Photophysical, Electrochemical, Density Functional Theory, and Spectroscopic Study of Some Oligothiophenes
Abstract
1. Introduction
1.1. Background and Motivation
1.2. Thiophene-Based Functional Materials
1.3. Controlling Solid-State Structure Through Crystal Engineering
1.4. Impact of Electron-Withdrawing Substituents
1.5. Previous Work on DCV- and TCV-Oligothiophenes
1.6. Synthetic Accessibility of Dicyanovinyl Thiophenes
1.7. Scope of This Work
2. Materials and Methods
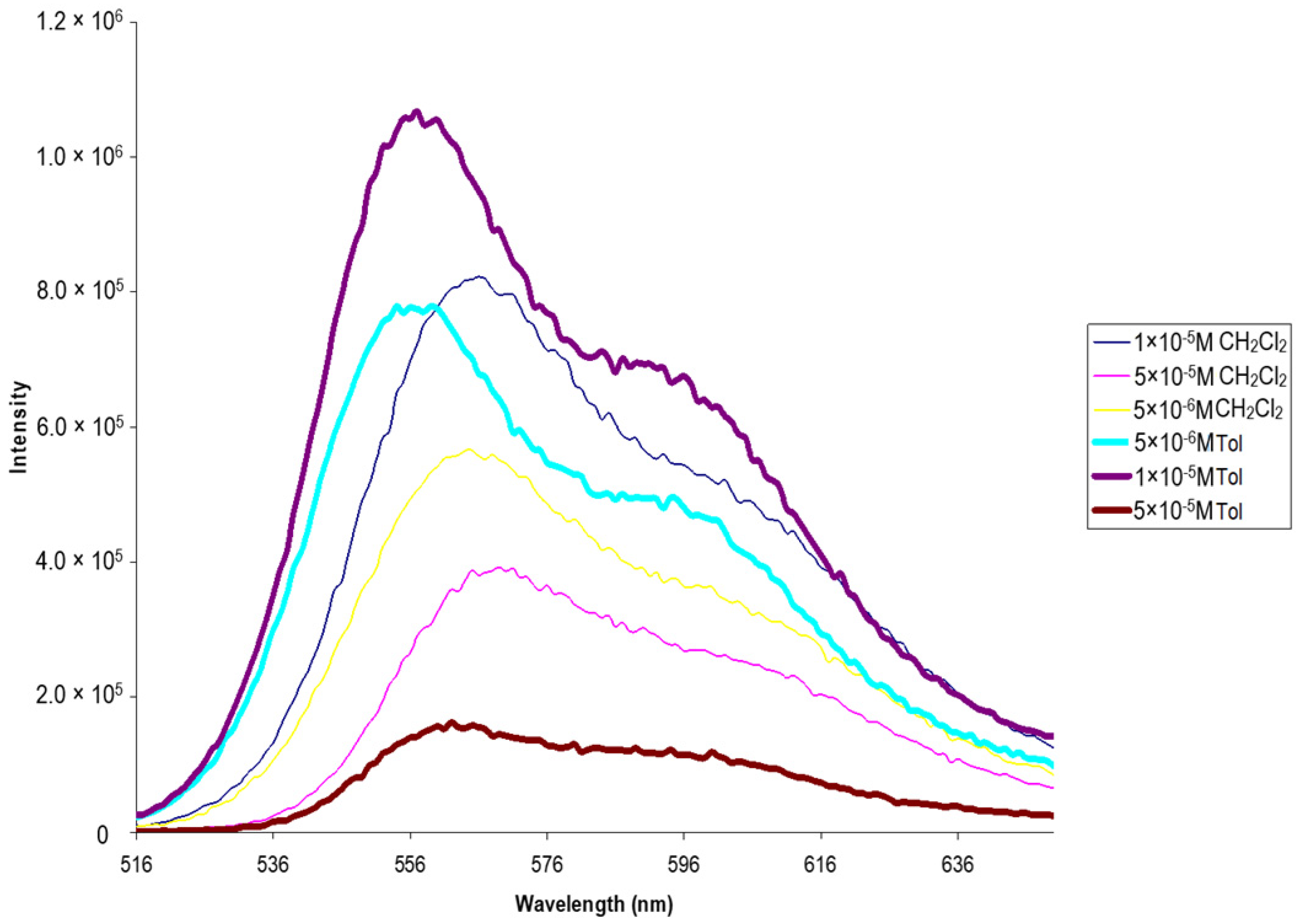
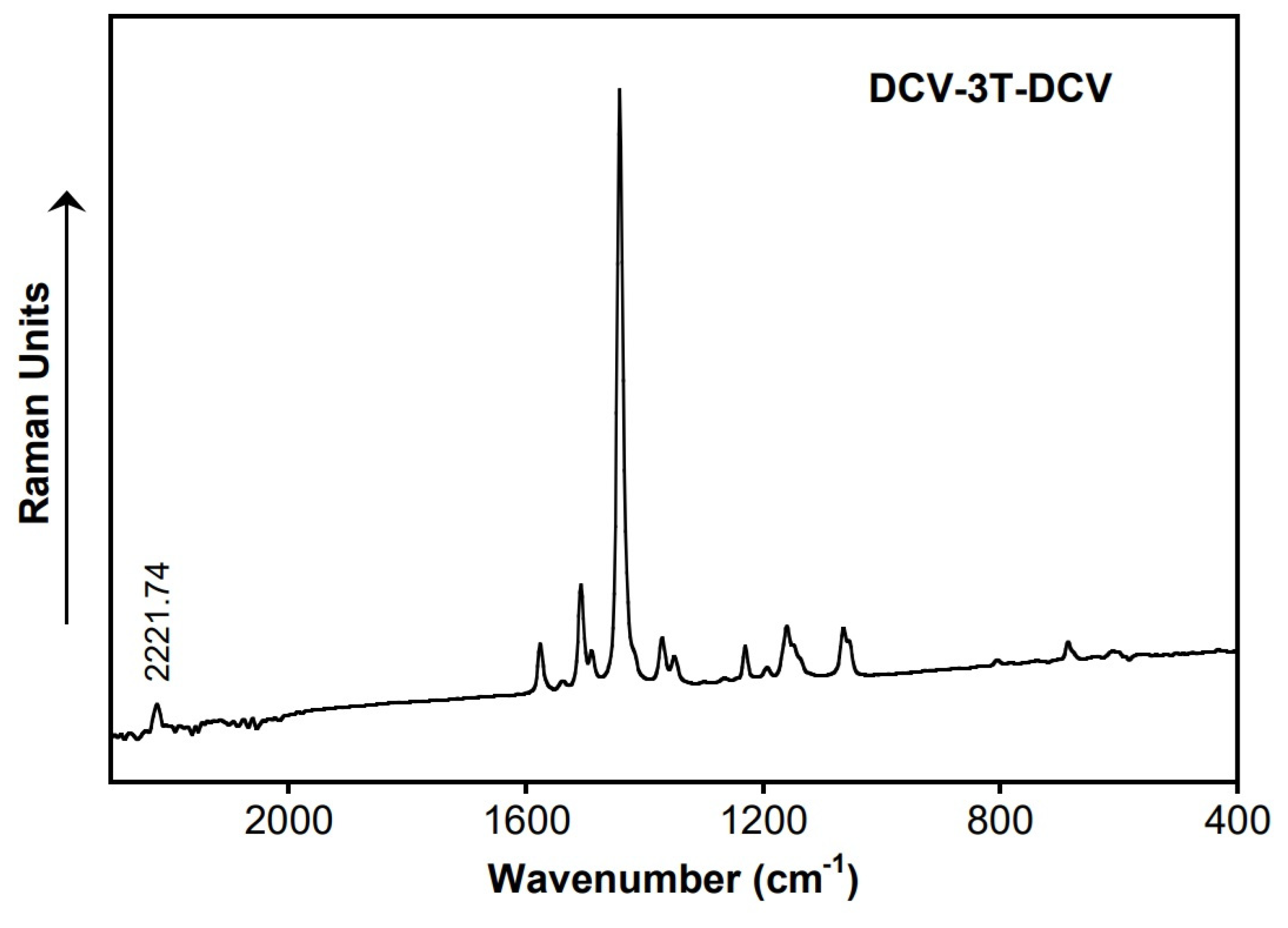
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kertesz, M.; Choi, C.H.; Yang, S. Conjugated Polymers and Aromaticity. Chem. Rev. 2005, 105, 3448–3481. [Google Scholar] [CrossRef]
- Roncali, J. Synthetic Principles for Bandgap Control in Linear π-Conjugated Systems. Chem. Rev. 1997, 97, 173–206. [Google Scholar] [CrossRef]
- Brédas, J.-L.; Beljonne, D.; Coropceanu, V.; Cornil, J. Charge-Transfer and Energy-Transfer Processes in π-Conjugated Oligomers and Polymers: A Molecular Picture. Chem. Rev. 2004, 104, 4971–5004. [Google Scholar] [CrossRef] [PubMed]
- Friederich, P.; Fediai, A.; Kaiser, S.; Konrad, M.; Jung, N.; Wenzel, W. Toward Design of Novel Materials for Organic Electronics. Adv. Mater. 2019, 31, 1808256. [Google Scholar] [CrossRef] [PubMed]
