Association between Physical Activity and Seasonal Variations in Metabolic and Vascular Function in Adults
Abstract
1. Introduction
2. Seasonal Variations in Metabolic and Vascular Function
2.1. Metabolic Function
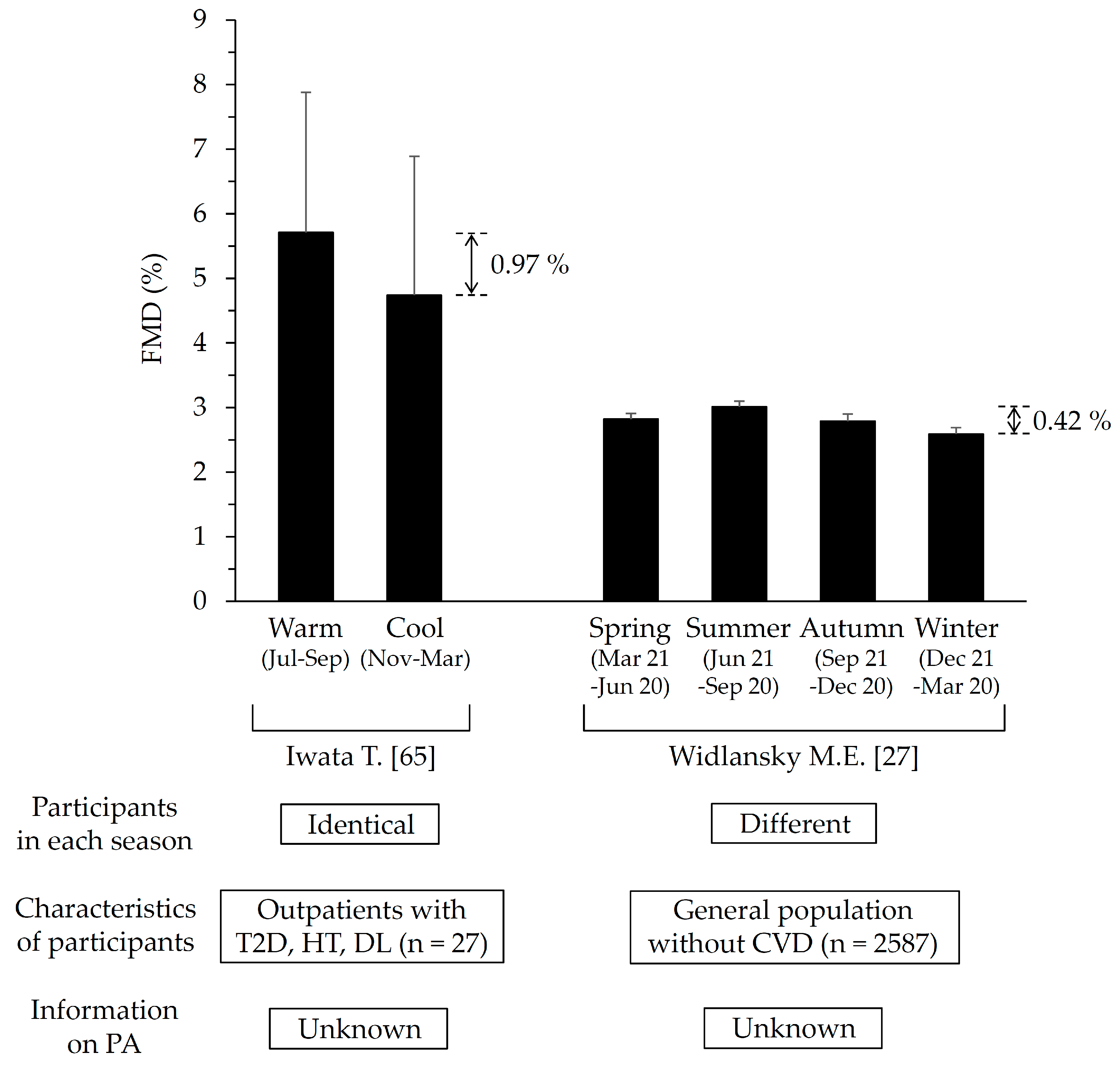
2.2. Vascular Function
3. Seasonal Variations in PA
4. Managing PA for Circumventing Seasonal Variations in Metabolic and Vascular Function
4.1. Effect of PA on Metabolic and Vascular Function
4.2. Influence of PA on Seasonal Variations in Metabolic and Vascular Function
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e563–e595. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- DECODE Study Group, the European Diabetes Epidemiology Group. Glucose tolerance and cardiovascular mortality: Comparison of fasting and 2-hour diagnostic criteria. Arch. Intern. Med. 2001, 161, 397–405. [Google Scholar] [CrossRef]
- Nakagami, T.; Qiao, Q.; Tuomilehto, J.; Balkau, B.; Tajima, N.; Hu, G.; Borch-Johnsen, K. Screen-detected diabetes, hypertension and hypercholesterolemia as predictors of cardiovascular mortality in five populations of Asian origin: The DECODA study. Eur. J. Cardiovasc. Prev. Rehabil. 2006, 13, 555–561. [Google Scholar] [CrossRef]
- Neunteufl, T.; Heher, S.; Katzenschlager, R.; Wolfl, G.; Kostner, K.; Maurer, G.; Weidinger, F. Late prognostic value of flow-mediated dilation in the brachial artery of patients with chest pain. Am. J. Cardiol. 2000, 86, 207–210. [Google Scholar] [CrossRef]
- Lerman, A.; Zeiher, A.M. Endothelial function: Cardiac events. Circulation 2005, 111, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Gokce, N.; Keaney, J.F., Jr.; Hunter, L.M.; Watkins, M.T.; Menzoian, J.O.; Vita, J.A. Risk stratification for postoperative cardiovascular events via noninvasive assessment of endothelial function: A prospective study. Circulation 2002, 105, 1567–1572. [Google Scholar] [CrossRef] [PubMed]
- Yeboah, J.; Folsom, A.R.; Burke, G.L.; Johnson, C.; Polak, J.F.; Post, W.; Lima, J.A.; Crouse, J.R.; Herrington, D.M. Predictive value of brachial flow-mediated dilation for incident cardiovascular events in a population-based study: The multi-ethnic study of atherosclerosis. Circulation 2009, 120, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Gaede, P.; Lund-Andersen, H.; Parving, H.H.; Pedersen, O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N. Engl. J. Med. 2008, 358, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Gaede, P.; Vedel, P.; Larsen, N.; Jensen, G.V.; Parving, H.H.; Pedersen, O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N. Engl. J. Med. 2003, 348, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Higueras-Fresnillo, S.; Cabanas-Sanchez, V.; Lopez-Garcia, E.; Esteban-Cornejo, I.; Banegas, J.R.; Sadarangani, K.P.; Rodriguez-Artalejo, F.; Martinez-Gomez, D. Physical Activity and Association Between Frailty and All-Cause and Cardiovascular Mortality in Older Adults: Population-Based Prospective Cohort Study. J. Am. Geriatr. Soc. 2018, 66, 2097–2103. