Effect of Parmigiano Reggiano Consumption on Blood Pressure of Spontaneous Hypertensive Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Determination of PR Cheese Chemical Composition and Mineral Elements Content
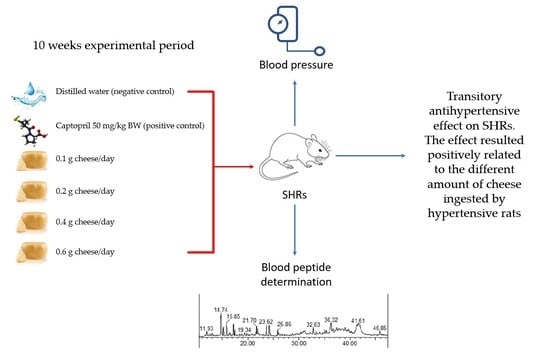
2.2. Animals and Experimental Design
2.3. Blood Pressure Measurements
2.4. Blood Collection
2.5. Biochemical Determinations
2.5.1. Reagents and Solvents
2.5.2. Peptide Synthesis
2.6. UHPLC/ESI-MS/MS Analysis
2.7. Statistical Analyses
3. Results
3.1. Body Weight
3.2. Blood Pressures
3.3. Biochemical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sánchez, A.; Vázquez, A. Bioactive peptides: A review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Bhandari, D.; Rafiq, S.; Gat, Y.; Gat, P.; Waghmare, R.; Kumar, V. A Review on Bioactive Peptides: Physiological Functions, Bioavailability and Safety. Int. J. Pept. Res. Ther. 2019, 26, 139–150. [Google Scholar] [CrossRef]
- Bhat, Z.F.; Kumar, S.; Bhat, H.F. Bioactive peptides of animal origin: A review. J. Food Sci. Technol. 2015, 52, 5377–5392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beltrán-Barrientos, L.M.; Hernández-Mendoza, A.; Torres-Llanez, M.J.; González-Córdova, A.F.; Vallejo-Córdoba, B. Invited review: Fermented milk as antihypertensive functional food. J. Dairy Sci. 2016, 99, 4099–4110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daliri, E.B.M.; Oh, D.H.; Lee, B.H. Bioactive peptides. Foods 2017, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Bello-Pérez, E.; Márquez-Hernández, R.I.; Hernández-Castellano, L.E. Bioactive peptides from milk: Animal determinants and their implications in human health. J. Dairy Res. 2019, 86, 136–144. [Google Scholar] [CrossRef] [Green Version]
- Kearney, P.M.; Whelton, M.; Reynolds, K.; Muntner, P.; Whelton, P.K.; He, J. Global burden of hypertension: Analysis of worldwide data. Lancet 2005, 365, 217–223. [Google Scholar] [CrossRef]
- Danaei, G.; Lu, Y.; Singh, G.; Carnahan, E.; Stevens, G.A.; Cowan, M.J.; Farzadfar, F.; Lin, J.K.; Finucane, M.M.; Rao, M.; et al. Cardiovascular disease, chronic kidney disease, and diabetes mortality burden of cardiometabolic risk factors from 1980 to 2010: A comparative risk assessment Global burden. Lancet Diabetes Endocrinol. 2014, 2, 634–647. [Google Scholar]
- Hamrahian, S.M. Management of Hypertension in Patients with Chronic Kidney Disease. Curr. Hypertens. Rep. 2017, 19, 43. [Google Scholar] [CrossRef]
- Jäkälä, P.; Vapaatalo, H. Antihypertensive Peptides from Milk Proteins. Pharmaceuticals 2010, 3, 251–272. [Google Scholar] [CrossRef] [Green Version]
- Rafiq, S.; Gulzar, N.; Sameen, A.; Huma, N.; Hayat, I.; Ijaz, R. Functional role of bioactive peptides with special reference to cheeses. Int. J. Dairy Technol. 2020, 74, 1–16. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yamamoto, N.; Sakai, K.; Takano, T. Antihypertensive Effect of Sour Milk and Peptides Isolated from It That are Inhibitors to Angiotensin I-Converting Enzyme. J. Dairy Sci. 1995, 78, 1253–1257. [Google Scholar] [CrossRef]
