Uncertainty-Aware Virtual Physics-Based Chloride Resistance Analysis of Metakaolin-Blended Concrete
Abstract
1. Introduction
2. Methodology
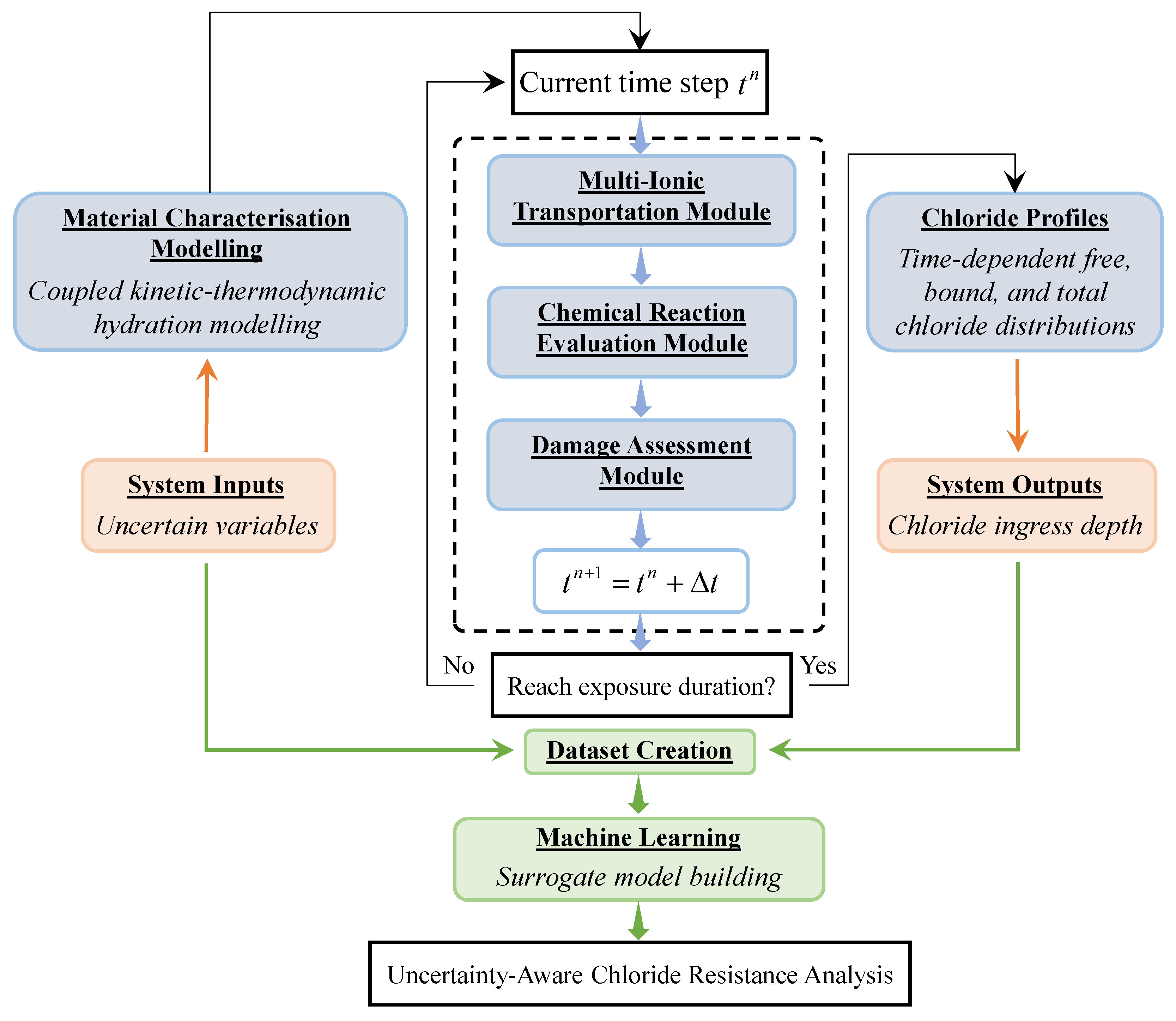
2.1. Approach Overview and Computational Framework
2.2. Material Characterisation Modelling
2.3. Reactive Transport Modelling for Chloride Ingress
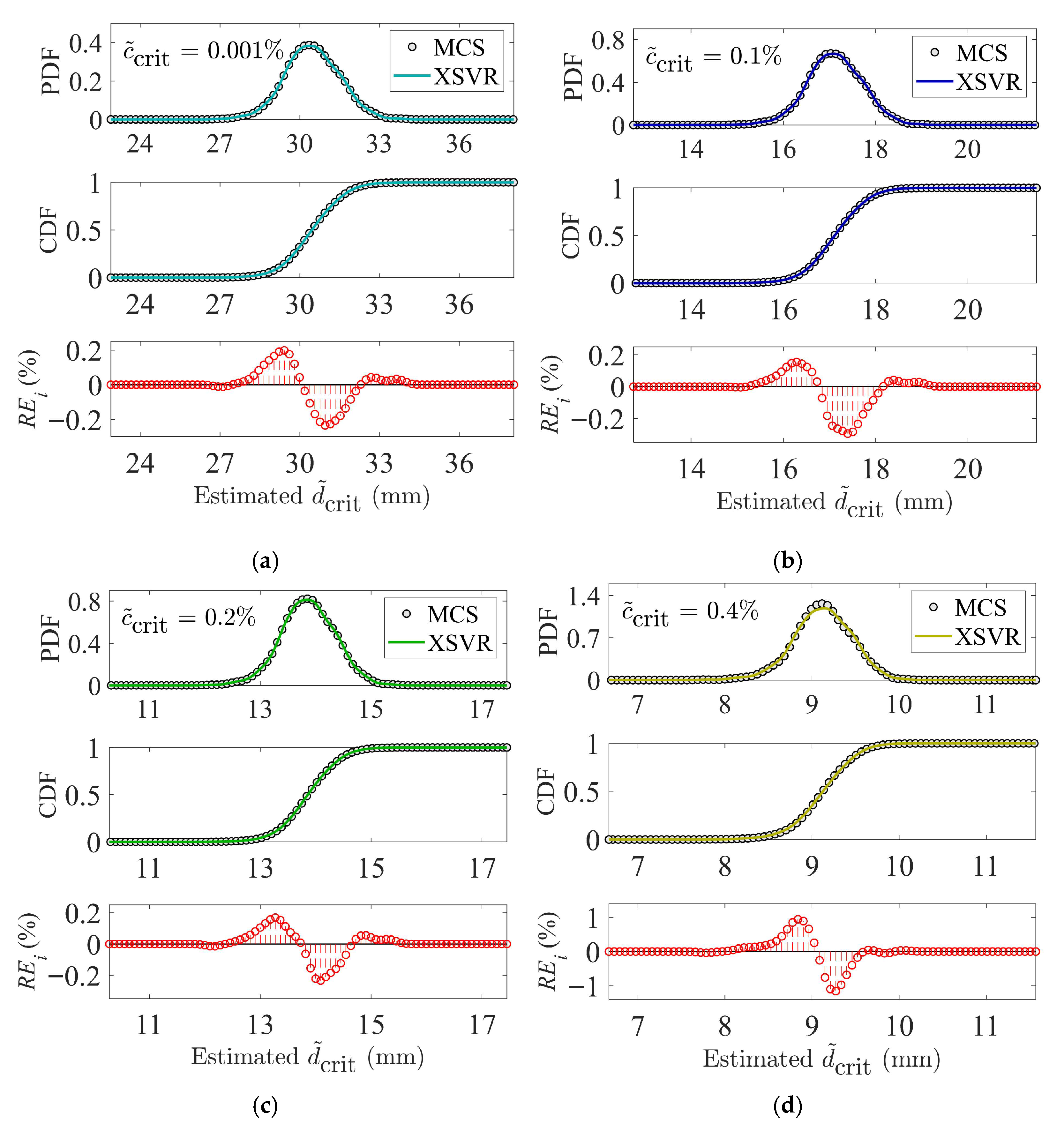
2.4. Extended Support Vector Regression (XSVR)
3. Model Examination and Analysis
3.1. Problem Description
3.2. Influence of Uncertainty
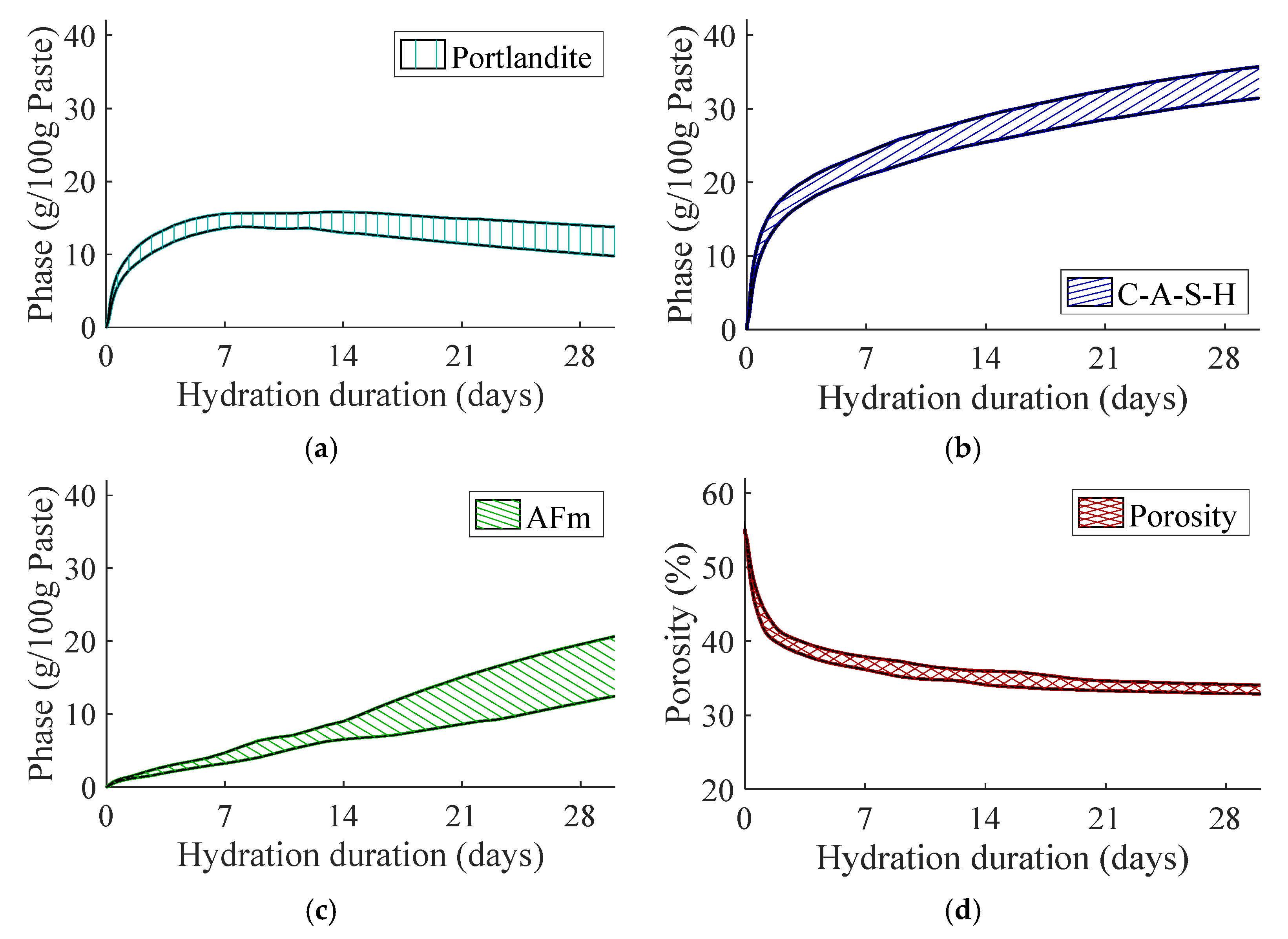
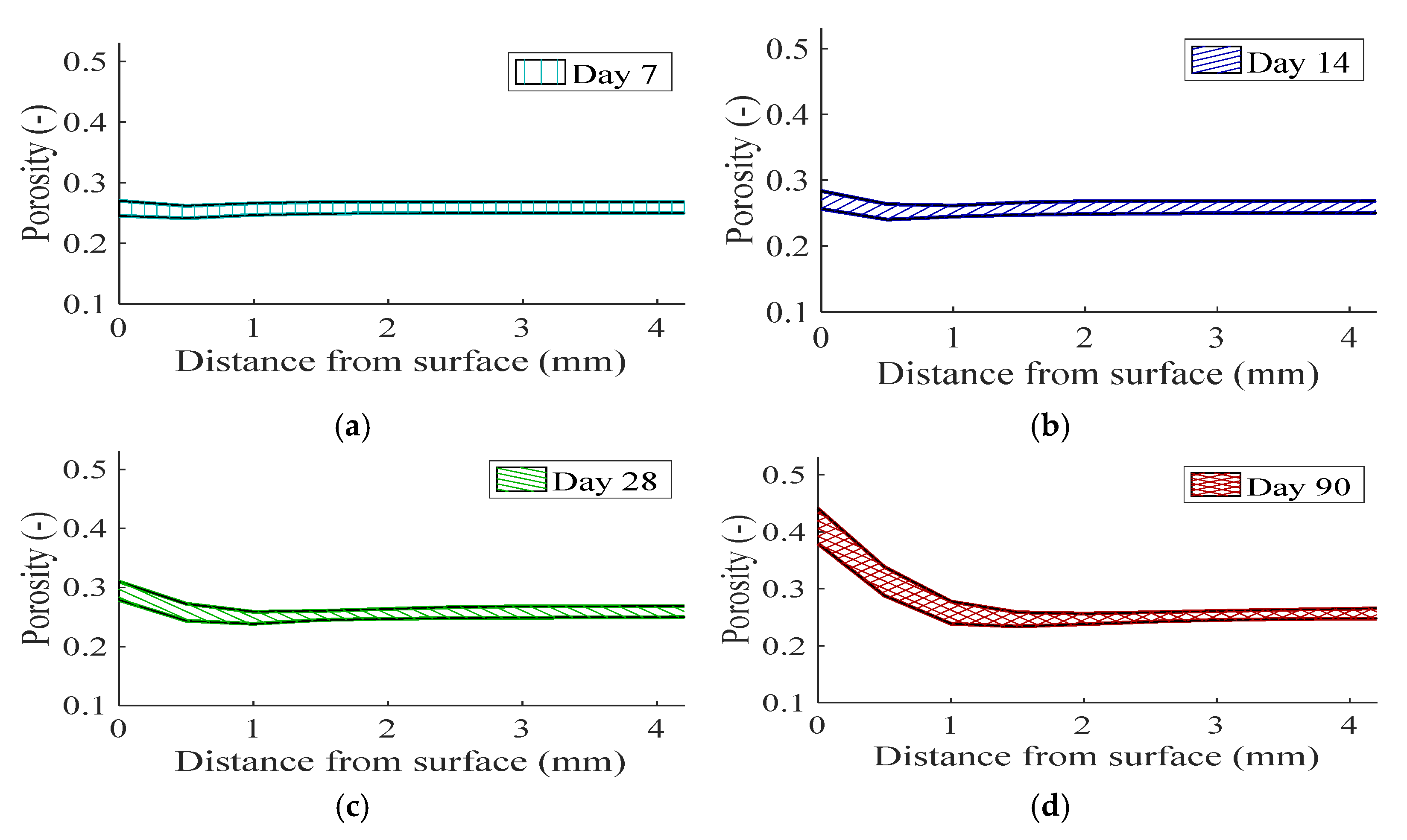
3.2.1. Impact on Hydrated Metakaolin-Blended Concrete
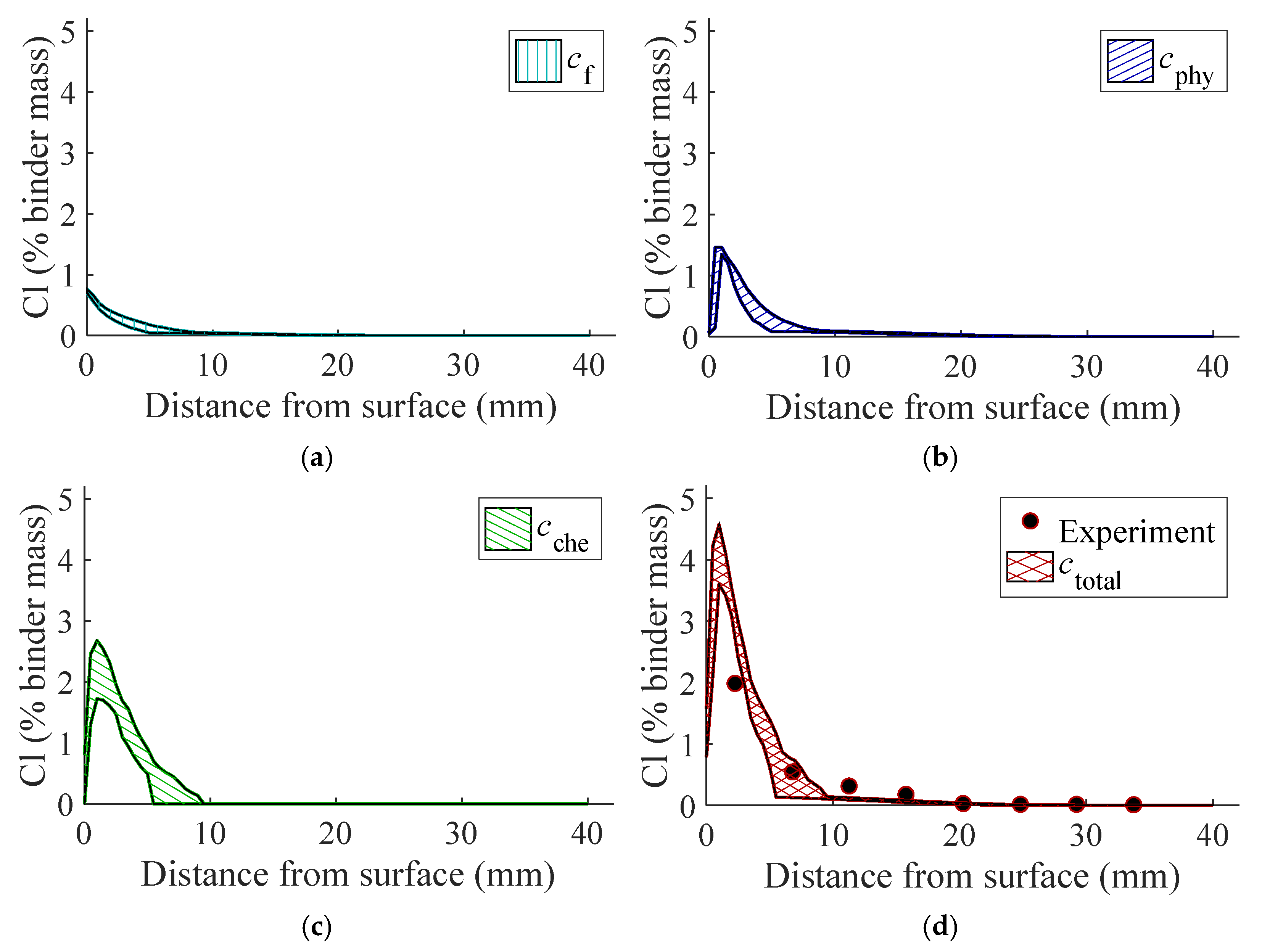
3.2.2. Impact on Chloride Penetration
3.3. Machine Learning-Aided Chloride Resistance Analysis
3.3.1. On Concrete Without Metakaolin Blend
3.3.2. On Concrete with Metakaolin Blend
4. Implications for Durable Design Optimisation
5. Conclusions
- (1)
- The influence of material uncertainty on binder hydration propagates as curing time. The stochastic response in terms of phase assemblage suggests notable impact on the amount of alumina-bearing hydrate, i.e., AFm phase, for the metakaolin-blended mix.
- (2)
- The MCS-guided physics-based stochastic chloride ingress analysis is capable of distinguishing different chloride profiles, i.e., , , , and . The metakaolin blend exhibits considerably higher binding capacity, hence slower chloride ingress process.
- (3)
- The proposed framework, featuring the XSVR, is demonstrated to be effective for correlating stochastic responses of chloride penetration depth with material randomness, significantly improving the efficiency of probabilistic modelling.
- (4)
