Robotic Systems in Knee Surgery: Current Concepts and Future Perspectives
Abstract
1. Introduction
2. Robotic-Assisted Surgeries: How Many Different Options?
3. MAKO
3.1. Principles
3.2. Surgical Technique
3.2.1. Unicompartmental Knee Arthroplasty (UKA)
3.2.2. Total Knee Arthroplasty (TKA)
3.3. Outcomes and Cost-Effectiveness
4. NAVIO
4.1. Principles
4.2. Surgical Technique
4.2.1. Unicompartimental Knee Arthroplasty (UKA)
4.2.2. Total Knee Arthroplasty (TKA)
4.3. Outcomes and Cost-Effectiveness
5. ROSA
5.1. Principles
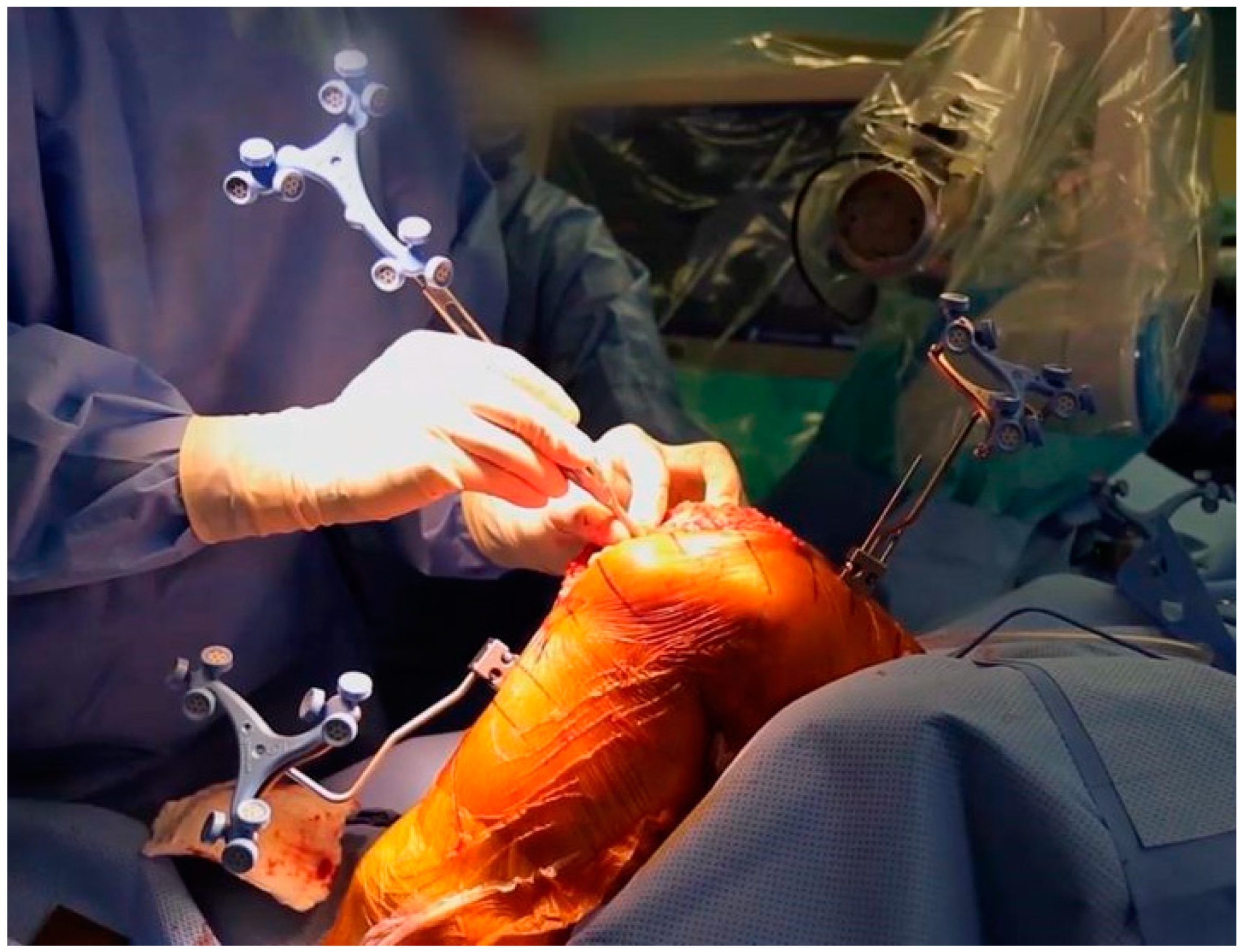
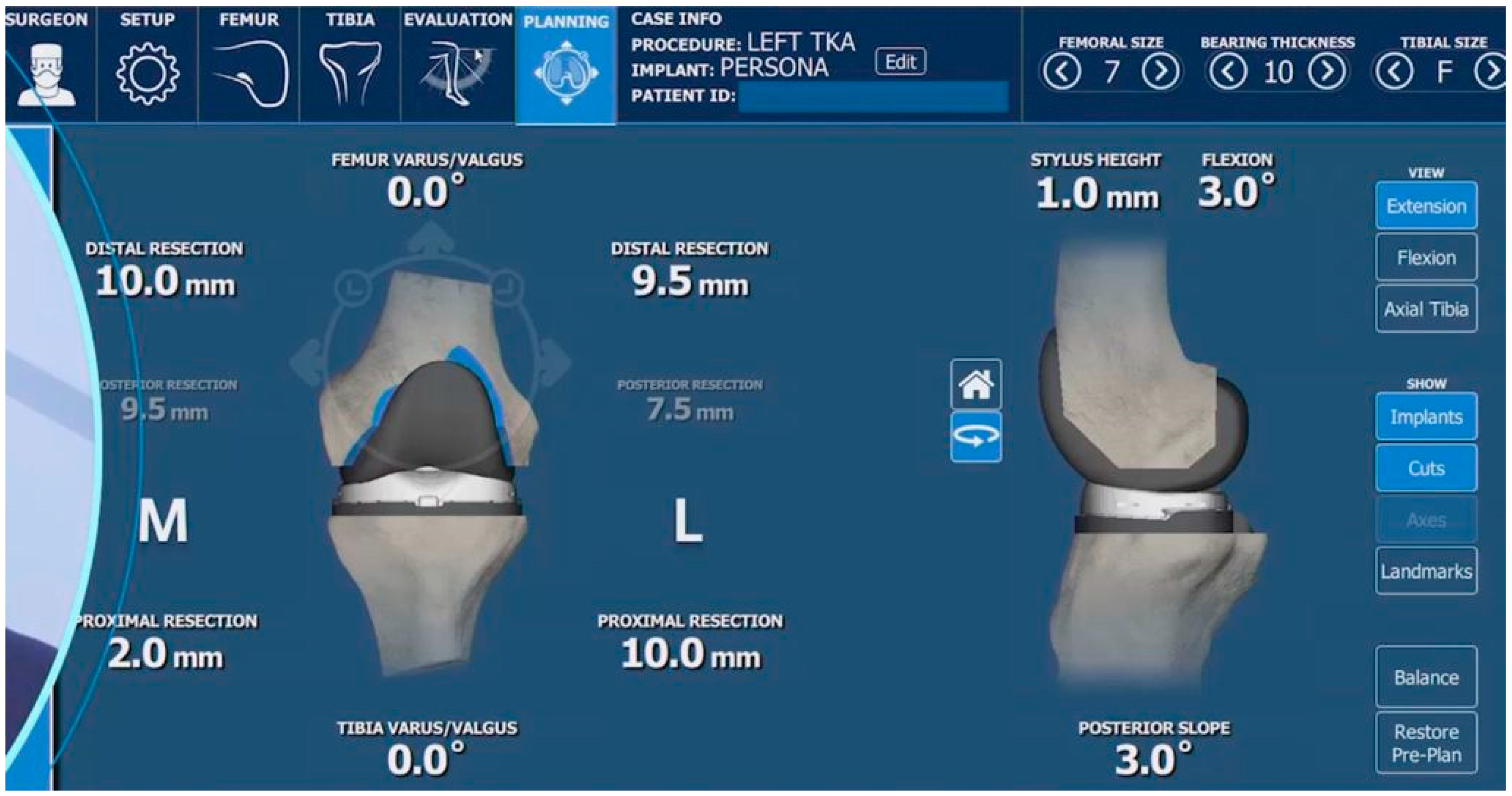
5.2. Surgical Technique for TKA
5.3. Outcomes and Cost-Effectiveness
6. Other Robotic Systems
6.1. OMNIbotics® System
6.2. CORI System
7. State of the Art and Future Perspectives
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Inacio, M.C.S.; Paxton, E.W.; Graves, S.E.; Namba, R.S.; Nemes, S. Projected increase in total knee arthroplasty in the United States—An alternative projection model. Osteoarthr. Cartil. 2017, 25, 1797–1803. [Google Scholar] [CrossRef]
- Sloan, M.; Premkumar, A.; Sheth, N.P. Projected Volume of Primary Total Joint Arthroplasty in the U.S., 2014 to 2030. J. Bone Jt. Surg. Am. 2018, 100, 1455–1460. [Google Scholar] [CrossRef]
- Home—The National Joint Registry. Available online: https://www.njrcentre.org.uk/ (accessed on 1 January 2023).
