A Pilot Randomised Controlled Trial Evaluating a Regenerative Epithelial Suspension for Medium-Size Partial-Thickness Burns in Children: The BRACS Trial
Abstract
1. Introduction
2. Materials and Methods
Statistical Methods
3. Results
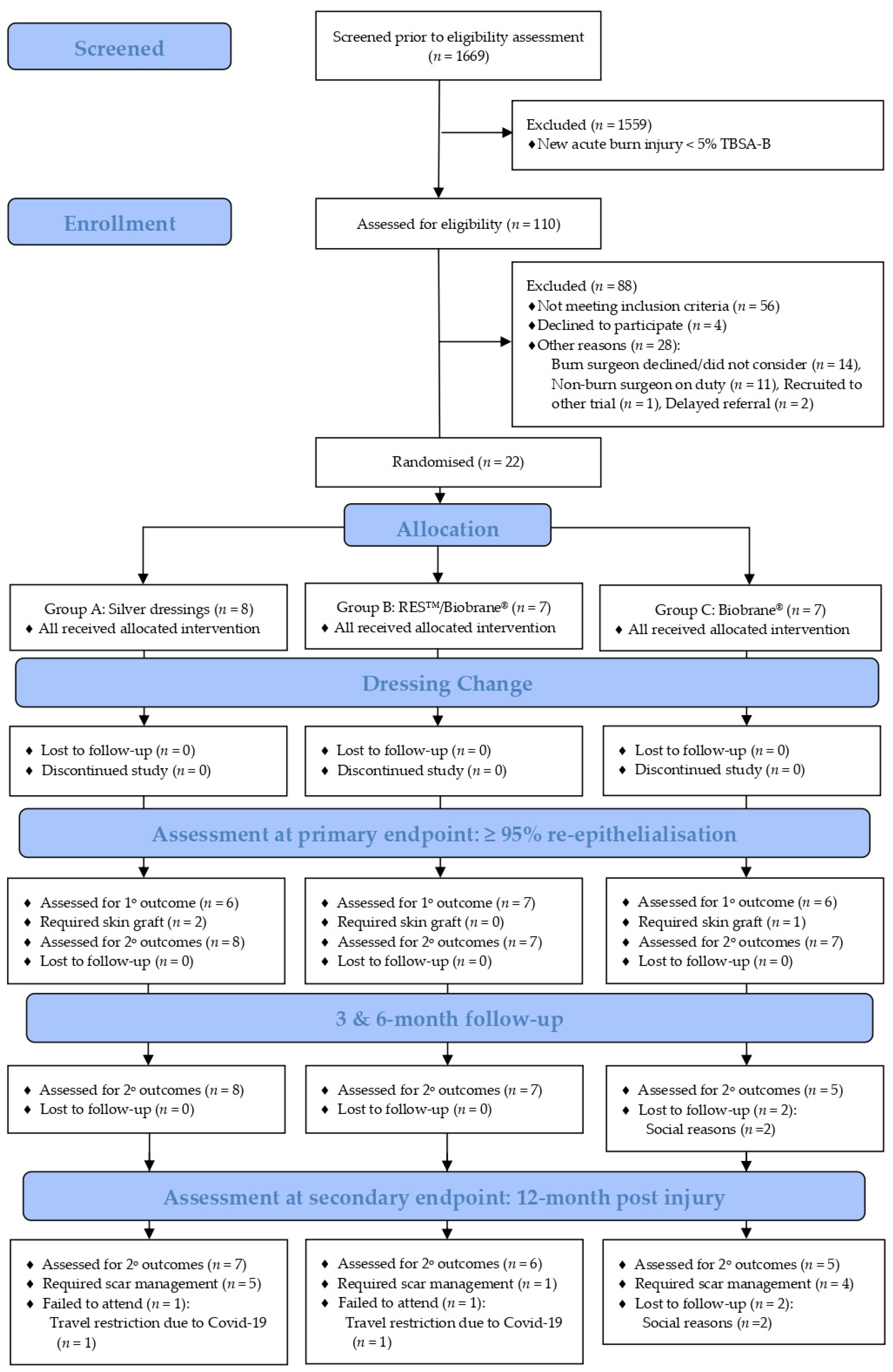
3.1. Participant Recruitment
3.2. Demographic Data
3.3. Primary Outcome: Time to Re-Epithelialisation
3.4. Secondary Outcomes
3.4.1. Pain
3.4.2. Itch Intensity
3.4.3. Ease of Dressing Application
3.4.4. Intervention Fidelity
3.4.5. Treatment Satisfaction
3.4.6. Scar Severity
| Outcome | Silver Dressings | RES™/Biobrane® | Biobrane® | |||
|---|---|---|---|---|---|---|
| Median (IQR) | Mean (SD) | Median (IQR) | Mean (SD) | Median (IQR) | Mean (SD) | |
| Scar Characteristics | ||||||
| Thickness (n, mm) | 5 | 4 | 4 | |||
| 1.32 (0.85–2.92) | 1.77 (1.22) | 1.08 (0.97–1.49) | 1.18 (0.29) | 1.59 (1.35–2.94) | 1.96 (0.94) | |
| Colour (n) | 4 | 4 | 5 | |||
| L*Scar | 32.10 (15.61–40.90) | 29.54 (14.06) | 35.78 (15.80–41.26) | 30.95 (14.46) | 39.13 (30.05–45.00) | 38.06 (8.18) |
| L*Normal | 29.84 (26.60–40.89) | 32.44 (8.12) | 32.18 (16.53–46.78) | 31.53 (16.10) | 43.73 (33.80–46.58) | 41.37 (7.26) |
| a*Scar | 14.23 (9.91–18.70) | 14.28 (4.62) | 13.35 (10.05–16.84) | 13.41 (3.55) | 15.12 (13.07–17.57) | 15.25 (2.34) |
| a*Normal | 13.53 (12.57–14.38) | 13.49 (0.98) | 13.24 (11.17–15.76) | 13.24 (2.57) | 11.23 (10.16–15.88) | 12.43 (3.40) |
| b*Scar | 13.94 (10.88–16.85) | 13.89 (3.09) | 14.29 (8.88–16.87) | 13.35 (4.30) | 9.35 (3.28–18.11) | 10.24 (7.84) |
| b*Normal | 16.87 (15.02–18.27) | 16.72 (1.73) | 15.61 (10.91–18.26) | 14.93 (3.95) | 14.85 (12.77–18.94) | 15.52 (3.33) |
| Clinician Scar Severity Report OSAS(n) ‡ | 5 | 4 | 4 | |||
| Overall Opinion | 2.00 (2.00–5.50) | 3.40 (3.13) | 2.00 (1.25–2.75) | 2.00 (0.82) | 2.50 (2.00–6.75) | 3.75 (2.87) |
| Treatment Satisfaction | ||||||
| Doctor (n ¥) | 7 | 9 | 7 | |||
| 8.00 (7.00–9.00) | 7.57 (2.23) | 9.00 (8.50–9.50) | 9.00 (0.71) | 5.00 (4.00–15.00) | 5.57 (1.90) | |
| Nurse (n ¥) | 0 | 1 | 0 | |||
| N/A | 9.00(9.00–9.00) | N/A | ||||
| Occupational Therapist (n ¥) | 1 | 1 | 3 | |||
| 8.00 (8.00–8.00) | 8.00 (_) | 7.00 (7.00–7.00) | 7.00 (_) | 8.00 (8.00–_) | 8.33 (0.58) | |
3.4.7. Health-Related Quality of Life
| Outcome | Silver Dressings | RES™/Biobrane® | Biobrane® | |||
|---|---|---|---|---|---|---|
| Median (IQR) | Mean (SD) | Median (IQR) | Mean (SD) | Median (IQR) | Mean (SD) | |
| POSAS † (n) | 7 | 5 | 4 | |||
| Overall Opinion | 3.00 (1.00–6.00) | 3.86 (2.97) | 2.00 (1.00–3.50) | 2.20 (1.64) | 3.50 (3.00–5.50) | 4.00(1.41) |
| POSAS Score | 14.00 (6.00–27.00) | 18.00 (14.06) | 9.00 (4.00–13.00) | 8.60 (5.18) | 22.50 (14.75–30.25) | 22.50(8.27) |
| BBSIP † (n) | 7 | 5 | 4 | |||
| Overall impact of burns | 1.13 (1.00–3.00) | 1.71 (0.95) | 1.00 (1.00–1.00) | 1.00 (0.00) | 1.38 (1.09–1.56) | 1.34 (0.26) |
| Treatment Satisfaction ≠ (n) | 10 | 8 | 6 | |||
| Parent/Guardian | 10.00 (8.25–10.00) | 9.10 (1.66) | 10.00 (10.00–10.00) | 10.00 (0.00) | 9.50 (9.00–10.00) | 9.50 (0.55) |
3.4.8. Health Resource Utilisation
