Abstract
Worldwide, the number of revisions to total knee arthroplasty procedures is increasing. Revision surgery is a challenging procedure, required for the management of bone loss after removal of the first implant. Although further long-term follow-up studies are needed, the use of cones in revisions of total knee arthroplasty yields reliability in fixation and stability to restore joint lines, especially in challenging surgeries with poor bone stock. The introduction of 3D-printed cones in revision surgery seems to be advantageous for AORI type III bone defects, especially in reducing intraoperative complications and procedure times. The aim of this study is to review the currently available literature to analyse clinical outcomes, complications, and radiographical results with the use of metaphyseal tantalum cones and new 3D-printed cones for the management of bone defects in the revision of total knee arthroplasty.
1. Introduction
Total knee arthroplasty (TKA) is the gold standard for treating late-stage knee osteoarthritis. The advancing age and the increasing level of obesity of the global population are the main risk factors for hip and knee osteoarthritis, which will become one of the leading causes of global disability [1].
Between 2001 and 2016 in Italy, total primary knee arthroplasty exhibited an increment of 262%, with an average annual growth rate of 6.6%. Romanini et al. calculated that in Italy by 2050, the incidence rate of TKA will rise by 45% with respect to 2017 [2]. Worldwide, an increase of 143% by 2050 is expected.
In the last forty years, innovation in the prosthetic field in terms of designs, materials, instrumentation, and surgical techniques has improved survivorship of TKA. Recent studies have demonstrated a positive survivorship of 25 years in 82.3% of first implants [3]. However, one in five patients are reportedly still not satisfied with the results of surgery [4]. This inevitably indicates increases in the revision total knee arthroplasty rate.
In the United States between 2005 and 2006, 60,355 revisions of TKA were performed; the major indications were periprosthetic infection, aseptic loosening, and implant failure. Subsequently, studies have demonstrated similar trends with substantial reductions in the number of failures secondary to polyethylene wear [5].
Revision surgery and the re-revision of TKA are very challenging procedures, fraught with problems and complications. Bone defects secondary to aseptic and septic failure, or iatrogenic bone loss after first implant removal, significantly jeopardize the stability of the implant and the final success of the revision surgery [6]. The introduction of metal augments for the treatment of bone defects seems to improve load distribution, granting better stability and fixation of the implant, and enhancing longevity. Nevertheless, periprosthetic infections remain the main cause for the failure and re-revision of TKA.
The aim of this study is to review the available literature and analyse clinical and radiographical outcomes with the use of standard cones and novel 3D-printed cones for the management of bone defects in the revision of total knee arthroplasty (TKA). The review will discuss the classification system and treatment options of bone defects in TKA; the material characteristics and mechanical properties of first- and second-generation metal cones; surgical techniques for the use of metaphyseal cones; and the clinical outcomes of 3D-printed cones in revision TKA with related cost analysis.
2. Materials and Methods
Electronic searches were conducted on the PubMed and Science Direct databases with different combinations of the following search terms: “cone”, “tantalum metal cone”, “metal cone”, “metaphyseal cone”, “total knee arthroplasty”, “revision total knee arthroplasty”, “3-d printed”, and “titanium cone”. The time interval was 1990 to March 2023. The search was restricted to articles written in English related to the use of metal augments in total knee arthroplasty revision surgery. Only articles published in peer-reviewed journals were included. The search included literature reviews and prospective and retrospective studies. Articles written in other languages without available abstracts and those related to other fields were excluded.
3. Bone Defects: Classification System
Key challenges in revision surgery are bone defects and their management.
In addition to preoperative evaluation of the patient with historical and clinical examination, the assessment and classification of bone loss before surgery are vital, especially to predict outcomes, to choose the right type of implant, and to avoid unforeseen intraoperative problems. Radiological studies are required, such as a weight bearing imaging with anteroposterior, lateral, and Merchant patellar views. CT scans may be useful to estimate bone loss and bone prosthesis interfaces more accurately [7]. A proper classification system could help surgeons evaluate bone defects before surgery to decide on the best option for management and treatment.
In the contemporary literature, the most commonly used classification, clinical trials, and treatment guideline protocols on knee revision surgery is the Anderson Orthopaedic Research Institute (AORI), described by Engh in 1997. It describes bone loss according to the size, localization, and soft tissue involvement from preoperative radiographs and is either confirmed or changed intraoperatively (Table 1). Type I are lesions are limited to cancellous bone with intact metaphysis and do not compromise the stability of the component. Type II involves metaphysis, affecting one condyle or tibial plateau (IIA) or two condyles or tibial plateaus (IIB). Type III lesions, beyond the metaphyseal bone which is deficient, involve the attachments of collateral ligaments and patellar tendon [8,9].

Table 1.
Description, characteristics, and treatment options of bone deficiencies in rTKA by Anderson Orthopaedic Research Institute (AORI) classification.
Although the AORI classification is the gold standard for the treatment choice and is simple to use, it has inherent limitations: AORI classifications typically underestimate bone loss [10]. This method does not quantify diaphyseal bone loss and only partially quantifies the metaphyseal area [11].
For this reason, several authors have proposed new classification systems or modified AORI classifications, although most of them are complex and have not been widely adopted. Rosso et al. modified the AORI classification with the intraoperative evaluation of epiphysis and metaphysis bone, distinguishing good bone quality (G), i.e., whether there was good cancellous bone and bleeding after bone preparation; sclerotic bone (S), i.e., whether there was a marble aspect of the bone with poor vascularization; and osteoporotic bone (O), i.e., poor bone quality [6].
Stambough et al. proposed an AORI modification in which Type IIA and Type III tibial defects are subdivided into contained and uncontained defects [12].
According to the concept of fixation, proposed by Morgan-Jones, there are three anatomical zones (epiphysis, metaphysis, and diaphysis) that are important for stability in revision surgery. In most revisions, the epiphysis and metaphysis are compromised, and the choice of an appropriate revision implant require a multizone approach [13].
Based on this concept, Jang S.J. et al. proposed an algorithm that identifies bone revision zones and cone placements with excellent accuracy and reliability through the use of deep learning, a subset of artificial intelligence [14].
Despite the efforts and the proposed modification, AORI classification remains a reliable system for femoral bone loss and substantial tibial bone loss [10,15].