- Evans, J.T.; Walker, R.W.; Evans, J.P.; Blom, A.W.; Sayers, A.; Whitehouse, M.R. How long does a knee replacement last? A systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet 2019, 393, 655–663. [Google Scholar] [CrossRef]
- Vertullo, C.J.; Lewis, P.L.; Lorimer, M.; Graves, S.E. The Effect on Long-Term Survivorship of Surgeon Preference for Posterior-Stabilized or Minimally Stabilized Total Knee Replacement: An Analysis of 63,416 Prostheses from the Australian Orthopaedic Association National Joint Replacement Registry. J. Bone Jt. Surg. Am. 2017, 99, 1129–1139. [Google Scholar] [CrossRef]
- Scott, C.E.H.; Turnbull, G.S.; MacDonald, D.; Breusch, S.J. Activity levels and return to work following total knee arthroplasty in patients under 65 years of age. Bone Jt. J. 2017, 99-B, 1037–1046. [Google Scholar] [CrossRef]
- Luna, I.E.; Kehlet, H.; Peterson, B.; Wede, H.R.; Hoevsgaard, S.J.; Aasvang, E.K. Early patient-reported outcomes versus objective function after total hip and knee arthroplasty: A prospective cohort study. Bone Jt. J. 2017, 99-B, 1167–1175. [Google Scholar] [CrossRef]
- Kayani, B.; Konan, S.; Ayuob, A.; Onochie, E.; Al-Jabri, T.; Haddad, F.S. Robotic technology in total knee arthroplasty: A systematic review. EFORT Open Rev. 2019, 4, 611–617. [Google Scholar] [CrossRef]
- Batailler, C.; Fernandez, A.; Swan, J.; Servien, E.; Haddad, F.S.; Catani, F.; Lustig, S. MAKO CT-based robotic arm-assisted system is a reliable procedure for total knee arthroplasty: A systematic review. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 3585–3598. [Google Scholar] [CrossRef]
- Abdel, M.P.; Ledford, C.K.; Kobic, A.; Taunton, M.J.; Hanssen, A.D. Contemporary failure aetiologies of the primary, posterior-stabilised total knee arthroplasty. Bone Jt. J. 2017, 99-B, 647–652. [Google Scholar] [CrossRef]
- Kutzner, I.; Bender, A.; Dymke, J.; Duda, G.; von Roth, P.; Bergmann, G. Mediolateral force distribution at the knee joint shifts across activities and is driven by tibiofemoral alignment. Bone Jt. J. 2017, 99-B, 779–787. [Google Scholar] [CrossRef]
- Shatrov, J.; Parker, D. Computer and robotic—Assisted total knee arthroplasty: A review of outcomes. J. Exp. Orthop. 2020, 7, 70. [Google Scholar] [CrossRef]
- Chin, B.Z.; Tan, S.S.H.; Chua, K.C.X.; Budiono, G.R.; Syn, N.L.-X.; O’Neill, G.K. Robot-Assisted versus Conventional Total and Unicompartmental Knee Arthroplasty: A Meta-analysis of Radiological and Functional Outcomes. J. Knee Surg. 2021, 34, 1064–1075. [Google Scholar] [CrossRef]
- Haddad, F.S. Evolving techniques: The need for better technology. Bone Jt. J. 2017, 99-B, 145–146. [Google Scholar] [CrossRef]
- Kayani, B.; Haddad, F.S. Robotic total knee arthroplasty: Clinical outcomes and directions for future research. Bone Jt. Res. 2019, 8, 438–442. [Google Scholar] [CrossRef]
- Khan, M.; Osman, K.; Green, G.; Haddad, F.S. The epidemiology of failure in total knee arthroplasty: Avoiding your next revision. Bone Jt. J. 2016, 98-B, 105–112. [Google Scholar] [CrossRef]
- Hampp, E.L.; Chughtai, M.; Scholl, L.Y.; Sodhi, N.; Bhowmik-Stoker, M.; Jacofsky, D.J.; Mont, M.A. Robotic-Arm Assisted Total Knee Arthroplasty Demonstrated Greater Accuracy and Precision to Plan Compared with Manual Techniques. J. Knee Surg. 2019, 32, 239–250. [Google Scholar] [CrossRef]
- Jacofsky, D.J.; Allen, M. Robotics in Arthroplasty: A Comprehensive Review. J. Arthroplasty 2016, 31, 2353–2363. [Google Scholar] [CrossRef]
- Bautista, M.; Manrique, J.; Hozack, W.J. Robotics in Total Knee Arthroplasty. J. Knee Surg. 2019, 32, 600–606. [Google Scholar] [CrossRef]
- Sultan, A.A.; Samuel, L.T.; Khlopas, A.; Sodhi, N.; Bhowmik-Stoker, M.; Chen, A.; Orozco, F.; Kolisek, F.; Mahoney, O.; Smith, L.; et al. Robotic-Arm Assisted Total Knee Arthroplasty More Accurately Restored the Posterior Condylar Offset Ratio and the Insall-Salvati Index Compared to the Manual Technique; A Cohort-Matched Study. Surg. Technol. Int. 2019, 34, 409–413. [Google Scholar]
- Figueroa, F.; Parker, D.; Fritsch, B.; Oussedik, S. New and evolving technologies for knee arthroplasty—Computer navigation and robotics: State of the art. J. ISAKOS 2018, 3, 46–54. [Google Scholar] [CrossRef]
- Kayani, B.; Konan, S.; Pietrzak, J.R.T.; Haddad, F.S. Iatrogenic Bone and Soft Tissue Trauma in Robotic-Arm Assisted Total Knee Arthroplasty Compared With Conventional Jig-Based Total Knee Arthroplasty: A Prospective Cohort Study and Validation of a New Classification System. J. Arthroplast. 2018, 33, 2496–2501. [Google Scholar] [CrossRef]
- Khlopas, A.; Chughtai, M.; Hampp, E.L.; Scholl, L.Y.; Prieto, M.; Chang, T.-C.; Abbasi, A.; Bhowmik-Stoker, M.; Otto, J.; Jacofsky, D.J.; et al. Robotic-Arm Assisted Total Knee Arthroplasty Demonstrated Soft Tissue Protection. Surg. Technol. Int. 2017, 30, 441–446. [Google Scholar]
- Stryker. Mako™ Partial Knee | Surgical Reference Guide. Available online: https://www.strykermeded.com/media/2238/mako-partial-knee_medial-unicondylar-resurfacing_surgical-reference-guide.pdf. (accessed on 1 January 2016).