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Tanaka, S.; Heianza, Y.; Fujihara, K.; Horikawa, C.; Shimano, H.; Saito, K.; Yamada, N.; Ohashi, Y.; Sone, H. Association between physical activity and risk of all-cause mortality and cardiovascular disease in patients with diabetes: A meta-analysis. Diabetes Care 2013, 36, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Mumu, S.J.; Fahey, P.P.; Ali, L.; Rahman, A.; Merom, D. Seasonal Variations in Physical Activity Domains among Rural and Urban Bangladeshis Using a Culturally Relevant Past Year Physical Activity Questionnaire (PYPAQ). J. Environ. Public Health 2019, 2019, 2375474. [Google Scholar] [CrossRef] [PubMed]
- Pivarnik, J.M.; Reeves, M.J.; Rafferty, A.P. Seasonal variation in adult leisure-time physical activity. Med. Sci. Sports Exerc. 2003, 35, 1004–1008. [Google Scholar] [CrossRef]
- Uitenbroek, D.G. Seasonal variation in leisure time physical activity. Med. Sci. Sports Exerc. 1993, 25, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Hermann, J.M.; Rosenbauer, J.; Dost, A.; Steigleder-Schweiger, C.; Kiess, W.; Schofl, C.; Holl, R.W.; Initiative, D.P.V. Seasonal Variation in Blood Pressure in 162,135 Patients with Type 1 or Type 2 Diabetes Mellitus. J. Clin. Hypertens. 2016, 18, 270–278. [Google Scholar] [CrossRef]
- Marti-Soler, H.; Gubelmann, C.; Aeschbacher, S.; Alves, L.; Bobak, M.; Bongard, V.; Clays, E.; de Gaetano, G.; Di Castelnuovo, A.; Elosua, R.; et al. Seasonality of cardiovascular risk factors: An analysis including over 230 000 participants in 15 countries. Heart 2014, 100, 1517–1523. [Google Scholar] [CrossRef]
- Matthews, C.E.; Freedson, P.S.; Hebert, J.R.; Stanek, E.J., 3rd; Merriam, P.A.; Rosal, M.C.; Ebbeling, C.B.; Ockene, I.S. Seasonal variation in household, occupational, and leisure time physical activity: Longitudinal analyses from the seasonal variation of blood cholesterol study. Am. J. Epidemiol. 2001, 153, 172–183. [Google Scholar] [CrossRef]
- Fares, A. Winter cardiovascular diseases phenomenon. N. Am. J. Med. Sci. 2013, 5, 266–279. [Google Scholar] [CrossRef]
- Atsumi, A.; Ueda, K.; Irie, F.; Sairenchi, T.; Iimura, K.; Watanabe, H.; Iso, H.; Ota, H.; Aonuma, K. Relationship between cold temperature and cardiovascular mortality, with assessment of effect modification by individual characteristics: Ibaraki Prefectural Health Study. Circ. J. 2013, 77, 1854–1861. [Google Scholar] [CrossRef]
- Liu, C.; Yavar, Z.; Sun, Q. Cardiovascular response to thermoregulatory challenges. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1793–H1812. [Google Scholar] [CrossRef]
- Miah, A.H.; Sutradhar, S.R.; Ahmed, S.; Bhattacharjee, M.; Alam, M.K.; Bari, M.A.; Tariquzzaman, M.; Mondol, G.D.; Khan, N.A.; Bari, M.S.; et al. Seasonal variation in types of stroke and its common risk factors. Mymensingh Med. J. 2012, 21, 13–20. [Google Scholar]
- Widlansky, M.E.; Vita, J.A.; Keyes, M.J.; Larson, M.G.; Hamburg, N.M.; Levy, D.; Mitchell, G.F.; Osypiuk, E.W.; Vasan, R.S.; Benjamin, E.J. Relation of season and temperature to endothelium-dependent flow-mediated vasodilation in subjects without clinical evidence of cardiovascular disease (from the Framingham Heart Study). Am. J. Cardiol. 2007, 100, 518–523. [Google Scholar] [CrossRef]