- Bader, M.M.; Carvalho, T.; Li, H.; Tarter, S.; Spangler, C.W. Design and Synthesis of New Acceptor Molecules for Photoinduced Electron Transfer Reverse Saturable Absorption. In Linear and Nonlinear Optics of Organic Materials, Proceedings of the International Symposium on Optical Science and Technology, 2001, San Diego, CA, USA, 29 July–3 August 2001; SPIE: Bellingham, WA, USA, 2001; Volume 4461. [Google Scholar] [CrossRef]
- Mishra, A.; Ma, C.-Q.; Bäuerle, P. Functional Oligothiophenes: Molecular Design for Multidimensional Nanoarchitectures and Their Applications. Chem. Rev. 2009, 109, 1141–1276. [Google Scholar] [CrossRef] [PubMed]
- Perepichka, I.F.; Perepichka, D.F. Handbook of Thiophene-Based Materials: Applications in Organic Electronics and Photonics, 2 Volumes; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Pham, P.-T.T.; Bader, M.M. Inter- and Intramolecular Interactions in Some Bromo- and Tricyanovinyl-Substituted Thiophenes and Ethylenedioxythiophenes. Cryst. Growth Des. 2014, 14, 916–922. [Google Scholar] [CrossRef]
- Casado, J.; Pappenfus, T.M.; Miller, L.L.; Mann, K.R.; Ortí, E.; Viruela, P.M.; Pou-Amérigo, R.; Hernández, V.; López Navarrete, J.T. Nitro-Functionalized Oligothiophenes as a Novel Type of Electroactive Molecular Material: Spectroscopic, Electrochemical, and Computational Study. J. Am. Chem. Soc. 2003, 125, 2524–2534. [Google Scholar] [CrossRef]