- Quirós, A.; Ramos, M.; Muguerza, B.; Delgado, M.A.; Miguel, M.; Aleixandre, A.; Recio, I. Identification of novel antihypertensive peptides in milk fermented with Enterococcus faecalis. Int. Dairy J. 2007, 17, 33–41. [Google Scholar] [CrossRef]
- Stuknytė, M.; Cattaneo, S.; Masotti, F.; De Noni, I. Occurrence and fate of ACE-inhibitor peptides in cheeses and in their digestates following in vitro static gastrointestinal digestion. Food Chem. 2015, 168, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Basiricò, L.; Catalani, E.; Morera, P.; Cattaneo, S.; Stuknytė, M.; Bernabucci, U.; De Noni, I.; Nardone, A. Release of angiotensin converting enzyme-inhibitor peptides during in vitro gastrointestinal digestion of Parmigiano Reggiano PDO cheese and their absorption through an in vitro model of intestinal epithelium. J. Dairy Sci. 2015, 98, 7595–7601. [Google Scholar] [CrossRef] [Green Version]
- Majumder, K.; Wu, J. Molecular Targets of Antihypertensive Peptides: Understanding the Mechanisms of Action Based on the Pathophysiology of Hypertension. Int. J. Mol. Sci. 2014, 16, 256–283. [Google Scholar] [CrossRef] [Green Version]
- Cicero, A.F.; Colletti, A.; Rosticci, M.; Cagnati, M.; Urso, R.; Giovannini, M.; Borghi, C.; D’Addato, S. Effect of lactotripeptides isoleucine-proline-proline/ valine-proline-proline on blood pressure and arterial stiffness changes in subjects with suboptimal blood pressure control and metabolic syndrome: A double-blind, randomized, crossover clinical trial. Metab. Syndr. Relat. Disord. 2016, 14, 161–166. [Google Scholar] [CrossRef]
- Smacchi, E.; Gobbetti, M. Peptides from several italian cheeses inhibitory to proteolytic enzymes of lactic acid bacteria, pseudomonas fluorescens ATCC 948 and to the angiotensin I-converting enzyme. Enzym. Microb. Technol. 1998, 22, 687–694. [Google Scholar] [CrossRef]
- Bernabucci, U.; Catalani, E.; Basiricò, L.; Morera, P.; Nardone, A. In vitro ACE-inhibitory activity and in vivo antihypertensive effects of water-soluble extract by Parmigiano Reggiano and Grana Padano cheeses. Int. Dairy J. 2014, 37, 16–19. [Google Scholar] [CrossRef]
- Gómez-Ruiz, J.Á.; Taborda, G.; Amigo, L.; Recio, I.; Ramos, M. Identification of ACE-inhibitory peptides in different Spanish cheeses by tandem mass spectrometry. Eur. Food Res. Technol. 2006, 223, 595–601. [Google Scholar] [CrossRef]
- Saito, T.; Nakamura, T.; Kitazawa, H.; Kawai, Y.; Itoh, T. Isolation and Structural Analysis of Antihypertensive Peptides That Exist Naturally in Gouda Cheese. J. Dairy Sci. 2000, 83, 1434–1440. [Google Scholar] [CrossRef]