- Makinen, T.M.; Mantysaari, M.; Paakkonen, T.; Jokelainen, J.; Palinkas, L.A.; Hassi, J.; Leppaluoto, J.; Tahvanainen, K.; Rintamaki, H. Autonomic nervous function during whole-body cold exposure before and after cold acclimation. Aviat. Space Environ. Med. 2008, 79, 875–882. [Google Scholar] [CrossRef]
- Mourot, L.; Bouhaddi, M.; Regnard, J. Effects of the cold pressor test on cardiac autonomic control in normal subjects. Physiol. Res. 2009, 58, 83–91. [Google Scholar] [CrossRef]
- Alperovitch, A.; Lacombe, J.M.; Hanon, O.; Dartigues, J.F.; Ritchie, K.; Ducimetiere, P.; Tzourio, C. Relationship between blood pressure and outdoor temperature in a large sample of elderly individuals: The Three-City study. Arch. Intern. Med. 2009, 169, 75–80. [Google Scholar] [CrossRef]
- Kimura, T.; Senda, S.; Masugata, H.; Yamagami, A.; Okuyama, H.; Kohno, T.; Hirao, T.; Fukunaga, M.; Okada, H.; Goda, F. Seasonal blood pressure variation and its relationship to environmental temperature in healthy elderly Japanese studied by home measurements. Clin. Exp. Hypertens. 2010, 32, 8–12. [Google Scholar] [CrossRef]
- Cuspidi, C.; Ochoa, J.E.; Parati, G. Seasonal variations in blood pressure: A complex phenomenon. J. Hypertens. 2012, 30, 1315–1320. [Google Scholar] [CrossRef]
- Hanna, J.M. Climate, altitude, and blood pressure. Hum. Biol. 1999, 71, 553–582. [Google Scholar]
- Lim, Y.H.; Park, M.S.; Kim, Y.; Kim, H.; Hong, Y.C. Effects of cold and hot temperature on dehydration: A mechanism of cardiovascular burden. Int. J. Biometeorol. 2015, 59, 1035–1043. [Google Scholar] [CrossRef]
- Kashiwazaki, H.; Dejima, Y.; Suzuki, T. Influence of upper and lower thermoneutral room temperatures (20 degrees C and 25 degrees C) on fasting and post-prandial resting metabolism under different outdoor temperatures. Eur. J. Clin. Nutr. 1990, 44, 405–413. [Google Scholar]
- Kershenbaum, A.; Kershenbaum, A.; Tarabeia, J.; Stein, N.; Lavi, I.; Rennert, G. Unraveling seasonality in population averages: An examination of seasonal variation in glucose levels in diabetes patients using a large population-based data set. Chronobiol. Int. 2011, 28, 352–360. [Google Scholar] [CrossRef]
- Nedergaard, J.; Bengtsson, T.; Cannon, B. Unexpected evidence for active brown adipose tissue in adult humans. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E444–E452. [Google Scholar] [CrossRef]
- Tseng, C.L.; Brimacombe, M.; Xie, M.; Rajan, M.; Wang, H.; Kolassa, J.; Crystal, S.; Chen, T.C.; Pogach, L.; Safford, M. Seasonal patterns in monthly hemoglobin A1c values. Am. J. Epidemiol. 2005, 161, 565–574. [Google Scholar] [CrossRef]
- Carney, T.A.; Guy, S.P.; Helliwell, C.D. Seasonal variation in HbA1c in patients with Type 2 diabetes mellitus. Diabet. Med. 2000, 17, 554–555. [Google Scholar] [CrossRef]
- Sohmiya, M.; Kanazawa, I.; Kato, Y. Seasonal changes in body composition and blood HbA1c levels without weight change in male patients with type 2 diabetes treated with insulin. Diabetes Care 2004, 27, 1238–1239. [Google Scholar] [CrossRef]