- Hapiot, P.; Demanze, F.; Yassar, A.; Garnier, F. Molecular Engineering of Band-Level Energies in Oligothiophenes through Cyano-Substitutions. J. Phys. Chem. 1996, 100, 8397–8401. [Google Scholar] [CrossRef]
- Zhan, X.; Zhang, W.; Li, Y.; Zhang, X.; Xie, Z.; Zhan, Q.; Zhan, Y. Dicyanovinyl-Substituted Oligothiophenes as Materials for Organic Photovoltaic Cells. J. Am. Chem. Soc. 2011, 133, 6787–6795. [Google Scholar]
- Bader, M.M.; Pham, P.-T.; Alrifai, S.M.T.; Younas, S.H.; Albusaili, J.A. Molecular Design and Crystal Engineering of Oligothiophenes Using Dicyanovinyl- (DCV) and Tricyanovinyl- (TCV) Groups. CrystEngComm 2025, 27, 5558–5570. [Google Scholar] [CrossRef]
- Cai, X.; Burand, M.W.; Newman, C.R.; da Silva Filho, D.A.; Pappenfus, T.M.; Bader, M.M.; Brédas, J.-L.; Mann, K.R.; Frisbie, C.D.J. N-and P-channel transport behavior in thin film transistors based on tricyanovinyl-capped oligothiophenes. Phys. Chem. B 2006, 110, 14590–14597. [Google Scholar] [CrossRef]
- Bader, M.M.; Pham, P.-T.T.; Nassar, B.R.; Lin, H.; Xia, Y.; Frisbie, C.D. Extended 7,7,8,8-Tetracyano-p-quinodimethane-Based Acceptors: How Molecular Shape and Packing Impact Electron Accepting Behavior. Cryst. Growth Des. 2009, 9, 4599–4601. [Google Scholar] [CrossRef]
- Spartan, Version 24; Wavefunction Inc: Irvine, CA, USA, 2024.
