercise and GLUT4 expression in human skeletal muscle: Influence of exercise intensity. J. Appl. Physiol. 2006, 101, 934–937. [Google Scholar] [CrossRef] [Green Version]
- Lund, S.; Holman, G.D.; Schmitz, O.; Pedersen, O. Contraction stimulates translocation of glucose transporter GLUT4 in skeletal muscle through a mechanism distinct from that of insulin. Proc. Natl. Acad. Sci. USA 1995, 92, 5817–5821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holloszy, J.O. Exercise-induced increase in muscle insulin sensitivity. J. Appl. Physiol. 2005, 99, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Sjøberg, K.A.; Frøsig, C.; Kjøbsted, R.; Sylow, L.; Kleinert, M.; Betik, A.C.; Shaw, C.S.; Kiens, B.; Wojtaszewski, J.F.P.; Rattigan, S.; et al. Exercise increases human skeletal muscle insulin sensitivity via coordinated increases in microvascular perfusion and molecular signaling. Diabetes 2017, 66, 1501–1510. [Google Scholar] [CrossRef] [Green Version]
- Richter, E.A.; Hargreaves, M. Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol. Rev. 2013, 93, 993–1017. [Google Scholar] [CrossRef] [Green Version]
- Flores-Opazo, M.; McGee, S.L.; Hargreaves, M. Exercise and GLUT4. Exerc. Sport Sci. Rev. 2020, 48, 110–118. [Google Scholar] [CrossRef]
- Derave, W.; Lund, S.; Holman, G.D.; Wojtaszewski, J.; Pedersen, O.; Richter, E.A. Contraction-stimulated muscle glucose transport and GLUT-4 surface content are dependent on glycogen content. Am. J. Physiol. 1999, 277, E1103–E1110. [Google Scholar] [CrossRef]
- Katz, A.; Sahlin, K.; Broberg, S. Regulation of glucose utilization in human skeletal muscle during moderate dynamic exercise. Am. J. Physiol. 1991, 260, E411–E415. [Google Scholar] [CrossRef] [PubMed]
- Merz, K.E.; Thurmond, D.C. Role of skeletal muscle in insulin resistance and glucose uptake. Compr. Physiol. 2020, 10, 785–809. [Google Scholar] [CrossRef]
- McConell, G.K.; Sjøberg, K.A.; Ceutz, F.; Gliemann, L.; Nyberg, M.; Hellsten, Y.; Frøsig, C.; Kiens, B.; Wojtaszewski, J.F.P.; Richter, E.A. Insulin-induced membrane permeability to glucose in human muscles at rest and following exercise. J. Physiol. 2020, 598, 303–315. [Google Scholar] [CrossRef]
- Ojuka, E.O.; Goyaram, V.; Smith, J.A.H. The role of CaMKII in regulating GLUT4 expression in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E322–E331. [Google Scholar] [CrossRef]
- Smith, J.A.H.; Kohn, T.A.; Chetty, A.K.; Ojuka, E.O. CaMK activation during exercise is required for histone hyperacetylation and MEF2A binding at the MEF2 site on the Glut4 gene. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E698–E704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Wen, L.; Zhou, S.; Zhang, Y.; Wang, X.-H.; He, Y.-Y.; Davie, A.; Broadbent, S. Effects of four weeks intermittent hypoxia intervention on glucose homeostasis, insulin sensitivity, GLUT4 translocation, insulin receptor phosphorylation, and Akt activity in skeletal muscle of obese mice with type 2 diabetes. PLoS ONE 2018, 13, e0203551. [Google Scholar] [CrossRef] [PubMed]
- Wallberg-Henriksson, H.; Constable, S.H.; Young, D.A.; Holloszy, J.O. Glucose transport into rat skeletal muscle: Interaction between exercise and insulin. J. Appl. Physiol. 1988, 65, 909–913. [Google Scholar] [CrossRef]