- Ong, L.; Shah, N.P. Influence of probiotic Lactobacillus acidophilus and Lactobacillus helveticus on proteolysis, organic acid profiles, and ACE-inhibitory activity of cheddar cheeses ripened at 4, 8, and 12 °C. J. Food Sci. 2008, 73, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Bütikofer, U.; Meyer, J.; Sieber, R.; Walther, B.; Wechsler, D. Occurrence of the Angiotensin-Converting Enzyme–Inhibiting Tripeptides Val-Pro-Pro and Ile-Pro-Pro in Different Cheese Varieties of Swiss Origin. J. Dairy Sci. 2008, 91, 29–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pripp, A.H.; Sørensen, R.; Stepaniak, L.; Sørhaug, T. Relationship between proteolysis and angiotensin-I-converting enzyme inhibition in different cheeses. LWT 2006, 39, 677–683. [Google Scholar] [CrossRef]
- Meyer, J.; Bütikofer, U.; Walther, B.; Wechsler, D.; Sieber, R. Hot topic: Changes in angiotensin-converting enzyme inhibition and concentrations of the tripeptides Val-Pro-Pro and Ile-Pro-Pro during ripening of different Swiss cheese varieties. J. Dairy Sci. 2009, 92, 826–836. [Google Scholar] [CrossRef] [Green Version]
- Sieber, R.; Bütikofer, U.; Egger, C.; Portmann, R.; Walther, B.; Wechsler, D. ACE-inhibitory activity and ACE-inhibiting peptides in different cheese varieties. Dairy Sci. Technol. 2009, 90, 47–73. [Google Scholar] [CrossRef]
- Bottesini, C.; Paolella, S.; Lambertini, F.; Galaverna, G.; Tedeschi, T.; Dossena, A.; Marchelli, R.; Sforza, S. Antioxidant capacity of water soluble extracts from Parmigiano-Reggiano cheese. Int. J. Food Sci. Nutr. 2013, 64, 953–958. [Google Scholar] [CrossRef]
- Parrot, S.; Degraeve, P.; Curia, C.; Martial-Gros, A. In vitro study on digestion of peptides in Emmental cheese: Analytical evaluation and influence on angiotensin I converting enzyme inhibitory peptides. Nahrung/Food 2003, 47, 87–94. [Google Scholar] [CrossRef]
- Sforza, S.; Cavatorta, V.; Lambertini, F.; Galaverna, G.; Dossena, A.; Marchelli, R. Cheese peptidomics: A detailed study on the evolution of the oligopeptide fraction in Parmigiano-Reggiano cheese from curd to 24 months of aging. J. Dairy Sci. 2012, 95, 3514–3526. [Google Scholar] [CrossRef] [Green Version]
- Summer, A.; Formaggioni, P.; Franceschi, P.; Di Frangia, F.; Righi, F.; Malacarne, M. Cheese as Functional Food: The Example of Parmigiano Reggiano and Grana Padano. Food Technol. Biotechnol. 2017, 55, 277–289. [Google Scholar] [CrossRef]
- AOAC. Method 920.123. In Official Methods of Analysis, 16th ed.; AOAC International: Gaithersburg, MD, USA, 1995. [Google Scholar]
- AOAC. Method 933.05. In Official Methods of Analysis (2012), 19th ed.; AOAC International: Gaithersburg, MD, USA, 2012. [Google Scholar]
- Bradley, R.L.; Vanderwarn, M.A. Determination of Moisture in Cheese and Cheese Products. J. AOAC Int. 2001, 84, 570–592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- AOAC. Method 935.42. Ash of Cheese. In Official Methods of Analysis, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2000. [Google Scholar]