- Nordfeldt, S.; Ludvigsson, J. Seasonal variation of HbA1c in intensive treatment of children with type 1 diabetes. J. Pediatr. Endocrinol. Metab. 2000, 13, 529–535. [Google Scholar] [CrossRef]
- Walker, B.R.; Best, R.; Noon, J.P.; Watt, G.C.; Webb, D.J. Seasonal variation in glucocorticoid activity in healthy men. J. Clin. Endocrinol. Metab. 1997, 82, 4015–4019. [Google Scholar] [CrossRef]
- Plasqui, G.; Kester, A.D.; Westerterp, K.R. Seasonal variation in sleeping metabolic rate, thyroid activity, and leptin. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E338–E343. [Google Scholar] [CrossRef][Green Version]
- Gordon, D.J.; Trost, D.C.; Hyde, J.; Whaley, F.S.; Hannan, P.J.; Jacobs, D.R., Jr.; Ekelund, L.G. Seasonal cholesterol cycles: The Lipid Research Clinics Coronary Primary Prevention Trial placebo group. Circulation 1987, 76, 1224–1231. [Google Scholar] [CrossRef]
- Grimes, D.S.; Hindle, E.; Dyer, T. Sunlight, cholesterol and coronary heart disease. QJM 1996, 89, 579–589. [Google Scholar] [CrossRef]
- Kamezaki, F.; Sonoda, S.; Tomotsune, Y.; Yunaka, H.; Otsuji, Y. Seasonal variation in metabolic syndrome prevalence. Hypertens. Res. 2010, 33, 568–572. [Google Scholar] [CrossRef]
- Nadif, R.; Goldberg, S.; Gourmelen, J.; Ozguler, A.; Goldberg, M.; Zins, M.; Henny, J. Seasonal variations of lipid profiles in a French cohort. Atherosclerosis 2019, 286, 181–183. [Google Scholar] [CrossRef]
- Vedel-Krogh, S.; Kobylecki, C.J.; Nordestgaard, B.G.; Langsted, A. The Christmas holidays are immediately followed by a period of hypercholesterolemia. Atherosclerosis 2019, 281, 121–127. [Google Scholar] [CrossRef]
- Skutecki, R.; Cymes, I.; Draganska, E.; Glinska-Lewczuk, K.; Bucinski, A.; Drozdowski, M.; Romaszko, J. Are the Levels of Lipid Parameters Associated with Biometeorological Conditions? Int. J. Environ. Res. Public Health 2019, 16, 4636. [Google Scholar] [CrossRef] [PubMed]
- Yoneshiro, T.; Aita, S.; Matsushita, M.; Kayahara, T.; Kameya, T.; Kawai, Y.; Iwanaga, T.; Saito, M. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Investig. 2013, 123, 3404–3408. [Google Scholar] [CrossRef]
- Shimamoto, H.; Adachi, Y.; Tanaka, K. Seasonal variation of alterations in exercise-induced body composition in obese Japanese women. Eur. J. Appl. Physiol. 2002, 86, 382–387. [Google Scholar] [CrossRef]
- Johnson, F.; Mavrogianni, A.; Ucci, M.; Vidal-Puig, A.; Wardle, J. Could increased time spent in a thermal comfort zone contribute to population increases in obesity? Obes. Rev. 2011, 12, 543–551. [Google Scholar] [CrossRef]
- Valdes, S.; Doulatram-Gamgaram, V.; Lago, A.; Garcia Torres, F.; Badia-Guillen, R.; Olveira, G.; Goday, A.; Calle-Pascual, A.; Castano, L.; Castell, C.; et al. Ambient temperature and prevalence of diabetes and insulin resistance in the Spanish population: Di@bet.es study. Eur. J. Endocrinol. 2019, 180, 273–280. [Google Scholar] [CrossRef]
- Suga, H.; Asakura, K.; Sasaki, S.; Nojima, M.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Effect of seasonality on the estimated mean value of nutrients and ranking ability of a self-administered diet history questionnaire. Nutr. J. 2014, 13, 51. [Google Scholar] [CrossRef][Green Version]