3.4.9. Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Registration
Protocol
Abbreviations
| ACHD | Autologous Cell Harvesting Device |
| ASCS | Autologous Skin Cell Suspension |
| AUD | Australian Dollar |
| BBSIP | Brisbane Burn Scar Impact Profile |
| BRACS | Biobrane®, RECELL® Autologous Skin Cell Suspension and Silver Dressings |
| CHU9D | Child Health Utility 9D |
| COD | Change of Dressing |
| FLACC | Face, Legs, Activity, Cry, Consolability |
| FPS-R | Faces Pain Scale-Revised |
| HREC | Human Research Ethics Committee |
| IQR | Interquartile Range |
| MAX | Maximum |
| MIN | Minimum |
| NRS-I | Numeric Rating Scale—Itch |
| NRS-I Proxy | Numeric Rating Scale—Itch Proxy |
| NRS-P | Numeric Rating Scale—Pain |
| NRS-P Proxy | Numeric Rating Scale—Pain Proxy |
| OSAS | Observer Scar Assessment Scale |
| POSAS | Patient and Observer Scar Assessment Scale |
| QUT | Queensland University of Technology |
| REDCap | Research Electronic Data Capture |
| RES™ | Regenerative Epidermal Suspension |
| SSA | Site Specific Approval |
| TBSA-B | Burn Total Body Surface Area |
| TTRE | Time to re-epithelialisation |
References
- Rayner, R.; Prentice, J. Paediatric Burns: A Brief Global Review. Wound Pract. Res. J. Aust. Wound Manag. Assoc. 2011, 19, 39–46. [Google Scholar]
- Stockton, K.A.; Harvey, J.; Kimble, R.M. A prospective observational study investigating all children presenting to a specialty paediatric burns centre. Burns 2015, 41, 476–483. [Google Scholar] [CrossRef]
- Albertyn, R.; Bickler, S.W.; Rode, H. Paediatric burn injuries in Sub Saharan Africa—An overview. Burns 2006, 32, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.J.; Kimble, R.M.; Gramotnev, G.; Rodger, S.; Cuttle, L. Predictors of re-epithelialization in pediatric burn. Burns 2014, 40, 751–758. [Google Scholar] [CrossRef]
- Deitch, E.A.; Wheelahan, T.M.; Rose, M.P.; Clothier, J.; Cotter, J. Hypertrophic burn scars: Analysis of variables. J. Trauma 1983, 23, 895–898. [Google Scholar] [CrossRef] [PubMed]
- Lonie, S.; Baker, P.; Teixeira, R.P. Healing time and incidence of hypertrophic scarring in paediatric scalds. Burns 2017, 43, 509–513. [Google Scholar] [CrossRef]
- Cubison, T.C.; Pape, S.A.; Parkhouse, N. Evidence for the link between healing time and the development of hypertrophic scars (HTS) in paediatric burns due to scald injury. Burns 2006, 32, 992–999. [Google Scholar] [CrossRef] [PubMed]
- Chipp, E.; Charles, L.; Thomas, C.; Whiting, K.; Moiemen, N.; Wilson, Y. A prospective study of time to healing and hypertrophic scarring in paediatric burns: Every day counts. Burn. Trauma 2017, 5, 3. [Google Scholar] [CrossRef]
- Perkins, M.; Wood, F.; Griffin, B.; Gus, E.; Carney, B.; Teague, W.; Tracy, L. Variation in burn wound management approaches for paediatric burn patients in Australia and New Zealand. ANZ J. Surg. 2022, 92, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Gee Kee, E.L.; Kimble, R.M.; Cuttle, L.; Khan, A.; Stockton, K.A. Randomized controlled trial of three burns dressings for partial thickness burns in children. Burns 2015, 41, 946–955. [Google Scholar] [CrossRef]