4. Treatment Options
The purpose of classification systems is to suggest an appropriate treatment based on the bone defect. Cement should be used in cases of limited bone defects (AORI type I), peripheral deficiency up to 10%, central and cystic defects, and especially in cancellous non-sclerotic bone. The use of screws in addition to cement unloads the joint line and the cement–bone interface. Unfortunately, the outcomes of this technique are not satisfying; the use of a relatively large amount of cement could lead to thermal necrosis of the bone interface. Furthermore, the cement mass may exhibit failure due to its low resistance to stress and compression [16]. Autograft or allograft bones should be used in small and contained defects, especially in young patients, due to their osteoconductive properties. Both exhibit better load transfer compared with cement. Autografts are only used for small defects due to their limited availability. The use of allografts showed great results in terms of stability and survivorship for the management of severe tibial bone defect in revision surgery [17]; however, since large allografts have numerous disadvantages such as a lack of revascularization and inadequate remodelling [18], the introduction of other treatment options in revision surgery has become necessary.
The surgical choice for small uncontained defects (AORI IIa–IIb) is based on the size and location of the bone defect. Modular metal augments in wedges or blocks could be used for defects of up to 25 mm, and are particularly useful in tibial plateau deflections or posterior femoral condyles bone defects. These augments provide prompt support but often require additional resection to fit to the host bone [19].
Nowadays, there are outstanding options for the treatment of large, uncontained bone defects (AORI Type III); the use of tantalum or titanium cones and sleeves is widespread in knee surgery revisions. Sleeves and cones are metal devices that induce stability in the prosthesis with a cementless metaphyseal anchorage (Figure 1). Cones are first implanted press-fit into the defect after its preparation; then, the prosthesis is cemented into the cone [8]. Sleeves are bonded to the stem with a morse taper and inserted as a press-fit implant after preparing the host bone with a broach. The choice between one or the other is dictated by surgeon preference, shape, and size of the defect [20]. On the other hand, surgical techniques implanting the sleeve require the use of broaches that achieve alignment of the implant by the intramedullary channel are difficult to manage in patients with canal–epiphyseal mismatch. Moreover, metaphyseal sleeves need strong contact with the host bone, which is difficult and highly surgery demanding in cases of uncontained defects [21].

Figure 1.
Metaphyseal modular implants for bone defection repair: (a) porous metal cones (Stryker, Mahwah, NJ, USA); (b) porous metal sleeves (DePuy Orthopedics, Warsaw, IN, USA).
5. Metallic Cones and Materials
First-generation metaphyseal cones are metal cone-shaped constructs composed of trabecular tantalum, which is a highly porous metal with excellent properties such as porosity, strength, flexibility, and biocompatibility. Tantalum was introduced in 1997. Since then, it has been widely used in hip orthopaedic reconstructive surgery and, in recent decades, in reconstructive surgery of the knee [22,23]. Porous tantalum is an excellent biomaterial with mechanical and biological properties for augmentation because of its similar elasticity to subchondral bone, low stiffness, high coefficient of friction that enables early weight bearing, and its excellent structure with a stable oxide that interacts with the surrounding tissue, facilitating osteoconduction and the development of bone-like apatite layers [24,25]. Moreover, porous tantalum foam seems to provide initial implant stability on loads while bone ingrowth occurs [26,27]. Bobyn et al. studied canine models of the characteristics of bone ingrowth using porous tantalum biomaterial and concluded that porous tantalum functions as a scaffold. In fact, the regions of bone-to-implant contact increase over time, producing osteons within the pores at a later time [23]. The elastic modulus of tantalum varies based on the porosity, and in general ranges between 2 and 20 GPa, imitating a cortical bone modulus of 3–30 GPa [28]. Metallic properties such as the surface area, porosity, hydrophobicity, chemistry, and microenvironmental factors are fundamental to surgery outcomes because they interact with the host bone and blood cells. Pure tantalum increases host white blood cell activation and seems to have a “bacteriophobic effect”, reducing S. aureus adhesion compared with titanium alloy, polished stainless steel, and tantalum-coated stainless steel implants, according to studies conducted by Schildhauer [29,30].
In the last decade, further improvements have been reached for the treatment of severe bone defects, with the production of 3D-printed cones. Second-generation cones are highly porous taper titanium cones produced from titanium powder using 3D-printed technology based on a computed tomography database. These cones have porous surface layers that provide bone ingrowth and reduce implant–bone mismatch. The implantation is made easier by specific instrumentation that enables the preparation of medial and lateral lobed portions to achieve a better fit [31,32]. These second-generation cones are implanted with a facilitated system of cannulated ream to prepare host bone to better fit the cone compared with the first-generation cones. Titanium and tantalum could also be coated with calcium phosphate to improve osseointegration and the bioactivity of augments [33]. According to Rambani R. et al., tantalum seems to be superior to titanium in terms of fewer radiolucencies, survivorship, osteointegration, decreasing osteolysism, and mechanical loosening [34]. On the other hand, titanium and its alloys reduce stress shielding compared with Co–Cr alloys because of their lower elastic moduli; however, this can cause stiffness mismatch to bone. The use of porous material overcomes this drawback and achieves stable long-term fixation [35].
Modern technology is constantly evolving; thus, surgeons have the possibility to use patient-specific 3D-printed titanium cones. After a knee CT or MRI, a computer reconstruction is created, and planning is performed with specialized computer-aided design software. A virtual model is used to mark the areas that could be removed or smoothed during surgery to facilitate the implantation. The model is then converted into a series of sliced 2D layers. Subsequently, the cones are printed layer by layer from titanium powder using a computer-controlled 3D printer. Grinding, coating, and/or surface oxidation are conducted in the postprocessing step. The production time of patient-specific cones could necessitate a variable time spanning from 2 to 8 weeks, including design, production, and logistics [36].
To date, several methods are used for titanium alloy 3D printing. The electron beam melting (EBM) technique is the most common 3D printing technique used to produce orthopaedic devices with high strength and low impurities. Other common techniques are selective laser sintering (SLS), which is the fastest production method in the industry but requires a longer post-printing process; and the selective laser melting (SLM) method, which yields higher accuracy but lower speed compared with SLS [37].
6. Surgical Technique
Even though the use of cones showed excellent results, the limited sizes and geometries of these devices could make the surgery technically challenging in matching the cone with the defect.
Surgical techniques that use metaphyseal standard cones consist of preparing the host bone with high-speed round or pencil-tip burrs and selecting the best size and shape of the cone using plastic trials. The resections are hand-made; therefore, care should be taken not to over-resect the bone. The definitive cone is then press-fitted into the metaphyseal bone and the stability of the porous tantalum is assessed during impaction. Figure 1 depicts voids between the host bone and the external surface of the cone then being filled with morselized cancellous bone grafts or demineralized bone matrixes [24,38]. Stepped cones are available when the bone defect is not centred, and could be used in combination with offset stems to restore the correct alignment [39]. Even though commercially available cones come in different shapes and measures, one of the major critiques in the use of metal standard cones is fitting to the remaining bone after TKA removal; in fact, standard cones usually do not fit to the individual patient’s gap and anatomy. The preparation of the host bone with burr in addition to removing remaining bone stock to adapt the cone makes the surgical technique more demanding and increases the risk of intraoperative fractures.