- Janecki, J.M. Cholesterol level in human serum: Seasonal variations and differences in 14 distant regions. Ann. Clin. Lab. Sci. 2013, 43, 407–413. [Google Scholar] [PubMed]
- Maruhashi, T.; Soga, J.; Fujimura, N.; Idei, N.; Mikami, S.; Iwamoto, Y.; Kajikawa, M.; Matsumoto, T.; Hidaka, T.; Kihara, Y.; et al. Relationship between flow-mediated vasodilation and cardiovascular risk factors in a large community-based study. Heart 2013, 99, 1837–1842. [Google Scholar] [CrossRef]
- Cheng, X.; Su, H. Effects of climatic temperature stress on cardiovascular diseases. Eur. J. Intern. Med. 2010, 21, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Ebner, A.; Poitz, D.M.; Alexiou, K.; Deussen, A. Secretion of adiponectin from mouse aorta and its role in cold storage-induced vascular dysfunction. Basic Res. Cardiol. 2013, 108, 390. [Google Scholar] [CrossRef]
- Ebner, A.; Poitz, D.M.; Augstein, A.; Strasser, R.H.; Deussen, A. Functional, morphologic, and molecular characterization of cold storage injury. J. Vasc. Surg. 2012, 56, 189–198 e183. [Google Scholar] [CrossRef][Green Version]
- Hopstock, L.A.; Wilsgaard, T.; Njolstad, I.; Mannsverk, J.; Mathiesen, E.B.; Lochen, M.L.; Bonaa, K.H. Seasonal variation in incidence of acute myocardial infarction in a sub-Arctic population: The Tromso Study 1974–2004. Eur. J. Cardiovasc. Prev. Rehabil. 2011, 18, 320–325. [Google Scholar] [CrossRef]
- Rumana, N.; Kita, Y.; Turin, T.C.; Murakami, Y.; Sugihara, H.; Morita, Y.; Tomioka, N.; Okayama, A.; Nakamura, Y.; Ueshima, H. Seasonal pattern of incidence and case fatality of acute myocardial infarction in a Japanese population (from the Takashima AMI Registry, 1988 to 2003). Am. J. Cardiol. 2008, 102, 1307–1311. [Google Scholar] [CrossRef]
- Ryti, N.R.I.; Makikyro, E.M.S.; Antikainen, H.; Hookana, E.; Junttila, M.J.; Ikaheimo, T.M.; Kortelainen, M.L.; Huikuri, H.V.; Jaakkola, J.J.K. Risk of sudden cardiac death in relation to season-specific cold spells: A case-crossover study in Finland. BMJ Open 2017, 7, e017398. [Google Scholar] [CrossRef]
- Toro, K.; Bartholy, J.; Pongracz, R.; Kis, Z.; Keller, E.; Dunay, G. Evaluation of meteorological factors on sudden cardiovascular death. J. Forensic Leg. Med. 2010, 17, 236–242. [Google Scholar] [CrossRef]
- Wang, H.; Sekine, M.; Chen, X.; Kagamimori, S. A study of weekly and seasonal variation of stroke onset. Int. J. Biometeorol. 2002, 47, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Iwata, M.; Miyashita, Y.; Kumagai, H. Seasonal variation of endothelium-dependent flow-mediated vasodilation measured in the same subjects. Am. J. Cardiovasc. Dis. 2012, 2, 111–115. [Google Scholar]
- Shirai, K.; Hiruta, N.; Song, M.; Kurosu, T.; Suzuki, J.; Tomaru, T.; Miyashita, Y.; Saiki, A.; Takahashi, M.; Suzuki, K.; et al. Cardio-ankle vascular index (CAVI) as a novel indicator of arterial stiffness: Theory, evidence and perspectives. J. Atheroscler. Thromb. 2011, 18, 924–938. [Google Scholar] [CrossRef] [PubMed]