- Karlsson, M.; Elmasry, M.; Steinvall, I.; Sjoberg, F.; Olofsson, P.; Thorfinn, J. Superiority of silver-foam over porcine xenograft dressings for treatment of scalds in children: A prospective randomised controlled trial. Burns 2019, 45, 1401–1409. [Google Scholar] [CrossRef]
- Hyland, E.J.; D’Cruz, R.; Menon, S.; Harvey, J.G.; La Hei, E.; Lawrence, T.; Waddell, K.; Nash, M.; Holland, A.J. BiobraneTM versus acticoatTM for the treatment of mid-dermal pediatric burns: A prospective randomized controlled pilot study. Int. J. Burn. Trauma 2018, 8, 63–67. [Google Scholar]
- Gotschall, C.; Morrison, M.; Eichelberger, M. Prospective, Randomized Study of the Efficacy of Mepitel*on Children With Partial-Thickness Scalds. J. Burn. Care Rehabil. 1998, 19, 279–283. [Google Scholar] [CrossRef]
- Bairagi, A.; Griffin, B.; Tyack, Z.; Vagenas, D.; McPhail, S.M.; Kimble, R. Comparative effectiveness of Biobrane(R), RECELL(R) Autologous skin Cell suspension and Silver dressings in partial thickness paediatric burns: BRACS randomised trial protocol. Burn. Trauma 2019, 7, 33. [Google Scholar]
- Bairagi, A.; Griffin, B.; Banani, T.; McPhail, S.M.; Kimble, R.; Tyack, Z. A systematic review and meta-analysis of randomized trials evaluating the efficacy of autologous skin cell suspensions for re-epithelialization of acute partial thickness burn injuries and split-thickness skin graft donor sites. Burns 2021, 47, 1225–1240. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Pek, C.H.; Por, Y.C.; Lim, G.J.S. Biobrane dressing for paediatric burns in Singapore: A retrospective review. Singap. Med. J. 2018, 59, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Wood, F.; Martin, L.; Lewis, D.; Rawlins, J.; McWilliams, T.; Burrows, S.; Rea, S. A prospective randomised clinical pilot study to compare the effectiveness of Biobrane(R) synthetic wound dressing, with or without autologous cell suspension, to the local standard treatment regimen in paediatric scald injuries. Burns 2012, 38, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Campanella, S.D.; Rapley, P.; Ramelet, A.S. A randomised controlled pilot study comparing Mepitel((R)) and SurfaSoft((R)) on paediatric donor sites treated with Recell((R)). Burns 2011, 37, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.S. Use of Biobrane in pediatric scald burf Biobrane in 106 children. Burns 1998, 24, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, C.; Peter, S.D.S.; Lacey, S.; Beery, M.; Ward-Smith, P.; Sharp, R.J.; Ostlie, D.J. Biobrane versus duoderm for the treatment of intermediate thickness burns in children: A prospective, randomized trial. Burns 2005, 31, 890–893. [Google Scholar]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. BMJ 2016, 355, i5239. [Google Scholar] [CrossRef]
- Management NIoTaI. NSW Trauma App Analysis Report 21 August–8 November 2015 NSW, Australia: Institute of Trauma and Injury Management. 2015. Available online: https://aci.health.nsw.gov.au/__data/assets/pdf_file/0007/349063/NSW_Trauma_App_Analysis_Report_August_2015_-_August_2016.pdf (accessed on 27 January 2016).