Surgical techniques for the implantation of 3D cones consist of preparing the intramedullary canal with sequential reaming until a correct fit is achieved. Then, the reamer is left in the canal and used as a guide for a conical reamer of the corresponding cone size. Single conical reamers could be used for symmetric defects or an instrumented system to fill medial or lateral defects. After choosing the right cone that matches the bone defect, the definitive cone is impacted (Figure 2). Rarely is a bone graft needed [40].
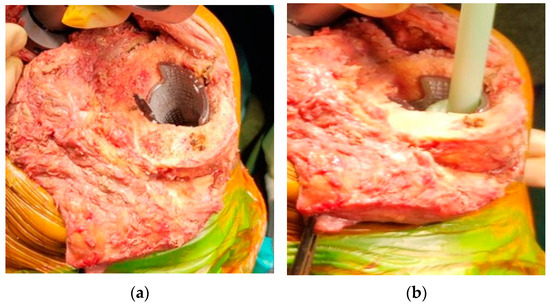
Figure 2.
Surgical technique: (a) the definitive cone is press-fitted into the metaphyseal bone; (b) the prosthesis is then cemented into the cone.
7. Outcomes and Complications
The number of TKA revisions is destined to increase; therefore, it is necessary to evaluate which type of implant would be correct to implant based on the bone defect. There are several studies in the literature comparing outcomes and survivorship of the use of cones and other treatments in revision surgery.
Outcomes for the use of standard cones in rTKA are very promising even though only few studies have employed a long-term follow-up. Sandiford et al. compared trabecular metal cones and femoral head allografts in rTKA, concluding that there were no differences between the two groups in terms of clinical outcome, survivorship of the implant at 5-year follow-up, component alignment, and the number of complications. However, they declared that TM were used for relatively easier, symmetric defects [41]. On the other hand, Beckmann et al. found that aseptic loosening occurred 75% less frequently in TKA revision with the use of metal cones compared with the use of structural or bulk allografts due to poor revascularization and remodelling [42]. Several studies have demonstrated that there is not a significant difference in the use of sleeves and cones in TKA revision surgery in terms of aseptic revision, survivorship, and postoperative clinical outcomes [43,44]. Fisher et al. demonstrated that in both cases, the use of metal devices improved KSS and KSFS scores postoperatively. Although periprosthetic infections appear to be more frequent with the use of cones, a possible explanation could be found in different metal properties of the devices: sleeves have a dense surface, while cones have a trabecular pored surface [45]. Mancuso et al. suggested that tibial sleeves have a slightly lower survivorship. The reason could be in a more modular construct customized with cones that confer greater stability compared with sleeve constructs. On the other hand, cones are cemented while sleeves are press-fitted at their morse–taper junction, which could contribute to the difference in survivorship [8].
In the past, the major cause of primary TKA failure has been polyethylene wear; however, with the improvements in surgical techniques and prosthesis designs, the major overall causes of failure have become aseptic loosening, infection, instability, patellofemoral complication, and pain [46]. Several studies at mid-term follow-up have demonstrated excellent osteointegration with reactive osseous trabeculation at the cone–bone interface with excellent survivorship and no evidence of loosening or migration of the construct [38,47]. Bohl et al. compared the results of rTKA using porous metaphyseal tantalum cones to traditional hybrid stem fixation with clinical and radiographic outcomes at a minimum two-year follow-up. The hybrid technique consists of only using the cement for the metaphysis and undersurface of the component. At follow-up radiographs, osteointegration was apparent in all the porous cones [48]. At mid-term follow-up, good osteointegration and stability were evaluated by following radiographs with the absence of loosening, migration, and radiolucent lines in several studies [49,50]. Potter et al. confirm the trend in the literature with their study in which only 4% of the cones required revisions due to aseptic loosening at five-year follow-up [51]. In contrast, Girerd found that all the tibial cones that needed to be removed secondary to PJI were well-fixed, while all the femoral cones were loose and readily extracted. Although radiographic outcomes, complications, and clinical outcomes showed good KSSs after revision surgery, quality of life scores were low compared with analogue studies [52].
One of the main complications and reasons for re-revision in rTKA is periprosthetic joint infection (PJI). Among patient’s characteristics, a prior history of septic revision is associated with an increased risk of following surgery with removal of the cone. Brown et al. identified a postoperative complication rate of almost 45%, although the two main complications were PJI and stiffness [53]. Abdelaziz conducted a study analysing complications and clinical and radiographical outcomes in patients who underwent one-stage rTKA with tantalum cone for periprosthetic infection. Compared with a previous study that analysed complications in one-stage rTKA for infection without the use of the cone, the rate of re-revision due to reinfection was higher, but the rate of aseptic loosening was 2%. In most cases, the reinfection was caused by a new pathogen [54,55]. Even though most periprosthetic joint infection complications were in patients who required a revision of TKA due to a primarily infection, some studies have demonstrated that PJI is the most frequent complication even when excluding those patients [56]. In their retrospective study that included 94 tantalum implants during rTKA, Burastero et al. concluded that the use of tantalum cones could also be considered a safe and effective treatment of bone defects in terms of clinical and radiological mid-term outcomes in the primary septic failure of TKA. Moreover, 67.5% of the patients received 75 mm stems [57].
No studies have clearly defined what is the best implant or stem length to use in combination with metaphyseal fixation. Hybrid fixation with uncemented long-stem and porous tantalum cones demonstrated great survivorship at a short-term follow-up [58]. Anderson et al. found that, despite an excellent metaphyseal fixation provided, the use of cones with cementless stems may increase the risk of malalignment compared with cemented stems [59]. Rossi et al. found a greater number of failures and aseptic loosening with consequent re-revisions in patients who underwent revision with a lower joint constraint (PS vs. CCK implants) [60]. The use of tantalum cones with short, cemented stems in rTKA seems to have comparable or even better outcomes in terms of stability, quality of life, and function compared with re-revision with TM and long partially cemented stems. Jacquets et al. also highlighted lower survivorship in TM and long-stem groups [56,61]. Moreover, the use of short stems avoids the risk of prosthesis malalignment in patients with previous distal femoral or proximal tibial osteotomy, fracture during intramedullary canal preparation, or end-of-stem pain. The use of metaphyseal cones also facilitates the combination of several techniques such as diaphyseal impaction grafting, as described by Ullmark and Hovelius [62], with excellent survival rates free from aseptic loosening and improved diaphyseal bone stock useful for further revision [63].