- Inaba, H.; Takeshita, K.; Uchida, Y.; Hayashi, M.; Okumura, T.; Hirashiki, A.; Yoshikawa, D.; Ishii, H.; Yamamoto, K.; Nakayama, T.; et al. Recovery of flow-mediated vasodilatation after repetitive measurements is involved in early vascular impairment: Comparison with indices of vascular tone. PLoS ONE 2014, 9, e83977. [Google Scholar] [CrossRef] [PubMed]
- Igaki, M.; Honda, H.; Komatsu, M.; Sakaue, M. Effect of exercise therapy on flow-mediated dilation in type 2 diabetes mellitus and non-diabetes patients—Relationship with changes in body composition, glycemic control—(In Japanese). JAHS 2017, 8, 23–29. [Google Scholar]
- Kudo, N.; Nishide, R.; Mizutani, M.; Ogawa, S.; Tanimura, S. Association between the type of physical activity and metabolic syndrome in middle-aged and older adult residents of a semi-mountainous area in Japan. Environ. Health Prev. Med. 2021, 26, 46. [Google Scholar] [CrossRef]
- Tyrovolas, S.; Chalkias, C.; Morena, M.; Kalogeropoulos, K.; Tsakountakis, N.; Zeimbekis, A.; Gotsis, E.; Metallinos, G.; Bountziouka, V.; Lionis, C.; et al. High relative environmental humidity is associated with diabetes among elders living in Mediterranean islands. J. Diabetes Metab. Disord. 2014, 13, 25. [Google Scholar] [CrossRef][Green Version]
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1.9 million participants. Lancet Glob. Health 2018, 6, e1077–e1086. [Google Scholar] [CrossRef]
- Juna, C.F.; Cho, Y.H.; Ham, D.; Joung, H. Associations of Relative Humidity and Lifestyles with Metabolic Syndrome among the Ecuadorian Adult Population: Ecuador National Health and Nutrition Survey (ENSANUT-ECU) 2012. Int. J. Environ. Res. Public Health 2020, 17, 9023. [Google Scholar] [CrossRef]
- Togo, F.; Watanabe, E.; Park, H.; Shephard, R.J.; Aoyagi, Y. Meteorology and the physical activity of the elderly: The Nakanojo Study. Int. J. Biometeorol. 2005, 50, 83–89. [Google Scholar] [CrossRef]
- Dannenberg, A.L.; Keller, J.B.; Wilson, P.W.; Castelli, W.P. Leisure time physical activity in the Framingham Offspring Study. Description, seasonal variation, and risk factor correlates. Am. J. Epidemiol. 1989, 129, 76–88. [Google Scholar] [CrossRef]
- Shephard, R.J.; Aoyagi, Y. Seasonal variations in physical activity and implications for human health. Eur. J. Appl. Physiol. 2009, 107, 251–271. [Google Scholar] [CrossRef]
- Chan, C.B.; Ryan, D.A.; Tudor-Locke, C. Relationship between objective measures of physical activity and weather: A longitudinal study. Int. J. Behav. Nutr. Phys. Act. 2006, 3, 21. [Google Scholar] [CrossRef]
- Hoaas, H.; Zanaboni, P.; Hjalmarsen, A.; Morseth, B.; Dinesen, B.; Burge, A.T.; Cox, N.S.; Holland, A.E. Seasonal variations in objectively assessed physical activity among people with COPD in two Nordic countries and Australia: A cross-sectional study. Int. J. Chron. Obstruct. Pulmon. Dis. 2019, 14, 1219–1228. [Google Scholar] [CrossRef]
- Klompstra, L.; Jaarsma, T.; Stromberg, A.; van der Wal, M.H.L. Seasonal variation in physical activity in patients with heart failure. Heart Lung 2019, 48, 381–385. [Google Scholar] [CrossRef]
