Recent Progress in Stem Cell Research of the Pituitary Gland and Pituitary Adenoma
Abstract
1. Introduction
1.1. Pituitary Gland
1.2. Pituitary Adenoma
1.3. Tissue and Cancer Stem Cells
2. Pituitary Gland Stem Cells
2.1. Historical Background
2.2. Markers
2.3. Stem Cell Niche
2.4. Differentiation
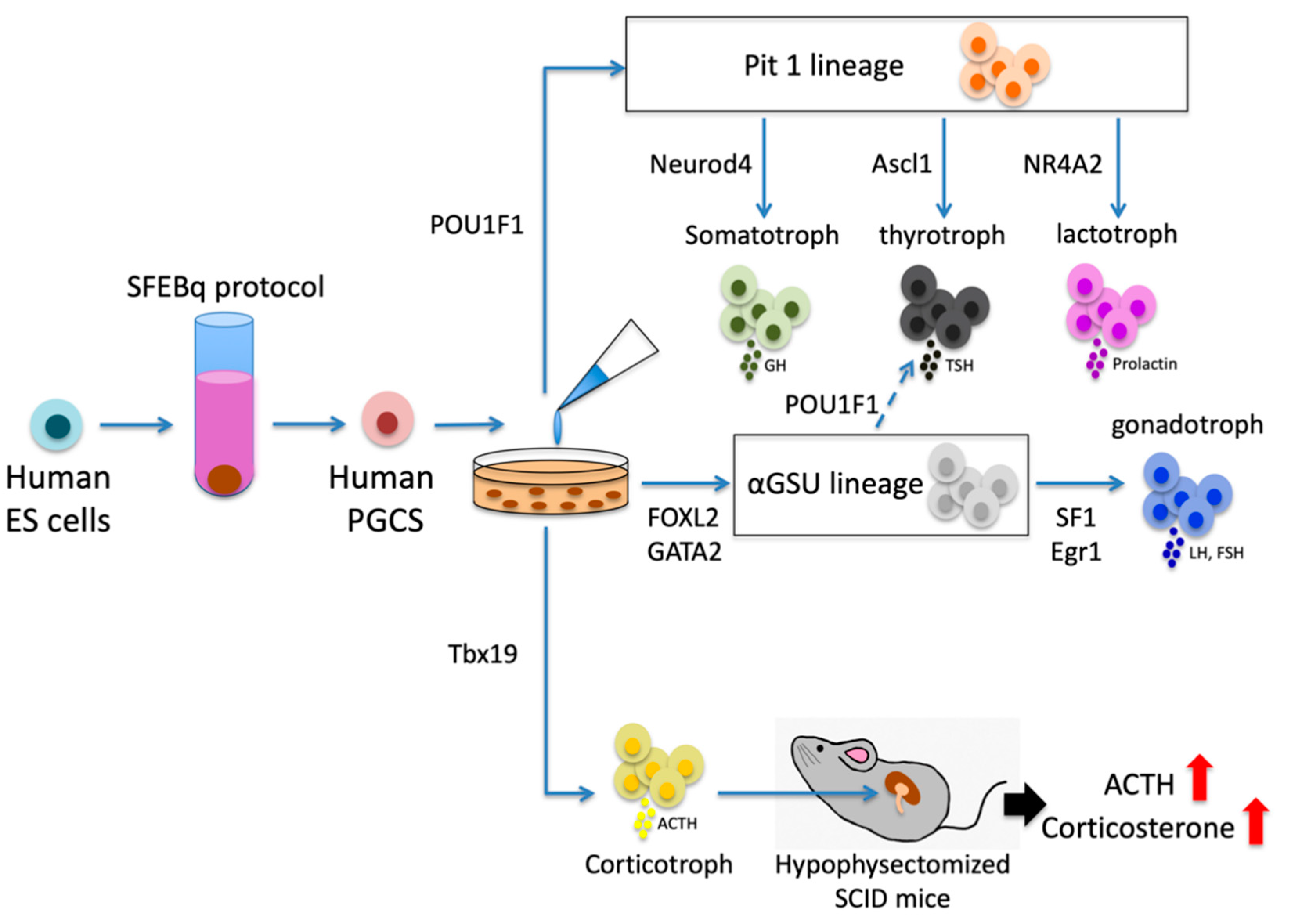
2.5. Treatment Strategies Using PGSCs
3. Pituitary Adenoma Stem Cells
3.1. Tumor-Initiating Capacity
3.2. Treatment Strategies for PASCs
4. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bancalari, R.E.; Gregory, L.C.; McCabe, M.J.; Dattani, M.T. Pituitary gland development: An update. Endocr. Dev. 2012, 23, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.D.; Stone, B.M.; Raetzman, L.T.; Camper, S.A. Cell proliferation and vascularization in mouse models of pituitary hormone deficiency. Mol. Endocrinol. 2006, 20, 1378–1390. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Zhang, J.; Tollkuhn, J.; Ohsawa, R.; Bresnick, E.H.; Guillemot, F.; Kageyama, R.; Rosenfeld, M.G. Sustained Notch signaling in progenitors is required for sequential emergence of distinct cell lineages during organogenesis. Genes Dev. 2006, 20, 2739–2753. [Google Scholar] [CrossRef] [PubMed]
- Potok, M.A.; Cha, K.B.; Hunt, A.; Brinkmeier, M.L.; Leitges, M.; Kispert, A.; Camper, S.A. WNT signaling affects gene expression in the ventral diencephalon and pituitary gland growth. Dev. Dyn. 2008, 237, 1006–1020. [Google Scholar] [CrossRef] [PubMed]
- Molitch, M.E. Diagnosis and Treatment of Pituitary Adenomas: A Review. Jamanetwork 2017, 317, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Melmed, S. Pathogenesis of pituitary tumors. Nat. Rev. Endocrinol. 2011, 7, 257–266. [Google Scholar] [CrossRef]
- Melmed, S. Pituitary tumors. Endocrinol. Metab. Clin. N. Am. 2015, 44, 1–9. [Google Scholar] [CrossRef]
- Mete, O.; Lopes, M.B. Overview of the 2017 WHO Classification of Pituitary Tumors. Endocr. Pathol. 2017, 28, 228–243. [Google Scholar] [CrossRef]
- Loeffler, M.; Roeder, I. Tissue stem cells: Definition, plasticity, heterogeneity, self-organization and models—A conceptual approach. Cells Tissues Organs 2002, 171, 8–26. [Google Scholar] [CrossRef]
- Lapidot, T.; Sirard, C.; Vormoor, J.; Murdoch, B.; Hoang, T.; Caceres-Cortes, J.; Minden, M.; Paterson, B.; Caligiuri, M.A.; Dick, J.E. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994, 367, 645–648. [Google Scholar] [CrossRef]
- Batlle, E.; Clevers, H. Cancer stem cells revisited. Nat. Med. 2017, 23, 1124–1134. [Google Scholar] [CrossRef]