- Huss, A.S.; Pappenfus, T.; Bohnsack, J.; Burand, M.; Mann, K.R.; Blank, D.A. The Influence of Internal Charge Transfer on Nonradiative Decay in Substituted Terthiophenes. J. Phys. Chem. A 2009, 113, 10202–10210. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Kong, J.; Zhang, W.; Kuang, Z.; Zhou, M. Triplet Excited-State Dynamics in Benzothiadiazole-Based Thermally Activated Delayed Fluorescence Compound. J. Phys. Chem. Lett. 2024, 15, 2885–2892. [Google Scholar] [CrossRef] [PubMed]
- Stoltzfus, D.M.; Donaghey, J.E.; Armin, A.; Shaw, P.E.; Burn, P.L.; Meredith, P. Charge Generation Pathways in Organic Solar Cells: Assessing the Contribution from the Electron Acceptor. Chem. Rev. 2016, 116, 12920–12955. [Google Scholar] [CrossRef] [PubMed]
- Effenberger, F.; Wuerthner, F.; Steybe, F. Synthesis and Solvatochromic Properties of Donor–Acceptor–Substituted Oligothiophenes. J. Org. Chem. 1995, 60, 2082–2091. [Google Scholar] [CrossRef]
- Bogner, L.; Yang, Z.; Baum, S.; Corso, M.; Fitzner, R.; Bäuerle, P.; Franke, K.J.; Pascual, J.I.; Tegeder, P. Electronic States and Exciton Dynamics in Dicyanovinyl-Sexithiophene on Au(111). J. Phys. Chem. C 2016, 120, 27268–27275. [Google Scholar] [CrossRef]
- Yang, T.; Tang, Y.; Wei, Y.; Xie, L.; Huang, W. Molecular Gridization of Organic Semiconducting π Backbones. Acc. Chem. Res. 2025, 13, 1748–1751. [Google Scholar] [CrossRef]
- Harpham, M.R.; Süzer, Ö.; Ma, C.-Q.; Bäuerle, P.; Goodson, T., III. Thiophene Dendrimers as Entangled Photon Sensor Materials. J. Am. Chem. Soc. 2009, 131, 973–979. [Google Scholar] [CrossRef]
- Li, S.; Cooper, V.R.; Thonhauser, T.; Lundqvist, B.I.; Langreth, D.C. Stacking Interactions and DNA Intercalation. J. Phys. Chem. B 2009, 113, 11166–11172. [Google Scholar] [CrossRef]
- Ganji, M.; Kim, S.H.; van der Torre, J.; Abbondanzieri, E.; Dekker, C. Intercalation-Based Single-Molecule Fluorescence Assay To Study DNA Supercoil Dynamics. Nano Lett. 2016, 16, 4699–4707. [Google Scholar] [CrossRef]
