A Standardized Protocol for Post-Partum Salpingectomy with Suture Ligation: A Prospective Feasibility Study
Abstract
:1. Introduction
2. Materials and Methods
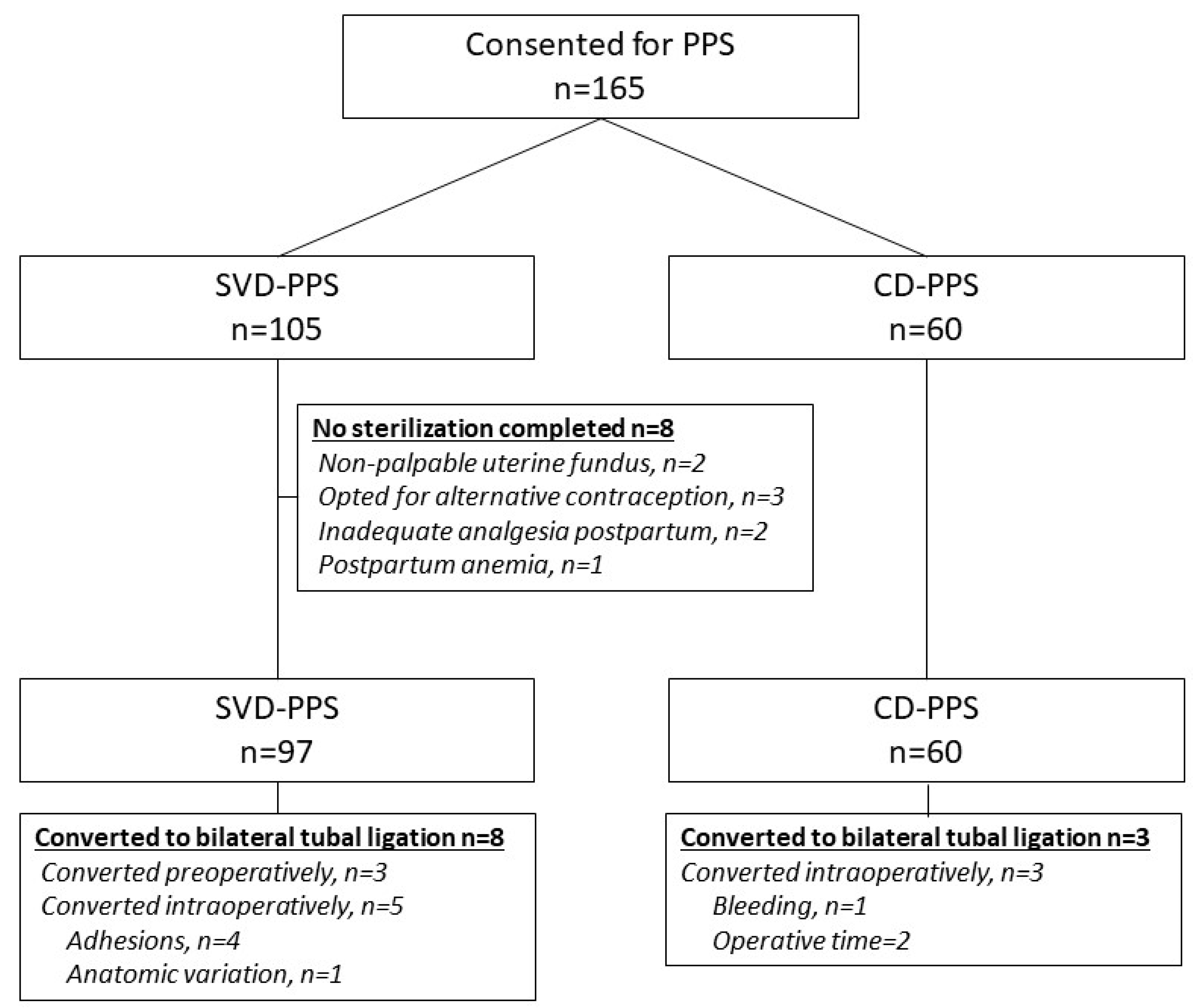
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [Green Version]
- Lowe, K.A.; Chia, V.M.; Taylor, A.; O’Malley, C.; Kelsh, M.; Mohamed, M.; Mowat, F.S.; Goff, B. An international assessment of ovarian cancer incidence and mortality. Gynecol. Oncol. 2013, 130, 107–114. [Google Scholar] [CrossRef]
- Carlson, J.W.; Jarboe, E.A.; Kindelberger, D.; Nucci, M.R.; Hirsch, M.S.; Crum, C.P. Serous Tubal Intraepithelial Carcinoma: Diagnostic Reproducibility and its Implications. Int. J. Gynecol. Pathol. 2010, 29, 310–314. [Google Scholar] [CrossRef]
- Erickson, B.K.; Conner, M.G.; Landen, C.N. The role of the fallopian tube in the origin of ovarian cancer. Am. J. Obstet. Gynecol. 2013, 209, 409–414. [Google Scholar] [CrossRef] [Green Version]
- Corzo, C.; Iniesta, M.D.; Patrono, M.G.; Lu, K.H.; Ramirez, P.T. Role of Fallopian Tubes in the Development of Ovarian Cancer. J. Minim. Invasive Gynecol. 2017, 24, 230–234. [Google Scholar] [CrossRef]
- Gynecologists, A.C. of O. and Hereditary Breast and Ovarian Cancer Syndrome. Obstet. Gynecol. 2020, 133, 168–186. [Google Scholar]
- Statement, S.G.O.C. Society of Gynecologic Oncologists Clinical Practice Committee Statement on Prophylactic Salpingo-oophorectomy. Gynecol. Oncol. 2005, 98, 179–181. [Google Scholar] [CrossRef]
- Boerner, T.; Long Roche, K. Salpingectomy for the Risk Reduction of Ovarian Cancer: Is It Time for a Salpingectomy-alone Approach? J. Minim. Invasive Gynecol. 2021, 28, 403–408. [Google Scholar] [CrossRef]
- Walker, J.L.; Powell, C.B.; Chen, L.M.; Carter, J.; Bae Jump, V.L.; Parker, L.P.; Borowsky, M.E.; Gibb, R.K. Society of Gynecologic Oncology recommendations for the prevention of ovarian cancer. Cancer 2015, 121, 2108–2120. [Google Scholar] [CrossRef]
