Comparative Analysis of Bio- and Chemo-Catalysts for the Synthesis of Flavour Compound Hexanal from Linoleic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis and Enrichment of Linoleic Acid
2.3. Preparation of LOX-Containing Soybean Flour Suspension
2.4. Hydroperoxidation of Linoleic Acid
2.5. Synthesis of Hexanal and 12-Oxo-9(Z)-dodecenoic Acid with Papaya Hydroperoxide Lyase HPLCP-N
2.6. Cleavage of HPODE Using Lewis Acids
2.7. Chemo-Catalytic Cleavage of HPODE Using Zeolites in an Autoclave
2.8. UV-Photometric Analyses of Enzyme Activities and HPODE Content
2.9. HPLC Analyses of HPODE Regioisomers
2.10. GC Analyses of Linoleic Acid, HPODE and Cleavage Products
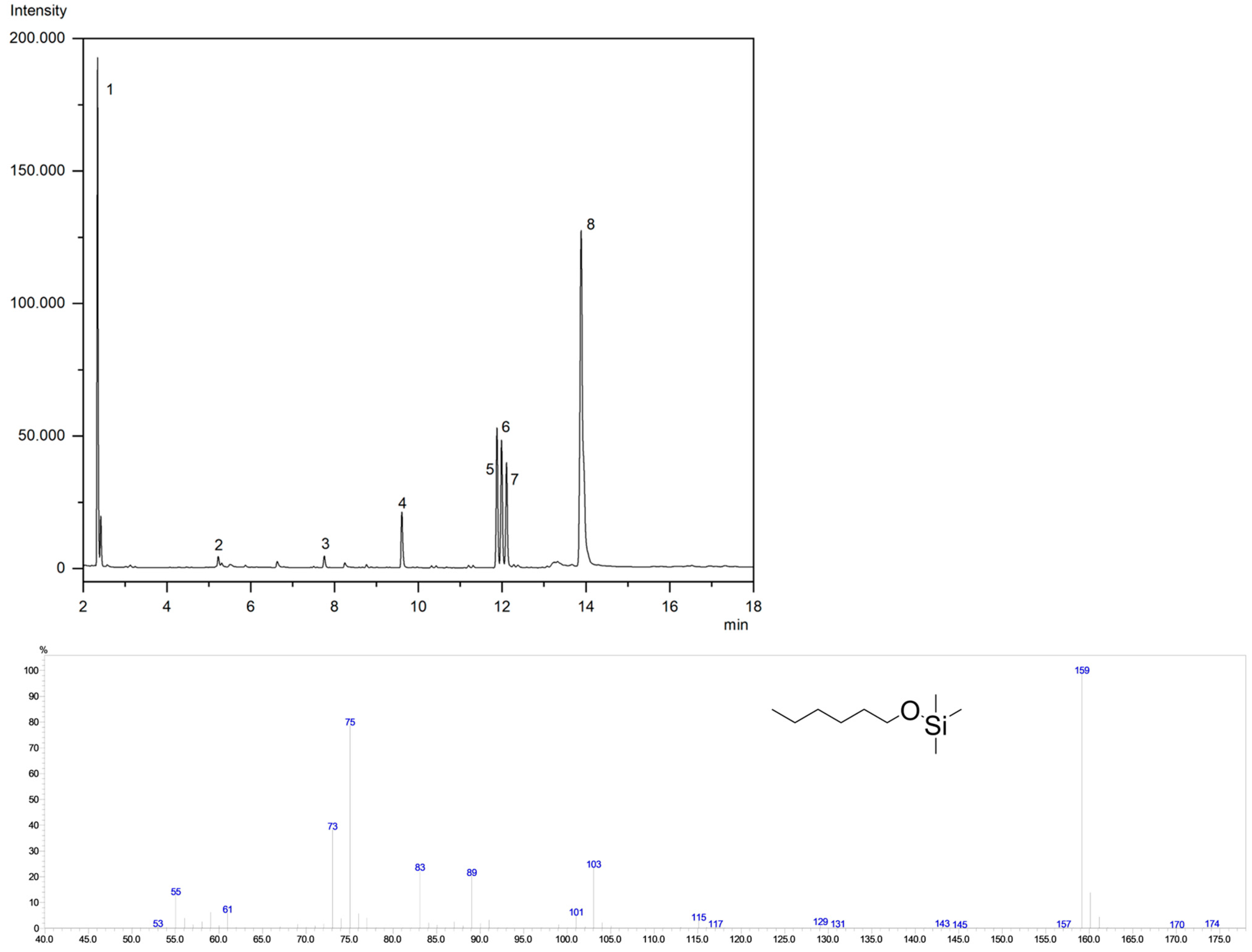
2.11. GC-MS Analysis of HPODE Cleavage Products
3. Results and Discussion
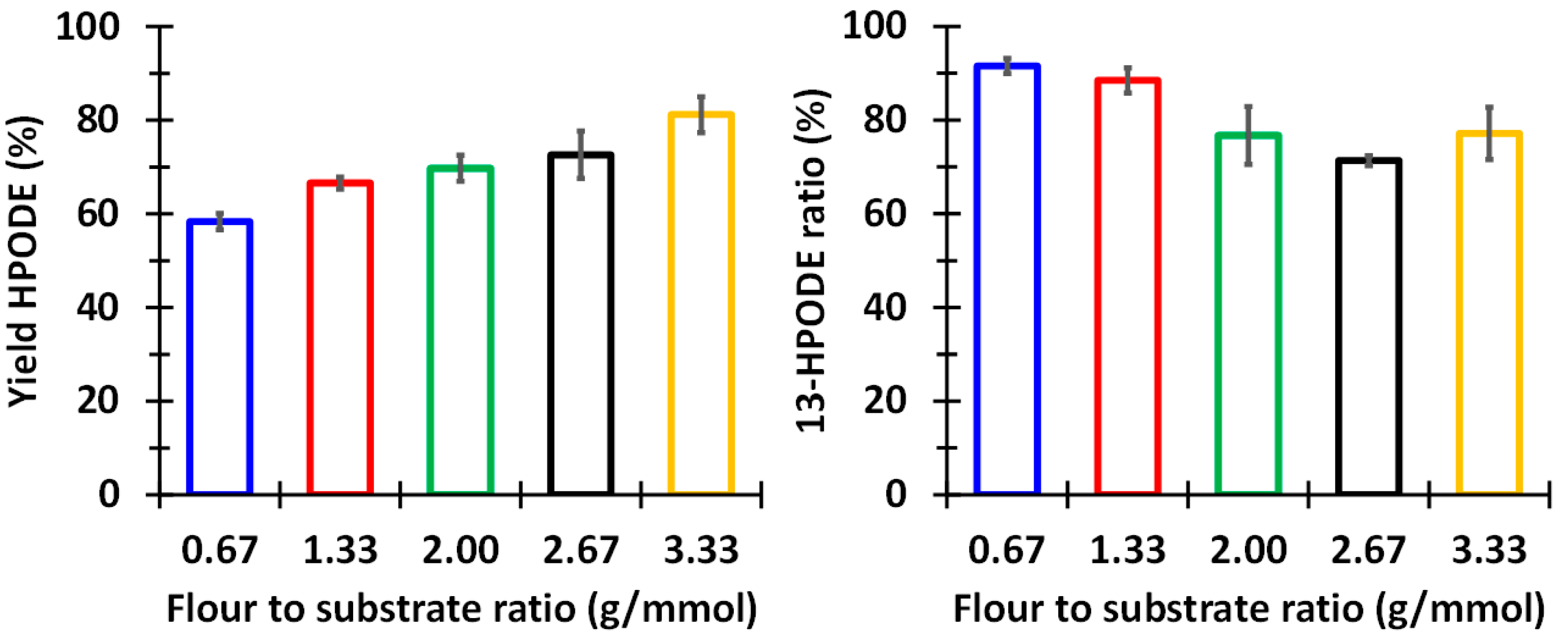
3.1. Biocatalytic Synthesis of 13-HPODE
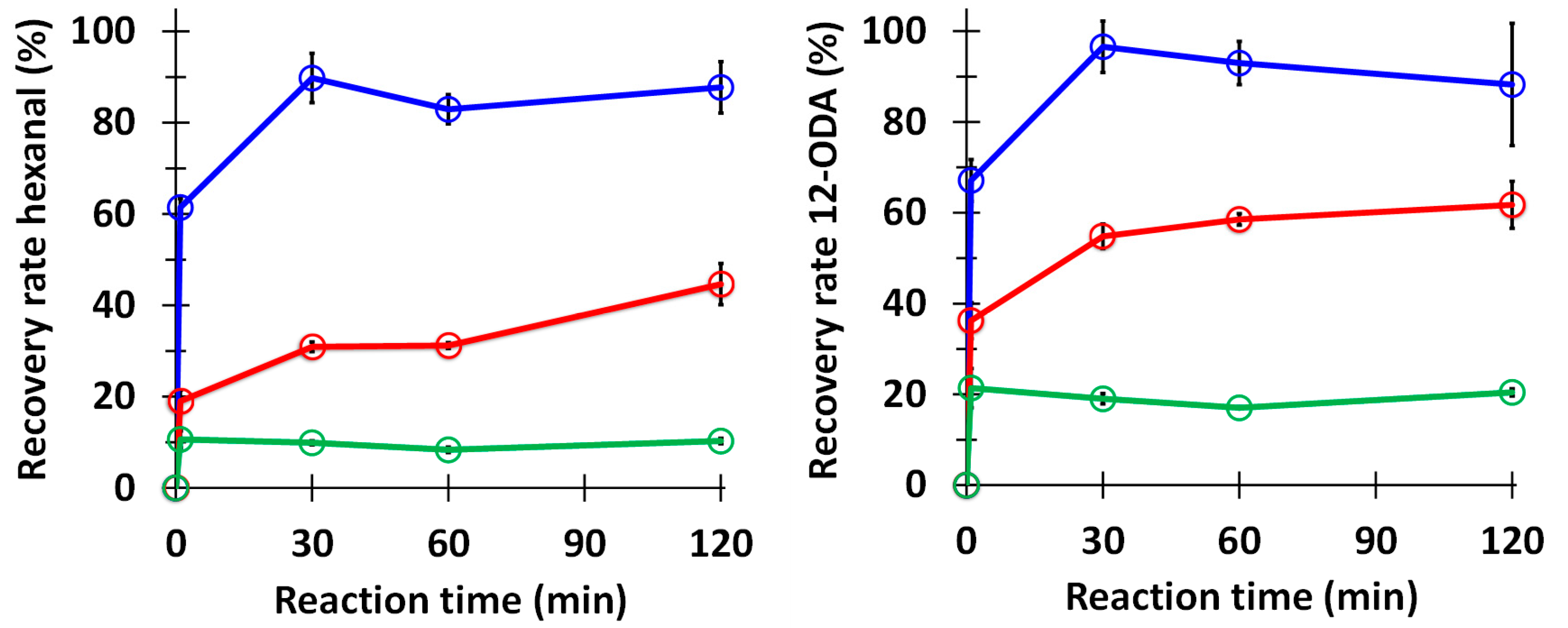
3.2. Hydroperoxide Lyase Splitting of 13-HPODE
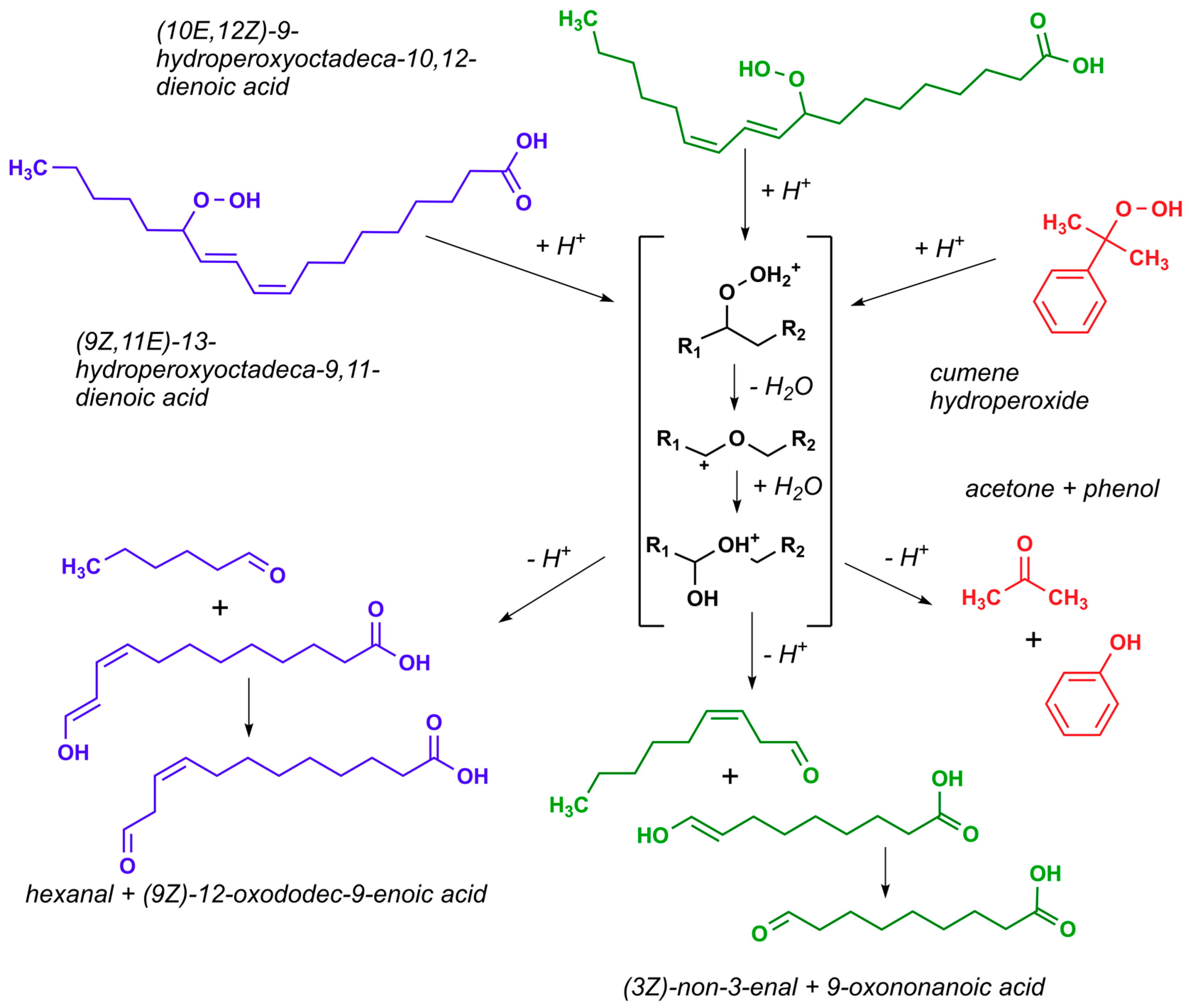
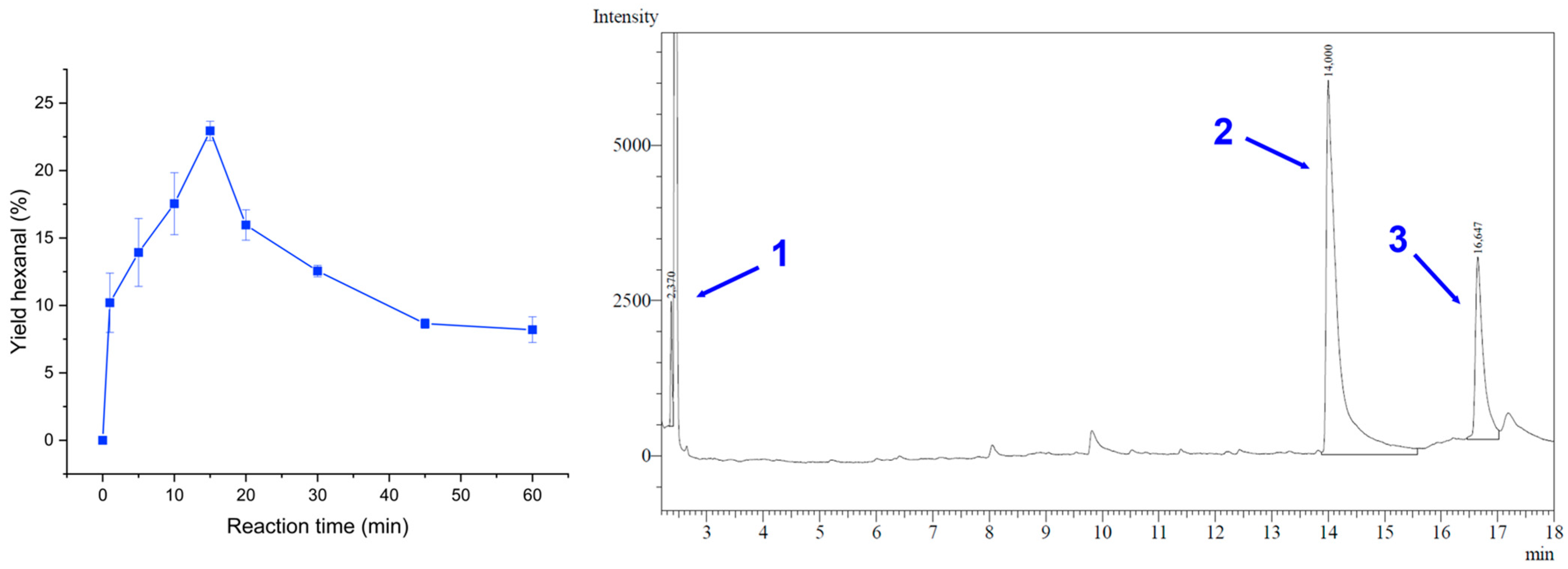
3.3. Lewis Acid Catalysed Cleavage of HPODE to Hexanal
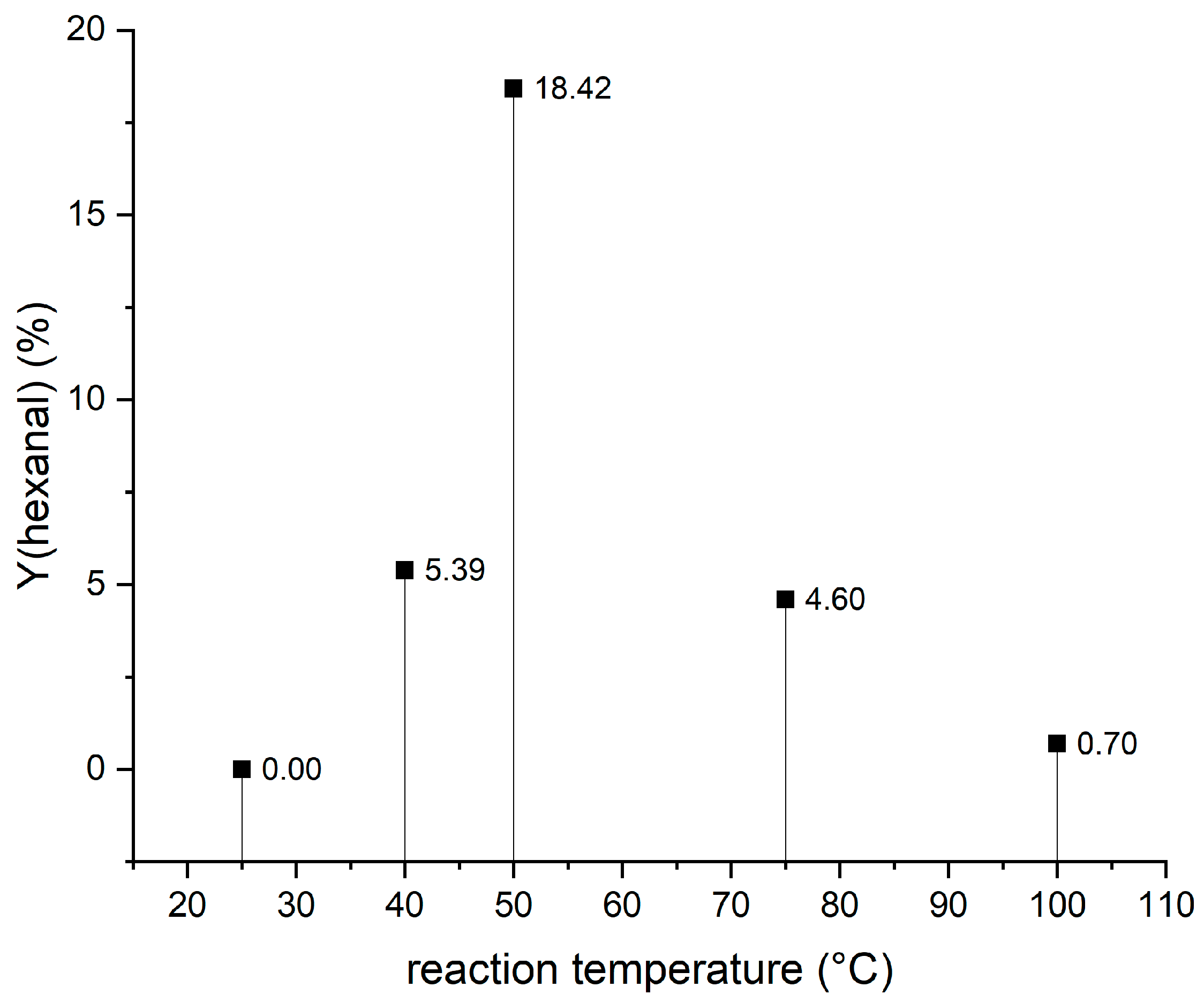
3.4. Chemo-Catalytic Cleavage of HPODE Using Zeolites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lee, S.Y.; Kim, H.U.; Chae, T.U.; Cho, J.S.; Kim, J.W.; Shin, J.H.; Kim, D.I.; Ko, Y.-S.; Jang, W.D.; Jang, Y.-S. A comprehensive metabolic map for production of bio-based chemicals. Nat. Catal. 2019, 2, 18–33. [Google Scholar] [CrossRef]