- In the case of dealing with multiple sources of uncertainty, the XSVR showcases advanced performance compared to conventional SVR, GPR, and Kriging methods.
- (5)
- Regardless of the purity of metakaolin resources, it is concluded that grinding for finer particle size is considered beneficial for achieving greater chloride resistance of the metakaolin-blended concrete.
- (6)
- Future studies should focus on generalisation of the affinity parameter for the proposed approach to be more versatile and accurate in reflecting the varied physical binding capacities of metakaolin-blended concrete, considering uncertainty in resources.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFm | Alumina Ferrite Monosulfate |
| AS2 | Al2O3⋅2SiO2 |
| Al/Si | Aluminium-to-Silicate Ratio |
| Ca/Si | Calcium-to-Silicate Ratio |
| CDF | Cumulative Distribution Function |
| CH | Portlandite |
| C-A-S-H | Calcium-Aluminium-Silicate-Hydrate |
| CoV | Coefficient of Variation |
| GGK | Generalised Gegenbauer kernel |
| GPR | Gaussian Process Regression |
| MK | Metakaolin |
| MCS | Monte Carlo Simulation |
| PDE | Partial Differential Equation |
| Probability Density Function | |
| PNP | Poisson–Nernst–Planck |
| R2 | Coefficient of Determination |
| RE | Relative Error |
| RMSE | Root Mean Square Error |
| SCM | Supplementary Cementitious Material |
| SVR | Support Vector Regression |
| XSVR | Extended Support Vector Regression |
Appendix A
| Portlandite | |||
| −897.01 | −984.68 | 83.39 | |
| Calcite | |||
| −1129.18 | −1207.41 | 92.68 | |
| Gypsum | |||
| −1797.76 | −2023.36 | 193.80 | |
| Monocarbonate | |||
| −7337.46 | −8250 | 657 | |
| Hemicarbonate | |||
| −7335.97 | −8270 | 712.63 | |
| Hydrogarnet | |||
| −5008.2 | −5537.3 | 422 | |
| −4479.90 | −4823 | 840 | |
| −4926 | −5335 | 619 | |
| AFm | |||
| −7778.40 | −8758.60 | 791.60 | |