- AOAC. Method 985.35. In Official Methods of Analysis, 15th ed.; AOAC International: Washington, DC, USA, 1990. [Google Scholar]
- Bianchi, G.; Ferrari, P.; Cusi, D.; Salardi, S.; Guidi, E.; Niutta, E.; Tripodi, G. Genetic and Experimental Hypertension in the Animal Model-Similarities and Dissimilarities to the Development of Human Hypertension. J. Cardiovasc. Pharmacol. 1986, 8, S64–S70. [Google Scholar] [CrossRef] [PubMed]
- Praagman, J.; Franco, O.H.; Ikram, M.A.; Soedamah-Muthu, S.S.; Engberink, M.F.; Van Rooij, F.J.; Hofman, A.; Geleijnse, J.M. Dairy products and the risk of stroke and coronary heart disease: The Rotterdam Study. Eur. J. Nutr. 2015, 54, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, B.J.H.; Gruppen, H. Prediction of Molar Extinction Coefficients of Proteins and Peptides Using UV Absorption of the Constituent Amino Acids at 214 nm To Enable Quantitative Reverse Phase High-Performance Liquid Chromatography−Mass Spectrometry Analysis. J. Agric. Food Chem. 2007, 55, 5445–5451. [Google Scholar] [CrossRef]
- Foltz, M.; van der Pijl, P.C.; Duchateau, G.S.M.J.E. Current In Vitro Testing of Bioactive Peptides Is Not Valuable. J. Nutr. 2010, 140, 117–118. [Google Scholar] [CrossRef] [Green Version]
- Costa, V.A.; Vianna, L.M.; Aguila, M.B.; Mandarim-De-Lacerda, C.A. Alpha-tocopherol supplementation favorable effects on blood pressure, blood viscosity and cardiac remodeling of spontaneously hypertensive rats. J. Nutr. Biochem. 2005, 16, 251–256. [Google Scholar] [CrossRef]
- Boelsma, E.; Kloek, J. Lactotripeptides and antihypertensive effects: A critical review. Br. J. Nutr. 2008, 101, 776–786. [Google Scholar] [CrossRef] [Green Version]
- Engberink, M.F.; Schouten, E.; GKok, F.J.; van Mierlo, L.A.J.; Brouwer, I.A.; Geleijnse, J.M. Lactotripeptides show no effect on human blood pressure Results from a double-blind randomized controlled trial. Hypertension 2008, 51, 399–405. [Google Scholar] [CrossRef]
- FitzGerald, R.J.; Meisel, H. Milk protein-derived peptide inhibitors of angiotensin-I-converting enzyme. Br. J. Nutr. 2000, 84, S33–S37. [Google Scholar] [CrossRef] [Green Version]
- van der Zander, K.; Bots, M.L.; Bak, A.A.A.; Koning, M.M.G.; de Leeuw, P.W. Enzymatically hydrolyzed lactotripeptides do not lower blood pressure in mildly hypertensive subjects. Am. J. Clin. Nutr. 2008, 88, 1697–1702. [Google Scholar] [CrossRef]
- Vermeirssen, V.; Van Camp, J.; Verstraete, W. Bioavailability of angiotensin I converting enzyme inhibitory peptides. Br. J. Nutr. 2004, 92, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Maqueda, D.; Miralles, B.; Recio, I.; Hernández-Ledesma, B. Antihypertensive peptides from food proteins: A review. Food Funct. 2012, 3, 350–361. [Google Scholar] [CrossRef] [PubMed]
- FitzGeral, R.J.; Meisel, H. Milk protein hydrolysates and bioactive peptides. In Advanced Dairy Chemistry; Fox, P.F., McSweeney, P.L.H., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2003; pp. 675–698. [Google Scholar]