- Agosti, E.; Rivola, M.; Hernandez, V.; Del Zoppo, M.; Zerbi, G. Electronic and Dynamical Effects from the Unusual Features of the Raman Spectra of Oligo and Polythiophenes. Synth. Met. 1999, 100, 101–112. [Google Scholar] [CrossRef]
- Esposti, A.D.; Moze, O.; Taliani, C.; Tomkinson, J.; Zamboni, R.; Zerbetto, F. The Intramolecular Vibrations of Prototypical Polythiophenes. J. Chem. Phys. 1996, 104, 9704–9718. [Google Scholar] [CrossRef]
- Milani, A.; Brambilla, L.; Del Zoppo, M.; Zerbi, G. Raman Dispersion and Intermolecular Interactions in Unsubstituted Thiophene Oligomers. J. Phys. Chem. B 2007, 111, 1271–1276. [Google Scholar] [CrossRef]
- Ortí, E.; Viruela, P.M.; Viruela, R.; Effenberger, F.; Hernández, V.; López Navarrete, J.T. Raman and Theoretical Study of the Solvent Effects on the Sizable Intramolecular Charge Transfer in the Push–Pull 5-(Dimethylamino)-5′-nitro-2,2′-bithiophene. J. Phys. Chem. A 2005, 109, 8724–8731. [Google Scholar] [CrossRef]