- Stryker. Mako TKA Surgical Guide. Available online: https://www.strykermeded.com/media/2223/mako-tka-surgical-guide.pdf (accessed on 1 January 2016).
- Marchand, R.C.; Sodhi, N.; Khlopas, A.; Sultan, A.A.; Harwin, S.F.; Malkani, A.L.; Mont, M.A. Patient Satisfaction Outcomes after Robotic Arm-Assisted Total Knee Arthroplasty: A Short-Term Evaluation. J. Knee Surg. 2017, 30, 849–853. [Google Scholar] [CrossRef]
- Mont, M.; Kinsey, T.; Zhang, J.; Bhowmik-Stoker, M.; Chen, A.; Orozco, F.; Hozack, W.; Mahoney, O. Robotic-assisted total knee arthroplasty demonstrates greater component placement accuracy compared with manual instrumentation: Initial results of a prospective multicentre evaluation. Orthop. Proc. 2020, 102-B, 433. [Google Scholar] [CrossRef]
- Bhimani, S.J.; Bhimani, R.; Smith, A.; Eccles, C.; Smith, L.; Malkani, A. Robotic-assisted total knee arthroplasty demonstrates decreased postoperative pain and opioid usage compared to conventional total knee arthroplasty. Bone Jt. Open 2020, 1, 8–12. [Google Scholar] [CrossRef]
- Sires, J.D.; Craik, J.D.; Wilson, C.J. Accuracy of Bone Resection in MAKO Total Knee Robotic-Assisted Surgery. J. Knee Surg. 2021, 34, 745–748. [Google Scholar] [CrossRef]
- Sires, J.D.; Wilson, C.J. CT Validation of Intraoperative Implant Position and Knee Alignment as Determined by the MAKO Total Knee Arthroplasty System. J. Knee Surg. 2021, 34, 1133–1137. [Google Scholar] [CrossRef]
- Marchand, R.C.; Khlopas, A.; Sodhi, N.; Condrey, C.; Piuzzi, N.S.; Patel, R.; Delanois, R.E.; Mont, M.A. Difficult Cases in Robotic Arm-Assisted Total Knee Arthroplasty: A Case Series. J. Knee Surg. 2018, 31, 27–37. [Google Scholar] [CrossRef]
- Kayani, B.; Konan, S.; Huq, S.S.; Tahmassebi, J.; Haddad, F.S. Robotic-arm assisted total knee arthroplasty has a learning curve of seven cases for integration into the surgical workflow but no learning curve effect for accuracy of implant positioning. Knee Surg. Sports Traumatol. Arthrosc. 2019, 27, 1132–1141. [Google Scholar] [CrossRef]
- Naziri, Q.; Cusson, B.C.; Chaudhri, M.; Shah, N.V.; Sastry, A. Making the transition from traditional to robotic-arm assisted TKA: What to expect? A single-surgeon comparative-analysis of the first-40 consecutive cases. J. Orthop. 2019, 16, 364–368. [Google Scholar] [CrossRef]
- Varughese, I.; Whitehouse, S.L.; Donnelly, W.J.; Crawford, R.W. The Cost Effectiveness of Unicompartmental versus Total Knee Arthroplasty. J. Knee Surg. 2022. [Google Scholar] [CrossRef]
- Stauss, R.; Savov, P.; Tuecking, L.-R.; Windhagen, H.; Ettinger, M. Robotic-assisted TKA reduces surgery duration, length of stay and 90-day complication rate of complex TKA to the level of noncomplex TKA. Arch. Orthop. Trauma Surg. 2022, 143, 3423–3430. [Google Scholar] [CrossRef]
- Marchand, R.C.; Sodhi, N.; Bhowmik-Stoker, M.; Scholl, L.; Condrey, C.; Khlopas, A.; Sultan, A.A.; Newman, J.M.; Mont, M.A. Does the Robotic Arm and Preoperative CT Planning Help with 3D Intraoperative Total Knee Arthroplasty Planning? J. Knee Surg. 2019, 32, 742–749. [Google Scholar] [CrossRef]
- Onggo, J.R.; Onggo, J.D.; De Steiger, R.; Hau, R. Robotic-assisted total knee arthroplasty is comparable to conventional total knee arthroplasty: A meta-analysis and systematic review. Arch. Orthop. Trauma Surg. 2020, 140, 1533–1549. [Google Scholar] [CrossRef]
- Marchand, R.C.; Sodhi, N.; Anis, H.K.; Ehiorobo, J.; Newman, J.M.; Taylor, K.; Condrey, C.; Hepinstall, M.S.; Mont, M.A. One-Year Patient Outcomes for Robotic-Arm-Assisted versus Manual Total Knee Arthroplasty. J. Knee Surg. 2019, 32, 1063–1068. [Google Scholar] [CrossRef]
- Agarwal, N.; To, K.; McDonnell, S.; Khan, W. Clinical and Radiological Outcomes in Robotic-Assisted Total Knee Arthroplasty: A Systematic Review and Meta-Analysis. J. Arthroplast. 2020, 35, 3393–3409.e2. [Google Scholar] [CrossRef]
- Cotter, E.J.; Wang, J.; Illgen, R.L. Comparative Cost Analysis of Robotic-Assisted and Jig-Based Manual Primary Total Knee Arthroplasty. J. Knee Surg. 2020, 35, 176–184. [Google Scholar] [CrossRef]
- Kayani, B.; Konan, S.; Tahmassebi, J.; Pietrzak, J.R.T.; Haddad, F.S. Robotic-arm assisted total knee arthroplasty is associated with improved early functional recovery and reduced time to hospital discharge compared with conventional jig-based total knee arthroplasty: A prospective cohort study. Bone Jt. J. 2018, 100-B, 930–937. [Google Scholar] [CrossRef]
- Marchand, K.B.; Moody, R.; Scholl, L.Y.; Bhowmik-Stoker, M.; Taylor, K.B.; Mont, M.A.; Marchand, R.C. Results of Robotic-Assisted Versus Manual Total Knee Arthroplasty at 2-Year Follow-up. J. Knee Surg. 2023, 36, 159–166. [Google Scholar] [CrossRef]
- NAVIO Surgical System—Smith & Nephew. Available online: http://www.smithnephewlivesurgery.com/navio-surgical-system (accessed on 1 March 2020).