- Plasqui, G.; Westerterp, K.R. Seasonal variation in total energy expenditure and physical activity in Dutch young adults. Obes. Res. 2004, 12, 688–694. [Google Scholar] [CrossRef]
- Kimura, T.; Kobayashi, H.; Nakayama, E.; Kakihana, W. Seasonality in physical activity and walking of healthy older adults. J. Physiol. Anthropol. 2015, 34, 33. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, V.A.; Smart, N.A. Exercise training for blood pressure: A systematic review and meta-analysis. J. Am. Heart Assoc. 2013, 2, e004473. [Google Scholar] [CrossRef]
- Roberts, C.K.; Hevener, A.L.; Barnard, R.J. Metabolic syndrome and insulin resistance: Underlying causes and modification by exercise training. Compr. Physiol. 2013, 3, 1–58. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Missbach, B.; Dias, S.; Konig, J.; Hoffmann, G. Impact of different training modalities on glycaemic control and blood lipids in patients with type 2 diabetes: A systematic review and network meta-analysis. Diabetologia 2014, 57, 1789–1797. [Google Scholar] [CrossRef]
- American Diabetes Association. 5. Facilitating Behavior Change and Well-being to Improve Health Outcomes: Standards of Medical Care in Diabetes-2021. Diabetes Care 2021, 44, S53–S72. [Google Scholar] [CrossRef]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J. Nitric oxide as a unique signaling molecule in the vascular system: A historical overview. J. Physiol. Pharmacol. 2002, 53, 503–514. [Google Scholar]
- Qiu, S.; Cai, X.; Yin, H.; Sun, Z.; Zugel, M.; Steinacker, J.M.; Schumann, U. Exercise training and endothelial function in patients with type 2 diabetes: A meta-analysis. Cardiovasc. Diabetol. 2018, 17, 64. [Google Scholar] [CrossRef] [PubMed]
- Jakicic, J.M.; Kraus, W.E.; Powell, K.E.; Campbell, W.W.; Janz, K.F.; Troiano, R.P.; Sprow, K.; Torres, A.; Piercy, K.L.; 2018 Physical Activity Guidelines Advisory Committee. Association between Bout Duration of Physical Activity and Health: Systematic Review. Med. Sci. Sports Exerc. 2019, 51, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Stergiou, G.S.; Myrsilidi, A.; Kollias, A.; Destounis, A.; Roussias, L.; Kalogeropoulos, P. Seasonal variation in meteorological parameters and office, ambulatory and home blood pressure: Predicting factors and clinical implications. Hypertens. Res. 2015, 38, 869–875. [Google Scholar] [CrossRef]
- Mavri, A.; Guzic-Salobir, B.; Salobir-Pajnic, B.; Keber, I.; Stare, J.; Stegnar, M. Seasonal variation of some metabolic and haemostatic risk factors in subjects with and without coronary artery disease. Blood Coagul. Fibrinolysis 2001, 12, 359–365. [Google Scholar] [CrossRef]
- Woodhouse, P.R.; Khaw, K.T.; Plummer, M. Seasonal variation of blood pressure and its relationship to ambient temperature in an elderly population. J. Hypertens. 1993, 11, 1267–1274. [Google Scholar] [CrossRef]
- Ryu, O.H.; Lee, S.; Yoo, H.J.; Choi, M.G. Seasonal variations in glycemic control of type 2 diabetes in Korean women. J. Endocrinol. Investig. 2014, 37, 575–581. [Google Scholar] [CrossRef]