| −7325.7 | −8262.4 | 831.5 | |
| AFt | |||
| −15,205.94 | −17,535 | 1900 | |
| −14,728.10 | −16,950.20 | 1792.40 | |
| −14,565.64 | −16,792 | 1858 | |
| −14,282.36 | −16,600 | 1937 | |
| Friedel’s salt | |||
| −6810.90 | −7604 | 731 | |
| Kuzel’s salt | |||
| −7533.40 | −8472.01 | 820 | |
| C-A-S-H | |||
| −2560.00 | −2831.4 | 152.8 | |
| −2342.90 | −2551.3 | 154.5 | |
| −2452.46 | −2642.0 | 185.6 | |
| −2474.28 | −2666.7 | 198.4 | |
| −2516.90 | −2780.3 | 159.9 | |
| −2292.82 | −2491.3 | 163.1 | |
| −2381.81 | −2568.7 | 195.0 | |
| −2465.40 | −2720.7 | 167.0 |
References
- Snellings, R.; Suraneni, P.; Skibsted, J. Future and emerging supplementary cementitious materials. Cem. Concr. Res. 2023, 171, 107199. [Google Scholar] [CrossRef]
- Scrivener, K.; Martirena, F.; Bishnoi, S.; Maity, S. Calcined clay limestone cements (LC3). Cem. Concr. Res. 2018, 114, 49–56. [Google Scholar] [CrossRef]
- Yu, Y.; Gunasekara, C.; Elakneswaran, Y.; Robert, D.; Law, D.W.; Setunge, S. Unified hydration model for multi-blend fly ash cementitious systems of wide-range replacement rates. Cem. Concr. Res. 2024, 180, 107487. [Google Scholar] [CrossRef]
- Pillai, R.G.; Gettu, R.; Santhanam, M.; Rengaraju, S.; Dhandapani, Y.; Rathnarajan, S.; Basavaraj, A.S. Service life and life cycle assessment of reinforced concrete systems with limestone calcined clay cement (LC3). Cem. Concr. Res. 2019, 118, 111–119. [Google Scholar] [CrossRef]
- Avet, F.; Scrivener, K. Investigation of the calcined kaolinite content on the hydration of Limestone Calcined Clay Cement (LC3). Cem. Concr. Res. 2018, 107, 124–135. [Google Scholar] [CrossRef]
- Sharma, M.; Bishnoi, S.; Martirena, F.; Scrivener, K. Limestone calcined clay cement and concrete: A state-of-the-art review. Cem. Concr. Res. 2021, 149, 106564. [Google Scholar] [CrossRef]