- Hayes, D.G.; Smith, G.A. Chapter 1—Biobased Surfactants: Overview and Industrial State of the Art. In Biobased Surfactants, 2nd ed.; Hayes, D.G., Solaiman, D.K.Y., Ashby, R.D., Eds.; AOCS Press: Urbana, IL, USA, 2019; pp. 3–38. [Google Scholar] [CrossRef]
- Buchhaupt, M.; Guder, J.C.; Etschmann, M.M.W.; Schrader, J. Synthesis of green note aroma compounds by biotransformation of fatty acids using yeast cells coexpressing lipoxygenase and hydroperoxide lyase. Appl. Microbiol. Biotechnol. 2012, 93, 159–168. [Google Scholar] [CrossRef]
- Vincenti, S.; Mariani, M.; Alberti, J.-C.; Jacopini, S.; Brunini-Bronzini de Caraffa, V.; Berti, L.; Maury, J. Biocatalytic Synthesis of Natural Green Leaf Volatiles Using the Lipoxygenase Metabolic Pathway. Catalysts 2019, 9, 873. [Google Scholar] [CrossRef]
- Gigot, C.; Ongena, M.; Fauconnier, M.L.; Wathelet, J.P.; Jardin, P.d.; Thonart, P. The lipoxygenase metabolic pathway in plants: Potential for industrial production of natural green leaf volatiles. Biotechnol. Agron. Soc. Environ. 2010, 14, 451–460. [Google Scholar]
- Fauconnier, M.L.; Marlier, M. An efficient procedure for the production of fatty acid hydroperoxides from hydrolyzed flax seed oil and soybean lipoxygenase. Biotechnol. Tech. 1996, 10, 839–844. [Google Scholar] [CrossRef]
- Gala Marti, V.; Coenen, A.; Schörken, U. Synthesis of Linoleic Acid 13-Hydroperoxides from Safflower Oil Utilizing Lipoxygenase in a Coupled Enzyme System with In-Situ Oxygen Generation. Catalysts 2021, 11, 1119. [Google Scholar] [CrossRef]
- Gargouri, M.; Legoy, M.D. Bienzymatic reaction for hydroperoxide production in a multiphasic system. Enzym. Microb. Technol. 1997, 21, 79–84. [Google Scholar] [CrossRef]