- Levy, A. Physiological Implications of Pituitary Trophic Activity. J. Endocrinol. 2002, 174, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Nishimura, N.; Ueharu, H.; Kanno, N.; Higuchi, M.; Horiguchi, K.; Kato, T.; Kato, Y. Isolation of adult pituitary stem/progenitor cell clusters located in the parenchyma of the rat anterior lobe. Stem Cell Res. 2016, 17, 318–329. [Google Scholar] [CrossRef] [PubMed]
- Tunici, P.; Yu, J.S. Pituitary Adenoma Stem Cells. Methods Mol. Biol. 2009, 568, 195–201. [Google Scholar] [CrossRef]
- Chen, J.; Gremeaux, L.; Fu, Q.; Liekens, D.; Van Laere, S.; Vankelecom, H. Pituitary progenitor cells tracked down by side population dissection. Stem Cells 2009, 27, 1182–1195. [Google Scholar] [CrossRef] [PubMed]
- Rizzoti, K.; Akiyama, H.; Lovell-Badge, R. Mobilized adult pituitary stem cells contribute to endocrine regeneration in response to physiological demand. Cell Stem Cell 2013, 13, 419–432. [Google Scholar] [CrossRef]
- Yoshimura, F.; Ishikawa, H. Identification of the thyrotrophs with the gonadotrophs in the anterior pituitaries of thyroidectomized rats. Endocrinol. Jpn. 1969, 16, 69–85. [Google Scholar] [CrossRef][Green Version]
- Fauquier, T.; Rizzoti, K.; Dattani, M.; Lovell-Badge, R.; Robinson, I.C. SOX2-expressing progenitor cells generate all of the major cell types in the adult mouse pituitary gland. Proc. Natl. Acad. Sci. USA 2008, 105, 2907–2912. [Google Scholar] [CrossRef]
- Andoniadou, C.L.; Matsushima, D.; Mousavy Gharavy, S.N.; Signore, M.; Mackintosh, A.I.; Schaeffer, M.; Gaston-Massuet, C.; Mollard, P.; Jacques, T.S.; Le Tissier, P.; et al. Sox2(+) stem/progenitor cells in the adult mouse pituitary support organ homeostasis and have tumor-inducing potential. Cell Stem Cell 2013, 13, 433–445. [Google Scholar] [CrossRef]
- Yoshida, S.; Kato, T.; Higuchi, M.; Yako, H.; Chen, M.; Kanno, N.; Ueharu, H.; Kato, Y. Rapid transition of NESTIN-expressing dividing cells from PROP1-positive to PIT1-positive advances prenatal pituitary development. J. Neuroendocrinol. 2013, 25, 779–791. [Google Scholar] [CrossRef]
- Mantovani, G.; Giardino, E.; Treppiedi, D.; Catalano, R.; Mangili, F.; Spada, A.; Arosio, M.; Peverelli, E. Stem Cells in Pituitary Tumors: Experimental Evidence Supporting Their Existence and Their Role in Tumor Clinical Behavior. Front. Endocrinol. 2019, 10, 745. [Google Scholar] [CrossRef]
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim. Biophys. Acta 2014, 1840, 2506–2519. [Google Scholar] [CrossRef] [PubMed]