- Nguyen, Q.D.; Castel, A. Reinforcement corrosion in limestone flash calcined clay cement-based concrete. Cem. Concr. Res. 2020, 132, 106051. [Google Scholar] [CrossRef]
- Maraghechi, H.; Avet, F.; Wong, H.; Kamyab, H.; Scrivener, K. Performance of Limestone Calcined Clay Cement (LC3) with various kaolinite contents with respect to chloride transport. Mater. Struct. 2018, 51, 125. [Google Scholar] [CrossRef]
- Wang, T.; Zheng, Y.; Qian, H.; Shi, Z.; Medepalli, S.; Zhou, J.; He, F.; Ishida, T.; Hou, D.; Zhang, G.; et al. Effects of Al in C–A–S–H gel on the chloride binding capacity of blended cement paste. Cem. Concr. Res. 2025, 190, 107805. [Google Scholar] [CrossRef]
- Song, Q.; Yu, R.; Shui, Z.; Wang, Y.; Rao, S.; Wu, S.; He, Y. Physical and chemical coupling effect of metakaolin induced chloride trapping capacity variation for Ultra High Performance Fibre Reinforced Concrete (UHPFRC). Constr. Build. Mater. 2019, 223, 765–774. [Google Scholar] [CrossRef]
- Abdulalim Alabdullah, A.; Iqbal, M.; Zahid, M.; Khan, K.; Nasir Amin, M.; Jalal, F.E. Prediction of rapid chloride penetration resistance of metakaolin based high strength concrete using light GBM and XGBoost models by incorporating SHAP analysis. Constr. Build. Mater. 2022, 345, 128296. [Google Scholar] [CrossRef]
- Gbozee, M.; Zheng, K.; He, F.; Zeng, X. The influence of aluminum from metakaolin on chemical binding of chloride ions in hydrated cement pastes. Appl. Clay Sci. 2018, 158, 186–194. [Google Scholar] [CrossRef]
- Al-alaily, H.S.; Hassan, A.A.A. Time-dependence of chloride diffusion for concrete containing metakaolin. J. Build. Eng. 2016, 7, 159–169. [Google Scholar] [CrossRef]