- Cartee, G.D.; Young, D.A.; Sleeper, M.D.; Zierath, J.; Wallberg-Henriksson, H.; Holloszy, J.O. Prolonged increase in insulin-stimulated glucose transport in muscle after exercise. Am. J. Physiol. 1989, 256, E494–E499. [Google Scholar] [CrossRef]
- McCoy, M.; Proietto, J.; Hargreves, M. Effect of detraining on GLUT-4 protein in human skeletal muscle. J. Appl. Physiol. 1994, 77, 1532–1536. [Google Scholar] [CrossRef]
- Dela, F.; Mikines, K.J.; von Linstow, M.; Secher, N.H.; Galbo, H. Effect of training on insulin-mediated glucose uptake in human muscle. Am. J. Physiol. 1992, 263, E1134–E1143. [Google Scholar] [CrossRef]
- Barbour, L.A.; McCurdy, C.E.; Hernandez, T.L.; Friedman, J.E. Chronically increased S6K1 is associated with impaired IRS1 signaling in skeletal muscle of GDM women with impaired glucose tolerance postpartum. J. Clin. Endocrinol. Metab. 2011, 96, 1431–1441. [Google Scholar] [CrossRef]
- Damm, P.; Handberg, A.; Kühl, C.; Beck-Nielsen, H.; Mølsted-Pedersen, L. Insulin receptor binding and tyrosine kinase activity in skeletal muscle from normal pregnant women and women with gestational diabetes. Obstet. Gynecol. 1993, 82, 251–259. [Google Scholar] [PubMed]
- Ferrari, F.; Bock, P.M.; Motta, M.T.; Helal, L. Biochemical and molecular mechanisms of glucose uptake stimulated by physical exercise in insulin resistance state: Role of inflammation. Arq. Bras. Cardiol. 2019, 113, 1139–1148. [Google Scholar] [CrossRef]
- Klip, A.; McGraw, T.E.; James, D.E. Thirty sweet years of GLUT4. J. Biol. Chem. 2019, 294, 11369–11381. [Google Scholar] [CrossRef] [Green Version]
- O’Gorman, D.J.; Karlsson, H.K.R.; McQuaid, S.; Yousif, O.; Rahman, Y.; Gasparro, D.; Glund, S.; Chibalin, A.V.; Zierath, J.R.; Nolan, J.J. Exercise training increases insulin-stimulated glucose disposal and GLUT4 (SLC2A4) protein content in patients with type 2 diabetes. Diabetologia 2006, 49, 2983–2992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dastbarhagh, H.; Kargarfard, M.; Abedi, H.; Bambaeichi, E.; Nazarali, P. Effects of food restriction and/or aerobic exercise on the GLUT4 in type 2 diabetic male rats. Int. J. Prev. Med. 2019, 10, 139. [Google Scholar] [CrossRef]
- Søgaard, D.; Lund, M.T.; Scheuer, C.M.; Dehlbaek, M.S.; Dideriksen, S.G.; Abildskov, C.V.; Christensen, K.K.; Dohlmann, T.L.; Larsen, S.; Vigelsø, A.H.; et al. High-intensity interval training improves insulin sensitivity in older individuals. Acta Physiol. 2018, 222, e13009. [Google Scholar] [CrossRef]
- Rattigan, S.; Wallis, M.G.; Youd, J.M.; Clark, M.G. Exercise training improves insulin-mediated capillary recruitment in association with glucose uptake in rat hindlimb. Diabetes 2001, 50, 2659–2665. [Google Scholar] [CrossRef] [Green Version]
- Scalzo, R.L.; Schauer, I.E.; Rafferty, D.; Knaub, L.A.; Kvaratskhelia, N.; Johnson, T.K.; Pott, G.B.; Abushamat, L.A.; Whipple, M.O.; Huebschmann, A.G.; et al. Single-leg exercise training augments in vivo skeletal muscle oxidative flux and vascular content and function in adults with type 2 diabetes. J. Physiol. 2021. [Google Scholar] [CrossRef]
- Axelrod, C.L.; Fealy, C.E.; Mulya, A.; Kirwan, J.P. Exercise training remodels human skeletal muscle mitochondrial fission and fusion machinery towards a pro-elongation phenotype. Acta Physiol. 2019, 225, e13216. [Google Scholar] [CrossRef]
- Mishra, P.; Varuzhanyan, G.; Pham, A.H.; Chan, D.C. Mitochondrial dynamics is a distinguishing feature of skeletal muscle fiber types and regulates organellar compartmentalization. Cell Metab. 2015, 22, 1033–1044. [Google Scholar] [CrossRef] [Green Version]