Long-term results are yet to be determined because only a few studies in the literature have analysed long-term outcomes and the survivorship of tantalum cones. De Martino et al. studied 28 patients who underwent rTKA with the use of tantalum cones with a minimum follow-up of 10 years. Six patients of the study underwent re-revision and only three of them, at time of reoperation, exhibited loosening of the cone, demonstrating excellent survivorship and fixation in the long term [64]. On the other hand, Abdelaziz et al. found an overall re-revision rate of 52% at a minimum of 10-year follow-up; half of the surviving patients without re-revision still suffered from knee pain [65].
Although first-generation cones showed excellent results, there are some limitations regarding inaccurate bone preparations and limited sizes and shapes. Hand-made resections of bone to fit the standard cone increase the risk of intraoperative fractures. Villanueva-Martinez et al. reported an intraoperative fracture rate of 24% related to cone implantation, most which were femoral [66]. Compared with standard cones, intraoperative-cone-related fractures were less frequent in TKA revision with the use of 3D cones, probably due to the non-necessity of additional reshaping of the host bone [20]. Gibon et al. compared tantalum cones and titanium cones and sleeves with a minimum 1-year follow-up. Intraoperative fractures occurred in five patients of the TM group compared with none in the other groups. The revision rate was lower in the Ti cone group. Despite these trends, there were no statistically significant differences across groups, demonstrating that all three technologies had excellent mid-term revision-free survival rates [67].
At short-term follow up, the use of 3D-printed cones and tantalum cones in rTKA showed good osteointegration in radiographic analyses, and close to a 100% survival rate for aseptic loosening and improvements in functional results; unfortunately, the main cause of failure and reoperation is infection [31,40,68,69,70]. In this study, 3D-printed titanium metaphyseal cones were investigated, analysing survivorship, radiological osteointegration, complications, and clinical outcomes. Preoperative planning consisted of clinical examination and radiographs to identify bone defects, although the effective defects were assessed intraoperatively. At the end of follow-up, five cones had been revised (three PJI, one periprosthetic fracture, and one aseptic loosening). At two-year follow-up, 100% of the cones were free of aseptic loosening.
Developing 3D or custom-made cones grows from the limited geometries available is necessary to fill bone gaps. There were three intraoperative fractures during cone impaction. There were four aseptic loosening events in total: only one was revised, while the other three were diagnosed radiologically [71]. Burastero et al. replicated in silico geometric and implants during surgery to analyse biomechanics during daily activities of three different revision knee techniques. They concluded that the use of custom-made porous titanium metaphyseal cones and stems that were thinner than the diaphyseal canals resulted in better distribution of the stress on both the tibia and femur compared with the use of cement stems or press-fit stems alone. Custom devices, in fact, provide stability in the region between the pure metaphyseal zone and a pure diaphyseal zone [72]. Faizan et al. demonstrated that new porous titanium cones compared with traditional porous cones exhibited significantly less micromotion of the implant under physiologic loading during daily activities. Furthermore, these new devices are designed to better fit to bone defect anatomy and adopt simplified instrumentation that maximizes surgeon efficiency during challenging procedures [73]. Shichman et al. compared functional outcomes and survival in rTKA with the use of porous titanium cones with stems of variable length and fixation, particularly the hybrid cementation technique and the cemented technique. At 2 years of follow-up, the whole cohort demonstrated an excellent survivorship, with numerically higher freedom from all-cause re-revision for the cemented group. Furthermore, the proportion of revisions for malposition of the component was higher in the hybrid cementation group. The rate of radiographic cone osteointegration was 99.4% [74]. Behery et al. demonstrated 100% survivorship free of aseptic loosening at mid-term follow-up using 3D-printed porous titanium cones with short, cemented stems. This construct avoids the use of long-stem fixation associated with more challenges in the case of re-revision, end-of-stem pain, and inherent coronal malalignment conditioned by diaphyseal engaging stems [75]. The use of a novel cone with a thinner wall enables the use of cementless stems and offsets. Aseptic survivorship of the cones was 95.9% at 2-year follow-up, with an improvement of functional outcomes [76].
Studying the use of patient-specific cones, Cherny et al. described the technique and prospectively evaluated the outcomes. In this case, the surfaces of the printed patient-specific cones were roughened in the zones in contact with the bone to improve primary fixation, while the surface in contact with soft tissue was smoothened, reducing adhesions or irritation. Intraoperatively, the 3D-printed cones adapted their size and shape to fit the bone gap, and radiographic signs of osteointegration were detected within the first six months after surgery. At one-year follow-up, none of the patients exhibited component loosening [36]. Despite the laborious production process, Li Y. et al. found that with the use of PS metallic cones, the procedure is simplified, and the operating time is decreased [77].
8. Costs
According to a French study, the average additional cost related to the cones was 2709 ± 1138 EUR/stay, which is approximately 35% of the total cost of the mean stay for a rTKA. The economic impact of the use of porous metal cone is significant and unfavourable compared with the use of allograft; thus, obtaining adequate funding for such devices is a key obstacle for French hospitals [78]. The mean average selling price of tibial metal cones on the market (produced by Zimmer Biomet Holdings, Inc., Warsaw, IN, USA;, Smith & Nephew, Inc., Memphis, TN, USA, and Stryker Orthopaedics, Mahwah, NJ, USA) is USD 4.201, the cost of a TKA revision for aseptic loosening is USD 29.802, and the average cost of TKA re-revision for aseptic loosening is USD 34.874. The average hospitalization cost of re-revisions for all other causes is USD 35.808. Based on a literature review, the annual re-revision rate for aseptic tibial cone loosening is 0.14%, while the rate for aseptic tibial component loosening without cone is 0.76%. The surgical and hospitalization costs of a TKA revision with the use of metaphyseal cone exceeded USD 2026 on average compared with the cost of a TKA revision without cone. Regarding cost-effectiveness, based on the lifetime, patients with loosening rates of approximately 0.89 at 40 years old and 4.38% at 90 years old may benefit from cone augmentation. High rates of aseptic loosening are associated with AORI Type III defects, increased implant constraints, smoking, age < 65 years, obesity, and initial revisions for aseptic loosening [79]. In THA reconstruction surgery, Migaud et al. estimated the cost of classical reconstruction using an allograft with a reinforcement cage compared with modular materials and custom-made materials. The costs were considerably higher for custom-made materials, but the reduced surgery time with positive outcomes could reflect in reduced costs in the long term [80,81]. The use of metaphyseal implants can result in increases in costs ranging from 25% with the use of a single sleeve to 300% with the use of tibial and femoral cones, which does not justify the routine use of metal meta-diaphyseal implants [44].
The costs of single 3D-printed implants tend to be higher than standard cones, and vary between developer, country, and commercial agreements. This must be attributed to the novelty of this technique, which has been developed within the last two decades and requires additional efforts in the production process. As the productivity of 3D implants improves, costs are expected to decrease [82]. Nevertheless, cost–benefit analyses should account for production costs, surgical times, operating room turnover times, transfusion rates and blood loss, hospital stays, and the rate of revision surgeries. Based on a financial analysis on the literature conducted by Ballard et al., the use of 3D-printed constructs could reduce operating room costs and procedure times [83].