- Sun, Y.-M.; Liang, M.-T.; Chang, T.-P. Time/depth dependent diffusion and chemical reaction model of chloride transportation in concrete. Appl. Math. Model. 2012, 36, 1114–1122. [Google Scholar] [CrossRef]
- Yu, X.; Hu, T.; Khodadadi, N.; Liu, Q.; Nanni, A. Modeling chloride ion diffusion in recycled aggregate concrete: A fuzzy neural network approach integrating material and environmental factors. Structures 2025, 73, 108372. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Y.X.; Khennane, A. Numerical modelling of degradation of cement-based materials under leaching and external sulfate attack. Comput. Struct. 2015, 158, 1–14. [Google Scholar] [CrossRef]
- Yu, Y.; Dong, B.; Gao, W.; Sofi, A. Physics-based stochastic aging corrosion analysis assisted by machine learning. Probabilistic Eng. Mech. 2022, 69, 103270. [Google Scholar] [CrossRef]
- Tian, Y.; Feng, Y.; Gao, W. Virtual Modelling Framework-Based Inverse Study for the Mechanical Metamaterials with Material Nonlinearity. Modelling 2025, 6, 24. [Google Scholar] [CrossRef]
- Feng, Y.; Wu, D.; Stewart, M.G.; Gao, W. Past, current and future trends and challenges in non-deterministic fracture mechanics: A review. Comput. Methods Appl. Mech. Eng. 2023, 412, 116102. [Google Scholar] [CrossRef]
- Yu, Y.; Gunasekara, C.; Elakneswaran, Y.; Robert, D.; Law, D.W.; Setunge, S. On the hydration of limestone calcined kaolinitic clay cement and energy-efficient production. Cem. Concr. Compos. 2024, 153, 105698. [Google Scholar] [CrossRef]
- Wagner, T.; Kulik, D.A.; Hingerl, F.F.; Dmytrieva, S.V. GEM-Selektor geochemical modeling package: TSolMod library and data interface for multicomponent phase models. Can. Mineral. 2012, 50, 1173–1195. [Google Scholar] [CrossRef]