- Committee on Gynecologic Practice Opportunistic Salpingectomy as an Ovarian Cancer Primary Prevention Strategy. Obstet. Gynecol. 2017, 67, 243–246. [CrossRef]
- Jacobson, M.; Bernardini, M.; Sobel, M.L.; Kim, R.H.; McCuaig, J.; Allen, L. No. 366-Gynaecologic Management of Hereditary Breast and Ovarian Cancer. J. Obstet. Gynaecol. Can. 2018, 40, 1497–1510. [Google Scholar] [CrossRef]
- Powell, C.B.; Alabaster, A.; Simmons, S.; Garcia, C.; Martin, M.; McBride-Allen, S.; Littell, R.D. Salpingectomy for sterilization: Change in practice in a large integrated health care system, 2011–2016. Obstet. Gynecol. 2017, 130, 961–967. [Google Scholar] [CrossRef]
- Morelli, M.; Venturella, R.; Mocciaro, R.; Di Cello, A.; Rania, E.; Lico, D.; D’Alessandro, P.; Zullo, F. Prophylactic salpingectomy in premenopausal low-risk women for ovarian cancer: Primum non nocere. Gynecol. Oncol. 2013, 129, 448–451. [Google Scholar] [CrossRef] [Green Version]
- Kwon, J.S.; Tinker, A.; Pansegrau, G.; McAlpine, J.; Housty, M.; McCullum, M.; Gilks, C.B. Prophylactic salpingectomy and delayed oophorectomy as an alternative for BRCA mutation carriers. Obstet. Gynecol. 2013, 121, 14–24. [Google Scholar] [CrossRef]
- Hanley, G.E.; Kwon, J.S.; McAlpine, J.N.; Huntsman, D.G.; Finlayson, S.J.; Miller, D. Examining indicators of early menopause following opportunistic salpingectomy: A cohort study from British Columbia, Canada. Am. J. Obstet. Gynecol. 2020, 223, 221.e1–221.e11. [Google Scholar] [CrossRef] [Green Version]
- Dilley, S.E.; Straughn, J.M.; Leath, C.A. The evolution of and evidence for opportunistic salpingectomy. Obstet. Gynecol. 2017, 130, 814–824. [Google Scholar] [CrossRef]
- Kotsopoulos, J.; Narod, S.A. Prophylactic salpingectomy for the prevention of ovarian cancer: Who should we target? Int. J. Cancer 2020, 147, 1245–1251. [Google Scholar] [CrossRef]
- Dilley, S.E.; Havrilesky, L.J.; Bakkum-Gamez, J.; Cohn, D.E.; Michael Straughn, J.; Caughey, A.B.; Rodriguez, M.I. Cost-effectiveness of opportunistic salpingectomy for ovarian cancer prevention. Gynecol. Oncol. 2017, 146, 373–379. [Google Scholar] [CrossRef]
