Hydrothermal Synthesis of Vanadium Oxide Microstructures with Mixed Oxidation States
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
4.1. Vanadium Oxide ((CnH(2n+1)NH3)2V7O16 Nanotubes (VOx-NTs)
4.2. Vanadium Oxide ((CnH(2n+1)NH3)2V7O16 Nanourchins (VOx-NU)



4.3. Vanadium Oxide (NH4)2V7O16 Micro-Squares (VOx-MSQ) and VO2 Micro-Crosses (VOx-MC)







4.4. Vanadium Oxide V6O11 Rotationally Symetric Nano Six-folds (VOx-NC) and Nano Star Fruits (VOx-NSF)



5. Mixed Oxidation States Rate Control
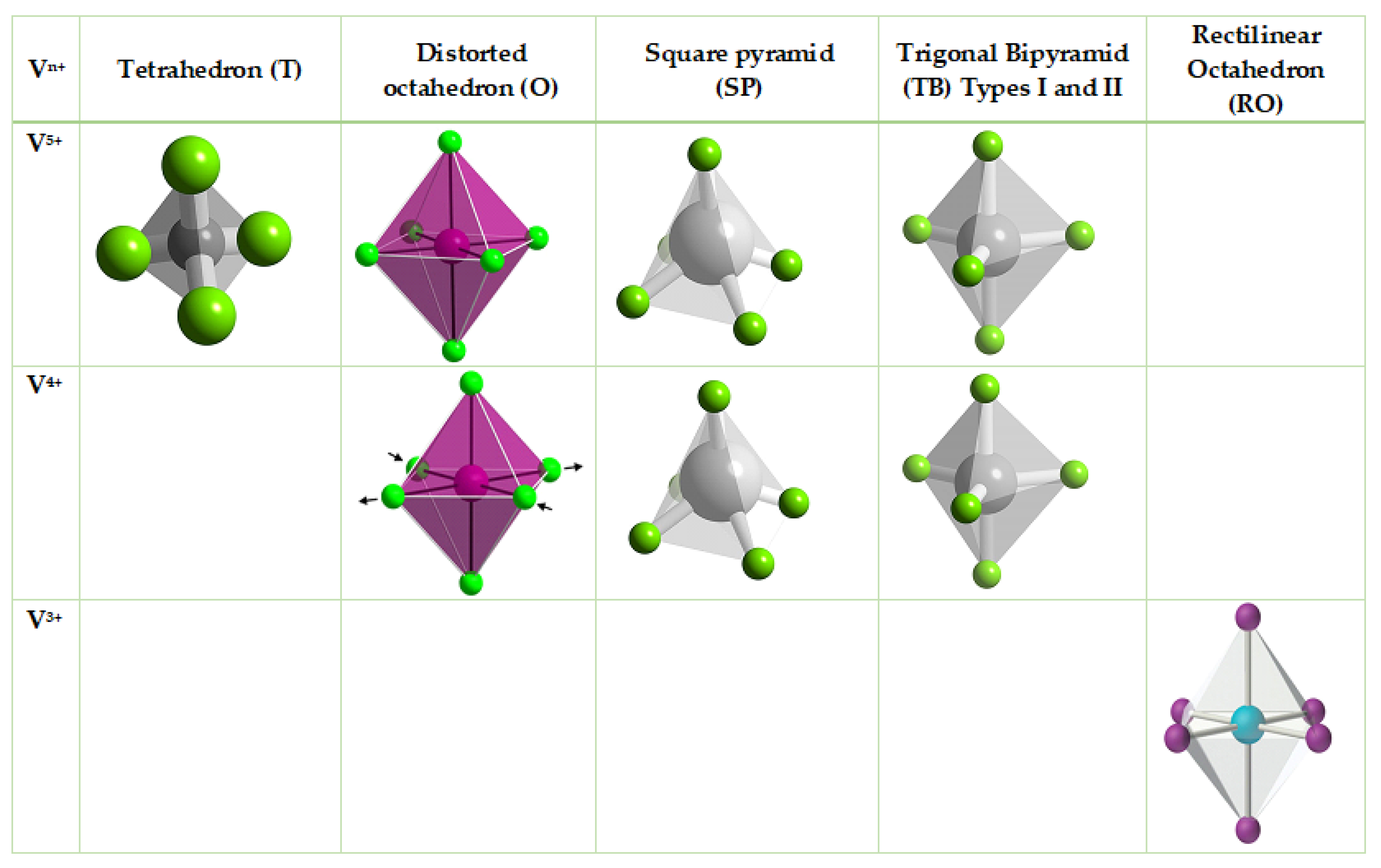
6. Morphologic Defects
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mounasamy, V.; Mani, G.K.; Madanagurusamy, S. Vanadium oxide nanostructures for chemiresistive gas and vapour sensing: A review on state of the art. Microchim. Acta 2020, 187, 1–29. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, Y.; Xia, Y.; Xiang, S.; Han, S.; Cai, C.; Wang, Q.; Wang, Y.; Gu, M. Fast Zn2+ kinetics of vanadium oxide nanotubes in high-performance rechargeable zinc-ion batteries. J. Power Sources 2020, 451, 227767. [Google Scholar] [CrossRef]
- Mousavi, S.M. Vanadium oxide nanotubes for selective catalytic reduction of NOx with NH3. Chin. J. Chem. Eng. 2016, 24, 914–919. [Google Scholar] [CrossRef]
- Zhou, X.; He, T.; Chen, X.; Zhao, Z.; Guo, X.; Sun, L.; Liu, Z. Synthesis and lithium storage properties of vanadium oxide nanotubes (VOx-NTs)-Polyaniline nanocomposite as cathode material for lithium ion batteries. J. Mater. Sci. Mater. Electron. 2017, 28, 11098–11107. [Google Scholar] [CrossRef]
- Pinheiro, A.L.G.; do Carmo, J.V.C.; Carvalho, D.C.; Oliveira, A.C.; Rodríguez-Castellón, E.; Tehuacanero-Cuapa, S.; Otubo, L.; Lang, R. Bio-additive fuels from glycerol acetalization over metals-containing vanadium oxide nanotubes (MeVOx-NT in which, Me = Ni, Co, or Pt). Fuel Processing Technol. 2019, 184, 45–56. [Google Scholar] [CrossRef]
- Fang, D.; Li, L.; Xu, W.; Li, G.; Luo, Z.; Liang, C.; Ji, Y.; Xu, J.; Xiong, C. Self-assembled hairy ball-like V2O5 nanostructures for lithium ion batteries. RSC Adv. 2014, 4, 25205–25209. [Google Scholar] [CrossRef]
- Yu, W.; Wang, J.; Gou, Z.; Zeng, W.; Guo, W.; Lin, L. Hydrothermal synthesis of vanadium pentoxide nanostructures and their morphology control. Ceram. Int. 2013, 39, 2639–2643. [Google Scholar] [CrossRef]
- Shoorangiz, M.; Shariatifard, L.; Roshan, H.; Mirzaei, A. Hydrothermally synthesized flower-like vanadium oxide nanostructures for ethanol sensing studies. Mater. Sci. Semicond. Processing 2022, 137, 106241. [Google Scholar] [CrossRef]
- Nefzi, H.; Sediri, F. Vanadium oxide nanourchins: Hydrothermal synthesis, characterization, frequency dependent electrical properties. Ceram. Int. 2015, 41, 1391–1399. [Google Scholar] [CrossRef]