- Wang, J.; Li, K.; He, Y.; Liu, X.; Wang, P.; Xu, L.; Yan, J.; Yan, Y. Bi-enzyme directed self-assembled system toward biomimetic synthesis of fatty acid hydroperoxides like soybean. Compos. Part B Eng. 2021, 222, 109091. [Google Scholar] [CrossRef]
- Gala Marti, V.; Müller, C.; Spektor, V.; Schörken, U. Immobilization of Lipoxygenase, Catalase, and Lipase for a Reactor Design Targeting Linoleic Acid Hydroperoxidation. Eur. J. Lipid Sci. Technol. 2023, 125, 2200140. [Google Scholar] [CrossRef]
- Stolterfoht, H.; Rinnofner, C.; Winkler, M.; Pichler, H. Recombinant Lipoxygenases and Hydroperoxide Lyases for the Synthesis of Green Leaf Volatiles. J. Agric. Food Chem. 2019, 67, 13367–13392. [Google Scholar] [CrossRef]
- Brühlmann, F.; Bosijokovic, B. Efficient Biochemical Cascade for Accessing Green Leaf Alcohols. Org. Process Res. Dev. 2016, 20, 1974–1978. [Google Scholar] [CrossRef]
- Kaur, I.; Korrapati, N.; Bonello, J.; Mukherjee, A.; Rishi, V.; Bendigiri, C. Biosynthesis of natural aroma compounds using recombinant whole-cell tomato hydroperoxide lyase biocatalyst. J. Biosci. 2022, 47, 37. [Google Scholar] [CrossRef]
- Coenen, A.; Ferrer, M.; Jaeger, K.-E.; Schörken, U. Synthesis of 12-aminododecenoic acid by coupling transaminase to oxylipin pathway enzymes. Appl. Microbiol. Biotechnol. 2023, 107, 2209–2221. [Google Scholar] [CrossRef] [PubMed]
- Coenen, A.; Marti, V.G.; Müller, K.; Sheremetiev, M.; Finamore, L.; Schörken, U. Synthesis of Polymer Precursor 12-Oxododecenoic Acid Utilizing Recombinant Papaya Hydroperoxide Lyase in an Enzyme Cascade. Appl. Biochem. Biotechnol. 2022, 194, 6194–6212. [Google Scholar] [CrossRef] [PubMed]