- Olson, L.E.; Tollkuhn, J.; Scafoglio, C.; Krones, A.; Zhang, J.; Ohgi, K.A.; Wu, W.; Taketo, M.M.; Kemler, R.; Grosschedl, R.; et al. Homeodomain-mediated beta-catenin-dependent switching events dictate cell-lineage determination. Cell 2006, 125, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Peel, M.T.; Ho, Y.; Liebhaber, S.A. Transcriptome Analyses of Female Somatotropes and Lactotropes Reveal Novel Regulators of Cell Identity in the Pituitary. Endocrinology 2018, 159, 3965–3980. [Google Scholar] [CrossRef] [PubMed]
- Ellsworth, B.S.; Egashira, N.; Haller, J.L.; Butts, D.L.; Cocquet, J.; Clay, C.M.; Osamura, R.Y.; Camper, S.A. FOXL2 in the pituitary: Molecular, genetic, and developmental analysis. Mol. Endocrinol. 2006, 20, 2796–2805. [Google Scholar] [CrossRef] [PubMed]
- Egashira, N.; Takekoshi, S.; Takei, M.; Teramoto, A.; Osamura, R.Y. Expression of FOXL2 in human normal pituitaries and pituitary adenomas. Mod. Pathol. 2011, 24, 765–773. [Google Scholar] [CrossRef]
- Lamolet, B.; Poulin, G.; Chu, K.; Guillemot, F.; Tsai, M.J.; Drouin, J. Tpit-independent function of NeuroD1(BETA2) in pituitary corticotroph differentiation. Mol. Endocrinol. 2004, 18, 995–1003. [Google Scholar] [CrossRef]
- Ozone, C.; Suga, H.; Eiraku, M.; Kadoshima, T.; Yonemura, S.; Takata, N.; Oiso, Y.; Tsuji, T.; Sasai, Y. Functional anterior pituitary generated in self-organizing culture of human embryonic stem cells. Nat. Commun. 2016, 7, 10351. [Google Scholar] [CrossRef]
- Watanabe, K.; Kamiya, D.; Nishiyama, A.; Katayama, T.; Nozaki, S.; Kawasaki, H.; Watanabe, Y.; Mizuseki, K.; Sasai, Y. Directed differentiation of telencephalic precursors from embryonic stem cells. Nat. Neurosci. 2005, 8, 288–296. [Google Scholar] [CrossRef]
- Eiraku, M.; Watanabe, K.; Matsuo-Takasaki, M.; Kawada, M.; Yonemura, S.; Matsumura, M.; Wataya, T.; Nishiyama, A.; Muguruma, K.; Sasai, Y. Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 2008, 3, 519–532. [Google Scholar] [CrossRef]
- Lafont, C.; Desarménien, M.G.; Cassou, M.; Molino, F.; Lecoq, J.; Hodson, D.; Lacampagne, A.; Mennessier, G.; El Yandouzi, T.; Carmignac, D.; et al. Cellular in vivo imaging reveals coordinated regulation of pituitary microcirculation and GH cell network function. Proc. Natl. Acad. Sci. USA 2010, 107, 4465–4470. [Google Scholar] [CrossRef]
- Fu, Q.; Gremeaux, L.; Luque, R.M.; Liekens, D.; Chen, J.; Buch, T.; Waisman, A.; Kineman, R.; Vankelecom, H. The adult pituitary shows stem/progenitor cell activation in response to injury and is capable of regeneration. Endocrinology 2012, 153, 3224–3235. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, B.; Piao, J.; Ramnarine, K.; Tomishima, M.J.; Tabar, V.; Studer, L. Derivation of Diverse Hormone-Releasing Pituitary Cells from Human Pluripotent Stem Cells. Stem Cell Rep. 2016, 6, 858–872. [Google Scholar] [CrossRef]