9. Conclusions
The use of metallic metaphyseal cones has changed the surgical approach to bone defects in total knee arthroplasty revisions, in particular improving implant stability. The introduction of titanium 3D-printed metaphyseal cones in comparison to first-generation cones facilitated enhanced surgical techniques, improved implant adaptability, and reduced intraoperative complications. The use of titanium 3D-printed metaphyseal cones in total knee arthroplasty revisions seems to be advantageous in the management of considerable bone defects (AORI IIb–III). Short- and mid-term studies have demonstrated excellent outcomes in terms of clinical and radiographical outcomes, osteointegration, and reductions in complications. Although the reported outcomes are favourable for the use of cones in revision surgery, the literature is very limited due to the lack of long-term studies or assessments of large groups of patients. To date, long-term studies have not presented unique results on the benefits of using metallic cones; further observations are needed. Moreover, the increased challenges of performing surgery in cases of implant revisions with a well-fixed cone should not be underestimated. Surgeons should consider various techniques for reconstruction depending on patient age, life expectancy, the degree of bone loss, the amount of augmentation required, the usage of long or offset stems, and custom or hinge/tumour prostheses.
Author Contributions
Conceptualization, M.B. and G.M.; methodology, M.B. and G.M.; formal analysis, M.B., G.P. and A.C.; resources, A.C.; data curation, M.B., G.M. and G.P.; writing—original draft preparation, M.B., G.M. and G.P.; writing—review and editing, D.K. and M.V.; supervision, A.C., C.Z., D.K. and M.V.; project administration, G.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Romanini, E.; Decarolis, F.; Luzi, I.; Zanoli, G.; Venosa, M.; Laricchiuta, P.; Carrani, E.; Torre, M. Total knee arthroplasty in Italy: Reflections from the last fifteen years and projections for the next thirty. Int. Orthop. (SICOT) 2019, 43, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.T.; Walker, R.W.; Evans, J.P.; Blom, A.W.; Sayers, A.; Whitehouse, M.R. How long does a knee replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet 2019, 393, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Bourne, R.B.; Chesworth, B.M.; Davis, A.M.; Mahomed, N.N.; Charron, K.D. Patient satisfaction after total knee arthroplasty: Who is satisfied and who is not? Clin. Orthop. Relat. Res. 2010, 468, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Bozic, K.J.; Kurtz, S.M.; Lau, E.; Ong, K.; Chiu, V.; Vail, T.P.; Rubash, H.E.; Berry, D.J. The epidemiology of revision total knee arthroplasty in the United States. Clin. Orthop. Relat. Res. 2010, 468, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Rosso, F.; Cottino, U.; Dettoni, F.; Bruzzone, M.; Bonasia, D.E.; Rossi, R. Revision total knee arthroplasty (TKA): Mid-term outcomes and bone loss/quality evaluation and treatment. J. Orthop. Surg. Res. 2019, 14, 280. [Google Scholar] [CrossRef]
- Gonzalez, M.H.; Mekhail, A.O. The Failed Total Knee Arthroplasty: Evaluation and Etiology. J. Am. Acad. Orthop. Surg. 2004, 12, 436–446. [Google Scholar] [CrossRef]
- Mancuso, F.; Beltrame, A.; Colombo, E.; Miani, E.; Bassini, F. Management of metaphyseal bone loss in revision knee arthroplasty. Acta Biomed. 2017, 88, 98–111. [Google Scholar]
- Engh, G.A. Classification of Bone Defects Femur and Tibia. In Knee Arthroplasty Handbook; Scuderi, G.R., Tria, A.J., Eds.; Springer: New York, NY, USA, 2006; pp. 116–132. [Google Scholar] [CrossRef]
- Mulhall, K.J.; Ghomrawi, H.M.; Engh, G.A.; Clark, C.R.; Lotke, P.; Saleh, K.J. Radiographic Prediction of Intraoperative Bone Loss in Knee Arthroplasty Revision. Clin. Orthop. Relat. Res. 2006, 446, 51–58. [Google Scholar] [CrossRef]
- Belt, M.; Smulders, K.; van Houten, A.; Wymenga, A.; Heesterbeek, P.; van Hellemondt, G. What Is the Reliability of a New Classification for Bone Defects in Revision TKA Based on Preoperative Radiographs? Clin. Orthop. Relat. Res. 2020, 478, 2057–2064. [Google Scholar] [CrossRef]
- Stambough, J.B.; Haynes, J.A.; Barrack, R.L.; Nunley, R.M. Acetabular wedge augments for uncontained tibial plateau defects in revision total knee arthroplasty. Arthroplast. Today 2018, 4, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Morgan-Jones, R.; Oussedik, S.I.; Graichen, H.; Haddad, F.S. Zonal fixation in revision total knee arthroplasty. Bone Jt. J. 2015, 97-B, 147–149. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.J.; Flevas, D.A.; Kunze, K.N.; Anderson, C.G.; Fontana, M.A.; Boettner, F.; Sculco, T.P.; Baldini, A.; Sculco, P.K. Standardized Fixation Zones and Cone Assessments for Revision Total Knee Arthroplasty Using Deep Learning. J. Arthroplast. 2023, 38, S259–S265. [Google Scholar] [CrossRef]
- Khan, Y.; Arora, S.; Kashyap, A.; Patralekh, M.K.; Maini, L. Bone defect classifications in revision total knee arthroplasty, their reliability and utility: A systematic review. Arch. Orthop. Trauma. Surg. 2023, 143, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Brooks, P.J.; Walker, P.S.; Scott, R.D. Tibial Component Fixation in Deficient Tibial Bone Stock. Clin. Orthop. Relat. Res. 1984, 184, 302–308. [Google Scholar] [CrossRef]