- Chandrappa, G.T.; Steunou, N.; Cassaignon, S.; Bauvais, C.; Biswas, P.K.; Livage, J. Vanadium Oxide: From Gels to Nanotubes. J. Sol-Gel Sci. Technol. 2003, 26, 593–596. [Google Scholar] [CrossRef]
- Liu, M.; Su, B.; Tang, Y.; Jiang, X.; Yu, A. Recent Advances in Nanostructured Vanadium Oxides and Composites for Energy Conversion. Adv. Energy Mater. 2017, 7, 1700885. [Google Scholar] [CrossRef] [Green Version]
- Sahraeian, N.; Esmaeilzadeh, F.; Mowla, D. Hydrothermal synthesis of V2O5 nanospheres as catalyst for hydrogen sulfide removal from sour water. Ceram. Int. 2021, 47, 923–934. [Google Scholar] [CrossRef]
- Faggio, G.; Modafferi, V.; Panzera, G.; Alfieri, D.; Santangelo, S. Micro-Raman and photoluminescence analysis of composite vanadium oxide/poly-vinyl acetate fibres synthesised by electro-spinning. J. Raman Spectrosc. 2012, 43, 761–768. [Google Scholar] [CrossRef]
- Bieri, F.; Krumeich, F.; Muhr, H.-J.; Nesper, R. The First Vanadium Oxide Nanotubes Containing an Aromatic Amine as Template. Helv. Chim. Acta 2001, 84, 3015–3022. [Google Scholar] [CrossRef]
- Niederberger, M.; Muhr, H.-J.; Krumeich, F.; Bieri, F.; Günther, D.; Nesper, R. Low-Cost Synthesis of Vanadium Oxide Nanotubes via Two Novel Non-Alkoxide Routes. Chem. Mater. 2000, 12, 1995–2000. [Google Scholar] [CrossRef]
- Chen, X.; Sun, X.; Li, Y. Self-Assembling Vanadium Oxide Nanotubes by Organic Molecular Templates. Inorg. Chem. 2002, 41, 4524–4530. [Google Scholar] [CrossRef]
- Zhang, Y. Synthesis and phase transition properties of VO2 (M) hollow spheres with large thermal hysteresis width. Micro Nano Lett. 2019, 14, 819–822. [Google Scholar] [CrossRef]
- Xu, Y.; Deng, X.; Li, Q.; Zhang, G.; Xiong, F.; Tan, S.; Wei, Q.; Lu, J.; Li, J.; An, Q.; et al. Vanadium Oxide Pillared by Interlayer Mg2+ Ions and Water as Ultralong-Life Cathodes for Magnesium-Ion Batteries. Chem 2019, 5, 1194–1209. [Google Scholar] [CrossRef]
- Inagaki, M.; Nakamura, T.; Shimizu, A. Soft process for the intercalation of ammonium cations into vanadium oxide. J. Mater. Res. 1998, 13, 896–900. [Google Scholar] [CrossRef]
- Rojas-Cervantes, M.L.; Casal, B.; Aranda, P.; Savirón, M.; Galván, J.C.; Ruiz-Hitzky, E. Hybrid materials based on vanadium pentoxide intercalation complexes. Colloid Polym. Sci. 2001, 279, 990–1004. [Google Scholar] [CrossRef]
- Clites, M.; Pomerantseva, E. Bilayered vanadium oxides by chemical pre-intercalation of alkali and alkali-earth ions as battery electrodes. Energy Storage Mater. 2018, 11, 30–37. [Google Scholar] [CrossRef]