- Kwon, J.S.; Mcalpine, J.N.; Hanley, G.E.; Finlayson, S.J.; Cohen, T.; Miller, D.M.; Gilks, C.B.; Huntsman, D.G. Costs and benefits of opportunistic salpingectomy as an ovarian cancer prevention strategy. Obstet. Gynecol. 2015, 125, 338–345. [Google Scholar] [CrossRef]
- Subramaniam, A.; Blanchard, C.T.; Erickson, B.K.; Szychowski, J.; Leath, C.A.; Biggio, J.R.; Huh, W.K. Feasibility of complete salpingectomy compared with standard postpartum tubal ligation at cesarean delivery a randomized controlled trial. Obstet. Gynecol. 2018, 132, 20–27. [Google Scholar] [CrossRef]
- Wagar, M.K.; Godecker, A.; Landeros, M.V.; Williams, M. Postpartum Salpingectomy Compared with Standard Tubal Ligation after Vaginal Delivery. Obstet. Gynecol. 2021, 137, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Danis, R.B.; Della Badia, C.R.; Richard, S.D. Postpartum Permanent Sterilization: Could Bilateral Salpingectomy Replace Bilateral Tubal Ligation? J. Minim. Invasive Gynecol. 2016, 23, 928–932. [Google Scholar] [CrossRef] [PubMed]
- Chohan, L.; Richardson, D.L. Acog Committee Opinion Number 774 Opportunistic Salpingectomy as a Strategy for Epithelial Ovarian Cancer Prevention. Obstet. Gynecol. 2019, 133, E279–E284. [Google Scholar] [CrossRef]
- Dildy, G.A.; Paine, A.R.; George, N.C.; Velasco, C. Estimating blood loss: Can teaching significantly improve visual estimation? Obstet. Gynecol. 2004, 104, 601–606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parikh, P.; Kim, S.; Hathcock, M.; Torbenson, V.E.; Raju, R. Safety of salpingectomy at time of delivery. J. Matern. Neonatal Med. 2021, 34, 2765–2770. [Google Scholar] [CrossRef] [PubMed]
- Roeckner, J.T.; Sawangkum, P.; Sanchez-Ramos, L.; Duncan, J.R. Salpingectomy at the time of cesarean delivery: A systematic review and meta-analysis. Obstet. Gynecol. 2020, 135, 550–557. [Google Scholar] [CrossRef]