- Management NIoTaI. NSW Trauma App Analysis Report August 2015–August 2016 NSW, Australia: NSW Institute of Trauma and Injury Management. 2016. Available online: https://aci.health.nsw.gov.au/__data/assets/pdf_file/0007/349063/NSW_Trauma_App_Analysis_Report_August_2015_-_August_2016.pdf (accessed on 22 February 2017).
- Fontaine, M.; Ravat, F.; Latarjet, J. The e-burn application—A simple mobile tool to assess TBSA of burn wounds. Burns 2018, 44, 237–238. [Google Scholar] [CrossRef] [PubMed]
- Chong, H.P.; Quinn, L.; Jeeves, A.; Cooksey, R.; Lodge, M.; Carney, B.; Molony, D. A comparison study of methods for estimation of a burn surface area: Lund and Browder, e-burn and Mersey Burns. Burns 2020, 46, 483–489. [Google Scholar] [CrossRef]
- Holland, A.J.; Martin, H.C.; Cass, D.T. Laser Doppler imaging prediction of burn wound outcome in children. Burns 2002, 28, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Monstrey, S.; Hoeksema, H.; Baker, R.; Jeng, J.; Spence, R.; Wilson, D.; Pape, S. Burn wound healing time assessed by laser Doppler imaging. Part 2: Validation of a dedicated colour code for image interpretation. Burns 2011, 37, 249–256. [Google Scholar] [CrossRef]
- Pape, S.; Baker, R.; Wilson, D.; Hoeksema, H.; Jeng, J.; Spence, R.; Monstrey, S. Burn wound healing time assessed by laser Doppler imaging (LDI). Part 1: Derivation of a dedicated colour code for image interpretation. Burns 2012, 38, 187–194. [Google Scholar] [CrossRef]
- Merkel, S.; Voepel-Lewis, T.; Shayevitz, J.; Malviya, S. FLACC Pain Assessment Tool. Anesthesiology 1994, 81, A1360. [Google Scholar] [CrossRef]
- Page, M.G.; Katz, J.; Stinson, J.; Isaac, L.; Martin-Pichora, A.L.; Campbell, F. Validation of the numerical rating scale for pain intensity and unpleasantness in pediatric acute postoperative pain: Sensitivity to change over time. J. Pain 2012, 13, 359–369. [Google Scholar] [CrossRef]
- Huguet, A.; Stinson, J.N.; McGrath, P.J. Measurement of self-reported pain intensity in children and adolescents. J. Psychosom. Res. 2010, 68, 329–336. [Google Scholar] [CrossRef] [PubMed]
- McLaren, K. XIII—The Development of the CIE 1976 (L* a* b*) Uniform Colour Space and Colour-difference Formula. J. Soc. Dye. Colour. 2008, 92, 338–341. [Google Scholar] [CrossRef]
- van der Wal, M.; Bloemen, M.; Verhaegen, P.; Tuinebreijer, W.; de Vet, H.; van Zuijlen, P.; Middelkoop, E. Objective color measurements: Clinimetric performance of three devices on normal skin and scar tissue. J. Burn. Care Res. 2013, 34, e187–e194. [Google Scholar] [CrossRef] [PubMed]
- Nieuwendijk, S.M.P.; de Korte, I.J.; Pursad, M.M.; van Dijk, M.; Rode, H. Post burn pruritus in pediatric burn patients. Burns 2018, 44, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Tyack, Z.; Ziviani, J.; Kimble, R.; Plaza, A.; Jones, A.; Cuttle, L.; Simons, M. Measuring the impact of burn scarring on health-related quality of life: Development and preliminary content validation of the Brisbane Burn Scar Impact Profile (BBSIP) for children and adults. Burns 2015, 41, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Tyack, Z.; Kimble, R.; McPhail, S.; Plaza, A.; Simons, M. Psychometric properties of the Brisbane Burn Scar Impact Profile in adults with burn scars. PLoS ONE 2017, 12, e0184452. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Kimble, R.; McPhail, S.; Tyack, Z. The Brisbane Burn Scar Impact Profile (child and young person version) for measuring health-related quality of life in children with burn scars: A longitudinal cohort study of reliability, validity and responsiveness. Burns 2019, 45, 1537–1552. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Kimble, R.; McPhail, S.; Tyack, Z. The longitudinal validity, reproducibility and responsiveness of the Brisbane Burn Scar Impact Profile (caregiver report for young children version) for measuring health-related quality of life in children with burn scars. Burns 2019, 45, 1792–1809. [Google Scholar] [CrossRef]
- Tyack, Z.; Simons, M.; Kimble, R. Brisbane Burn Scar Impact Profile for Caregivers of Children Aged Less Than 8 Years Version 1.0. Queensland: The State of Queensland (Queensland Health). 2013. Available online: http://www.ccbtr.com.au/brisbane-burn-scar-impact-profile/ (accessed on 20 September 2017).