- Kulik, D.A.; Wagner, T.; Dmytrieva, S.V.; Kosakowski, G.; Hingerl, F.F.; Chudnenko, K.V.; Berner, U.R. GEM-Selektor geochemical modeling package: Revised algorithm and GEMS3K numerical kernel for coupled simulation codes. Comput. Geosci. 2013, 17, 1–24. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Park, K.-B. Analysis of compressive strength development of concrete containing high volume fly ash. Constr. Build. Mater. 2015, 98, 810–819. [Google Scholar] [CrossRef]
- Wang, X.-Y. Evaluation of the properties of cement–calcined Hwangtoh clay–limestone ternary blends using a kinetic hydration model. Constr. Build. Mater. 2021, 303, 124596. [Google Scholar] [CrossRef]
- Oh, B.H.; Jang, S.Y. Prediction of diffusivity of concrete based on simple analytic equations. Cem. Concr. Res. 2004, 34, 463–480. [Google Scholar] [CrossRef]
- Samson, E.; Marchand, J. Modeling the transport of ions in unsaturated cement-based materials. Comput. Struct. 2007, 85, 1740–1756. [Google Scholar] [CrossRef]
- Badogiannis, E.; Aggeli, E.; Papadakis, V.G.; Tsivilis, S. Evaluation of chloride-penetration resistance of metakaolin concrete by means of a diffusion–Binding model and of the k-value concept. Cem. Concr. Compos. 2015, 63, 1–7. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics; CRC Press: Gaithersburg, MD, USA, 2004. [Google Scholar]
- Lothenbach, B.; Kulik, D.A.; Matschei, T.; Balonis, M.; Baquerizo, L.; Dilnesa, B.; Miron, G.D.; Myers, R.J. Cemdata18: A chemical thermodynamic database for hydrated Portland cements and alkali-activated materials. Cem. Concr. Res. 2019, 115, 472–506. [Google Scholar] [CrossRef]
- Smola, A.J.; Schölkopf, B. A tutorial on support vector regression. Stat. Comput. 2004, 14, 199–222. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, J.; Zou, H. The doubly regularized support vector machine. Stat. Sin. 2006, 16, 589–615. [Google Scholar]