- Florio, T. Adult pituitary stem cells: From pituitary plasticity to adenoma development. Neuroendocrinology 2011, 94, 265–277. [Google Scholar] [CrossRef]
- Gleiberman, A.S.; Michurina, T.; Encinas, J.M.; Roig, J.L.; Krasnov, P.; Balordi, F.; Fishell, G.; Rosenfeld, M.G.; Enikolopov, G. Genetic approaches identify adult pituitary stem cells. Proc. Natl. Acad. Sci. USA 2008, 105, 6332–6337. [Google Scholar] [CrossRef]
- Mertens, F.; Gremeaux, L.; Chen, J.; Fu, Q.; Willems, C.; Roose, H.; Govaere, O.; Roskams, T.; Cristina, C.; Becu-Villalobos, D.; et al. Pituitary tumors contain a side population with tumor stem cell-associated characteristics. Endocr. Relat. Cancer 2015, 22, 481–504. [Google Scholar] [CrossRef]
- Xu, Q.; Yuan, X.; Tunici, P.; Liu, G.; Fan, X.; Xu, M.; Hu, J.; Hwang, J.Y.; Farkas, D.L.; Black, K.L.; et al. Isolation of tumour stem-like cells from benign tumours. Br. J. Cancer 2009, 101, 303–311. [Google Scholar] [CrossRef]
- Chen, L.; Ye, H.; Wang, X.; Tang, X.; Mao, Y.; Zhao, Y.; Wu, Z.; Mao, X.O.; Xie, L.; Jin, K.; et al. Evidence of brain tumor stem progenitor-like cells with low proliferative capacity in human benign pituitary adenoma. Cancer Lett. 2014, 349, 61–66. [Google Scholar] [CrossRef]
- Manoranjan, B.; Mahendram, S.; Almenawer, S.A.; Venugopal, C.; McFarlane, N.; Hallett, R.; Vijayakumar, T.; Algird, A.; Murty, N.K.; Sommer, D.D.; et al. The identification of human pituitary adenoma-initiating cells. Acta Neuropathol. Commun. 2016, 4, 125. [Google Scholar] [CrossRef]
- Wurth, R.; Thellung, S.; Corsaro, A.; Barbieri, F.; Florio, T. Experimental Evidence and Clinical Implications of Pituitary Adenoma Stem Cells. Front. Endocrinol. 2020, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Wurth, R.; Barbieri, F.; Pattarozzi, A.; Gaudenzi, G.; Gatto, F.; Fiaschi, P.; Ravetti, J.L.; Zona, G.; Daga, A.; Persani, L.; et al. Phenotypical and Pharmacological Characterization of Stem-Like Cells in Human Pituitary Adenomas. Mol. Neurobiol. 2017, 54, 4879–4895. [Google Scholar] [CrossRef] [PubMed]
- Perez-Moreno, M.A.; Locascio, A.; Rodrigo, I.; Dhondt, G.; Portillo, F.; Nieto, M.A.; Cano, A. A new role for E12/E47 in the repression of E-cadherin expression and epithelial-mesenchymal transitions. J. Biol. Chem. 2001, 276, 27424–27431. [Google Scholar] [CrossRef] [PubMed]