- Brühlmann, F.; Bosijokovic, B.; Ullmann, C.; Auffray, P.; Fourage, L.; Wahler, D. Directed evolution of a 13-hydroperoxide lyase (CYP74B) for improved process performance. J. Biotechnol. 2013, 163, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Kazimírová, V.; Zezulová, V.; Krasňan, V.; Štefuca, V.; Rebroš, M. Optimization of Hydroperoxide Lyase Production for Recombinant Lipoxygenase Pathway Cascade Application. Catalysts 2021, 11, 1201. [Google Scholar] [CrossRef]
- Yaremenko, I.A.; Vil, V.A.; Demchuk, D.V.; Terent’ev, A.O. Rearrangements of organic peroxides and related processes. Beilstein J. Org. Chem. 2016, 12, 1647–1748. [Google Scholar] [CrossRef]
- Drönner, J.; Hausoul, P.; Palkovits, R.; Eisenacher, M. Solid Acid Catalysts for the Hock Cleavage of Hydroperoxides. Catalysts 2022, 12, 91. [Google Scholar] [CrossRef]
- Gardner, H.W.; Plattner, R.D. Linoleate hydroperoxides are cleaved heterolytically into aldehydes by a Lewis acid in aprotic solvent. Lipids 1984, 19, 294–299. [Google Scholar] [CrossRef]
- Corma, A.; García, H. Lewis Acids: From Conventional Homogeneous to Green Homogeneous and Heterogeneous Catalysis. Chem. Rev. 2003, 103, 4307–4366. [Google Scholar] [CrossRef]
- Nikoofar, K.; Khademi, Z. A review on green Lewis acids: Zirconium(IV) oxydichloride octahydrate (ZrOCl2·8H2O) and zirconium(IV) tetrachloride (ZrCl4) in organic chemistry. Res. Chem. Intermed. 2016, 42, 3929–3977. [Google Scholar] [CrossRef]