- Philp, A.M.; Saner, N.J.; Lazarou, M.; Ganley, I.G.; Philp, A. The influence of aerobic exercise on mitochondrial quality control in skeletal muscle. J. Physiol. 2020. [Google Scholar] [CrossRef]
- Steinberg, G.R. Cellular energy sensing and metabolism—Implications for treating diabetes: The 2017 Outstanding scientific achievement award lecture. Diabetes 2018, 67, 169–179. [Google Scholar] [CrossRef] [Green Version]
- Halling, J.F.; Jessen, H.; Nøhr-Meldgaard, J.; Buch, B.T.; Christensen, N.M.; Gudiksen, A.; Ringholm, S.; Neufer, P.D.; Prats, C.; Pilegaard, H. PGC-1α regulates mitochondrial properties beyond biogenesis with aging and exercise training. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E513–E525. [Google Scholar] [CrossRef] [PubMed]
- Laker, R.C.; Drake, J.C.; Wilson, R.J.; Lira, V.A.; Lewellen, B.M.; Ryall, K.A.; Fisher, C.C.; Zhang, M.; Saucerman, J.J.; Goodyear, L.J.; et al. Ampk phosphorylation of Ulk1 is required for targeting of mitochondria to lysosomes in exercise-induced mitophagy. Nat. Commun. 2017, 8, 548. [Google Scholar] [CrossRef] [PubMed]
- Sidarala, V.; Pearson, G.L.; Parekh, V.S.; Thompson, B.; Christen, L.; Gingerich, M.A.; Zhu, J.; Stromer, T.; Ren, J.; Reck, E.C.; et al. Mitophagy protects beta cells from inflammatory damage in diabetes. JCI Insight 2020. [Google Scholar] [CrossRef]
- Heo, J.-W.; No, M.-H.; Park, D.-H.; Kang, J.-H.; Seo, D.Y.; Han, J.; Neufer, P.D.; Kwak, H.-B. Effects of exercise on obesity-induced mitochondrial dysfunction in skeletal muscle. Korean J. Physiol. Pharmacol. Off. J. Korean Physiol. Soc. Korean Soc. Pharmacol. 2017, 21, 567–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Hu, J.; Zhou, H. Macrophage and adipocyte mitochondrial dysfunction in obesity-induced metabolic diseases. World J. Mens. Health 2020. [Google Scholar] [CrossRef]
- Boyle, K.E.; Hwang, H.; Janssen, R.C.; DeVente, J.M.; Barbour, L.A.; Hernandez, T.L.; Mandarino, L.J.; Lappas, M.; Friedman, J.E. Gestational diabetes is characterized by reduced mitochondrial protein expression and altered calcium signaling proteins in skeletal muscle. PLoS ONE 2014, 9, e106872. [Google Scholar] [CrossRef] [Green Version]
- Kruse, R.; Sahebekhtiari, N.; Højlund, K. The mitochondrial proteomic signatures of human skeletal muscle linked to insulin resistance. Int. J. Mol. Sci. 2020, 21, 5374. [Google Scholar] [CrossRef]
- Benite-Ribeiro, S.A.; Lucas-Lima, K.L.; Jones, J.N.; Dos Santos, J.M. Transcription of mtDNA and dyslipidemia are ameliorated by aerobic exercise in type 2 diabetes. Mol. Biol. Rep. 2020, 47, 7297–7303. [Google Scholar] [CrossRef]
- Pino, M.F.; Stephens, N.A.; Eroshkin, A.M.; Yi, F.; Hodges, A.; Cornnell, H.H.; Pratley, R.E.; Smith, S.R.; Wang, M.; Han, X.; et al. Endurance training remodels skeletal muscle phospholipid composition and increases intrinsic mitochondrial respiration in men with Type 2 diabetes. Physiol. Genomics 2019, 51, 586–595. [Google Scholar] [CrossRef]
- Khalafi, M.; Symonds, M.E.; Akbari, A. The impact of exercise training versus caloric restriction on inflammation markers: A systemic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2021, 1–16. [Google Scholar] [CrossRef]