- Tamura, R.; Ohara, K.; Morimoto, Y.; Kosugi, K.; Oishi, Y.; Sato, M.; Yoshida, K.; Toda, M. PITX2 Expression in Non-functional Pituitary Neuroendocrine Tumor with Cavernous Sinus Invasion. Endocr. Pathol. 2019, 30, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Haston, S.; Pozzi, S.; Carreno, G.; Manshaei, S.; Panousopoulos, L.; Gonzalez-Meljem, J.M.; Apps, J.R.; Virasami, A.; Thavaraj, S.; Gutteridge, A.; et al. MAPK pathway control of stem cell proliferation and differentiation in the embryonic pituitary provides insights into the pathogenesis of papillary craniopharyngioma. Development 2017, 144, 2141–2152. [Google Scholar] [CrossRef] [PubMed]
- Carreno, G.; Gonzalez-Meljem, J.M.; Haston, S.; Martinez-Barbera, J.P. Stem cells and their role in pituitary tumorigenesis. Mol. Cell. Endocrinol. 2017, 445, 27–34. [Google Scholar] [CrossRef]
- Lasorella, A.; Rothschild, G.; Yokota, Y.; Russell, R.G.; Iavarone, A. Id2 mediates tumor initiation, proliferation, and angiogenesis in Rb mutant mice. Mol. Cell. Biol. 2005, 25, 3563–3574. [Google Scholar] [CrossRef]
- Elston, M.S.; Gill, A.J.; Conaglen, J.V.; Clarkson, A.; Shaw, J.M.; Law, A.J.; Cook, R.J.; Little, N.S.; Clifton-Bligh, R.J.; Robinson, B.G.; et al. Wnt pathway inhibitors are strongly down-regulated in pituitary tumors. Endocrinology 2008, 149, 1235–1242. [Google Scholar] [CrossRef]
- Donangelo, I.; Ren, S.G.; Eigler, T.; Svendsen, C.; Melmed, S. Sca1(+) murine pituitary adenoma cells show tumor-growth advantage. Endocr. Relat. Cancer 2014, 21, 203–216. [Google Scholar] [CrossRef][Green Version]
- Zubeldia-Brenner, L.; De Winne, C.; Perrone, S.; Rodriguez-Segui, S.A.; Willems, C.; Ornstein, A.M.; Lacau-Mengido, I.; Vankelecom, H.; Cristina, C.; Becu-Villalobos, D. Inhibition of Notch signaling attenuates pituitary adenoma growth in Nude mice. Endocr. Relat. Cancer 2019, 26, 13–29. [Google Scholar] [CrossRef]
- Morris, J.C.; Tan, A.R.; Olencki, T.E.; Shapiro, G.I.; Dezube, B.J.; Reiss, M.; Hsu, F.J.; Berzofsky, J.A.; Lawrence, D.P. Phase I study of GC1008 (fresolimumab): A human anti-transforming growth factor-beta (TGFβ) monoclonal antibody in patients with advanced malignant melanoma or renal cell carcinoma. PLoS ONE 2014, 9, e90353. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, B.; Zhou, L.; Yu, S.; Su, Z.; Song, J.; Sun, Q.; Sha, O.; Wang, X.; Jiang, W.; et al. Prodigiosin inhibits Wnt/beta-catenin signaling and exerts anticancer activity in breast cancer cells. Proc. Natl. Acad. Sci. USA 2016, 113, 13150–13155. [Google Scholar] [CrossRef] [PubMed]