- Engh, G.A.; Ammeen, D.J. Use of Structural Allograft in Revision Total Knee Arthroplasty in Knees with Severe Tibial Bone Loss. J. Bone Jt. Surg.-Am. Vol. 2007, 89, 2640–2647. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, S.; Li, X.Q.; Davy, D.T.; Klein, L.; Goldberg, V.M. Critical biological determinants of incorporation of non-vascularized cortical bone grafts. Quantification of a complex process and structure. J. Bone Jt. Surg. Am. 1997, 79, 1–16. [Google Scholar] [CrossRef]
- Sheth, N.P.; Bonadio, M.B.; Demange, M.K. Bone Loss in Revision Total Knee Arthroplasty: Evaluation and Management. J. Am. Acad. Orthop. Surg. 2017, 25, 348–357. [Google Scholar] [CrossRef]
- Zanirato, A.; Formica, M.; Cavagnaro, L.; Divano, S.; Burastero, G.; Felli, L. Metaphyseal cones and sleeves in revision total knee arthroplasty: Two sides of the same coin? Complications, clinical and radiological results—A systematic review of the literature. Musculoskelet. Surg. 2020, 104, 25–35. [Google Scholar] [CrossRef]
- Matar, H.E.; Bloch, B.V.; James, P.J. Role of metaphyseal sleeves in revision total knee arthroplasty: Rationale, indications and long-term outcomes. J. Orthop. 2021, 23, 107–112. [Google Scholar] [CrossRef]
- Christie, M.J. Clinical applications of Trabecular Metal. Am. J. Orthop. 2002, 31, 219–220. [Google Scholar] [PubMed]
- Bobyn, J.D.; Stackpool, G.J.; Hacking, S.A.; Tanzer, M.; Krygier, J.J. Characteristics of bone ingrowth and interface mechanics of a new porous tantalum biomaterial. J. Bone Jt. Surg. Br. Vol. 1999, 81-B, 907–914. [Google Scholar] [CrossRef]
- Lachiewicz, P.F.; Watters, T.S. Porous metal metaphyseal cones for severe bone loss: When only metal will do. Bone Jt. J. 2014, 96-B, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Sporer, S.; Valle, C.; Jacobs, J.; Paprosky, W. Porous Tantalum in Reconstructive Surgery of the Knee—A Review. J. Knee Surg. 2010, 20, 185–194. [Google Scholar] [CrossRef]
- Cohen, R. A porous tantalum trabecular metal: Basic science. Am. J. Orthop. 2002, 31, 216–217. [Google Scholar]
- Zardiackas, L.D.; Parsell, D.E.; Dillon, L.D.; Mitchell, D.W.; Nunnery, L.A.; Poggie, R. Structure, metallurgy, and mechanical properties of a porous tantalum foam. J. Biomed. Mater. Res. 2001, 58, 180–187. [Google Scholar] [CrossRef]
- Balla, V.K.; Bodhak, S.; Bose, S.; Bandyopadhyay, A. Porous tantalum structures for bone implants: Fabrication, mechanical and in vitro biological properties. Acta Biomater. 2010, 6, 3349–3359. [Google Scholar] [CrossRef]
- Schildhauer, T.A.; Peter, E.; Muhr, G.; Köller, M. Activation of human leukocytes on tantalum trabecular metal in comparison to commonly used orthopedic metal implant materials. J. Biomed. Mater. Res. 2009, 88A, 332–341. [Google Scholar] [CrossRef]
- Schildhauer, T.A.; Robie, B.; Muhr, G.; Koller, M. Bacterial Adherence to Tantalum Versus Commonly Used Orthopedic Metallic Implant Materials. J. Orthop. Trauma 2006, 20, 476–484. [Google Scholar] [CrossRef]
- Denehy, K.M.; Abhari, S.; Krebs, V.E.; Higuera-Rueda, C.A.; Samuel, L.T.; Sultan, A.A.; Mont, M.A.; Malkani, A.L. Metaphyseal Fixation Using Highly Porous Cones in Revision Total Knee Arthroplasty: Minimum Two Year Follow Up Study. J. Arthroplast. 2019, 34, 2439–2443. [Google Scholar] [CrossRef]
- Black, J. Biologic performance of tantalum. Clin. Mater. 1994, 16, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Ohlmeier, M.; Lausmann, C.; Wolff, M.; Abdelaziz, H.; Gehrke, T.; Citak, M. Preliminary clinical results of coated porous tibia cones in septic and aseptic revision knee arthroplasty. Arch. Orthop. Trauma. Surg. 2021, 141, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Rambani, R.; Nayak, M.; Aziz, M.S.; Almeida, K. Tantalum Versus Titanium Acetabular Cups in Primary Total Hip Arthroplasty: Current Concept and a Review of the Current Literature. Arch. Bone Jt. Surg. 2022, 10, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Ryan, G.; Pandit, A.; Apatsidis, D. Fabrication methods of porous metals for use in orthopaedic applications. Biomaterials 2006, 27, 2651–2670. [Google Scholar] [CrossRef] [PubMed]
- Cherny, A.A.; Kovalenko, A.N.; Kulyaba, T.A.; Kornilov, N.N. A prospective study on outcome of patient-specific cones in revision knee arthroplasty. Arch. Orthop. Trauma. Surg. 2021, 141, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; Wang, J.; Huang, H.; Liu, X.; Zhang, J.; Li, Z. 3D printing metal implants in orthopedic surgery: Methods, applications and future prospects. J. Orthop. Translat. 2023, 42, 94–112. [Google Scholar] [CrossRef] [PubMed]
- Meneghini, R.M.; Lewallen, D.G.; Hanssen, A.D. Use of Porous Tantalum Metaphyseal Cones for Severe Tibial Bone Loss During Revision Total Knee Replacement. J. Bone Jt. Surg.-Am. Vol. 2008, 90, 78–84. [Google Scholar] [CrossRef]
- Radnay, C.S.; Scuderi, G.R. Management of Bone Loss: Augments, Cones, Offset Stems. Clin. Orthop. Relat. Res. 2006, 446, 83–92. [Google Scholar] [CrossRef]
- Chalmers, B.P.; Malfer, C.M.; Mayman, D.J.; Westrich, G.H.; Sculco, P.K.; Bostrom, M.P.; Jerabek, S.A. Early Survivorship of Newly Designed Highly Porous Metaphyseal Tibial Cones in Revision Total Knee Arthroplasty. Arthroplast. Today 2021, 8, 5–10. [Google Scholar] [CrossRef]
- Sandiford, N.A.; Misur, P.; Garbuz, D.S.; Greidanus, N.V.; Masri, B.A. No Difference Between Trabecular Metal Cones and Femoral Head Allografts in Revision TKA: Minimum 5-year Followup. Clin. Orthop. Relat. Res. 2017, 475, 118–124. [Google Scholar] [CrossRef]