- Kawanishi, N.; Yano, H.; Yokogawa, Y.; Suzuki, K. Exercise training inhibits inflammation in adipose tissue via both suppression of macrophage infiltration and acceleration of phenotypic switching from M1 to M2 macrophages in high-fat-diet-induced obese mice. Exerc. Immunol. Rev. 2010, 16, 105–118. [Google Scholar] [PubMed]
- Kawanishi, N.; Mizokami, T.; Yano, H.; Suzuki, K. Exercise attenuates M1 macrophages and CD8+ T cells in the adipose tissue of obese mice. Med. Sci. Sports Exerc. 2013, 45, 1684–1693. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Polaki, V.; Chen, S.; Bihl, J.C. Exercise improves endothelial function associated with alleviated inflammation and oxidative stress of perivascular adipose tissue in type 2 diabetic mice. Oxid. Med. Cell. Longev. 2020, 2020, 8830537. [Google Scholar] [CrossRef] [PubMed]
- Dobi, A.; Rosanaly, S.; Devin, A.; Baret, P.; Meilhac, O.; Harry, G.J.; d’Hellencourt, C.L.; Rondeau, P. Advanced glycation end-products disrupt brain microvascular endothelial cell barrier: The role of mitochondria and oxidative stress. Microvasc. Res. 2021, 133, 104098. [Google Scholar] [CrossRef]
- Coker, L.H.; Wagenknecht, L.E. Advanced glycation end products, diabetes, and the brain. Neurology 2011, 77, 1326–1327. [Google Scholar] [CrossRef] [Green Version]
- Jash, K.; Gondaliya, P.; Kirave, P.; Kulkarni, B.; Sunkaria, A.; Kalia, K. Cognitive dysfunction: A growing link between diabetes and Alzheimer’s disease. Drug Dev. Res. 2020, 81, 144–164. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, K.; Lindquist, K.; Schwartz, A.V.; Vitartas, C.; Vittinghoff, E.; Satterfield, S.; Simonsick, E.M.; Launer, L.; Rosano, C.; Cauley, J.A.; et al. Advanced glycation end product level, diabetes, and accelerated cognitive aging. Neurology 2011, 77, 1351–1356. [Google Scholar] [CrossRef] [Green Version]
- Ding, Q.; Vaynman, S.; Souda, P.; Whitelegge, J.P.; Gomez-Pinilla, F. Exercise affects energy metabolism and neural plasticity-related proteins in the hippocampus as revealed by proteomic analysis. Eur. J. Neurosci. 2006, 24, 1265–1276. [Google Scholar] [CrossRef]
- Yook, J.S.; Rakwal, R.; Shibato, J.; Takahashi, K.; Koizumi, H.; Shima, T.; Ikemoto, M.J.; Oharomari, L.K.; McEwen, B.S.; Soya, H. Leptin in hippocampus mediates benefits of mild exercise by an antioxidant on neurogenesis and memory. Proc. Natl. Acad. Sci. USA 2019, 116, 10988–10993. [Google Scholar] [CrossRef] [Green Version]
- Radak, Z.; Kumagai, S.; Taylor, A.W.; Naito, H.; Goto, S. Effects of exercise on brain function: Role of free radicals. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2007, 32, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.; Tilmant, K.; Hagar, A.; Klaunig, J.E. Effect of endurance exercise training on liver gene expression in male and female mice. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2020. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, J.A.; Gray, L.J.; Bodicoat, D.H.; Willis, S.A.; Stensel, D.J.; Nimmo, M.A.; Aithal, G.P.; King, J.A. The effect of exercise training on intrahepatic triglyceride and hepatic insulin sensitivity: A systematic review and meta-analysis. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2018, 19, 1446–1459. [Google Scholar] [CrossRef] [PubMed]
- Motiani, K.K.; Collado, M.C.; Eskelinen, J.-J.; Virtanen, K.A.; LÖyttyniemi, E.; Salminen, S.; Nuutila, P.; Kalliokoski, K.K.; Hannukainen, J.C. Exercise training modulates gut microbiota profile and improves endotoxemia. Med. Sci. Sports Exerc. 2020, 52, 94–104. [Google Scholar] [CrossRef] [Green Version]