- Weitkamp, J. Zeolites and catalysis. Solid State Ion. 2000, 131, 175–188. [Google Scholar] [CrossRef]
- Drönner, J.; Bijerch, K.; Hausoul, P.; Palkovits, R.; Eisenacher, M. High-Temperature-Treated LTX Zeolites as Heterogeneous Catalysts for the Hock Cleavage. Catalysts 2023, 13, 202. [Google Scholar] [CrossRef]
- Baysal, T.; Demirdöven, A. Lipoxygenase in fruits and vegetables: A review. Enzym. Microb. Technol. 2007, 40, 491–496. [Google Scholar] [CrossRef]
- Andreou, A.; Feussner, I. Lipoxygenases—Structure and reaction mechanism. Phytochemistry 2009, 70, 1504–1510. [Google Scholar] [CrossRef]
- Tu, H.-A.T.; Dobson, E.P.; Henderson, L.C.; Barrow, C.J.; Adcock, J.L. Soy flour as an alternative to purified lipoxygenase for the enzymatic synthesis of resolvin analogues. New Biotechnol. 2018, 41, 25–33. [Google Scholar] [CrossRef]
- Berry, H.; Debat, H.; Larreta-Garde, V. Excess substrate inhibition of soybean lipoxygenase-1 is mainly oxygen-dependent. FEBS Lett. 1997, 408, 324–326. [Google Scholar] [CrossRef]
- Touchy, A.S.; Rashed, M.N.; Huang, M.; Toyao, T.; Shimizu, K.-i.; Siddiki, S.M.A.H. Lewis Acid Promoted Sustainable Transformation of Triglycerides to Fatty Acids Using a Water-Tolerant Nb2O5 Catalyst. ACS Sustain. Chem. Eng. 2022, 10, 11791–11799. [Google Scholar] [CrossRef]
- Kahr, G.; Madsen, F.T. Determination of the cation exchange capacity and the surface area of bentonite, illite and kaolinite by methylene blue adsorption. Appl. Clay Sci. 1995, 9, 327–336. [Google Scholar] [CrossRef]
- Sigma-Aldrich. Product Specification of Montmorillonite K10; Sigma-Aldrich: St. Louis, MO, USA, 2023. [Google Scholar]