- Thakker, R.V.; Newey, P.J.; Walls, G.V.; Bilezikian, J.; Dralle, H.; Ebeling, P.R.; Melmed, S.; Sakurai, A.; Tonelli, F.; Brandi, M.L. Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1). J. Clin. Endocrinol. Metab. 2012, 97, 2990–3011. [Google Scholar] [CrossRef]
- Verges, B.; Boureille, F.; Goudet, P.; Murat, A.; Beckers, A.; Sassolas, G.; Cougard, P.; Chambe, B.; Montvernay, C.; Calender, A. Pituitary disease in MEN type 1 (MEN1): Data from the France-Belgium MEN1 multicenter study. J. Clin. Endocrinol. Metab. 2002, 87, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Stalberg, P.; Santesson, M.; Ekeblad, S.; Lejonklou, M.H.; Skogseid, B. Recognizing genes differentially regulated in vitro by the multiple endocrine neoplasia type 1 (MEN1) gene, using RNA interference and oligonucleotide microarrays. Surgery 2006, 140, 921–929; discussion 929–931. [Google Scholar] [CrossRef]
- Kaji, H.; Canaff, L.; Lebrun, J.-J.; Goltzman, D.; Hendy, G.N. Inactivation of menin, a Smad3−interacting protein, blocks transforming growth factor type b signaling. Proc. Natl. Acad. Sci. USA 2001, 98, 3837–3842. [Google Scholar] [CrossRef]
- Ellis, P.; Fagan, B.M.; Magness, S.T.; Hutton, S.; Taranova, O.; Hayashi, S.; McMahon, A.; Rao, M.; Pevny, L. SOX2, a persistent marker for multipotential neural stem cells derived from embryonic stem cells, the embryo or the adult. Dev. Neurosci. 2004, 26, 148–165. [Google Scholar] [CrossRef]
- Horiguchi, K.; Fujiwara, K.; Yoshida, S.; Nakakura, T.; Arae, K.; Tsukada, T.; Hasegawa, R.; Takigami, S.; Ohsako, S.; Yashiro, T.; et al. Isolation and characterisation of CD9-positive pituitary adult stem/progenitor cells in rats. Sci. Rep. 2018, 8, 5533. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toda, M.; Tamura, R.; Toda, M. Recent Progress in Stem Cell Research of the Pituitary Gland and Pituitary Adenoma. Endocrines 2020, 1, 49-57. https://doi.org/10.3390/endocrines1010006
Toda M, Tamura R, Toda M. Recent Progress in Stem Cell Research of the Pituitary Gland and Pituitary Adenoma. Endocrines. 2020; 1(1):49-57. https://doi.org/10.3390/endocrines1010006
Chicago/Turabian StyleToda, Masataro, Ryota Tamura, and Masahiro Toda. 2020. "Recent Progress in Stem Cell Research of the Pituitary Gland and Pituitary Adenoma" Endocrines 1, no. 1: 49-57. https://doi.org/10.3390/endocrines1010006
APA StyleToda, M., Tamura, R., & Toda, M. (2020). Recent Progress in Stem Cell Research of the Pituitary Gland and Pituitary Adenoma. Endocrines, 1(1), 49-57. https://doi.org/10.3390/endocrines1010006