- Zhang, J.; Ng, N.; Scott, C.E.H.; Blyth, M.J.G.; Haddad, F.S.; Macpherson, G.J.; Patton, J.T.; Clement, N.D. Robotic arm-assisted versus manual unicompartmental knee arthroplasty: A systematic review and meta-analysis of the MAKO robotic system. Bone Jt. J. 2022, 104-B, 541–548. [Google Scholar] [CrossRef]
- Cool, C.L.; Jacofsky, D.J.; Seeger, K.A.; Sodhi, N.; Mont, M.A. A 90-day episode-of-care cost analysis of robotic-arm assisted total knee arthroplasty. J. Comp. Eff. Res. 2019, 8, 327–336. [Google Scholar] [CrossRef]
- Collins, K.; Agius, P.A.; Fraval, A.; Petterwood, J. Initial Experience with the NAVIO Robotic-Assisted Total Knee Replacement-Coronal Alignment Accuracy and the Learning Curve. J. Knee Surg. 2022, 35, 1295–1300. [Google Scholar] [CrossRef]
- Bollars, P.; Boeckxstaens, A.; Mievis, J.; Janssen, D. The Learning Curve and Alignment Assessment of an Image-Free Handheld Robot in TKA: The First Patient Series in Europe. In EPiC Series in Health Sciences; EasyChair: Manchester, UK, 2019; Volume 3, pp. 49–52. [Google Scholar]
- Pastides, P.; Nathwani, D. The role of newer technologies in knee arthroplasty. Orthop. Trauma 2017, 31, 47–52. [Google Scholar] [CrossRef]
- Husain, A.; Lee, G.-C. Establishing Realistic Patient Expectations Following Total Knee Arthroplasty. J. Am. Acad. Orthop. Surg. 2015, 23, 707–713. [Google Scholar] [CrossRef]
- Sephton, B.M.; Shearman, A.; Nathwani, D. 24-hour discharge in unicompartmental knee arthroplasty using the navio◊ robotic system: A retrospective analysis. Orthop. Proc. 2021, 103-B, 26. [Google Scholar] [CrossRef]
- Lyu, H.; Wick, E.C.; Housman, M.; Freischlag, J.A.; Makary, M.A. Patient satisfaction as a possible indicator of quality surgical care. JAMA Surg. 2013, 148, 362–367. [Google Scholar] [CrossRef]
- Nherera, L.; Verma, S.; Trueman, P. MT1 cost-effectiveness of robotic-assisted unicompartmental knee arthroplasty (R-UKA)* compared to traditional unicompartmental knee arthroplasty (T-UKA). Value Health 2019, 22, S41. [Google Scholar] [CrossRef]
- Parratte, S.; Price, A.J.; Jeys, L.M.; Jackson, W.F.; Clarke, H.D. Accuracy of a New Robotically Assisted Technique for Total Knee Arthroplasty: A Cadaveric Study. J. Arthroplast. 2019, 34, 2799–2803. [Google Scholar] [CrossRef]
- ROSA- User Manual and Surgical Technique. Available online: https://www.zimmerbiomet.eu/content/dam/zb-corporate/en-eu/surgical-techniques/mymobility/2300.5-GLBL-en%20ROSA%20Knee.pdf (accessed on 17 June 2021).
- Batailler, C.; Hannouche, D.; Benazzo, F.; Parratte, S. Concepts and techniques of a new robotically assisted technique for total knee arthroplasty: The ROSA knee system. Arch. Orthop. Trauma Surg. 2021, 141, 2049–2058. [Google Scholar] [CrossRef]
- Klag, E.A.; Lizzio, V.A.; Charters, M.A.; Ayoola, A.S.; Wesemann, L.; Banka, T.R.; North, W.T. Increased Accuracy in Templating for Total Knee Arthroplasty Using 3D Models Generated from Radiographs. J. Knee Surg. 2023, 36, 837–842. [Google Scholar] [CrossRef]
- ROSA® Knee System Brochure. Available online: https://www.zimmerbiomet.com/content/dam/zimmer-biomet/medical-professionals/robotics/rosa-knee/2455.1-GLBL-en%20ROSA%20Knee%20Brochure-V7-OK.pdf (accessed on 1 January 2019).
- ROSA® Knee System detailed brochure. Available online: https://www.zimmerbiomet.com/content/dam/zb-corporate/en/products/specialties/knee/rosa-knee/3076.5-GLBL-en%20ROSA%20Knee%20Detailed%20Brochure.pdf (accessed on 1 January 2023).
- Seidenstein, A.; Birmingham, M.; Foran, J.; Ogden, S. Better accuracy and reproducibility of a new robotically-assisted system for total knee arthroplasty compared to conventional instrumentation: A cadaveric study. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 859–866. [Google Scholar] [CrossRef]
- Shin, C.; Crovetti, C.; Huo, E.; Lionberger, D. Unsatisfactory accuracy of recent robotic assisting system ROSA for total knee arthroplasty. J. Exp. Orthop. 2022, 9, 82. [Google Scholar] [CrossRef]
- Bolam, S.M.; Tay, M.L.; Zaidi, F.; Sidaginamale, R.P.; Hanlon, M.; Munro, J.T.; Monk, A.P. Introduction of ROSA robotic-arm system for total knee arthroplasty is associated with a minimal learning curve for operative time. J. Exp. Orthop. 2022, 9, 86. [Google Scholar] [CrossRef]
- Vanlommel, L.; Neven, E.; Anderson, M.B.; Bruckers, L.; Truijen, J. The initial learning curve for the ROSA® Knee System can be achieved in 6-11 cases for operative time and has similar 90-day complication rates with improved implant alignment compared to manual instrumentation in total knee arthroplasty. J. Exp. Orthop. 2021, 8, 119. [Google Scholar] [CrossRef]
- Smith, A.F.; Eccles, C.J.; Bhimani, S.J.; Denehy, K.M.; Bhimani, R.B.; Smith, L.S.; Malkani, A.L. Improved Patient Satisfaction following Robotic-Assisted Total Knee Arthroplasty. J. Knee Surg. 2021, 34, 730–738. [Google Scholar] [CrossRef]
- Leelasestaporn, C.; Tarnpichprasert, T.; Arirachakaran, A.; Kongtharvonskul, J. Comparison of 1-year outcomes between MAKO versus NAVIO robot-assisted medial UKA: Nonrandomized, prospective, comparative study. Knee Surg. Relat. Res. 2020, 32, 13. [Google Scholar] [CrossRef]
- Are, L.; De Mauro, D.; Rovere, G.; Fresta, L.; Tartarone, M.; Illuminati, A.; Smakaj, A.; Maccauro, G.; Liuzza, F. Robotic-assisted unicompartimental knee arthroplasty performed with Navio system: A systematic review. Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 2624–2633. [Google Scholar] [CrossRef]
- Turan, K.; Camurcu, Y.; Kezer, M.; Uysal, Y.; Kizilay, Y.O.; Temiz, A. Preliminary outcomes of kinematically aligned robot-assisted total knee arthroplasty with patient-specific cartilage thickness measurement. J. Robot. Surg. 2023, 17, 979–985. [Google Scholar] [CrossRef]
- Kenanidis, E.; Paparoidamis, G.; Milonakis, N.; Potoupnis, M.; Tsiridis, E. Comparative outcomes between a new robotically assisted and a manual technique for total knee arthroplasty in patients with osteoarthritis: A prospective matched comparative cohort study. Eur. J. Orthop. Surg. Traumatol. 2023, 33, 1231–1236. [Google Scholar] [CrossRef]
- Parratte, S.; Van Overschelde, P.; Bandi, M.; Ozturk, B.Y.; Batailler, C. An anatomo-functional implant positioning technique with robotic assistance for primary TKA allows the restoration of the native knee alignment and a natural functional ligament pattern, with a faster recovery at 6 months compared to an adjusted mechanical technique. Knee Surg. Sports Traumatol. Arthrosc. 2023, 31, 1334–1346. [Google Scholar] [CrossRef]
- Shatrov, J.; Murphy, G.T.; Duong, J.; Fritsch, B. Robotic-assisted total knee arthroplasty with the OMNIBot platform: A review of the principles of use and outcomes. Arch. Orthop. Trauma Surg. 2021, 141, 2087–2096. [Google Scholar] [CrossRef]
- OMNIBotics® Knee System–ARTTM KNEE Application. Available online: https://eifu.coringroup.com/downloads/revisions/1085/IFU-036%20REV%20K%20OMNIBotics%20Knee%20System%20-%20ART%20Knee%20Application%20IFU.pdf (accessed on 1 March 2021).
