Effect of High-Dose Intravenous Vitamin C on Postpartum Oxidative Stress in Severe Preeclampsia
Abstract
1. Introduction
2. Methods
2.1. Oxidative Stress Biomarker Measurements
2.2. Statistical Analysis
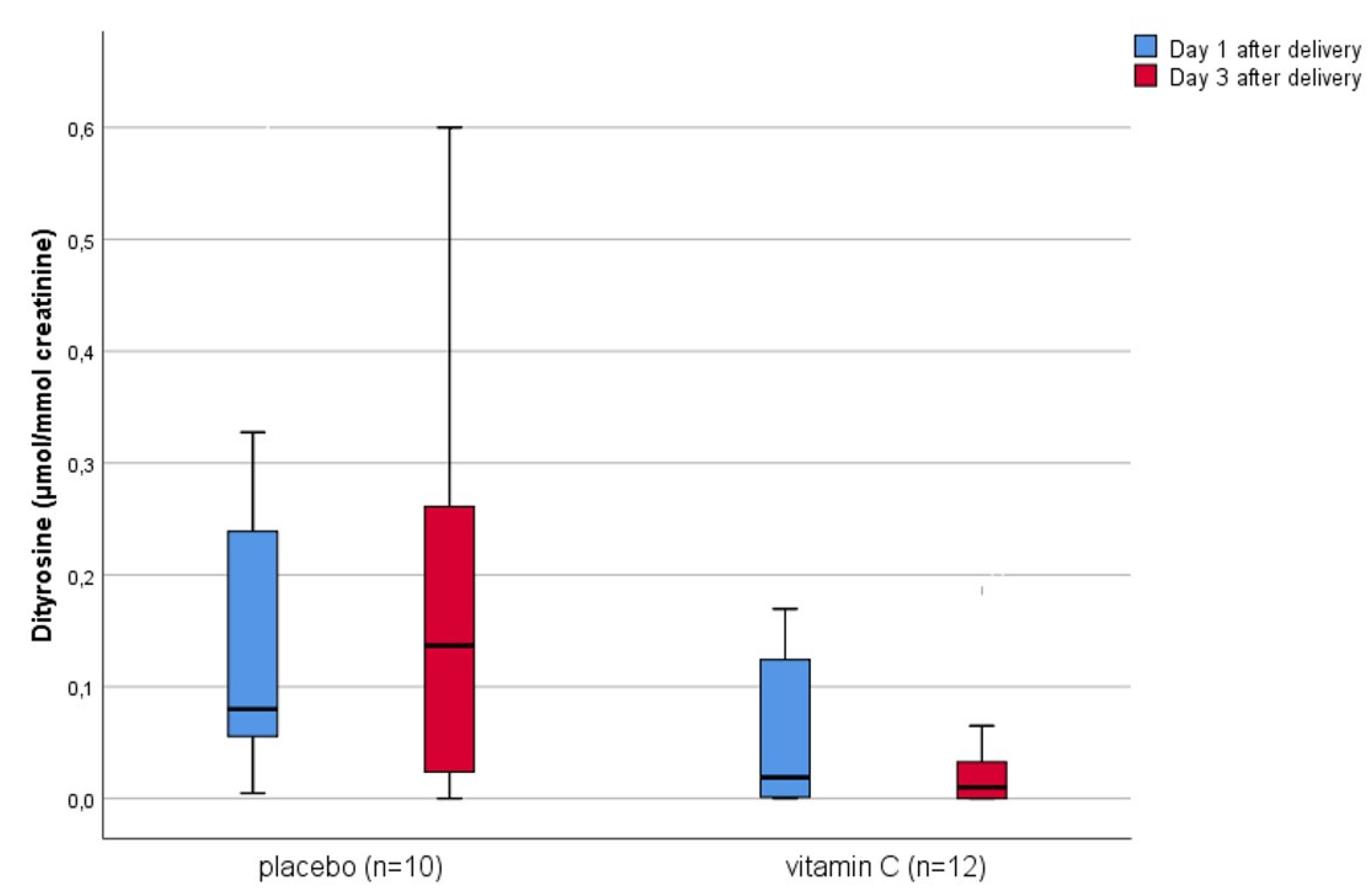
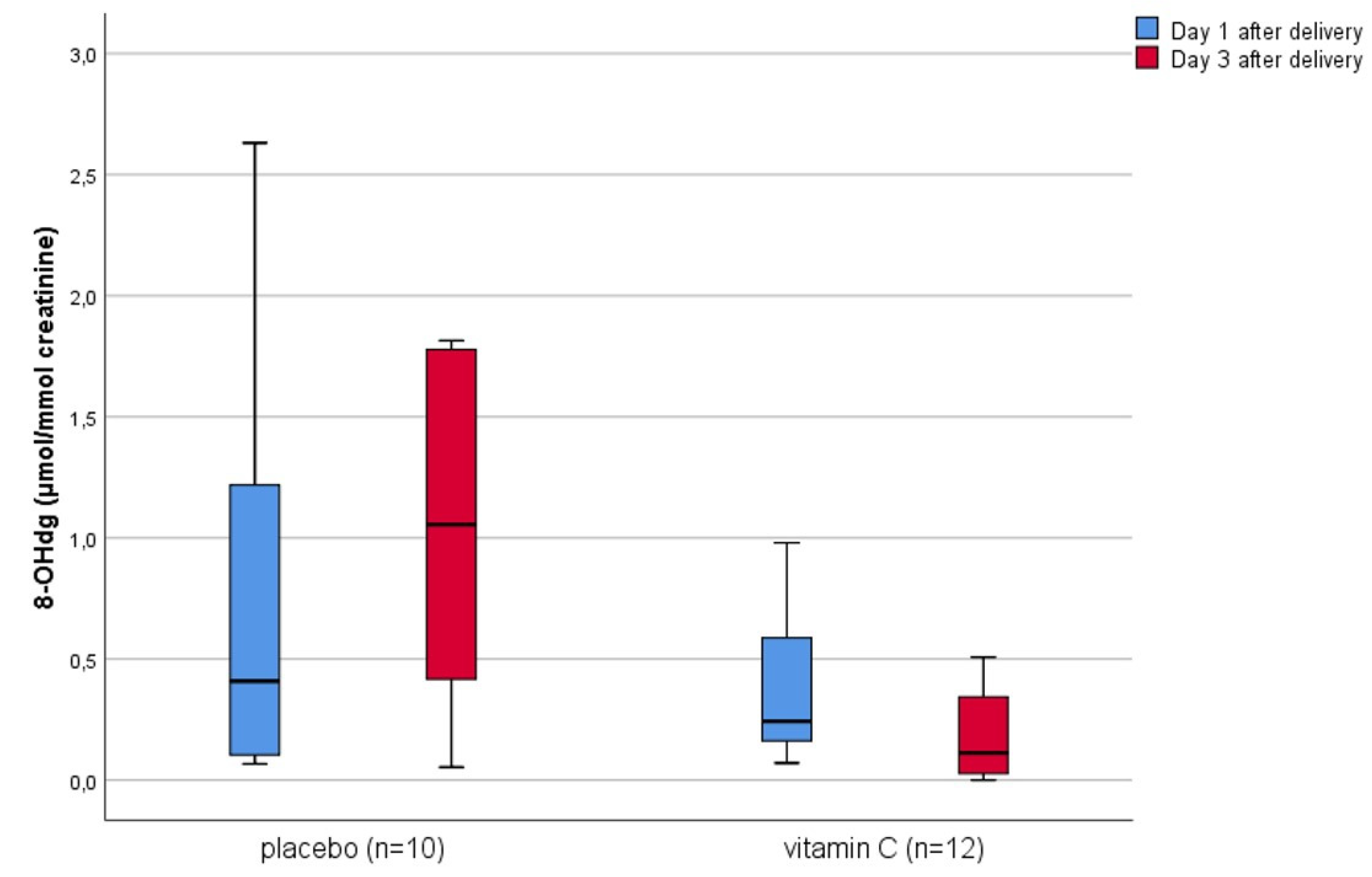
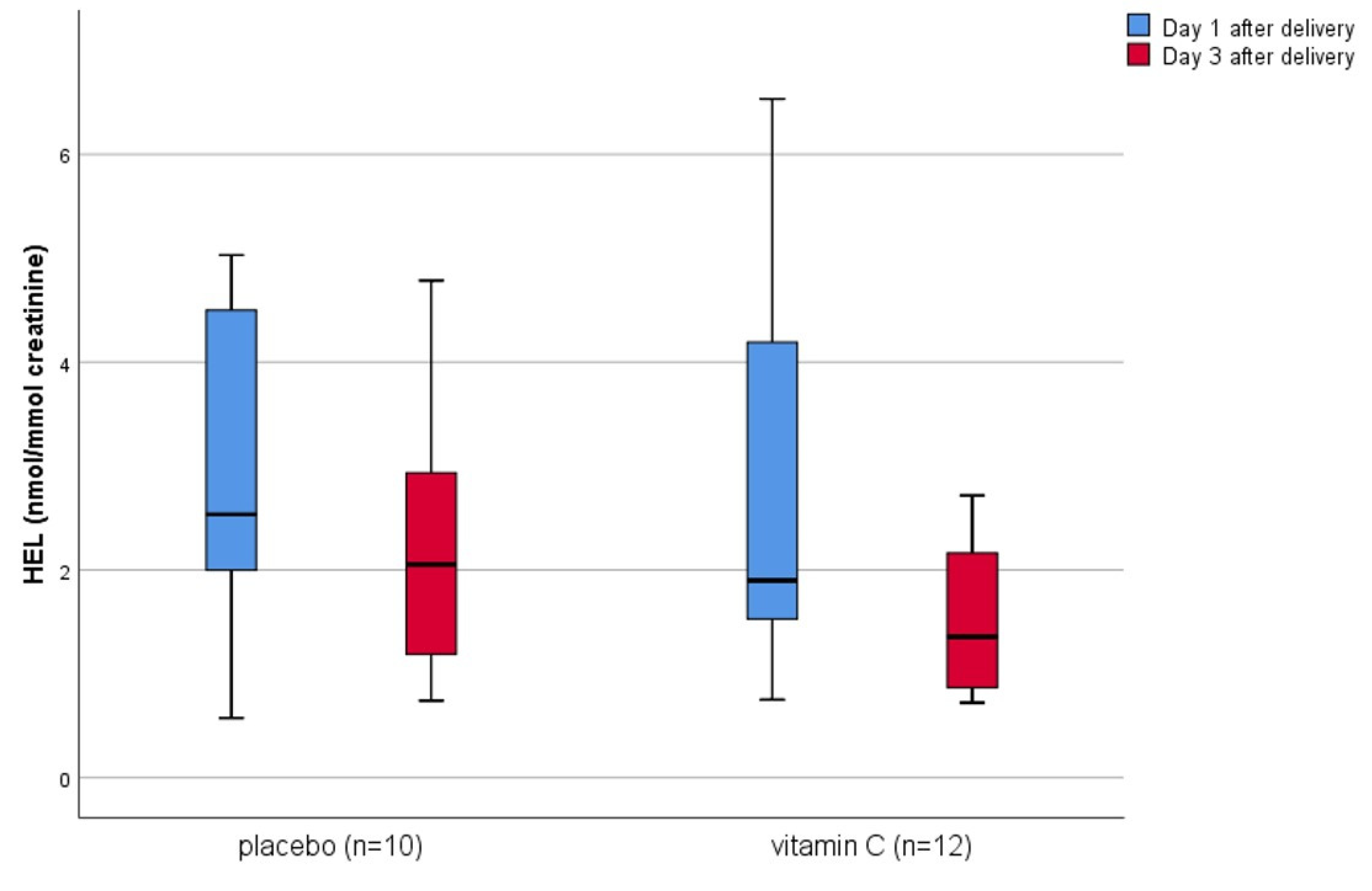
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Correction Statement
References
- Ananth, C.V.; Keyes, K.M.; Wapner, R.J. Pre-eclampsia rates in the United States, 1980–2010: Age-period-cohort analysis. BMJ 2013, 347, f6564. [Google Scholar] [CrossRef]
- Hernández-Díaz, S.; Toh, S.; Cnattingius, S. Risk of pre-eclampsia in first and subsequent pregnancies: Prospective cohort study. BMJ 2009, 338, b2255. [Google Scholar] [CrossRef] [PubMed]
- Hutcheon, J.A.; Lisonkova, S.; Joseph, K.S. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 391–403. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists; Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 2013, 122, 1122–1131. [Google Scholar] [CrossRef]
- World Health Organization. The World Health Report 2005: Make Every Mother and Child Count. 2005. Available online: https://www.who.int/whr/2005/whr2005_en.pdf?ua=1 (accessed on 28 May 2020).