- Tyack, Z.; Simons, M.; Kimble, R. Brisbane Burn Scar Impact Profile for Caregivers of Children 8 Years and Older. Version 1.0. Queensland: The State of Queensland (Queensland Health). 2013. Available online: http://www.ccbtr.com.au/brisbane-burn-scar-impact-profile/ (accessed on 20 September 2017).
- Tyack, Z.; Simons, M.; Kimble, R. Brisbane Burn Scar Impact Profile for Children 8 to 18 Years. Version 1.0. Queensland: The State of Queensland (Queensland Health). 2013. Available online: http://www.ccbtr.com.au/brisbane-burn-scar-impact-profile/ (accessed on 20 September 2017).
- Stevens, K.; Ratcliffe, J. Measuring and valuing health benefits for economic evaluation in adolescence: An assessment of the practicality and validity of the child health utility 9D in the Australian adolescent population. Value Health 2012, 15, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Stevens, K.J. Working with children to develop dimensions for a preference-based, generic, pediatric, health-related quality-of-life measure. Qual. Health Res. 2010, 20, 340–351. [Google Scholar] [CrossRef]
- Stevens, K. Developing a descriptive system for a new preference-based measure of health-related quality of life for children. Qual. Life Res. 2009, 18, 1105–1113. [Google Scholar] [CrossRef]
- Stevens, K. Assessing the performance of a new generic measure of health-related quality of life for children and refining it for use in health state valuation. Appl. Health Econ. Health Policy 2011, 9, 157–169. [Google Scholar] [CrossRef]
- Stevens, K. The Child Health Utility 9D(CHU9D)—A New Paediatric Preference Based Measure of Health Related Quality of Life. PRO Newsl. 2010, 43, 11–12. [Google Scholar]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Moiemen, N.; Mathers, J.; Jones, L.; Bishop, J.; Kinghorn, P.; Monahan, M.; Calvert, M.; Slinn, G.; Gardiner, F.; Bamford, A.; et al. Pressure garment to prevent abnormal scarring after burn injury in adults and children: The PEGASUS feasibility RCT and mixed-methods study. Health Technol. Assess. 2018, 22, 1–162. [Google Scholar] [CrossRef] [PubMed]
- Nunnally, J.C. Psychometric Theory, 3rd ed.; Bernstein, I.H., Ed.; McGraw-Hill: New York, NY, USA, 1994. [Google Scholar]
- Gee Kee, E.; Stockton, K.; Kimble, R.M.; Cuttle, L.; McPhail, S.M. Cost-effectiveness of silver dressings for paediatric partial thickness burns: An economic evaluation from a randomized controlled trial. Burns 2017, 43, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Frear, C.C.; Cuttle, L.; McPhail, S.M.; Chatfield, M.D.; Kimble, R.M.; Griffin, B.R. Randomized clinical trial of negative pressure wound therapy as an adjunctive treatment for small-area thermal burns in children. Br. J. Surg. 2020, 107, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Schreier, M. Qualitative Content Analysis; Sage: Thousand Oaks, CA, USA, 2014; pp. 170–183. [Google Scholar]
- Hsieh, H.F.; Shannon, S.E. Three approaches to qualitative content analysis. Qual. Health Res. 2005, 15, 1277–1288. [Google Scholar] [CrossRef]
- Bengtsson, M. How to plan and perform a qualitative study using content analysis. NursingPlus Open. 2016, 2, 8–14. [Google Scholar] [CrossRef]
- Elo, S.; Kyngas, H. The qualitative content analysis process. J. Adv. Nurs. 2008, 62, 107–115. [Google Scholar] [CrossRef]
- Willits, I.; Cole, H. The ReCell Spray-On Skin System for Treating Skin Loss, Scarring and Depigmentation after Burn Injury; NICE: Newcastle, UK, 2020. [Google Scholar]
- Griffin, B.R.; Frear, C.C.; Babl, F.; Oakley, E.; Kimble, R.M. Cool Running Water First Aid Decreases Skin Grafting Requirements in Pediatric Burns: A Cohort Study of Two Thousand Four Hundred Ninety-five Children. Ann. Emerg. Med. 2020, 75, 75–85. [Google Scholar] [CrossRef]
- Cuttle, L.; Kempf, M.; Kravchuk, O.; Phillips, G.E.; Mill, J.; Wang, X.-Q.; Kimble, R.M. The optimal temperature of first aid treatment for partial thickness burn injuries. Wound Repair Regen. 2008, 16, 626–634. [Google Scholar] [CrossRef]
- Piipponen, M.; Li, D.; Landen, N.X. The Immune Functions of Keratinocytes in Skin Wound Healing. Int. J. Mol. Sci. 2020, 21, 8790. [Google Scholar] [CrossRef] [PubMed]
- Wood, F.M.; Giles, N.; Stevenson, A.; Rea, S.; Fear, M. Characterisation of the cell suspension harvested from the dermal epidermal junction using a ReCell(R) kit. Burns 2012, 38, 44–51. [Google Scholar] [CrossRef] [PubMed]
- ACTRN12620000227998; The Effect of CUTICERIN® with or without a Regenerative Epithelial Suspension (RES™), on Pigmentation of Donor Sites in Children: The Pilot Randomised DRESSIng Trial. ANZCTR: Camperdown, NSW, Australia, 2020.