- CORI Surgical System|Smith+Nephew USA. Available online: https://www.smith-nephew.com/en-us/health-care-professionals/products/orthopaedics/cori#productfeatures (accessed on 1 January 2023).
- Vermue, H.; Batailler, C.; Monk, P.; Haddad, F.; Luyckx, T.; Lustig, S. The evolution of robotic systems for total knee arthroplasty, each system must be assessed for its own value: A systematic review of clinical evidence and meta-analysis. Arch. Orthop. Trauma Surg. 2023, 143, 3369–3381. [Google Scholar] [CrossRef]
- Hua, Y.; Salcedo, J. Cost-effectiveness analysis of robotic-arm assisted total knee arthroplasty. PLoS ONE 2022, 17, e0277980. [Google Scholar] [CrossRef]
- Tran, E.P.; Dingel, A.B.; Terhune, E.B.; Segovia, N.A.; Vuong, B.; Ganley, T.J.; Fabricant, P.D.; Green, D.W.; Stavinoha, T.J.; Shea, K.G. Anterior Cruciate Ligament Length in Pediatric Populations: An MRI Study. Orthop. J. Sports Med. 2021, 9, 23259671211002290. [Google Scholar] [CrossRef]
- Clement, N.D.; Deehan, D.J.; Patton, J.T. Robot-assisted unicompartmental knee arthroplasty for patients with isolated medial compartment osteoarthritis is cost-effective: A markov decision analysis. Bone Jt. J. 2019, 101-B, 1063–1070. [Google Scholar] [CrossRef]
- Rajan, P.V.; Khlopas, A.; Klika, A.; Molloy, R.; Krebs, V.; Piuzzi, N.S. The Cost-Effectiveness of Robotic-Assisted Versus Manual Total Knee Arthroplasty: A Markov Model-Based Evaluation. J. Am. Acad. Orthop. Surg. 2022, 30, 168–176. [Google Scholar] [CrossRef]
- DeFrance, M.J.; Yayac, M.F.; Courtney, P.M.; Squire, M.W. The Impact of Author Financial Conflicts on Robotic-Assisted Joint Arthroplasty Research. J. Arthroplasty 2021, 36, 1462–1469. [Google Scholar] [CrossRef]
- Kochanski, R.B.; Lombardi, J.M.; Laratta, J.L.; Lehman, R.A.; O’Toole, J.E. Image-Guided Navigation and Robotics in Spine Surgery. Neurosurgery 2019, 84, 1179–1189. [Google Scholar] [CrossRef]
- Zheng, G.; Nolte, L.-P. Computer-Aided Orthopaedic Surgery: State-of-the-Art and Future Perspectives. Adv. Exp. Med. Biol. 2018, 1093, 1–20. [Google Scholar] [CrossRef]
- Jivraj, J.; Deorajh, R.; Lai, P.; Chen, C.; Nguyen, N.; Ramjist, J.; Yang, V.X.D. Robotic laser osteotomy through penscriptive structured light visual servoing. Int. J. Comput. Assist. Radiol. Surg. 2019, 14, 809–818. [Google Scholar] [CrossRef]