- Hagen, J. Industrial Catalysis: A Practical Approach; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar]
- Rontani, J.F.; Aubert, C. Trimethylsilyl transfer during electron ionization mass spectral fragmentation of some omega-hydroxycarboxylic and omega-dicarboxylic acid trimethylsilyl derivatives and the effect of chain length. Rapid Commun. Mass Spectrom. 2004, 18, 1889–1895. [Google Scholar] [CrossRef]
- Rontani, J.F.; Aubert, C. Hydrogen and trimethylsilyl transfers during EI mass spectral fragmentation of hydroxycarboxylic and oxocarboxylic acid trimethylsilyl derivatives. J. Am. Soc. Mass Spectrom. 2008, 19, 66–75. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Solvent/Temperature | Catalyst (mM) Yield Hexanal (%) | Catalyst (mM) TOF (s−1) | ||||||
|---|---|---|---|---|---|---|---|---|
| (A) AlCl3 | 0.1 | 1 | 10 | 100 | 0.1 | 1 | 10 | 100 |
| MTBE 25 °C MTBE 50 °C | 2.4 ± 0.4 | 3.5 ± 0.3 | 11.4 ± 0.2 | 22.9 ± 0.7 10.5 ± 1.8 | 2.7 × 10−3 | 3.9 × 10−4 | 1.3 × 10−4 | 2.5 × 10−5 1.2 × 10−5 |
| THF 25 °C THF 50 °C | 4.6 ± 0.8 | 10.3 ± 1.1 10.5 ± 1.2 | 5.1 × 10−5 | 1.1 × 10−5 1.2 × 10−5 | ||||
| Diethyl ether 25 °C | 3.1 ± 0.1 | 3.8 ± 0.5 | 3.4 × 10−5 | 4.2 × 10−6 | ||||
| Methanol 25 °C | 7.3 ± 2.0 | 7.3 ± 2.0 | 8.7 ± 0.2 | 8.1 × 10−5 | 9.7 × 10−6 | |||
| Chloroform 25 °C | 1.3 ± 0.1 | 1.3 ± 0.1 | 5.9 ± 0.1 | 13.0 ± 2.1 | 1.4 × 10−3 | 1.4 × 10−4 | 6.6 × 10−5 | 1.4 × 10−5 |
| Heptane 25 °C | 0.5 ± 0.2 | 0.4 ± 0.2 | 2.8 ± 1.1 | 3.8 ± 1.3 | 5.6 × 10−4 | 4.4 × 10−5 | 3.1 × 10−5 | 4.2 × 10−6 |
| (B) ZrCl4 | 0.1 | 1 | 10 | 100 | 0.1 | 1 | 10 | 100 |