- Ma, Y.; Olendzki, B.C.; Li, W.; Hafner, A.R.; Chiriboga, D.; Hebert, J.R.; Campbell, M.; Sarnie, M.; Ockene, I.S. Seasonal variation in food intake, physical activity, and body weight in a predominantly overweight population. Eur. J. Clin. Nutr. 2006, 60, 519–528. [Google Scholar] [CrossRef]
- Ando, J.; Yamamoto, K. Effects of shear stress and stretch on endothelial function. Antioxid. Redox Signal. 2011, 15, 1389–1403. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Jimenez, M.; Morales-Palomo, F.; Ortega, J.F.; Moreno-Cabanas, A.; Guio de Prada, V.; Alvarez-Jimenez, L.; Mora-Rodriguez, R. Effects of Exercise Training during Christmas on Body Weight and Cardiometabolic Health in Overweight Individuals. Int. J. Environ. Res. Public Health 2020, 17, 4732. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, J.L.; Krishnan, S.; Stoner, M.A.; Goktas, Z.; Cooper, J.A. Effects of exercise during the holiday season on changes in body weight, body composition and blood pressure. Eur. J. Clin. Nutr. 2013, 67, 944–949. [Google Scholar] [CrossRef] [PubMed]
- Honda, H.; Igaki, M.; Komatsu, M.; Tanaka, S. Seasonal variations on endothelium-dependent flow-mediated vasodilation in adults with type 2 diabetes and nondiabetic adults with hypertension and/or dyslipidaemia who perform regular exercise. Endocrinol. Diabetes Metab. 2021, 4, e00168. [Google Scholar] [CrossRef]
| Study | Participants (Male/Female) | Study Design | Ex/Promoting PA (Proportion of Participants Fully Adherent) | Observation Period | Positive Effects in Ex/Promoting PA Group |
|---|---|---|---|---|---|
| Ramirez-Jimenez M. (2020) [91] | 38 overweight adults with metabolic syndrome (38/0) | Randomized control trial | HIIT: 43 min (4 × 4-min intervals at 90% HRmax with 3-min active recovery at 70% HRmax) (100%) | 3 weeks (during Christmas holidays) | BW, BP, mean arterial pressure, blood insulin, HOMA-IR, TC, LDL-C |
| Stevenson J.L. (2013) [92] | 148 healthy adults (48/100) | Prospective observational study | MIPA: ≥150 min/week (52%) | ≈2 months (from mid-November to early January) | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honda, H.; Igaki, M.; Komatsu, M.; Tanaka, S.-i. Association between Physical Activity and Seasonal Variations in Metabolic and Vascular Function in Adults. Endocrines 2021, 2, 150-159. https://doi.org/10.3390/endocrines2020015
Honda H, Igaki M, Komatsu M, Tanaka S-i. Association between Physical Activity and Seasonal Variations in Metabolic and Vascular Function in Adults. Endocrines. 2021; 2(2):150-159. https://doi.org/10.3390/endocrines2020015
Chicago/Turabian StyleHonda, Hiroto, Makoto Igaki, Motoaki Komatsu, and Shin-ichiro Tanaka. 2021. "Association between Physical Activity and Seasonal Variations in Metabolic and Vascular Function in Adults" Endocrines 2, no. 2: 150-159. https://doi.org/10.3390/endocrines2020015
APA StyleHonda, H., Igaki, M., Komatsu, M., & Tanaka, S.-i. (2021). Association between Physical Activity and Seasonal Variations in Metabolic and Vascular Function in Adults. Endocrines, 2(2), 150-159. https://doi.org/10.3390/endocrines2020015