- Lo, W.N.; Cheung, K.W.; Yung, S.H.; Chiu, K.H. Arthroscopy-assisted computer navigation in high tibial osteotomy for varus knee deformity. J. Orthop. Surg. 2009, 17, 51–55. [Google Scholar] [CrossRef]
- Saragaglia, D.; Chedal-Bornu, B.; Rouchy, R.C.; Rubens-Duval, B.; Mader, R.; Pailhé, R. Role of computer-assisted surgery in osteotomies around the knee. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3387–3395. [Google Scholar] [CrossRef]
- Bae, D.K.; Ko, Y.W.; Kim, S.J.; Baek, J.H.; Song, S.J. Computer-assisted navigation decreases the change in the tibial posterior slope angle after closed-wedge high tibial osteotomy. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3433–3440. [Google Scholar] [CrossRef]
- Zaffagnini, S.; Urrizola, F.; Signorelli, C.; Grassi, A.; Di Sarsina, T.R.; Lucidi, G.A.; Marcheggiani Muccioli, G.M.; Bonanzinga, T.; Marcacci, M. Current use of navigation system in ACL surgery: A historical review. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3396–3409. [Google Scholar] [CrossRef]
- Figueroa, F.; Figueroa, D.; Guiloff, R.; Putnis, S.; Fritsch, B.; Itriago, M. Navigation in anterior cruciate ligament reconstruction: State of the art. J. ISAKOS 2023, 8, 47–53. [Google Scholar] [CrossRef]
- Anthony, C.A.; Duchman, K.; McCunniff, P.; McDermott, S.; Bollier, M.; Thedens, D.R.; Wolf, B.R.; Albright, J.P. Double-bundle ACL reconstruction: Novice surgeons utilizing computer-assisted navigation versus experienced surgeons. Comput. Aided Surg. 2013, 18, 172–180. [Google Scholar] [CrossRef][Green Version]
- Ikuta, Y.; Nakamae, A.; Shimizu, R.; Ishikawa, M.; Nakasa, T.; Ochi, M.; Adachi, N. A Comparison of Central Anatomic Single-Bundle Reconstruction and Anatomic Double-Bundle Reconstruction in Anteroposterior and Rotational Knee Stability: Intraoperative Biomechanical Evaluation. J. Knee Surg. 2022, 35, 273–279. [Google Scholar] [CrossRef]
- Margier, J.; Tchouda, S.D.; Banihachemi, J.-J.; Bosson, J.-L.; Plaweski, S. Computer-assisted navigation in ACL reconstruction is attractive but not yet cost efficient. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 1026–1034. [Google Scholar] [CrossRef]