- Schölkopf, B.; Mika, S.; Burges, C.J.; Knirsch, P.; Müller, K.-R.; Rätsch, G.; Smola, A.J. Input space versus feature space in kernel-based methods. IEEE Trans. Neural Netw. 1999, 10, 1000–1017. [Google Scholar] [CrossRef]
- Yu, Y.; Wu, D.; Gao, W. Stochastic chemo-physical-mechanical degradation analysis on hydrated cement under acidic environments. Appl. Math. Model. 2020, 78, 75–97. [Google Scholar] [CrossRef]
- Feng, J.; Liu, L.; Wu, D.; Li, G.; Beer, M.; Gao, W. Dynamic reliability analysis using the extended support vector regression (X-SVR). Mech. Syst. Signal Process. 2019, 126, 368–391. [Google Scholar] [CrossRef]
- Ferreira, R.M.; Castro-Gomes, J.P.; Costa, P.; Malheiro, R. Effect of metakaolin on the chloride ingress properties of concrete. KSCE J. Civ. Eng. 2016, 20, 1375–1384. [Google Scholar] [CrossRef]
- Dong, B.; Yu, Y.; Gao, W.; Gunasekara, C.; Zhao, G.; Castel, A.; Setunge, S. Electro-chemo-physical analysis for long-term reinforcement corrosion within the reactive system of concrete. Cem. Concr. Compos. 2025, 155, 105846. [Google Scholar] [CrossRef]
- Dong, B.; Yu, Y.; Feng, Y.; Wu, D.; Zhao, G.; Liu, A.; Gao, W. Robust numerical solution for assessing corrosion of reinforced concrete structures under external power supply. Eng. Struct. 2023, 294, 116724. [Google Scholar] [CrossRef]
- Kijjanon, A.; Sumranwanich, T.; Tangtermsirikul, S. Influences of metakaolin and calcined clay blended cement on chloride resistance and electrical resistivity of concrete. Adv. Cem. Res. 2025, 37, 24–37. [Google Scholar] [CrossRef]
- Raheem, A.A.; Abdulwahab, R.; Kareem, M.A. Incorporation of metakaolin and nanosilica in blended cement mortar and concrete—A review. J. Clean. Prod. 2021, 290, 125852. [Google Scholar] [CrossRef]