- Beckmann, N.A.; Mueller, S.; Gondan, M.; Jaeger, S.; Reiner, T.; Bitsch, R.G. Treatment of Severe Bone Defects During Revision Total Knee Arthroplasty with Structural Allografts and Porous Metal Cones—A Systematic Review. J. Arthroplast. 2015, 30, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Small, I.; Meghpara, M.; Stein, J.; Goh, G.S.; Banerjee, S.; Courtney, P.M. Intermediate-Term Survivorship of Metaphyseal Cones and Sleeves in Revision Total Knee Arthroplasty. J. Arthroplast. 2022, 37, 1839–1843. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, M.J.; Lanting, B.A.; McCalden, R.W.; Naudie, D.D.; Howard, J.L.; MacDonald, S.J.; Vasarhelyi, E.M. Survivorship of Metaphyseal Cones and Sleeves in Revision Total Knee Arthroplasty. J. Arthroplast. 2022, 37, S263–S269. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.T.; Heinecke, M.; Röhner, E.; Schlattmann, P.; Matziolis, G. Cones and sleeves present good survival and clinical outcome in revision total knee arthroplasty: A meta-analysis. Knee Surg. Sports Traumatol. Arthrosc. 2022, 30, 2824–2837. [Google Scholar] [CrossRef] [PubMed]
- Inui, H.; Yamagami, R.; Kono, K.; Kawaguchi, K. What are the causes of failure after total knee arthroplasty? J. Jt. Surg. Res. 2023, 1, 32–40. [Google Scholar] [CrossRef]
- Erivan, R.; Tracey, R.; Mulliez, A.; Villatte, G.; Paprosky, W. Medium term clinical outcomes of tibial cones in revision knee arthroplasty. Arch. Orthop. Trauma. Surg. 2021, 141, 113–118. [Google Scholar] [CrossRef]
- Bohl, D.D.; Brown, N.M.; McDowell, M.A.; Levine, B.R.; Sporer, S.M.; Paprosky, W.G.; Della Valle, C.J. Do Porous Tantalum Metaphyseal Cones Improve Outcomes in Revision Total Knee Arthroplasty? J. Arthroplast. 2018, 33, 171–177. [Google Scholar] [CrossRef]
- Rajgopal, A.; Kumar, S.; Aggarwal, K. Midterm Outcomes of Tantalum Metal Cones for Severe Bone Loss in Complex Primary and Revision Total Knee Arthroplasty. Arthroplast. Today 2021, 7, 76–83. [Google Scholar] [CrossRef]
- Rao, B.M.; Kamal, T.T.; Vafaye, J.; Moss, M. Tantalum cones for major osteolysis in revision knee replacement. Bone Jt. J. 2013, 95-B, 1069–1074. [Google Scholar] [CrossRef]
- Potter, G.D.; Abdel, M.P.; Lewallen, D.G.; Hanssen, A.D. Midterm Results of Porous Tantalum Femoral Cones in Revision Total Knee Arthroplasty. J. Bone Jt. Surg. 2016, 98, 1286–1291. [Google Scholar] [CrossRef]
- Girerd, D.; Parratte, S.; Lunebourg, A.; Boureau, F.; Ollivier, M.; Pasquier, G.; Putman, S.; Migaud, H.; Argenson, J. Total knee arthroplasty revision with trabecular tantalum cones: Preliminary retrospective study of 51 patients from two centres with a minimal 2-year follow-up. Orthop. Traumatol. Surg. Res. 2016, 102, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.M.; Bell, J.A.; Jung, E.K.; Sporer, S.M.; Paprosky, W.G.; Levine, B.R. The Use of Trabecular Metal Cones in Complex Primary and Revision Total Knee Arthroplasty. J. Arthroplast. 2015, 30, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Zahar, A.; Kendoff, D.O.; Klatte, T.O.; Gehrke, T.A. Can Good Infection Control Be Obtained in One-stage Exchange of the Infected TKA to a Rotating Hinge Design? 10-year Results. Clin. Orthop. Relat. Res. 2016, 474, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Abdelaziz, H.; Biewald, P.; Anastasiadis, Z.; Haasper, C.; Gehrke, T.; Hawi, N.; Citak, M. Midterm Results After Tantalum Cones in 1-Stage Knee Exchange for Periprosthetic Joint Infection: A Single-Center Study. J. Arthroplast. 2020, 35, 1084–1089. [Google Scholar] [CrossRef]
- Jacquet, C.; Ros, F.; Guy, S.; Parratte, S.; Ollivier, M.; Argenson, J.-N. Trabecular Metal Cones Combined With Short Cemented Stem Allow Favorable Outcomes in Aseptic Revision Total Knee Arthroplasty. J. Arthroplast. 2021, 36, 657–663. [Google Scholar] [CrossRef]
- Burastero, G.; Cavagnaro, L.; Chiarlone, F.; Alessio-Mazzola, M.; Carrega, G.; Felli, L. The Use of Tantalum Metaphyseal Cones for the Management of Severe Bone Defects in Septic Knee Revision. J. Arthroplast. 2018, 33, 3739–3745. [Google Scholar] [CrossRef]
- Spinello, P.; Thiele, R.A.R.; Zepeda, K.; Giori, N.; Indelli, P.F. The use of tantalum cones and diaphyseal-engaging stems in tibial component revision: A consecutive series. Knee Surg. Relat. Res. 2022, 34, 12. [Google Scholar] [CrossRef]
- Anderson, L.A.; Christie, M.; Blackburn, B.E.; Mahan, C.; Earl, C.; Pelt, C.E.; Peters, C.L.; Gililland, J. 3D-printed titanium metaphyseal cones in revision total knee arthroplasty with cemented and cementless stems. Bone Jt. J. 2021, 103-B, 150–157. [Google Scholar] [CrossRef]
- Rossi, S.M.P.; Perticarini, L.; Ghiara, M.; Jannelli, E.; Cortesi, L.; Benazzo, F. High survival rate at mid-term follow up of porous tantalum cones for bone defects in revision total knee replacement: A 3–11 years follow up report. Knee 2022, 35, 175–182. [Google Scholar] [CrossRef]
- Batinica, B.; Bolam, S.M.; D’Arcy, M.; Zhu, M.; Monk, A.P.; Munro, J.T. Tibial metaphyseal cones combined with short stems perform as well as long stems in revision total knee arthroplasty. ANZ J. Surg. 2022, 92, 2254–2260. [Google Scholar] [CrossRef]
- Ullmark, G.; Hovelius, L. Impacted morsellized allograft and cement for revision total knee arthroplasty: A preliminary report of 3 cases. Acta Orthop. Scand. 1996, 67, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Bedard, N.A.; Cates, R.A.; Lewallen, D.G.; Sierra, R.J.; Hanssen, A.D.; Berry, D.J.; Abdel, M.P. Outcomes of a technique combining diaphyseal impaction grafting and metaphyseal cones for severe bone loss in revision total knee arthroplasty. Bone Jt. J. 2020, 102-B, 116–122. [Google Scholar] [CrossRef] [PubMed]