- López-Cabaña, Z.; Navas, D.; Benavente, E.; Ana, M.A.S.; Lavayen, V.; González, G. Hybrid Laminar Organic-Inorganic Semiconducting Nanocomposites. Mol. Cryst. Liq. Cryst. 2012, 554, 119–134. [Google Scholar] [CrossRef]
- Gannon, G.; O’Dwyer, C.; Larsson, J.A.; Thompson, D. Interdigitating Organic Bilayers Direct the Short Interlayer Spacing in Hybrid Organic–Inorganic Layered Vanadium Oxide Nanostructures. J. Phys. Chem. B 2011, 115, 14518–14525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krumeich, F.; Muhr, H.J.; Niederberger, M.; Bieri, F.; Reinoso, M.; Nesper, R. Vanadium Oxide Nanotubes with Diamine Templates. MRS Online Proc. Libr. 1999, 581, 393–398. [Google Scholar] [CrossRef]
- Durupthy, O.; Steunou, N.; Coradin, T.; Livage, J. Intercalation of dipeptides during V2O5•nH2O xerogel condensation. J. Phys. Chem. Solids 2006, 67, 944–949. [Google Scholar] [CrossRef]
- Zhang, Y.; Du, Y.; Song, B.; Wang, Z.; Wang, X.; Wan, F.; Ma, X. Manganese-ions and polyaniline co-intercalation into vanadium oxide for stable zinc-ion batteries. J. Power Sources 2022, 545, 231920. [Google Scholar] [CrossRef]
- Roy, A.; Pradhan, M.; Ray, C.; Sahoo, R.; Dutta, S.; Pal, T. Facile synthesis of pyridine intercalated ultra-long V2O5 nanowire from commercial V2O5: Catalytic applications in selective dye degradation. CrystEngComm 2014, 16, 7738–7744. [Google Scholar] [CrossRef]
- Zabrodina, G.S.; Makarov, S.G.; Kremlev, K.V.; Yunin, P.A.; Gusev, S.A.; Kaverin, B.S.; Kaverina, L.B.; Ketkov, S.Y. Novel hybrid materials based on the vanadium oxide nanobelts. Appl. Surf. Sci. 2016, 368, 395–402. [Google Scholar] [CrossRef]
- Zhou, X.; Chen, X.; He, T.; Bi, Q.; Sun, L.; Liu, Z. Fabrication of polypyrrole/vanadium oxide nanotube composite with enhanced electrochemical performance as cathode in rechargeable batteries. Appl. Surf. Sci. 2017, 405, 146–151. [Google Scholar] [CrossRef]
- Mai, L.; Chen, W.; Xu, Q.; Zhu, Q.; Han, C.; Peng, J. Cost-saving synthesis of vanadium oxide nanotubes. Solid State Commun. 2003, 126, 541–543. [Google Scholar] [CrossRef]
- Nadimicherla, R.; Liu, Y.; Chen, K.; Chen, W. Effect of polyethylene glycol on vanadium oxide nanotubes in lithium-ion batteries. Microelectron. Eng. 2014, 127, 81–85. [Google Scholar] [CrossRef]
- Kweon, H.; Lee, K.W.; Lee, C.E. Ferromagnetism in water-doped vanadium oxide nanotubes. J. Appl. Phys. 2010, 108, 023905. [Google Scholar] [CrossRef]
- Emmerich, J.; Dillen, M.; Kirschhock, C.E.A.; Martens, J.A. Fine-tuning of Vanadium Oxide Nanotubes. In Studies in Surface Science and Catalysis; Gaigneaux, E.M., Devillers, M., Hermans, S., Jacobs, P.A., Martens, J.A., Ruiz, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 175, pp. 249–252. [Google Scholar]