- Foo, W.Y.X.; Chou, A.C.C.; Lie, H.M.; Lie, D.T.T. Computer-assisted navigation in ACL reconstruction improves anatomic tunnel placement with similar clinical outcomes. Knee 2022, 38, 132–140. [Google Scholar] [CrossRef]
- Koulalis, D.; Di Benedetto, P.; Citak, M.; O’Loughlin, P.; Pearle, A.D.; Kendoff, D.O. Comparative study of navigated versus freehand osteochondral graft transplantation of the knee. Am. J. Sports Med. 2009, 37, 803–807. [Google Scholar] [CrossRef]
- Di Benedetto, P.; Citak, M.; Kendoff, D.; O’Loughlin, P.F.; Suero, E.M.; Pearle, A.D.; Koulalis, D. Arthroscopic mosaicplasty for osteochondral lesions of the knee: Computer-assisted navigation versus freehand technique. Arthroscopy 2012, 28, 1290–1296. [Google Scholar] [CrossRef]
- Young, S.W.; Safran, M.R.; Clatworthy, M. Applications of computer navigation in sports medicine knee surgery: An evidence-based review. Curr. Rev. Musculoskelet. Med. 2013, 6, 150–157. [Google Scholar] [CrossRef]
- Zaleski, M.; Hodel, S.; Fürnstahl, P.; Vlachopoulos, L.; Fucentese, S.F. Osteochondral Allograft Reconstruction of the Tibia Plateau for Posttraumatic Defects-A Novel Computer-Assisted Method Using 3D Preoperative Planning and Patient-Specific Instrumentation. Surg. J. 2021, 7, e289–e296. [Google Scholar] [CrossRef]
- Peng, X.; Wang, F.; Su, J. The value of computer-assisted navigation for bone reconstruction after tumor resection. Oncol. Lett. 2017, 14, 2771–2774. [Google Scholar] [CrossRef]
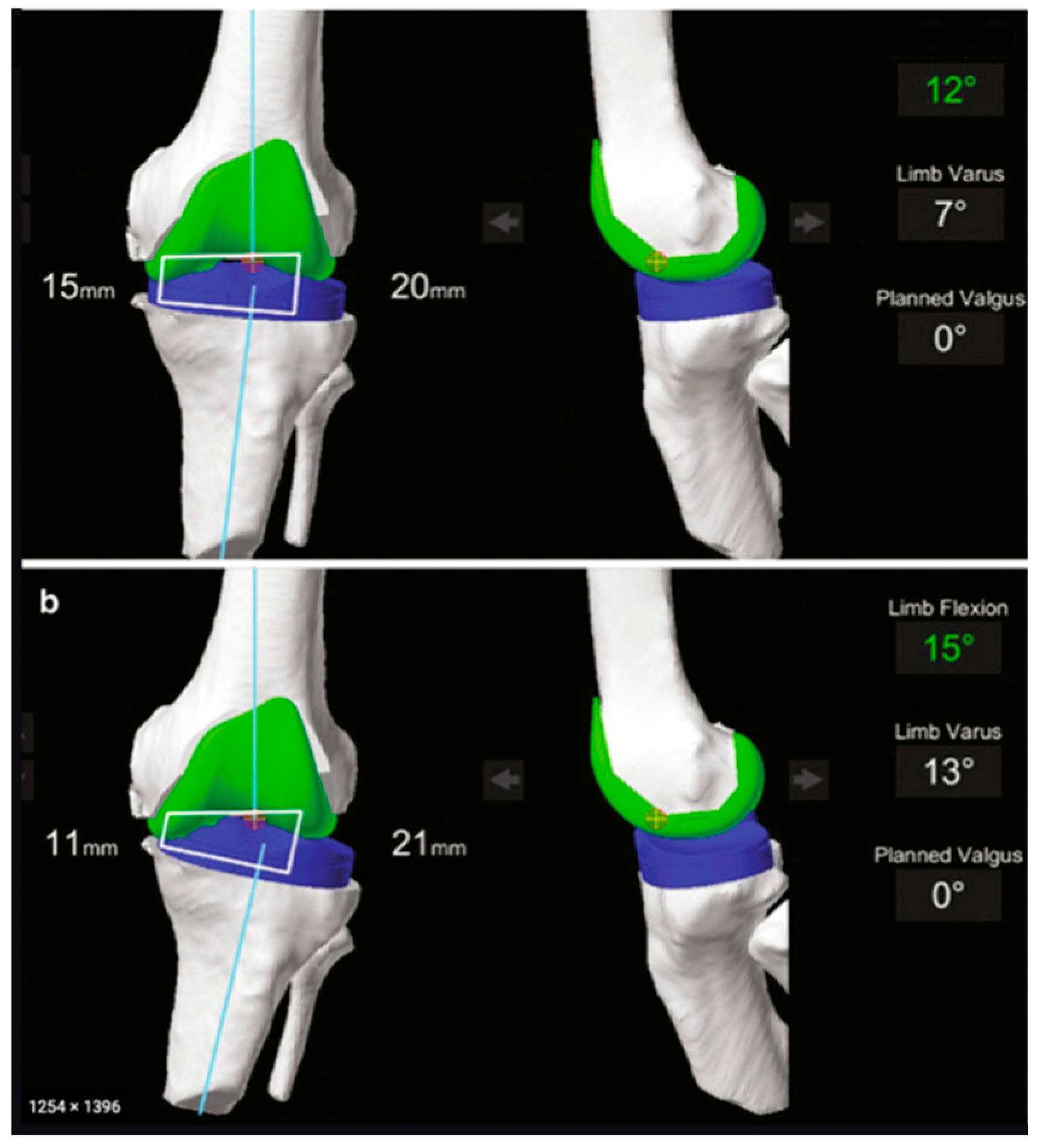
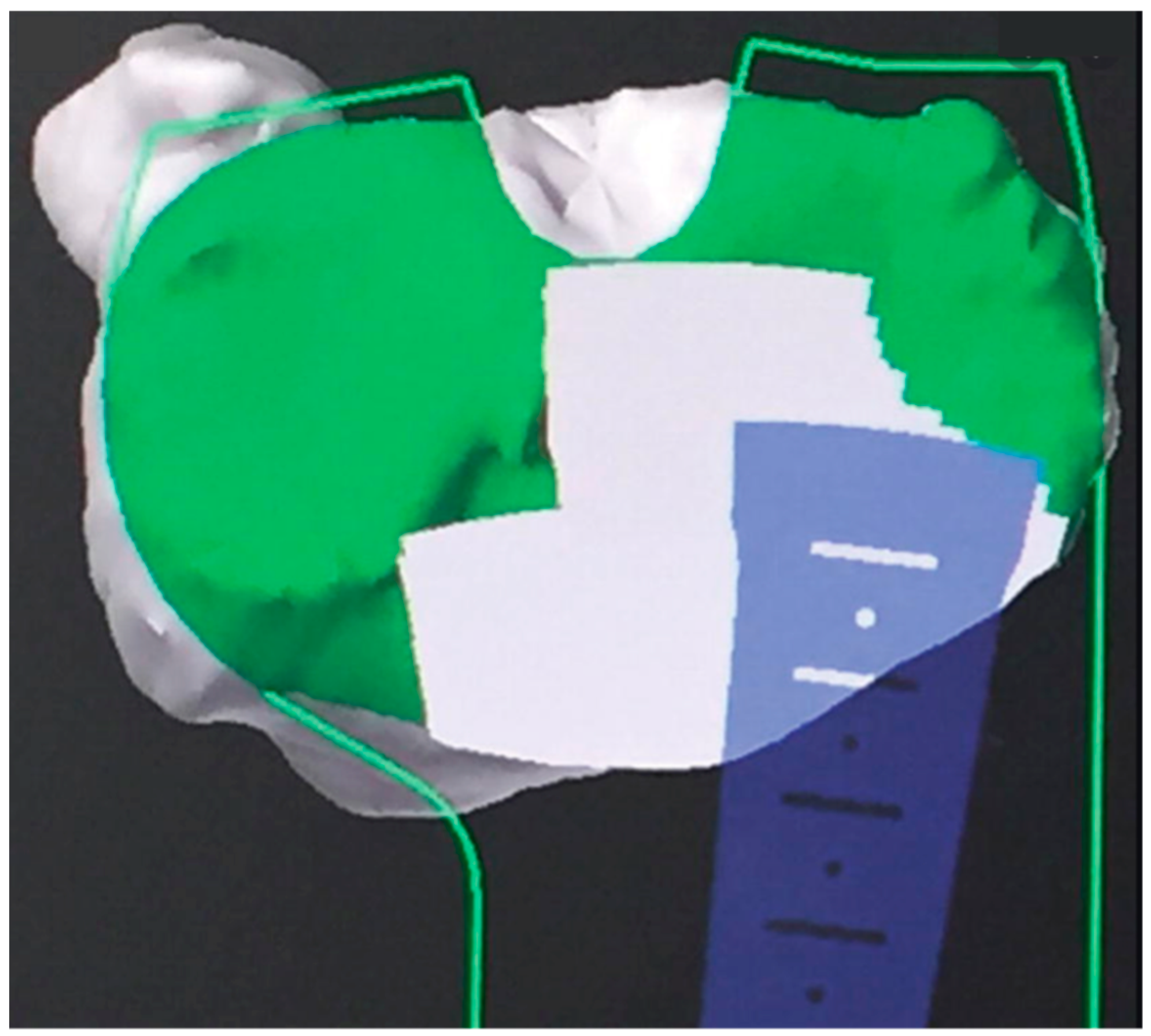
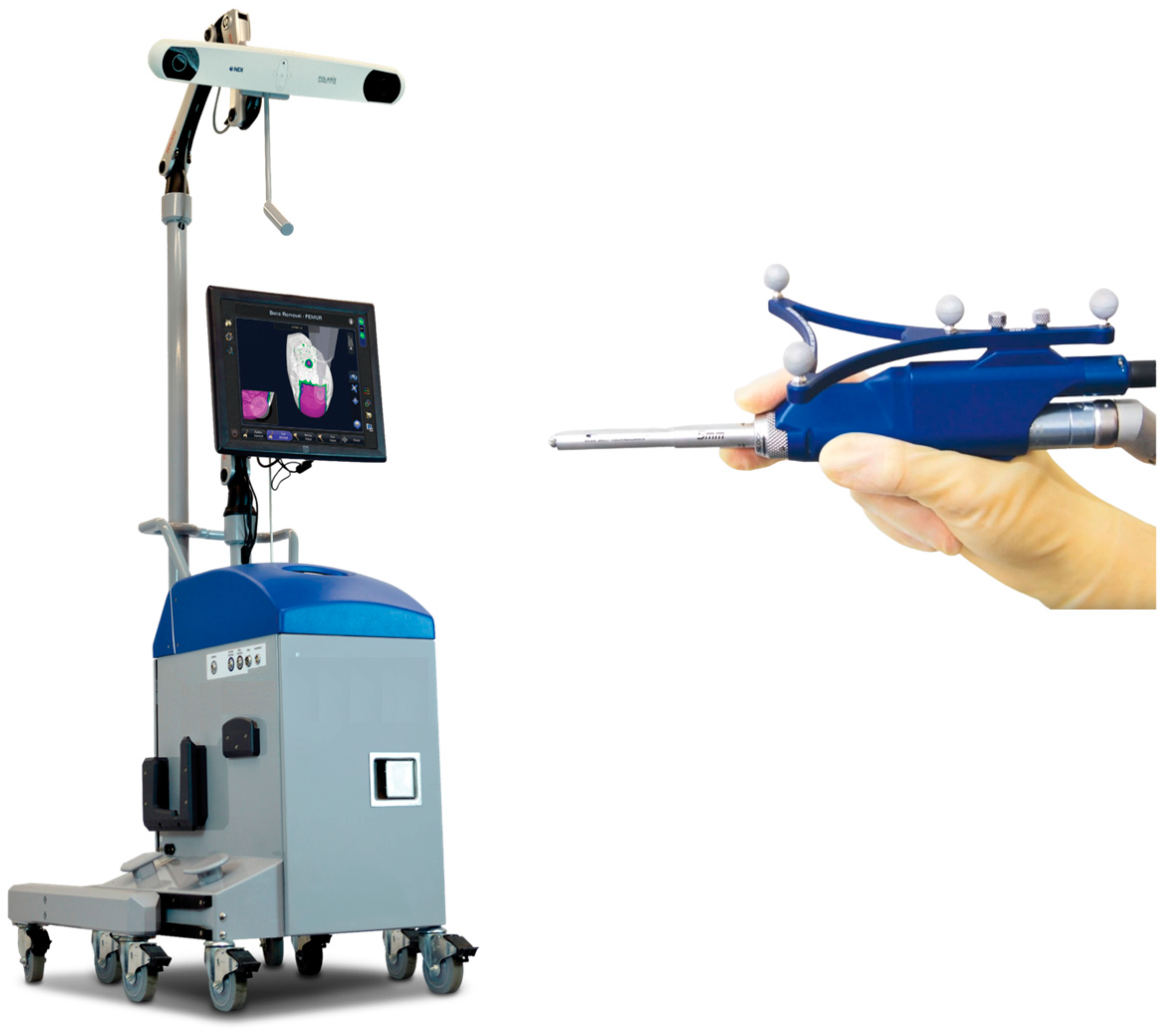
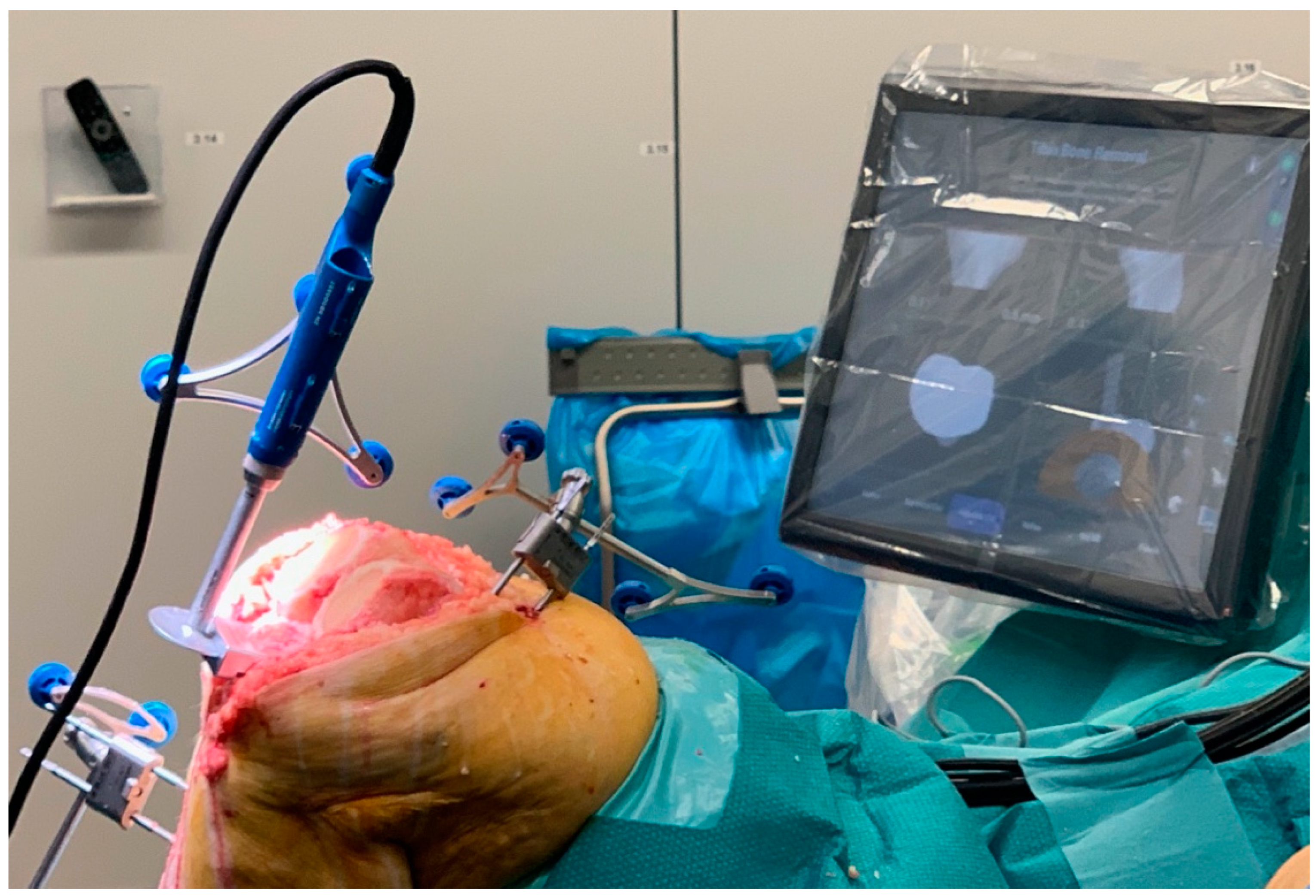

| MAKO® (Stryker Ltd., Kalamazoo, MI, USA) | NAVIO® (Smith & Nephew, Andover, TX, USA) | ROSA® (Zimmer Inc., Warsaw, IN, USA) | |
|---|---|---|---|
| level of surgeon involvement | Semi-active | Semi-active | Semi-active |
| image-based | CT scan | Imageless | X-rays (imageless possible) |
| UKA or TKA | Both | Both | TKA |
| implant choice | Closed platform-KINETIS® implant system (MAKO Surgical Corp., Ft. Lauderdale, FL, USA), TRIATHLON® (Stryker Ltd., Kalamazoo, MI, USA | Open platform | Closed platform-Persona® (Zimmer Inc., Warsaw, IN, USA), Vanguard® (Zimmer Inc., Warsaw, IN, USA) and Nexgen implants® (Zimmer Inc., Warsaw, IN, USA) |