- NCT03626701. RES® Prepared with RECELL® Compared to Standard of Care Dressings of Partial-Thickness Burns in Ages 1–16 Years. 2018. Available online: https://ClinicalTrials.gov/show/NCT03626701 (accessed on 15 February 2023).
- Griffin, B.; Bairagi, A.; Jones, L.; Dettrick, Z.; Holbert, M.; Kimble, R. Early non-excisional debridement of paediatric burns under general anaesthesia reduces time to re-epithelialisation and risk of skin graft. Sci. Rep. 2021, 11, 23753. [Google Scholar] [CrossRef] [PubMed]
- Bairagi, A.; Frear, C.; Sweeting, C.; Kimble, R.; Griffin, B. A pilot study comparing two burn wound stereophotogrammetry systems in a paediatric population. Burns 2022, 48, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Bairagi, A.; Griffin, B.; Banani, T.; McPhail, S.M.; Kimble, R.; Tyack, Z. Letter to the Editor and Author Response for “A systematic review and meta-analysis of randomized trials evaluating the efficacy of autologous skin cell suspensions for re-epithelialization of acute partial thickness burn injuries and split-thickness skin graft donor sites” by Bairagi; et al. Burns 2022, 48, 464–467. [Google Scholar]
- Bairagi, A.; Griffin, B.; McPhail, S.; Kimble, R.; Tyack, Z. Efficacy of Autologous Skin Cell Suspensions for Re-Epithelialisation of Acute Partial Thickness Burn Injuries and Split-Thickness Autograft Donor Sites: A Systematic Review and Meta-Analysis: PROSPERO International Prospective Register of Systematic Reviews. 2019. Available online: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=133171 (accessed on 15 February 2023).
- ACTRN12618000245291. The Biobrane®, RECELL® Autologous Cell Suspension and Silver Dressings Trial (BRACS Trial): A Three Arm Prospective Randomised Controlled Trial Comparing a Biological Dressing (Biobrane®) with Biobrane® and RECELL® and with Standard Silver Dressings in Children with a Significant Superficial Partial to Mid Dermal Thickness Burn: ANZCTR: Camperdown, NSW, Australia. 2018. Available online: http://www.anzctr.org.au/ACTRN12618000245291.aspx (accessed on 15 February 2018).