| MTBE 25 °C MTBE 50 °C | 1.2 ± 0.2 2.4 ± 0.5 | 1.7 ± 0.5 6.7 ± 1.1 | 7.3 ± 0.8 7.0 ± 0.4 | 11.9 ± 0.6 13.8 ± 1.6 | 1.3 × 10−3 2.7 × 10−3 | 1.9 × 10−4 7.4 × 10−4 | 8.1 × 10−5 7.8 × 10−5 | 1.3 × 10−5 1.5 × 10−5 |
| THF 25 °C | 2.3 ± 0.7 | 2.1 ± 0.4 | 2.1 ± 0.2 | 3.8 ± 0.1 | 2.6 × 10−3 | 2.3 × 10−4 | 2.3 × 10−5 | 4.2 × 10−6 |
| Chloroform 25 °C | 0.8 ± 0.2 | 4.6 ± 0.6 | 2.9 ± 0.6 | 9.0 ± 0.7 | 8.9 × 10−4 | 5.1 × 10−4 | 3.2 × 10−5 | 1.0 × 10−5 |
| Heptane 25 °C | 0.3 ± 0.2 | 0.4 ± 0.2 | 5.8 ± 1.7 | 7.5 ± 1.4 | 3.3 × 10−4 | 4.4 × 10−5 | 6.4 × 10−5 | 8.3 × 10−6 |
| Catalyst | Fragmentation Pattern | Y (Hexanal) /% |
|---|---|---|
| Beta Zeolite | HOCK | 18.33 |
| Zeolite USY | undefined | traces |
| ZSM-5 | undefined | traces |
| Zeolite Low Silica Linde Type X (uncalcined) | thermal decomposition | – |
| Zeolite Low Silica Linde Type X (calcinated) | thermal decomposition | – |
| Montmorillonite K10 | HOCK | 1.67 |
| Kaolinite natural | thermal decomposition | – |
| Zeolite | Beta | LSX Calcinated | LSX Uncalcinated | USY | ZSM-5 |
|---|---|---|---|---|---|
| Module | 18.2 | 0.87 | 1.24 | 3.30 | 13.80 |
| Pore size [Å] | 2.4 | 0.1 | 0.91 | 2.1 | 2.2 |
| BET SA [m2·g−1] | 573.945 | 0.1 | 417 | 586.13 | 325.72 |
| Compound | Ret. Time/min | m/z |
|---|---|---|
| Hexanal (1) | 2.34 | [M-174]+ 2.2; [M-159]+ 99.2; [M-75]+ 100.0; [M-174]+ 2.24 |
| Nonanal (2) | 5.22 | [M-201]+ 60.2; [M-103]+ 19.6; [M-75]+ 100.0 [M-73]+ 41.7 |
| 3-nonenal (3) | 7.55 | [M-199]+ 12.3; [M-129]+ 73.9; [M-75]+ 100.0 [M-73]+ 83.6 |
| 9-oxononanoic acid (4) | 9.60 | [M-303]+ 40.0; [M-213]+ 34.9; [M-147]+ 58.2; [M-73]+ 100 |