- Chaiworapongsa, T.; Chaemsaithong, P.; Yeo, L.; Romero, R. Pre-eclampsia part 1: Current understanding of its pathophysiology. Nat. Rev. Nephrol. 2014, 10, 466–480. [Google Scholar] [CrossRef] [PubMed]
- Cabello-Verrugio, C.; Simon, F.; Trollet, C.; Santibañez, J.F. Oxidative stress in disease and aging: Mechanisms and therapies 2016. Oxid. Med. Cell. Longev. 2017, 4310469. [Google Scholar] [CrossRef]
- McClure, J.H.; Cooper, G.M.; Clutton-Brock, T.H.; Centre for Maternal and Child Enquiries. Saving mothers’ lives: Reviewing maternal deaths to make motherhood safer: 2006-8: A review. Br. J. Anaesth. 2011, 107, 127–132. [Google Scholar] [CrossRef]
- Staff, A.C.; Benton, S.J.; von Dadelszen, P.; Roberts, J.M.; Taylor, R.N.; Powers, R.W.; Charnock-Jones, D.S.; Redman, C.W. Redefining preeclampsia using placenta-derived biomarkers. Hypertension 2013, 61, 932–942. [Google Scholar] [CrossRef]
- Mikhail, M.S.; Anyaegbunam, A.; Garfinkel, D.; Palan, P.R.; Basu, J.; Romney, S.L. Preeclampsia and antioxidant nutrients: Decreased plasma levels of reduced ascorbic acid, alpha-tocopherol, and beta-carotene in women with preeclampsia. Am. J. Obstet. Gynecol. 1994, 171, 150–157. [Google Scholar] [CrossRef]
- Honore, P.M.; Jacobs, R.; Hendrickx, I.; De Waele, E.; Spapen, H.D. Adjuvant vitamin C treatment in sepsis-how many oranges a day keep (vasopressor-dependent) septic shock away? J. Thorac. Dis. 2016, 8, E993–E995. [Google Scholar] [CrossRef]
- Lichtenstein, D.; Mézière, G.; Biderman, P.; Gepner, A.; Barré, O. The comet-tail artifact. An ultrasound sign of alveolar-interstitial syndrome. Am. J. Respir. Crit. Care Med. 1997, 156, 1640–1646. [Google Scholar] [CrossRef] [PubMed]
- Buehner, M.; Pamplin, J.; Studer, L.; Hughes, R.L.; King, B.T.; Graybill, J.C.; Chung, K.K. Oxalate nephropathy after continuous infusion of high-dose vitamin C as an adjunct to burn resuscitation. J. Burn Care Res. 2016, 37, e374–e379. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, J.; Hansen, J.; Ashmore, R.C. Postpartum preeclampsia complicated by acute pulmonary edema. Hawaii Med. J. 1994, 53, 248–249. [Google Scholar] [PubMed]
- Matthys, L.A.; Coppage, K.H.; Lambers, D.S.; Barton, J.R.; Sibai, B.M. Delayed postpartum preeclampsia: An experience of 151 cases. Am. J. Obstet. Gynecol. 2004, 190, 1464–1466. [Google Scholar] [CrossRef]
- Chappell, L.C.; Seed, P.T.; Briley, A.L.; Kelly, F.J.; Lee, R.; Hunt, B.J.; Parmar, K.; Bewley, S.J.; Shennan, A.H.; Steer, P.J.; et al. Effect of antioxidants on the occurrence of pre-eclampsia in women at increased risk: A randomised trial. Lancet 1999, 354, 810–816. [Google Scholar] [CrossRef]
- Beazley, D.; Ahokas, R.; Livingston, J.; Griggs, M.; Sibai, B.M. Vitamin C and E supplementation in women at high risk for preeclampsia: A double-blind, placebo-controlled trial. Am. J. Obstet. Gynecol. 2005, 192, 520–521. [Google Scholar] [CrossRef]