- Yamada, Y.; Matoba, N.; Usui, H.; Onishi, K.; Yoshikawa, M. Design of a Highly Potent Anti-hypertensive Peptide Based on Ovokinin(2-7). Biosci. Biotechnol. Biochem. 2002, 66, 1213–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segura-Campos, M.; Chel-Guerrero, L.; Betancur-Ancona, D.; Hernandez-Escalante, V.M. Bioavailability of Bioactive Peptides. Food Rev. Int. 2011, 27, 213–226. [Google Scholar] [CrossRef]
- Vermeirssen, V.; Van Camp, J.; Verstraete, W. Optimisation and validation of an angiotensin-converting enzyme inhibition assay for the screening of bioactive peptides. J. Biochem. Biophys. Methods 2002, 51, 75–87. [Google Scholar] [CrossRef]
- Gallego, M.; Grootaert, C.; Mora, L.; Aristoy, M.C.; Van Camp, J.; Toldrá, F. Transepithelial transport of dry-cured ham peptides with ACE inhibitory activity through a Caco-2 cell monolayer. J. Funct. Foods 2016, 21, 388–395. [Google Scholar] [CrossRef]
- van der Pijl, P.C.; Kies, A.K.; Ten Have, G.A.; Duchateau, G.S.; Deutz, N.E. Pharmacokinetics of proline-rich tripeptides in the pig. Peptides 2008, 29, 2196–2202. [Google Scholar] [CrossRef]
| Component | g/100 g of Cheese |
|---|---|
| Dry Matter | 66.57 ± 1.88 |
| Proteins | 31.33 ± 0.87 |
| Lipids | 30.34 ± 0.95 |
| Ash | 4.49 ± 0.50 |
| Na | 0.9 ± 0.25 |
| K | 1.6 ± 0.13 |
| Mg | 0.1 ± 0.03 |
| Ca | 2.7 ± 0.84 |
| Peptide | Precursor Ion (m/z) | Fragments (m/z) | Collision Energy (V) | S-Lens |
|---|---|---|---|---|
| VPP | 312.2 | 213.0 | 18 | 79.71 |
| 69.9 | 33 | |||
| IPP | 326.2 | 213.0 | 17 | 83.19 |
| 69.9 | 33 | |||
| RYLGY | 671.4 | 274.9 | 43 | 197.70 |
| 111.9 | 51 | |||
| RYLG | 508.3 | 275.0 | 34 | 165.01 |
| 69.9 | 43 | |||
| AYFYPEL | 902.4 | 357.9 | 24 | 200.54 |
| 234.8 | 46 | |||
| AYFYPE | 789.3 | 244.8 | 29 | 177.76 |
| 234.8 | 40 | |||
| LHLPLP | 689.4 | 364.0 | 27 | 167.08 |
| 250.9 | 39 | |||
| HLPLP | 576.4 | 109.9 | 47 | 155.00 |
| 251.0 | 26 |
| Peptide | Line Equation | R2 | LOD (nM) | LOQ (nM) |
|---|---|---|---|---|
| VPP | 0.9962 | 8.5 | 25.7 | |
| IPP | 0.9977 | 6.6 | 20.0 | |
| RYLGY | 0.9999 | 1.5 | 4.6 | |
| RYLG | 0.9881 | 15.2 | 46.0 | |
| AYFYPEL | 0.9754 | 21.9 | 66.4 | |
| AYFYPE | 0.9952 | 9.6 | 29.2 | |
| LHLPLP | 0.9749 | 22.2 | 67.1 | |
| HLPLP | 0.9952 | 9.6 | 29.1 |
| Days | Treatment | |||||
|---|---|---|---|---|---|---|
| 0.1 1 | 0.2 1 | 0.4 1 | 0.6 1 | Captopril 2 | Water 3 | |
| 0 | 294.8 | 298.4 | 296.2 | 297.0 | 296.2 | 298.2 |
| 11.0 | 12.7 | 10.7 | 10.2 | 10.1 | 12.5 | |
| 63 | 371.8 b | 370.0 b | 373.2 b | 368.2 b | 344.3 a | 363.4 b |
| 15.3 | 13.3 | 17.7 | 11.2 | 13.8 | 15.2 | |
| (63 − 0) | 77.0 | 71.6 | 77.0 | 71.2 | 48.1 | 65.2 |
| Days | Treatment | |||||
|---|---|---|---|---|---|---|
| 0.1 1 | 0.2 1 | 0.4 1 | 0.6 1 | Captopril 2 | Water 3 | |
| 0 | b 215.0 A | b 217.0 A | b 214.9 A | b 217.9 A | a 214.7 A | b 214.9 A |