- Bertrand, J.T.; Sullivan, T.M.; Knowles, E.A.; Zeeshan, M.F.; Shelton, J.D. Contraceptive method skew and shifts in method mix in low- and middle-income countries. Int. Perspect. Sex. Reprod. Health 2014, 40, 144–153. [Google Scholar] [CrossRef]
- Doh, A.S.; Kamdom-Moyo, J.; Kouam, L. Minilaparotomy tubal ligation in the immediate post-partum after vaginal delivery. A retrospective analysis of 253 cases in the maternity service of the Yaounde University Hospital Center (Cameroon). Contracept. Fertil. Sex. 1996, 24, 49–51. [Google Scholar]
- Andrade Zamora, J.G.; Andrade Zamora, B.N.; Aguirre Velázquez, R.M.; Vázquez Méndez, J.; García Flores, R.F. Postpartum sterilization in a program of medical training. Ginecol. Obstet. Mex. 1986, 54, 148–151. [Google Scholar]
- Keogh, S.C.; Fry, K.; Mbugua, E.; Ayallo, M.; Quinn, H.; Otieno, G.; Ngo, T.D. Vocal local versus pharmacological treatments for pain management in tubal ligation procedures in rural Kenya: A non-inferiority trial. BMC Women’s Health 2014, 14, 21. [Google Scholar] [CrossRef] [Green Version]
- Stafford, I.; Dildy, G.A.; Clark, S.L.; Belfort, M.A. Visually estimated and calculated blood loss in vaginal and cesarean delivery. Am. J. Obstet. Gynecol. 2008, 199, 519.e1–519.e7. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.L.; Schulkin, J.; Urban, R.R.; Wright, J.D.; Burke, W.M.; Hou, J.Y.; McElwain, C.A.; Tergas, A.I. Physicians’ Perspectives and Practice Patterns Toward Opportunistic Salpingectomy in High- and Low-Risk Women. Cancer Investig. 2017, 35, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Committee on Health Care for Underserved Women. Committee opinion No. 530: Access to postpartum sterilization. Obstet. Gynecol. 2012, 120, 212–215. [Google Scholar] [CrossRef] [PubMed]
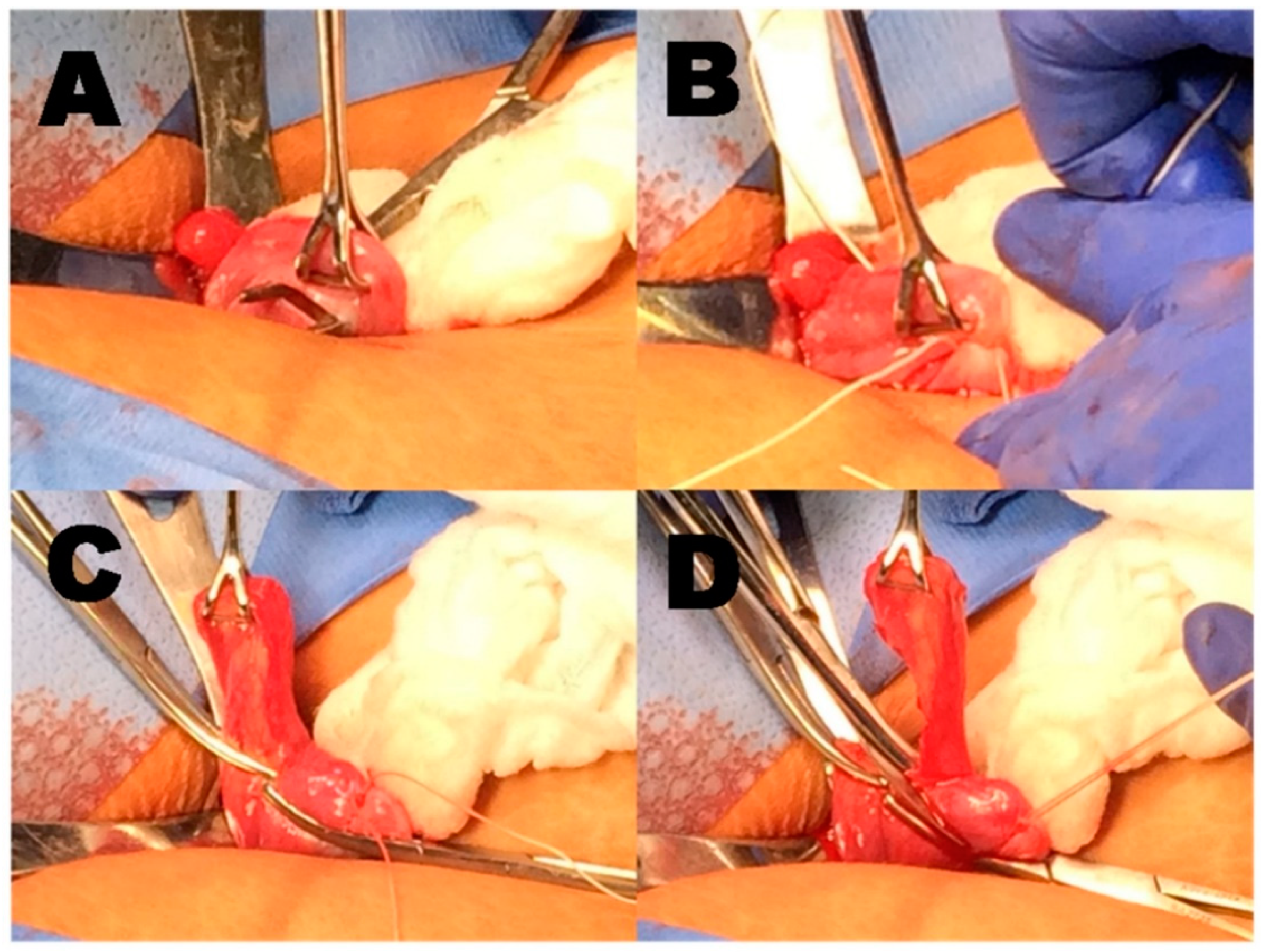
| Sterilized Post-Partum Days #0–2 | Sterilized at Cesarean | |||||
|---|---|---|---|---|---|---|
| SVD-PPS (n = 97) | SVD-BTL (n = 200) | p | CD-PPS (n = 60) | CD-BTL (n = 100) | p | |