| Parameter (n) | Silver Dressings ‡ (8) | RES™/Biobrane® (7) | Biobrane® (7) |
|---|---|---|---|
| Age (years) * | 1.50 (1.00–2.75) | 1.00 (1.00–2.00) | 2.00 (2.00–4.00) |
| Age (years, Min–Max) | 0.00–14.00 | 1.00–14.00 | 1.00–9.00 |
| Gender (n) | M4: F4 | M5: F2 | M1: F6 |
| Ethnicity (n) | |||
| European | 3 | 2 | 3 |
| Aboriginal or Torres Strait Islander | 1 | 1 | 2 |
| Asian | 2 | 2 | 1 |
| Middle Eastern | - | - | - |
| African | - | 1 | - |
| Other | 2 | 1 | 1 |
| Fitzpatrick Skin Type (n) | |||
| I | 1 | - | - |
| II | - | 1 | - |
| III | 2 | 2 | - |
| IV | 1 | 4 | 3 |
| V | 4 | - | 4 |
| VI | - | - | - |
| Co-morbidities (n) | |||
| Allergies | 1 | - | - |
| Skin disorders | 2 | - | 1 |
| Physical disability | - | - | - |
| Learning disability | 1 | 1 | 1 |
| Mental health illness | - | 1 | 1 |
| PTSS | 1 | - | - |
| Visual Impairment | - | - | - |
| Other | 2 | 1 | 3 |
| Language (n) | |||
| English | 4 | 3 | 6 |
| Multi-lingual, including English | 4 | 4 | 1 |
| Parameter (n) | Silver Dressings ‡ (8) | RES™/Biobrane® (7) | Biobrane® (7) |
|---|---|---|---|
| TBSA-B (%) | |||
| Median (IQR) | 11.5 (7.0–15.75) | 6.00 (5.0–20.0) | 10.0 (8.0–15.0) |
| Min–Max | 5–20 | 5–33 | 7–27 |
| Burn Depth (n) | |||
| Superficial thickness | 2 | 1 | 1 |
| Superficial partial thickness | 8 | 7 | 7 |
| Mid-Dermal partial thickness | 4 | 6 | 4 |
| Deep partial thickness | 1 | 1 | 2 |
| Injury mechanism (n) | |||
| Scald | 7 | 6 | 5 |
| Flame | - | 1 | 2 |
| Radiant Heat | 1 | - | - |
| Anatomical Location (n) | |||
| Head/Neck | 8 | 6 | 6 |
| Trunk | 8 | 7 | 7 |
| Upper limb | 5 | 5 | 7 |
| Hand | 3 | 2 | 2 |
| Lower limb | 1 | 3 | 4 |
| Foot | 8 | 2 | 7 |
| Appropriate First Aid (n) | 8 | 6 | 7 |
| Burn age at initial presentation (hours) * | 8.50 (2.25–21.00) | 9.00 (4.00–12.00) | 4.00 (3.00–6.00) |
| Burn age at initial dressing application (hours) * | 13.00 (6.25–24.75) | 14.00 (10.00–20.00) | 6.00 (4.00–18.00) |
| Outcome (n) | Silver Dressings ‡ (8) | RES™/Biobrane® (7) | Biobrane® (7) |
|---|---|---|---|
| Time to Re-epithelialisation (days) | |||
| Mean TTRE (SD) | 15.1 (±8.87) | 11.1 (±3.28) | 15.1 (±6.04) |
| Median TTRE (95%CI) | 12 (3.7–20.3) | 12 (5.6–18.4) | 14 (6.3–21.7) |
| Number of dressing applications (n) | |||
| Under general anaesthesia * | 3 (1.00–4.75) | 2 (1.00–3.00) | 3 (2.00–4.00) |
| Awake * | 2 (2.00–4.00) | 4 (3.00–4.00) | 2 (2.00–4.00) |
| Total * | 6 (3.00–7.75) | 6 (5.00–6.00) | 7 (5.00–8.00) |
| Pain * | |||
| Initial dressing application | |||
| Median pre-intervention FLACC score | 2.00 (0.00–2.75) | 0.00 (0.00–3.25) | 0.00 (0.00–0.00) |
| Median post-intervention FLACC score | 0.00 (0.00–0.00) | 0.00 (0.00–4.00) | 0.00 (0.00–2.50) |
| Median difference FLACC score | −2.00 | 0.00 | 0.00 |
| Median pre-intervention NRS-P Proxy score | 5.50 (3.00–9.50) | 7.00 (5.00–10.00) | 4.00 (3.00–4.75) |
| Median post intervention NRS-P Proxy score | 2.00 (1.00–4.00) | 4.00 (0.00–8.50) | 2.50 (0.00–10.00) |
| Median difference NRS-P Proxy score | −3.50 | −3.00 | −1.5 |
| Dressing Change #1 | |||
| Median pre-intervention FLACC score | 0.00 (0.00–1.75) | 0.00 (0.00–2.00) | 0.00 (0.00–0.00) |
| Median post-intervention FLACC score | 0.00 (0.00–3.50) | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) |
| Median difference FLACC score | 0.00 | 0.00 | 0.00 |
| Median pre-intervention NRS-P Proxy score | 1.00 (0.00–6.00) | 2.50 (0.00–5.50) | 1.00 (0.50–4.00) |
| Median post intervention NRS-P Proxy score | 4.00 (0.00–5.00) | 2.00 (1.75–3.50) | 1.00 (0.00–2.00) |
| Median difference NRS-P Proxy score | +3.00 | −0.50 | 0.00 |
| Itch * | |||
| Median NRS-I Proxy score | 6.00 (3.00–8.00) | 4.00 (2.25–7.00) | 4.00 (2.00–7.00) |
| Ease of Dressing Application * | |||
| Initial dressing application | |||