- Stevanović-Silva, J.; Beleza, J.; Coxito, P.; Pereira, S.; Rocha, H.; Gaspar, T.B.; Gärtner, F.; Correia, R.; Martins, M.J.; Guimarães, T.; et al. Maternal high-fat high-sucrose diet and gestational exercise modulate hepatic fat accumulation and liver mitochondrial respiratory capacity in mothers and male offspring. Metabolism 2021, 116, 154704. [Google Scholar] [CrossRef]
- Falcão-Tebas, F.; Marin, E.C.; Kuang, J.; Bishop, D.J.; McConell, G.K. Maternal exercise attenuates the lower skeletal muscle glucose uptake and insulin secretion caused by paternal obesity in female adult rat offspring. J. Physiol. 2020, 598, 4251–4270. [Google Scholar] [CrossRef]
- May, L. Cardiac physiology of pregnancy. Compr. Physiol. 2015, 5, 1325–1344. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.L.; Cotton, D.B.; Pivarnik, J.M.; Lee, W.; Hankins, G.D.; Benedetti, T.J.; Phelan, J.P. Position change and central hemodynamic profile during normal third-trimester pregnancy and post partum. Am. J. Obstet. Gynecol. 1991, 164, 883–887. [Google Scholar] [CrossRef]
- Barbour, L.A.; McCurdy, C.E.; Hernandez, T.L.; Kirwan, J.P.; Catalano, P.M.; Friedman, J.E. Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care 2007, 30 (Suppl. 2), S112–S119. [Google Scholar] [CrossRef] [Green Version]
- Zavalza-Gómez, A.B.; Anaya-Prado, R.; Rincón-Sánchez, A.R.; Mora-Martínez, J.M. Adipokines and insulin resistance during pregnancy. Diabetes Res. Clin. Pract. 2008, 80, 8–15. [Google Scholar] [CrossRef]
- Mumtaz, M. Gestational diabetes mellitus. Malays. J. Med. Sci. 2000, 7, 4–9. [Google Scholar]
- Butte, N.F. Carbohydrate and lipid metabolism in pregnancy: Normal compared with gestational diabetes mellitus. Am. J. Clin. Nutr. 2000, 71, 1256S–1261S. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, L.A.; Weissgerber, T.L. Clinical physiology of exercise in pregnancy: A literature review. J. Obstet. Gynaecol. Canada JOGC J. D’obstetrique Gynecol. du Canada JOGC 2003, 25, 473–483. [Google Scholar] [CrossRef]
- Nesti, L.; Pugliese, N.R.; Sciuto, P.; Natali, A. Type 2 diabetes and reduced exercise tolerance: A review of the literature through an integrated physiology approach. Cardiovasc. Diabetol. 2020, 19, 134. [Google Scholar] [CrossRef] [PubMed]
- Holwerda, S.W.; Restaino, R.M.; Manrique, C.; Lastra, G.; Fisher, J.P.; Fadel, P.J. Augmented pressor and sympathetic responses to skeletal muscle metaboreflex activation in type 2 diabetes patients. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H300–H309. [Google Scholar] [CrossRef]
- Karavelioglu, Y.; Karapinar, H.; Gul, İ.; Kucukdurmaz, Z.; Yilmaz, A.; Akpek, M.; Kaya, M.G. Blood pressure response to exercise is exaggerated in normotensive diabetic patients. Blood Press. 2013, 22, 21–26. [Google Scholar] [CrossRef]
- Kim, H.-K.; Hotta, N.; Ishizawa, R.; Iwamoto, G.A.; Vongpatanasin, W.; Mitchell, J.H.; Smith, S.A.; Mizuno, M. Exaggerated pressor and sympathetic responses to stimulation of the mesencephalic locomotor region and exercise pressor reflex in type 2 diabetic rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R270–R279. [Google Scholar] [CrossRef]
- Grotle, A.-K.; Macefield, V.G.; Farquhar, W.B.; O’Leary, D.S.; Stone, A.J. Recent advances in exercise pressor reflex function in health and disease. Auton. Neurosci. 2020, 228, 102698. [Google Scholar] [CrossRef]