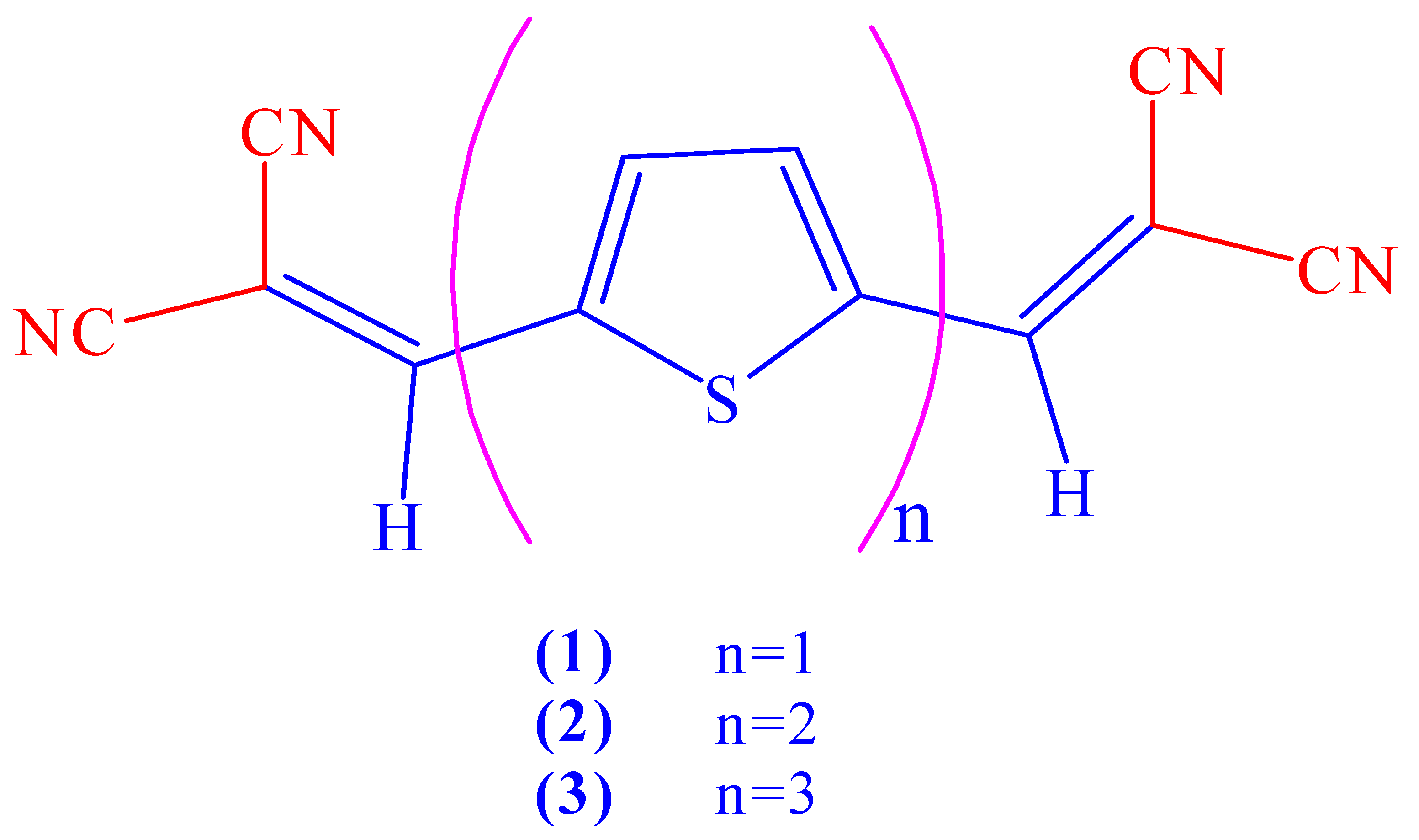
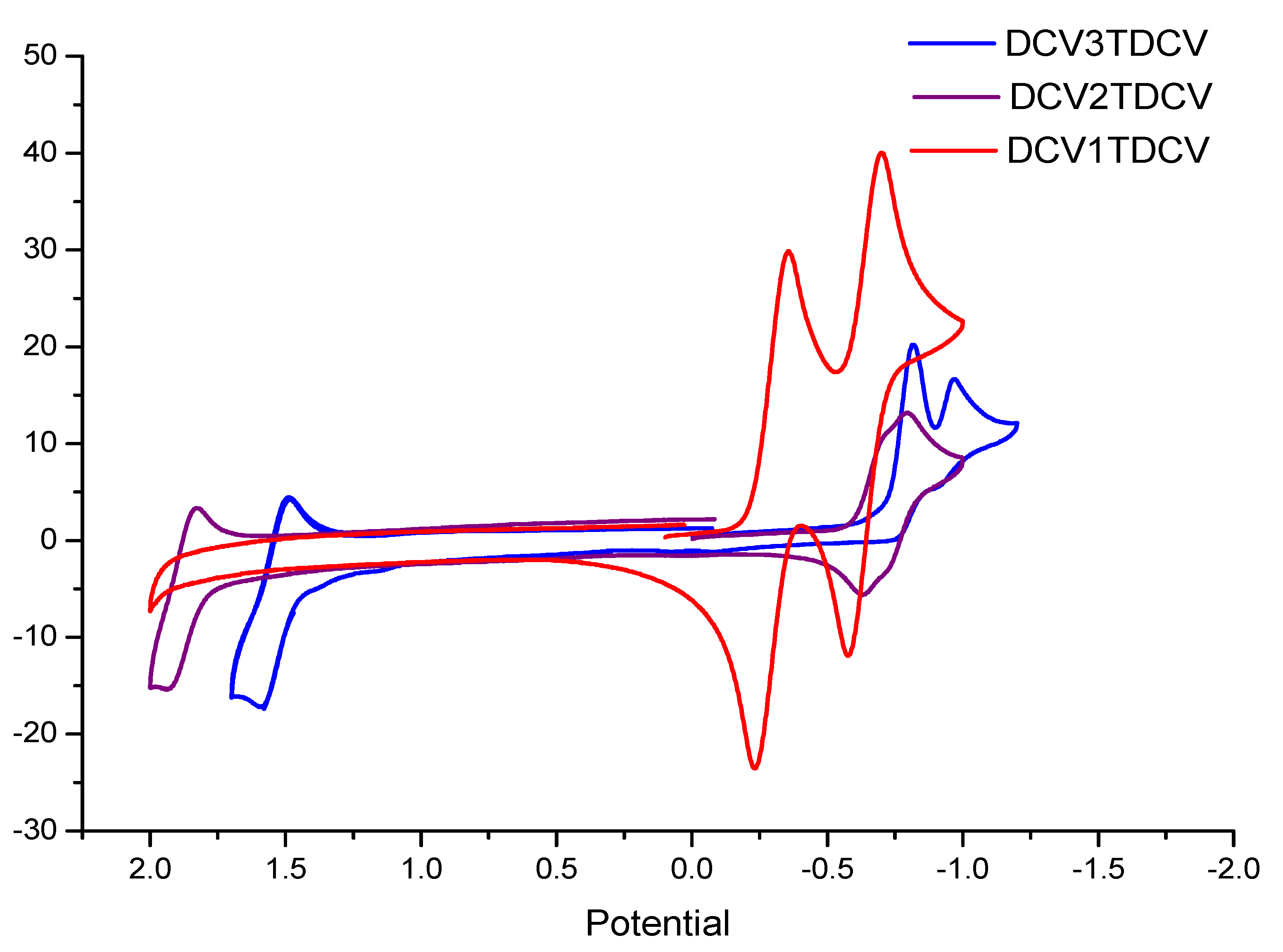
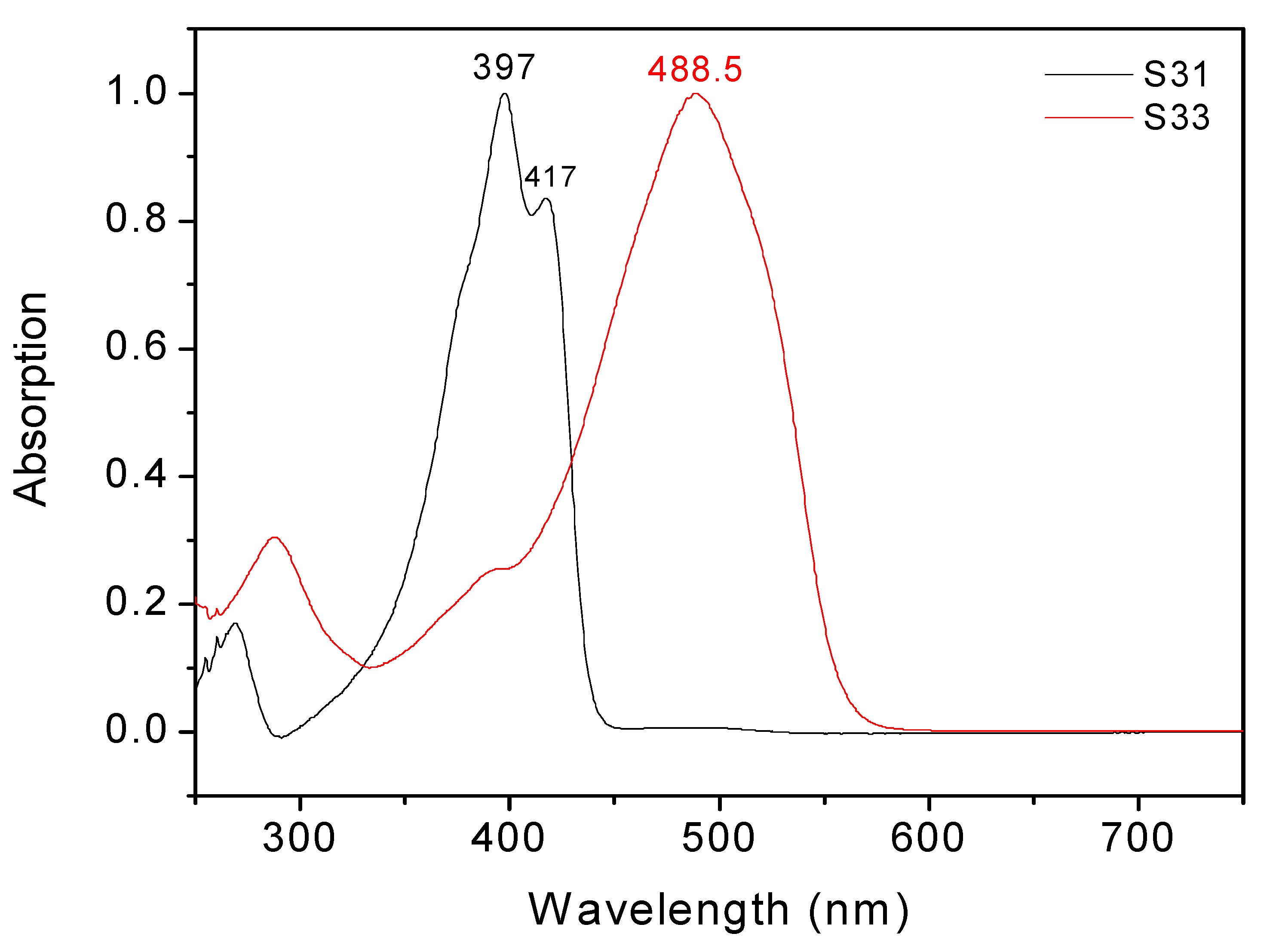
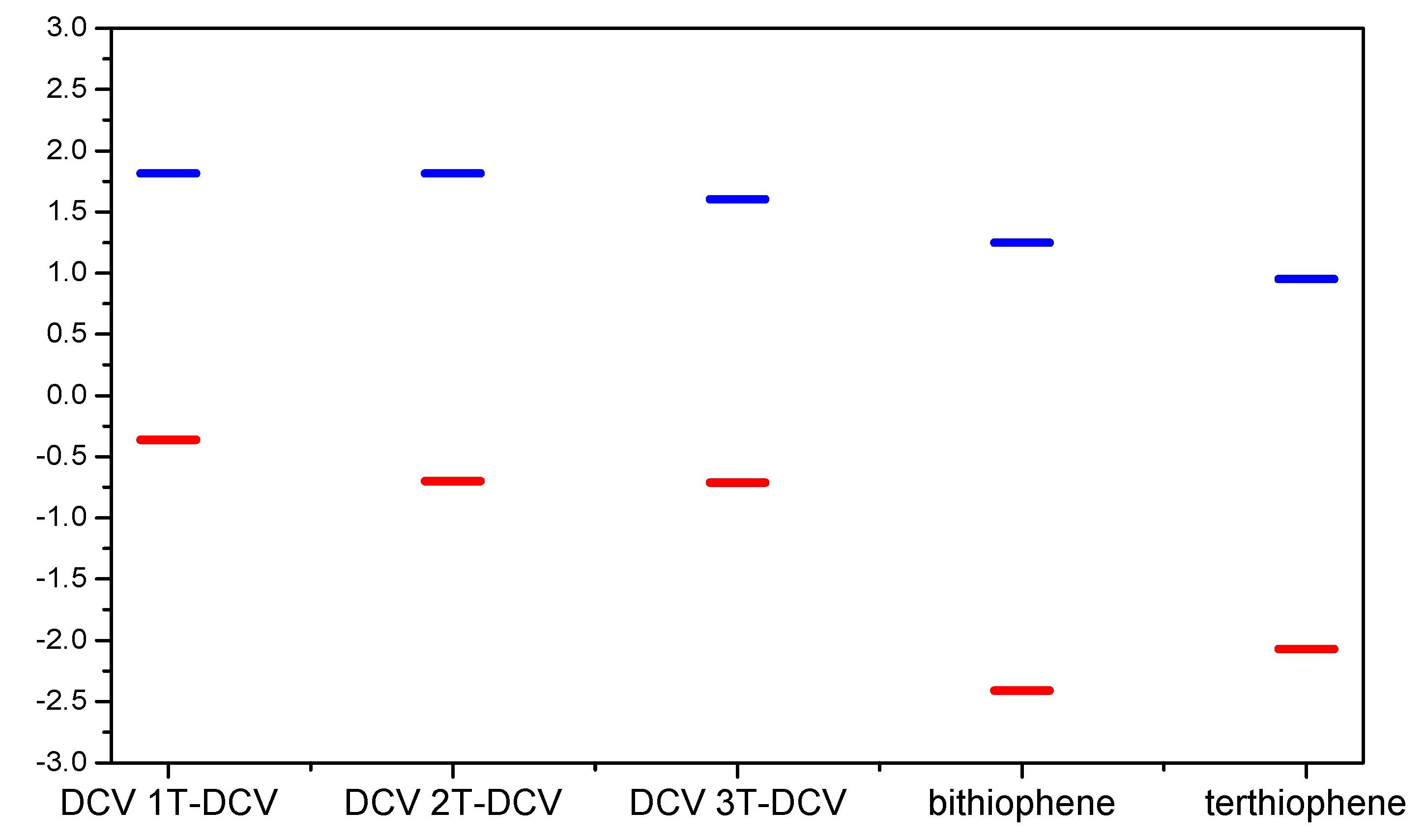


| Compound | Ep, ox | Ep, red | ΔEp | λmax in CH2Cl2 |
|---|---|---|---|---|
| DCV-T-DCV | not measurable | −0.358 (rev) −0.70 (rev) | - | 415 |
| DCV-2T-DCV | 1.83 | −0.796 | 2.556 | 451 |
| DCV-3T-DCV | 1.488 | −0.818 −0.972 | 2.296 | 502 |
| bithiophene | 1.25 | −2.41 −3.10 | 3.66 | 303.5 |
| terthiophene | 0.95 | −2.07 −2.47 | 3.02 | 353 |
| Tetracyanoethylene | Not measurable | −0.54 | ||
| TCV 2T-TCV b | 2.12 | −0.08 (rev) −0.37 (rev) −0.81 −1.47 | 2.20 | 504.0 (in acetonitrile) |
| cm−1 | ||
|---|---|---|
| Compound | IR | Raman |
| DCV-T-DCV | 2229.34 | 2232.2 |
| DCV-2T-DCV | 2223.56 | 2225.7 |
| DCV-3T-DCV | 2222.60 | 2221.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bader, M.M.; Pham, P.-T.T.; Busaili, J.A.; Alrifai, S.M.; Younas, S.H.; Elandaloussi, E.H. Photophysical, Electrochemical, Density Functional Theory, and Spectroscopic Study of Some Oligothiophenes. Optics 2025, 6, 56. https://doi.org/10.3390/opt6040056
Bader MM, Pham P-TT, Busaili JA, Alrifai SM, Younas SH, Elandaloussi EH. Photophysical, Electrochemical, Density Functional Theory, and Spectroscopic Study of Some Oligothiophenes. Optics. 2025; 6(4):56. https://doi.org/10.3390/opt6040056
Chicago/Turabian StyleBader, Mamoun M., Phuong-Truc T. Pham, Juri A. Busaili, Samar M. Alrifai, Sarah H. Younas, and El Hadj Elandaloussi. 2025. "Photophysical, Electrochemical, Density Functional Theory, and Spectroscopic Study of Some Oligothiophenes" Optics 6, no. 4: 56. https://doi.org/10.3390/opt6040056
APA StyleBader, M. M., Pham, P.-T. T., Busaili, J. A., Alrifai, S. M., Younas, S. H., & Elandaloussi, E. H. (2025). Photophysical, Electrochemical, Density Functional Theory, and Spectroscopic Study of Some Oligothiophenes. Optics, 6(4), 56. https://doi.org/10.3390/opt6040056