- De Martino, I.; Mancino, F.; Di Matteo, V.; Singlitico, A.; Maccauro, G.; Gasparini, G. Tantalum Cones for Severe Bone Defects in Revision Knee Arthroplasty: A Minimum 10-Year Follow-Up. J. Arthroplast. 2022, 38, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Abdelaziz, H.; Jaramillo, R.; Gehrke, T.; Ohlmeier, M.; Citak, M. Clinical Survivorship of Aseptic Revision Total Knee Arthroplasty Using Hinged Knees and Tantalum Cones at Minimum 10-Year Follow-Up. J. Arthroplast. 2019, 34, 3018–3022. [Google Scholar] [CrossRef]
- Villanueva-Martínez, M.; De la Torre-Escudero, B.; Rojo-Manaute, J.M.; Ríos-Luna, A.; Chana-Rodriguez, F. Tantalum Cones in Revision Total Knee Arthroplasty. A Promising Short-Term Result With 29 Cones in 21 Patients. J. Arthroplast. 2013, 28, 988–993. [Google Scholar] [CrossRef]
- Gibon, E.; Vasilopoulos, T.; Sipavicius, E.; Deen, J.T.; Prieto, H.A.; Gray, C.F.; Parvataneni, H.K.; Pulido, L. Surgical Management of Tibial Bone Loss in Revision Total Knee Arthroplasty: Clinical Outcomes and Radiographic Analysis of Tantalum Cones, Titanium Cones and Titanium Sleeves. Iowa Orthop. J. 2022, 42, 145–153. [Google Scholar]
- Kotrych, D.; Marcinkowski, S.; Brodecki, A.; Anuszkiewicz, M.; Kleszowski, J.; Bohatyrewicz, A.; Ciechanowicz, D. Does the use of 3D-printed cones give a chance to postpone the use of megaprostheses in patients with large bone defects in the knee joint? Open Med. 2022, 17, 1292–1298. [Google Scholar] [CrossRef]
- Tarabichi, S.; Grau, L.; Arshi, A.; Post, Z.; Ong, A.; Hozack, W.J. Clinical and Radiographic Outcomes of Novel 3D-Printed Highly Porous Knee Cone Design. Surg. Technol. Int. 2022, 40, sti40/1563. [Google Scholar] [CrossRef]
- Remily, E.A.; Castrodad, I.M.D.; Mohamed, N.S.; Wilkie, W.A.; Kelemen, M.N.; Delanois, R.E. Short-term Outcomes of 3D-Printed Titanium Metaphyseal Cones in Revision Total Knee Arthroplasty. Orthopedics 2021, 44, 43–47. [Google Scholar] [CrossRef]
- Tetreault, M.W.; Perry, K.I.; Pagnano, M.W.; Hanssen, A.D.; Abdel, M.P. Excellent two-year survivorship of 3D-printed metaphyseal cones in revision total knee arthroplasty: A reliable and safe reamer-based system. Bone Jt. J. 2020, 102-B, 107–115. [Google Scholar] [CrossRef]
- Burastero, G.; Pianigiani, S.; Zanvettor, C.; Cavagnaro, L.; Chiarlone, F.; Innocenti, B. Use of porous custom-made cones for meta-diaphyseal bone defects reconstruction in knee revision surgery: A clinical and biomechanical analysis. Arch. Orthop. Trauma. Surg. 2020, 140, 2041–2055. [Google Scholar] [CrossRef] [PubMed]
- Faizan, A.; Bhowmik-Stoker, M.; Alipit, V.; Kirk, A.E.; Krebs, V.E.; Harwin, S.F.; Meneghini, R.M. Development and Verification of Novel Porous Titanium Metaphyseal Cones for Revision Total Knee Arthroplasty. J. Arthroplast. 2017, 32, 1946–1953. [Google Scholar] [CrossRef] [PubMed]
- Shichman, I.; Oakley, C.; Willems, J.H.; van Hellemondt, G.G.; Heesterbeek, P.; Rozell, J.; Marwin, S.; Schwarzkopf, R. Novel metaphyseal porous titanium cones allow favorable outcomes in revision total knee arthroplasty. Arch. Orthop. Trauma. Surg. 2022, 143, 1537–1547. [Google Scholar] [CrossRef] [PubMed]
- Behery, O.A.; Shing, E.Z.; Yu, Z.; Springer, B.D.; Fehring, T.K.; Otero, J.E. Survivorship and Radiographic Evaluation of Metaphyseal Cones With Short Cemented Stems in Revision Total Knee Arthroplasty. J. Arthroplast. 2022, 37, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Guntin, J.; Bartosiak, K.A.; Della Valle, C.J.; Patel, A.; Gerlinger, T.L.; Nam, D. Radiographic and clinical analysis of a Porous-coated metaphyseal cone for revision total Knee arthroplasty. Knee 2022, 37, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, X.; Tian, H. Reconstruction for Massive Proximal Tibial Bone Defects Using Patient-Customized Three-Dimensional-Printed Metaphyseal Cones in Revision Total Knee Arthroplasty. Orthop. Surg. 2022, 14, 1071–1077. [Google Scholar] [CrossRef]
- Mékidèche, T.; Armoiry, X.; Sautet, P.; Dussart, C.; Mandy, B. Étude de l’impact économique des cônes en métal poreux dans les arthroplasties totales du genou. Ann. Pharm. Fr. 2022, 80, 81–88. [Google Scholar] [CrossRef]
- Serino, J.; Burnett, R.A.; Boniello, A.J.; Yang, J.; Sporer, S.M.; Della Valle, C.J. The Cost-Effectiveness of Tibial Metaphyseal Cones in Revision Total Knee Arthroplasty. J. Arthroplast. 2022, 37, S50–S55. [Google Scholar] [CrossRef]
- Malik, H.H.; Darwood, A.R.; Shaunak, S.; Kulatilake, P.; El-Hilly, A.A.; Mulki, O.; Baskaradas, A. Three-dimensional printing in surgery: A review of current surgical applications. J. Surg. Res. 2015, 199, 512–522. [Google Scholar] [CrossRef]
- Migaud, H.; Common, H.; Girard, J.; Huten, D.; Putman, S. Acetabular reconstruction using porous metallic material in complex revision total hip arthroplasty: A systematic review. Orthop. Traumatol. Surg. Res. 2019, 5, S53–S61. [Google Scholar] [CrossRef]
- Ballard, D.H.; Mills, P.; Duszak, R.; Weisman, J.A.; Rybicki, F.J.; Woodard, P.K. Medical 3D Printing Cost-Savings in Orthopedic and Maxillofacial Surgery: Cost Analysis of Operating Room Time Saved with 3D Printed Anatomic Models and Surgical Guides. Acad. Radiol. 2020, 27, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Keskinis, A.; Paraskevopoulos, K.; Diamantidis, D.E.; Ververidis, A.; Fiska, A.; Tilkeridis, K. The Role of 3D-Printed Patient-Specific Instrumentation in Total Knee Arthroplasty: A Literature Review. Cureus 2023, 15, e43321. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).