| 12-oxo-9(Z)-dodecenoic acid (5) | 11.88 | [M-343]+ 7.5; [M-147]+ 44.9; [M-129]+ 62.5; [M-73]+ 100 |
| 12-oxododecanoic acid (6) | 11.99 | [M-345]+ 34.2; [M-255]+ 41.6; [M-147]+ 38.5; [M-73]+ 100 |
| 12-oxo-(E)-10-dodecenoic acid (7) | 12.10 | [M-343]+ 2.1; [M-217]+ 16.4; [M-147]+ 24.8; [M-73]+ 100 |
| linoleic acid (8) | 13.89 | [M-337]+ 38.2; [M-262]+ 26.78; [M-75]+ 100.0; [M-67]+ 90.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drönner, J.; Marti, V.G.; Bandte, S.; Coenen, A.; Schörken, U.; Eisenacher, M. Comparative Analysis of Bio- and Chemo-Catalysts for the Synthesis of Flavour Compound Hexanal from Linoleic Acid. Reactions 2023, 4, 518-530. https://doi.org/10.3390/reactions4030031
Drönner J, Marti VG, Bandte S, Coenen A, Schörken U, Eisenacher M. Comparative Analysis of Bio- and Chemo-Catalysts for the Synthesis of Flavour Compound Hexanal from Linoleic Acid. Reactions. 2023; 4(3):518-530. https://doi.org/10.3390/reactions4030031
Chicago/Turabian StyleDrönner, Jan, Valentin Gala Marti, Simone Bandte, Anna Coenen, Ulrich Schörken, and Matthias Eisenacher. 2023. "Comparative Analysis of Bio- and Chemo-Catalysts for the Synthesis of Flavour Compound Hexanal from Linoleic Acid" Reactions 4, no. 3: 518-530. https://doi.org/10.3390/reactions4030031
APA StyleDrönner, J., Marti, V. G., Bandte, S., Coenen, A., Schörken, U., & Eisenacher, M. (2023). Comparative Analysis of Bio- and Chemo-Catalysts for the Synthesis of Flavour Compound Hexanal from Linoleic Acid. Reactions, 4(3), 518-530. https://doi.org/10.3390/reactions4030031