- Rumbold, A.R.; Crowther, C.A.; Haslam, R.R.; Dekker, G.A.; Robinson, J.S.; ACTS Study Group. Vitamins C and E and the risks of preeclampsia and perinatal complications. N. Engl. J. Med. 2006, 354, 1796–1806. [Google Scholar] [CrossRef]
- Matsuda, T.; Tanaka, H.; Hanumadass, M.; Gayle, R.; Yuasa, H.; Abcarian, H.; Matsuda, H.; Reyes, H. Effects of high-dose vitamin C administration on postburn microvascular fluid and protein flux. J. Burn Care Rehabil. 1992, 13, 560–566. [Google Scholar] [CrossRef]
- Matsuda, T.; Tanaka, H.; Reyes, H.M.; Richter, H.M., 3rd; Hanumadass, M.M.; Shimazaki, S.; Matsuda, H.; Nyhus, L.M. Antioxidant therapy using high dose vitamin C: Reduction of postburn resuscitation fluid volume requirements. World J. Surg. 1995, 19, 287–291. [Google Scholar] [CrossRef]
- Teng, J.; Pourmand, A.; Mazer-Amirshahi, M. Vitamin C: The next step in sepsis management? J. Crit. Care 2018, 43, 230–234. [Google Scholar] [CrossRef]
- Pryor, W.A. Oxy-radicals and related species: Their formation, lifetimes, and reactions. Annu. Rev. Physiol. 1986, 48, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef] [PubMed]
- Il’yasova, D.; Scarbrough, P.; Spasojevic, I. Urinary biomarkers of oxidative status. Clin. Chim. Acta 2012, 413, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Kino, S.; Masuda, A.; Takeuchi, M.; Ochi, T.; Osredkar, J.; Rejc, B.; Gersak, K.; Ramarathnam, N.; Kato, Y. Determination of HEL (Hexanoyl-lysine adduct): A novel biomarker for omega-6 PUFA oxidation. Subcell. Biochem. 2014, 77, 61–72. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Ogawa, Y.; Yoshida, R.; Shimamori, A.; Kasai, H.; Ohta, H. The stability of the oxidative stress marker, urinary 8-hydroxy-2′-deoxyguanosine (8-OHdG), when stored at room temperature. J. Occup. Health 2008, 50, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Czerska, M.; Mikołajewska, K.; Zieliński, M.; Gromadzińska, J.; Wąsowicz, W. Today’s oxidative stress markers. Med. Pr. 2015, 66, 393–405. [Google Scholar] [CrossRef]
- León-Reyes, G.; Maida-Claros, R.F.; Urrutia-Medina, A.X.; Jorge-Galarza, E.; Guzmán-Grenfell, A.M.; Fuentes-García, S.; Medina-Navarro, R.; Moreno-Eutimio, M.A.; Muñoz-Sánchez, J.L.; Hicks, J.J.; et al. Oxidative profiles of LDL and HDL isolated from women with preeclampsia. Lipids Health Dis. 2017, 16, 90. [Google Scholar] [CrossRef]
- Scholl, T.O.; Stein, T.P. Oxidant damage to DNA and pregnancy outcome. J. Matern. Fetal Med. 2001, 10, 182–185. [Google Scholar] [CrossRef]
- Barden, A.; Beilin, L.J.; Ritchie, J.; Croft, K.D.; Walters, B.N.; Michael, C.A. Plasma and urinary 8-iso-prostane as an indicator of lipid peroxidation in pre-eclampsia and normal pregnancy. Clin. Sci. 1996, 91, 711–718. [Google Scholar] [CrossRef]
- McKinney, E.T.; Shouri, R.; Hunt, R.S.; Ahokas, R.A.; Sibai, B.M. Plasma, urinary, and salivary 8-epi-prostaglandin f2alpha levels in normotensive and preeclamptic pregnancies. Am. J. Obstet. Gynecol. 2000, 183, 874–877. [Google Scholar] [CrossRef]