| Ionic Species | Radius (×10−10 m) | Intrinsic D (×10−9 m2/s) | Ionic Species | Radius (×10−10 m) | Intrinsic D (×10−9 m2/s) |
|---|---|---|---|---|---|
| 1.89 | 1.10 | 2.20 | 0.90 | ||
| 1.00 | 0.79 | 1.78 | 0.92 | ||
| 1.90 | 1.00 | 1.81 | 2.03 | ||
| 1.38 | 1.96 | 2.40 | 1.07 | ||
| 0.72 | 0.70 | 1.40 | 5.27 | ||
| 1.02 | 1.33 | 0.9 | 9.31 |
| Mix Identification | Mix Proportions (kg/m3) | |||
|---|---|---|---|---|
| Cement | Metakaolin | Aggregate | Water | |
| PC set | 440 | - | 1840 | 200 |
| MK set | 352 | 88 | ||
| Random Variables | Distribution | Mean | Coefficient of Variation (CoV) or Range |
|---|---|---|---|
| Lognormal | 4100 | 0.05 | |
| Lognormal | 15,000 | 0.05 | |
| (-) | Uniform | 0.9 | [0.85, 0.95] |
| Gumbel | 0.1 | ||
| Gumbel | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yu, Y.; Gardiner, D.; Sun, J.; Pasupathy, K. Uncertainty-Aware Virtual Physics-Based Chloride Resistance Analysis of Metakaolin-Blended Concrete. Modelling 2026, 7, 16. https://doi.org/10.3390/modelling7010016
Yu Y, Gardiner D, Sun J, Pasupathy K. Uncertainty-Aware Virtual Physics-Based Chloride Resistance Analysis of Metakaolin-Blended Concrete. Modelling. 2026; 7(1):16. https://doi.org/10.3390/modelling7010016
Chicago/Turabian StyleYu, Yuguo, David Gardiner, Jie Sun, and Kiru Pasupathy. 2026. "Uncertainty-Aware Virtual Physics-Based Chloride Resistance Analysis of Metakaolin-Blended Concrete" Modelling 7, no. 1: 16. https://doi.org/10.3390/modelling7010016
APA StyleYu, Y., Gardiner, D., Sun, J., & Pasupathy, K. (2026). Uncertainty-Aware Virtual Physics-Based Chloride Resistance Analysis of Metakaolin-Blended Concrete. Modelling, 7(1), 16. https://doi.org/10.3390/modelling7010016