- Dipla, K.; Kousoula, D.; Zafeiridis, A.; Karatrantou, K.; Nikolaidis, M.G.; Kyparos, A.; Gerodimos, V.; Vrabas, I.S. Exaggerated haemodynamic and neural responses to involuntary contractions induced by whole-body vibration in normotensive obese versus lean women. Exp. Physiol. 2016, 101. [Google Scholar] [CrossRef] [PubMed]
- Meher, M.; Panda, J.K. Impact of glycemic control over cardiac autonomic neuropathy. J. Diabetes Metab. Disord. 2020, 19, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, M.H.; Almby, K.; Pereira, M.J.; Eriksson, J.W. Altered hormonal and autonomic nerve responses to hypo- and hyperglycaemia are found in overweight and insulin-resistant individuals and may contribute to the development of type 2 diabetes. Diabetologia 2021, 64, 641–655. [Google Scholar] [CrossRef] [PubMed]
- McElwain, C.J.; Tuboly, E.; McCarthy, F.P.; McCarthy, C.M. Mechanisms of Endothelial Dysfunction in Pre-eclampsia and Gestational Diabetes Mellitus: Windows Into Future Cardiometabolic Health? Front. Endocrinol. 2020, 11, 655. [Google Scholar] [CrossRef] [PubMed]
- de Resende Guimarães, M.F.B.; Brandão, A.H.F.; de Lima Rezende, C.A.; Cabral, A.C.V.; Brum, A.P.; Leite, H.V.; Capuruço, C.A.B. Assessment of endothelial function in pregnant women with preeclampsia and gestational diabetes mellitus by flow-mediated dilation of brachial artery. Arch. Gynecol. Obstet. 2014, 290, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Dipla, K.; Triantafyllou, A.; Grigoriadou, I.; Kintiraki, E.; Triantafyllou, G.A.; Poulios, P.; Vrabas, I.S.; Zafeiridis, A.; Douma, S.; Goulis, D.G. Impairments in microvascular function and skeletal muscle oxygenation in women with gestational diabetes mellitus: Links to cardiovascular disease risk factors. Diabetologia 2017, 60. [Google Scholar] [CrossRef]
- Kelley, D.E.; Goodpaster, B.; Wing, R.R.; Simoneau, J.A. Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am. J. Physiol. 1999, 277, E1130–E1141. [Google Scholar] [CrossRef]
- Lee, H.; Song, W. Exercise and mitochondrial remodeling in skeletal muscle in type 2 diabetes. J. Obes. Metab. Syndr. 2018, 27, 150–157. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Seifert, T.; Brassard, P.; Rasmussen, P.; Vaag, A.; Nielsen, H.B.; Secher, N.H.; van Lieshout, J.J. Impaired cerebral blood flow and oxygenation during exercise in type 2 diabetic patients. Physiol. Rep. 2015, 3. [Google Scholar] [CrossRef]
- Kintiraki, E.; Dipla, K.; Triantafyllou, A.; Koletsos, N.; Grigoriadou, I.; Poulakos, P.; Sachpekidis, V.; Vrabas, I.S.; Zafeiridis, A.; Bili, E.; et al. Blunted cerebral oxygenation during exercise in women with gestational diabetes mellitus: Associations with macrovascular function and cardiovascular risk factors. Metabolism 2018, 83, 25–30. [Google Scholar] [CrossRef]
- Vounzoulaki, E.; Dipla, K.; Kintiraki, E.; Triantafyllou, A.; Grigoriadou, I.; Koletsos, N.; Zafeiridis, A.; Goulis, D.G.; Douma, S. Pregnancy and post-partum muscle and cerebral oxygenation during intermittent exercise in gestational diabetes: A pilot study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 232, 54–59. [Google Scholar] [CrossRef]