- Yancey, L.M.; Withers, E.; Bakes, K.; Abbott, J. Postpartum preeclampsia: Emergency department presentation and management. J. Emerg. Med. 2011, 40, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Dennis, A.T.; Solnordal, C.B. Acute pulmonary oedema in pregnant women. Anaesthesia 2012, 67, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, S.J.; Berg, C.; Bernstein, P.; Bingham, D.; Delgado, A.; Callaghan, W.M.; Harris, K.; Lanni, S.; Mahoney, J.; Main, E.; et al. Standardized severe maternal morbidity review: Rationale and process. Obstet. Gynecol. 2014, 124, 361–366. [Google Scholar] [CrossRef] [PubMed]


| Characteristic | Vitamin C (n = 12) | Placebo (n = 10) | p-Value |
|---|---|---|---|
| Maternal age (years) | 30 (22–41) | 30 (23–40) | 0.97 |
| Pre-pregnancy BMI (kg/m2) | 23 (17–35) | 26 (18–41) | 0.23 |
| BMI at delivery (kg/m2) | 29 (22–56) | 31 (20–49) | 0.69 |
| Nulliparity | 10 (83%) | 7 (70%) | 0.28 |
| Gestational age (weeks) | 33 (24–38) | 33 (26–39) | 0.38 |
| Caesarean delivery | 9 (75%) | 9 (90%) | 0.69 |
| Neonatal birth weight (g) | 1465 (490–3110) | 1820 (630–3870) | 0.33 |
| SGA | 8 (67%) | 4 (40%) | 0.15 |
| Systolic blood pressure ≥160 mm Hg at inclusion | 12 (100%) | 10 (100%) | / |
| Diastolic blood pressure ≥110 mm Hg at inclusion | 12 (100%) | 10 (100%) | / |
| Neurological symptoms | 3 (25%) | 4 (40%) | 0.21 |
| Serum creatinine >97 µmol/L | 1 (8%) | 0 (0%) | 0.33 |
| Elevated liver enzymes and/or pain in the right upper or middle upper abdomen | 8 (67%) | 3 (30%) | 0.09 |
| Thrombocytopenia | 3 (25%) | 2 (20%) | 0.78 |
| Urine output at inclusion (mL/kg/h) | 0.62 (0.40–1.17) | 0.52 (0.27–1.33) | 0.42 |
| Serum creatinine at inclusion (µmol/L) | 59 (48–133) | 55 (43–67) | 0.35 |
| sFlt-1/PlGF | 268 (152–2324) | 399 (111–998) | 0.12 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korenc, M.; Osredkar, J.; Gersak, K.; Kumer, K.; Fabjan, T.; Sterpin, S.; Lucovnik, M. Effect of High-Dose Intravenous Vitamin C on Postpartum Oxidative Stress in Severe Preeclampsia. Reprod. Med. 2020, 1, 122-131. https://doi.org/10.3390/reprodmed1020009
Korenc M, Osredkar J, Gersak K, Kumer K, Fabjan T, Sterpin S, Lucovnik M. Effect of High-Dose Intravenous Vitamin C on Postpartum Oxidative Stress in Severe Preeclampsia. Reproductive Medicine. 2020; 1(2):122-131. https://doi.org/10.3390/reprodmed1020009
Chicago/Turabian StyleKorenc, Monika, Joško Osredkar, Ksenija Gersak, Kristina Kumer, Teja Fabjan, Sasa Sterpin, and Miha Lucovnik. 2020. "Effect of High-Dose Intravenous Vitamin C on Postpartum Oxidative Stress in Severe Preeclampsia" Reproductive Medicine 1, no. 2: 122-131. https://doi.org/10.3390/reprodmed1020009
APA StyleKorenc, M., Osredkar, J., Gersak, K., Kumer, K., Fabjan, T., Sterpin, S., & Lucovnik, M. (2020). Effect of High-Dose Intravenous Vitamin C on Postpartum Oxidative Stress in Severe Preeclampsia. Reproductive Medicine, 1(2), 122-131. https://doi.org/10.3390/reprodmed1020009