| Age (years) | 32.94 ± 4.5 | 33.3 ± 4.4 | 0.556 | 34.2 ± 4.8 | 32.7 ± 4.8 | 0.043 |
| Gravidity | 4.67 ± 1.7 | 4.6 ± 1.6 | 0.661 | 4.3 ± 1.9 | 4.1 ± 1.6 | 0.592 |
| Parity | 4.1 ± 1.4 | 4.0 ± 1.3 | 0.802 | 3.8 ± 1.6 | 3.5 ± 1.3 | 0.209 |
| Prior abdominal surgery (median, IQR) | 0 (0–0) | 0 (0–0) | 0.0428 | 1 (1–2) | 2 (1–2) | 0.0257 |
| Race/ethnicity | ||||||
| Hispanic/Latina | 92 (94.8%) | 195 (97.5%) | 53 (88.3%) | 89 (88.1%) | ||
| Non-Hispanic White | 3 (3.1%) | 2 (1%) | 2 (3.3%) | 2 (2%) | ||
| Non-Hispanic Black | 2 (2.1%) | 2 (1%) | 5 (8.3%) | 9 (8.9%) | ||
| Non-Hispanic Asian | 0 (0%) | 1 (0.5%) | 0.427 | 0 (0%) | 1 (1%) | 0.83 |
| BMI (kg/m2) | 31.0 ± 5.0 | 31.9 ± 5.1 | 0.125 | 35.0 ± 6.2 | 34.0 ± 7.3 | 0.406 |
| Smoking | 1 (1%) | 11 (5.5%) | 0.067 | 3 (5%) | 8 (7.9%) | 0.478 |
| History of gonorrhea/chlamydia/ pelvic inflammatory disease | 10 (10.3%) | 15 (7.5%) | 0.414 | 6 (10%) | 8 (7.9%) | 0.651 |
| Diabetes, gestational or pre- gestational | 25 (25.8%) | 49 (24.5%) | 0.812 | 19 (31.7%) | 27 (26.7%) | 0.503 |
| Hypertension, including PIH | 15 (15.5%) | 32 (16.0%) | 0.82 | 19 (31.7%) | 21 (20.8%) | 0.123 |
| Family history of gynecologic or breast cancer | 7 (7.2%) | 13 (6.5%) | 0.817 | 5 (8.3%) | 8 (7.9%) | 0.926 |
| Chorioamnionitis | 2 (2.1%) | 1 (0.5%) | 0.207 | 1 (1.7%) | 0 (0%) | 0.193 |
| Sterilized Post-Partum Days #0–2 | Sterilized at Cesarean | |||||
|---|---|---|---|---|---|---|
| SVD-PPS | SVD-BTL | p | CD-PPS | CD-BTL | p | |
| (n = 97) | (n = 200) | (n = 60) | (n = 100) | |||
| Operative time (minutes) | 39.1 + 11.8 | 34.3 + 13.1 | 0.003 | 99.5 + 47.3 | 86.5 + 33.9 | 0.048 |
| Estimated blood loss (mL) | 21.0 + 22.0 | 13.4 + 17.3 | 0.001 | 994 + 622 | 988 + 661 | 0.935 |
| Skin incision | ||||||
| <5 cm | 95 (99.0%) | 197 (98.5%) | N/A | N/A | ||
| ≥5 cm | 1 (1.0%) | 3 (1.5%) | 0.749 | N/A | N/A | |
| Intra-operative blood transfusion | 0 | 0 | 3 (5.0%) | 2 (2.0%) | 0.286 | |
| Urinary tract injury | 0 | 0 | 1 (1.7%) | 1 (1.0%) | 0.708 | |