- McMurray, R.G.; Mottola, M.F.; Wolfe, L.A.; Artal, R.; Millar, L.; Pivarnik, J.M. Recent advances in understanding maternal and fetal responses to exercise. Med. Sci. Sports Exerc. 1993, 25, 1305–1321. [Google Scholar] [CrossRef] [PubMed]
- Persinger, R.; Foster, C.; Gibson, M.; Fater, D.C.W.; Porcari, J.P. Consistency of the talk test for exercise prescription. Med. Sci. Sports Exerc. 2004, 36, 1632–1636. [Google Scholar]
- Cremona, A.; O’Gorman, C.; Cotter, A.; Saunders, J.; Donnelly, A. Effect of exercise modality on markers of insulin sensitivity and blood glucose control in pregnancies complicated with gestational diabetes mellitus: A systematic review. Obes. Sci. Pract. 2018, 4, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, S.F.; Ferrara, A.; Hedderson, M.M.; Feng, J.; Neugebauer, R. Exercise during the first trimester of pregnancy and the risks of abnormal screening and gestational diabetes mellitus. Diabetes Care 2021, 44, 425–432. [Google Scholar] [CrossRef]
- Gulve, E.A. Exercise and glycemic control in diabetes: Benefits, challenges, and adjustments to pharmacotherapy. Phys Ther 2008, 88, 1297–1321. [Google Scholar] [CrossRef] [Green Version]
- Torlone, E.; Di Cianni, G.; Mannino, D.; Lapolla, A. Insulin analogs and pregnancy: An update. Acta Diabetol. 2009, 46, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Coe, D.P.; Conger, S.A.; Kendrick, J.M.; Howard, B.C.; Thompson, D.L.; Bassett, D.R.J.; White, J.D. Postprandial walking reduces glucose levels in women with gestational diabetes mellitus. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2018, 43, 531–534. [Google Scholar] [CrossRef] [Green Version]
- South-Paul, J.E.; Rajagopal, K.R.; Tenholder, M.F. The effect of participation in a regular exercise program upon aerobic capacity during pregnancy. Obstet. Gynecol. 1988, 71, 175–179. [Google Scholar]
- Santos, I.A.; Stein, R.; Fuchs, S.C.; Duncan, B.B.; Ribeiro, J.P.; Kroeff, L.R.; Carballo, M.T.; Schmidt, M.I. Aerobic exercise and submaximal functional capacity in overweight pregnant women: A randomized trial. Obstet. Gynecol. 2005, 106, 243–249. [Google Scholar] [CrossRef]
- Berghella, V.; Saccone, G. Exercise in pregnancy! Am. J. Obstet. Gynecol. 2017, 216, 335–337. [Google Scholar] [CrossRef]
- Huang, X.; Huang, J.; Wu, J.; Li, M.; Yang, Z.; Liu, L.; Lin, T.; Lan, Y.; Chen, K. Different exercises for pregnant women with gestational diabetes: A meta-analysis of randomized controlled trials. J. Sports Med. Phys. Fitness 2020, 60, 464–471. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dipla, K.; Zafeiridis, A.; Mintziori, G.; Boutou, A.K.; Goulis, D.G.; Hackney, A.C. Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus. Endocrines 2021, 2, 65-78. https://doi.org/10.3390/endocrines2020007
Dipla K, Zafeiridis A, Mintziori G, Boutou AK, Goulis DG, Hackney AC. Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus. Endocrines. 2021; 2(2):65-78. https://doi.org/10.3390/endocrines2020007
Chicago/Turabian StyleDipla, Konstantina, Andreas Zafeiridis, Gesthimani Mintziori, Afroditi K. Boutou, Dimitrios G. Goulis, and Anthony C. Hackney. 2021. "Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus" Endocrines 2, no. 2: 65-78. https://doi.org/10.3390/endocrines2020007
APA StyleDipla, K., Zafeiridis, A., Mintziori, G., Boutou, A. K., Goulis, D. G., & Hackney, A. C. (2021). Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus. Endocrines, 2(2), 65-78. https://doi.org/10.3390/endocrines2020007