| workflows | 1. Measured resections 2. Gap balancing | Only measured resection | 1. Measured resections 2. Gap balancing 3. Hybrid |
| bone cuts | Saw directly assembled on the system | Saw or handheld burr | The surgeon holds the external saw, and the system controls the cutting guides. |
| approval date | 2008 | 2012 | 2019 |
| MAKO® (Stryker Ltd., Kalamazoo, MI, USA) | NAVIO® (Smith & Nephew, Andover, TX, USA) | ROSA® (Zimmer Inc., Warsaw, IN, USA) | |
|---|---|---|---|
| ROM | 119° at 1 year PO | 130.3° at 1 year P.O. | 135.8 ± 10.2 at 1 year P.O. |
| satisfaction | 94% (vs. 82% cTKA) at 1 year P.O. | 81.8–82% at 1 year P.O. ARE | 95% vs. 92.5% (cTKA) at 1 year P.O. |
| womac | WOMAC score: 6 ± 6 vs. 9 ± 8 (cTKA, p < 0.05) | 87.05 ± 7.74 vs. 81.76 ± 8.95 cTKA (p < 0.0001) ADAMSKA CORI | N/A |
| KSS | F-KSS:80 (vs. 73 cTKA) at 1 year P.O. | F-KSS: 92.8–99.9 at 1 year P.O. | F-KSS 84.6 ± 15 vs. 79.1 ± 19 at 1 year P.O. |
| K-KSS:85 (vs. 82 cTKA)at 1 year P.O. | K-KSS: 91.9–96.9 at 1 year P.O. | K-KSS 92.3 ± 10 vs. 93.2 ± 6 at 1 year P.O. | |
| post operative vas pain score | VAS 2.6 (vs. 3.5–4.5 cTKA) at 6 weeks P.O. | VAS 2.5 ± 1.2 1 year P.O. TURAN | VAS 1 vs. 2 (cTKA) at 6 months |
| complications | no difference rTKA vs. cTKA | no difference rTKA vs. cTKA ARE | no difference rTKA vs. cTKA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantivalli, A.; Cottino, U.; Bonasia, D.E.; Rosso, F.; Rossi, R. Robotic Systems in Knee Surgery: Current Concepts and Future Perspectives. Prosthesis 2023, 5, 1257-1274. https://doi.org/10.3390/prosthesis5040086
Cantivalli A, Cottino U, Bonasia DE, Rosso F, Rossi R. Robotic Systems in Knee Surgery: Current Concepts and Future Perspectives. Prosthesis. 2023; 5(4):1257-1274. https://doi.org/10.3390/prosthesis5040086
Chicago/Turabian StyleCantivalli, Antonino, Umberto Cottino, Davide Edoardo Bonasia, Federica Rosso, and Roberto Rossi. 2023. "Robotic Systems in Knee Surgery: Current Concepts and Future Perspectives" Prosthesis 5, no. 4: 1257-1274. https://doi.org/10.3390/prosthesis5040086
APA StyleCantivalli, A., Cottino, U., Bonasia, D. E., Rosso, F., & Rossi, R. (2023). Robotic Systems in Knee Surgery: Current Concepts and Future Perspectives. Prosthesis, 5(4), 1257-1274. https://doi.org/10.3390/prosthesis5040086