| Lost pedicle or bleeding from the mesosalpinx | 3 (3.1%) | 5 (2.5%) | 0.767 | 1 (1.7%) | 2 (2.0 %) | 0.887 |
| Other injury to surrounding structures | 0 | 1 (0.5%) | 0.98 | 0 | 0 | |
| Procedure aborted | 5 (5.2%) | 2 (1%) | 0.027 | 3 (5.0%) | 5 (5.0%) | 0.989 |
| Sterilized Post-Partum Days #0–2 | Sterilized at Cesarean | |||||
|---|---|---|---|---|---|---|
| SVD-PPS | SVD-BTL | CD-PPS | CD-BTL | |||
| (n = 97) | (n = 200) | p | (n = 60) | (n = 100) | p | |
| Superficial wound separation | 0 | 0 | 0 | 3 (3.0%) | 0.18 | |
| Wound dehiscence | 0 | 0 | 0 | 0 | ||
| Re-admission | 1 (1%) | 2 (1%) | 0.98 | 0 | 0 | |
| Cellulitis | 0 | 0 | 0 | 2 (2.0%) | 0.27 | |
| Blood transfusion | 0 | 0 | 0 | 0 (0%) | ||
| Pathology confirmed permanent contraception | 97 (100%) | 198 (99.0%) | 60 (100.0) | 99 (99.0%) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsieh, G.L.; Wagar, M.K.; Antony, K.M.; Masand, R.; Anderson, M.L. A Standardized Protocol for Post-Partum Salpingectomy with Suture Ligation: A Prospective Feasibility Study. Reprod. Med. 2022, 3, 150-159. https://doi.org/10.3390/reprodmed3020013
Hsieh GL, Wagar MK, Antony KM, Masand R, Anderson ML. A Standardized Protocol for Post-Partum Salpingectomy with Suture Ligation: A Prospective Feasibility Study. Reproductive Medicine. 2022; 3(2):150-159. https://doi.org/10.3390/reprodmed3020013
Chicago/Turabian StyleHsieh, Gillian L., Matthew K. Wagar, Kathleen M. Antony, Ramya Masand, and Matthew L. Anderson. 2022. "A Standardized Protocol for Post-Partum Salpingectomy with Suture Ligation: A Prospective Feasibility Study" Reproductive Medicine 3, no. 2: 150-159. https://doi.org/10.3390/reprodmed3020013
APA StyleHsieh, G. L., Wagar, M. K., Antony, K. M., Masand, R., & Anderson, M. L. (2022). A Standardized Protocol for Post-Partum Salpingectomy with Suture Ligation: A Prospective Feasibility Study. Reproductive Medicine, 3(2), 150-159. https://doi.org/10.3390/reprodmed3020013