| Doctors (n = 48) | |||
| Application ease | 5.00 (2.00–6.00) | 6.50 (2.75–7.00) | 5.00 (3.00–7.00) |
| Dressing conformability | 3.00 (2.00–7.00) | 3.50 (2.00–7.50) | 4.50 (2.7–6.00) |
| Duration(minutes) | <60 | >60 | >60 |
| Nurses (n = 26) | |||
| Application ease | 2.00 (1.00–4.00) | 2.50 (1.00–8.75) | 1.00 (0.00–2.00) |
| Dressing conformability | 3.00 (1.00–3.50) | 2.00 (1.00–5.00) | 1.00 (0.50–1.50) |
| Duration(minutes) | <60 | <60 | <10 |
| Dressing Change | |||
| Doctors (n = 43) | |||
| Application ease | 7.00 (4.25–7.75) | 4.00 (2.00–8.00) | 3.00 (2.00–7.00) |
| Dressing conformability | 7.00 (5.00–7.00) | 2.00 (2.00–7.00) | 2.00 (2.00–7.00) |
| Duration(minutes) | <60 | <60 | 30>, <60 |
| Nurses (n = 101) | |||
| Application ease | 2.00 (1.00–3.00) | 2.00 (1.00–4.00) | 1.50 (0.00–3.00) |
| Dressing conformability | 2.00 (1.00–3.25) | 2.00 (0.25–4.25) | 2.00 (1.00–3.00) |
| Duration (minutes) | <60 | <60 | 30>, <60 |
| Intervention Fidelity * | |||
| Initial dressing application (%) | |||
| QV wash | 100 | 100 | 100 |
| Intervention | 87.50 | 83.61 | 97.62 |
| Dressing Change (%) | |||
| QV wash | 89.47 | 72.22 | 76.19 |
| Intervention | 94.08 | 90.79 | 100.00 |
| Treatment Satisfaction * | |||
| Staff | 9.00 (8.75–10.00) | 9.00 (8.00–9.75) | 8.00 (2.25–9.00) |
| Parent/Guardian | 10.00 (9.00–10.00) | 10.00 (10.00–10.00) | 10.00 (9.00–10.00) |
| Type of Adverse Event | Silver Dressings ‡ | RES™/Biobrane® | Biobrane® |
| (n) | (13) | (8) | (10) |
| Nil | 3 | 2 | 1 |
| Wound infection | 2 | 1 | 3 |
| Allergic reaction | 1 | 0 | 0 |
| Sepsis | 1 | 0 | 0 |
| Unplanned ICU admission | 2 | 1 | 2 |
| Burn depth progression | 2 | 1 | 3 |
| Required split-thickness skin graft | 2 | 0 | 1 |
| Other | |||
| Fever associated with teething | 0 | 1 | 0 |
| Unplanned ward admission | 0 | 1 | 0 |
| 12-h delay to theatre | 1 | 0 | 0 |
| Clavein–Dindo Grade of Complication | Silver Dressings ‡ | RES™/Biobrane® | Biobrane® |
| (n) | (10) | (6) | (9) |
| Grade I | 2 | 5 | 1 |
| Grade II | 0 | 0 | 2 |
| Grade IIIa | 0 | 0 | 0 |
| Grade IIIb | 4 | 0 | 3 |
| Grade IVa | 0 | 0 | 0 |
| Grade IVb | 4 | 1 | 3 |
| Grade V | 0 | 0 | 0 |
| Clavein–Dindo Grade of Most Severe Complication | Silver Dressings ‡ | RES™/Biobrane® | Biobrane® |
| (n) | (5) | (5) | (6) |
| Grade I | 1 | 4 | 1 |
| Grade II | 0 | 0 | 2 |
| Grade IIIa | 0 | 0 | 0 |
| Grade IIIb | 2 | 0 | 1 |
| Grade IVa | 0 | 0 | 0 |
| Grade IVb | 2 | 1 | 2 |
| Grade V | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bairagi, A.; Tyack, Z.; Kimble, R.; Vagenas, D.; McPhail, S.M.; Griffin, B. A Pilot Randomised Controlled Trial Evaluating a Regenerative Epithelial Suspension for Medium-Size Partial-Thickness Burns in Children: The BRACS Trial. Eur. Burn J. 2023, 4, 121-141. https://doi.org/10.3390/ebj4010012
Bairagi A, Tyack Z, Kimble R, Vagenas D, McPhail SM, Griffin B. A Pilot Randomised Controlled Trial Evaluating a Regenerative Epithelial Suspension for Medium-Size Partial-Thickness Burns in Children: The BRACS Trial. European Burn Journal. 2023; 4(1):121-141. https://doi.org/10.3390/ebj4010012
Chicago/Turabian StyleBairagi, Anjana, Zephanie Tyack, Roy Kimble, Dimitrios Vagenas, Steven M. McPhail, and Bronwyn Griffin. 2023. "A Pilot Randomised Controlled Trial Evaluating a Regenerative Epithelial Suspension for Medium-Size Partial-Thickness Burns in Children: The BRACS Trial" European Burn Journal 4, no. 1: 121-141. https://doi.org/10.3390/ebj4010012
APA StyleBairagi, A., Tyack, Z., Kimble, R., Vagenas, D., McPhail, S. M., & Griffin, B. (2023). A Pilot Randomised Controlled Trial Evaluating a Regenerative Epithelial Suspension for Medium-Size Partial-Thickness Burns in Children: The BRACS Trial. European Burn Journal, 4(1), 121-141. https://doi.org/10.3390/ebj4010012