| 6.0 | 4.7 | 4.1 | 4.1 | 4.7 | 4.2 | |
| 7 | b 210.2 A | b 212.4 A | b 217.3 A | b 212.9 A | b 180.1 B | b 215.3 A |
| 6.7 | 6.2 | 5.5 | 4.3 | 6.2 | 5.9 | |
| 21 | b 210.0 B | b 218.1 A | c 206.3 B | c 209.4 B | b 174.6 C | b 214.8 A |
| 2.4 | 4.5 | 3.2 | 2.9 | 3.6 | 1.7 | |
| 35 | b 211.9 A | c 209.0 A | b 214.2 A | c 204.9 B | b 175.0 C | b 211.5 A |
| 2.1 | 3.9 | 3.3 | 4.4 | 3.3 | 2.5 | |
| 49 | a 222.9 A | a 228.9 A | a 228.6 A | a 223.8 A | b 170.6 B | a 223.1 A |
| 3.2 | 1.8 | 2.3 | 3.0 | 3.2 | 3.4 | |
| 63 | a 224.9 A | a 227.8 A | a 237.8 A | a 226.8 A | b 179.8 B | a 227.4 A |
| 4.1 | 2.8 | 1.8 | 2.4 | 3.6 | 2.4 | |
| Overal mean | 214.3 A | 213.7 A | 215.2 A | 212.1 A | 180.3 B | 215.0 A |
| 1.6 | 1.4 | 1.4 | 1.4 | 1.4 | 1.2 | |
| Days | Treatment | |||||
|---|---|---|---|---|---|---|
| 0.1 1 | 0.2 1 | 0.4 1 | 0.6 1 | Captopril 2 | Water 3 | |
| 0 | a 193.5 A | b 192.9 A | b 196.8 A | ab 201.3 A | a 199.0 A | a 197.3 A |
| 9.2 | 7.4 | 7.15 | 6.3 | 7.2 | 6.5 | |
| 7 | a 195.1 A | b 193.5 A | b 190.7 A | b 192.5 A | b 170.2 B | a 196.3 A |
| 7.7 | 7.0 | 6.6 | 5.3 | 7.2 | 7.8 | |
| 21 | b 179.3 B | c 172.9 B | c 169.3 B | c 170.0 B | c 142.3 C | a 192.0 A |
| 4.7 | 12.8 | 6.9 | 5.5 | 7.7 | 7.0 | |
| 35 | a 189.2 A | b 191.0 A | b 188.7 A | c 172.9 B | c 145.6 C | a 194.4 A |
| 5.2 | 4.8 | 4.5 | 8.4 | 4.1 | 5.3 | |
| 49 | a 194.7 A | a 203.9 A | ab 199.6 A | a 197.1 A | c 141.4 B | a 203.8 A |
| 5.0 | 1.7 | 5.0 | 6.5 | 4.0 | 4.6 | |
| 63 | a 201.9 A | a 204.8 A | a 208.1 A | a 204.4 A | c 161.9 B | a 197.5 A |
| 5.0 | 3.7 | 2.3 | 3.8 | 4.0 | 3.5 | |
| Overal means | 188.6 A | 192.3 A | 191.6 A | 188.8 A | 151.9 B | 189.2 A |
| 2.1 | 2.1 | 1.9 | 2.1 | 2.0 | 2.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basiricò, L.; Morera, P.; Evangelista, C.; Galaverna, G.; Sforza, S.; Prandi, B.; Bernabucci, U.; Nardone, A. Effect of Parmigiano Reggiano Consumption on Blood Pressure of Spontaneous Hypertensive Rats. Dairy 2022, 3, 364-376. https://doi.org/10.3390/dairy3020028
Basiricò L, Morera P, Evangelista C, Galaverna G, Sforza S, Prandi B, Bernabucci U, Nardone A. Effect of Parmigiano Reggiano Consumption on Blood Pressure of Spontaneous Hypertensive Rats. Dairy. 2022; 3(2):364-376. https://doi.org/10.3390/dairy3020028
Chicago/Turabian StyleBasiricò, Loredana, Patrizia Morera, Chiara Evangelista, Gianni Galaverna, Stefano Sforza, Barbara Prandi, Umberto Bernabucci, and Alessandro Nardone. 2022. "Effect of Parmigiano Reggiano Consumption on Blood Pressure of Spontaneous Hypertensive Rats" Dairy 3, no. 2: 364-376. https://doi.org/10.3390/dairy3020028
APA StyleBasiricò, L., Morera, P., Evangelista, C., Galaverna, G., Sforza, S., Prandi, B., Bernabucci, U., & Nardone, A. (2022). Effect of Parmigiano Reggiano Consumption on Blood Pressure of Spontaneous Hypertensive Rats. Dairy, 3(2), 364-376. https://doi.org/10.3390/dairy3020028