- Nordlinder, S.; Augustsson, A.; Schmitt, T.; Guo, J.; Duda, L.C.; Nordgren, J.; Gustafsson, T.; Edström, K. Redox Behavior of Vanadium Oxide Nanotubes As Studied by X-ray Photoelectron Spectroscopy and Soft X-ray Absorption Spectroscopy. Chem. Mater. 2003, 15, 3227–3232. [Google Scholar] [CrossRef]
- Nefzi, H.; Sediri, F. Vanadium oxide nanotubes VOx-NTs: Hydrothermal synthesis, characterization, electrical study and dielectric properties. J. Solid State Chem. 2013, 201, 237–243. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Shi, Z. Hydrothermal synthesis of nanowires, nanobelts, and nanotubes of vanadium oxides from one reaction system. Int. J. Mater. Res. 2013, 104, 567–572. [Google Scholar] [CrossRef]
- Huang, Z.; Zeng, H.; Xue, L.; Zhou, X.; Zhao, Y.; Lai, Q. Synthesis of vanadium oxide, V6O13 hollow-flowers materials and their application in electrochemical supercapacitors. J. Alloy. Compd. 2011, 509, 10080–10085. [Google Scholar] [CrossRef]
- Wu, C.; Wei, H.; Ning, B.; Xie, Y. New Vanadium Oxide Nanostructures: Controlled Synthesis and Their Smart Electrical Switching Properties. Adv. Mater. 2010, 22, 1972–1976. [Google Scholar] [CrossRef]
- Zavalij, P.Y.; Whittingham, M.S. Structural chemistry of vanadium oxides with open frameworks. Acta Crystallogr. Sect. B 1999, 55, 627–663. [Google Scholar] [CrossRef] [Green Version]
- Prześniak-Welenc, M.; Łapiński, M.; Lewandowski, T.; Kościelska, B.; Wicikowski, L.; Sadowski, W. The Influence of Thermal Conditions on V2O5 Nanostructures Prepared by Sol-Gel Method. J. Nanomater. 2015, 2015. [Google Scholar] [CrossRef] [Green Version]
- Petkov, V.; Trikalitis, P.N.; Bozin, E.S.; Billinge, S.J.L.; Vogt, T.; Kanatzidis, M.G. Structure of V2O5•nH2O Xerogel Solved by the Atomic Pair Distribution Function Technique. J. Am. Chem. Soc. 2002, 124, 10157–10162. [Google Scholar] [CrossRef]
- Timmerman, M.A.; Xia, R.; Le, P.T.P.; Wang, Y.; ten Elshof, J.E. Metal Oxide Nanosheets as 2D Building Blocks for the Design of Novel Materials. Chem.—A Eur. J. 2020, 26, 9084–9098. [Google Scholar] [CrossRef] [PubMed]
- Saini, M.; Dehiya, B.S.; Umar, A.; Goyat, M.S. Phase modulation in nanocrystalline vanadium di-oxide (VO2) nanostructures using citric acid via one pot hydrothermal method. Ceram. Int. 2019, 45, 18452–18461. [Google Scholar] [CrossRef]
- Soltane, L.; Sediri, F. Hydrothermal synthesis and characterization of mesoporous rod-like hybrid organic-inorganic nanocrystalline based vanadium oxide. Ceram. Int. 2014, 40, 1531–1538. [Google Scholar] [CrossRef]
- Liang, X.; Gao, G.; Wu, G.; Yang, H. Synthesis and characterization of novel hierarchical starfish-like vanadium oxide and their electrochemical performance. Electrochim. Acta 2016, 188, 625–635. [Google Scholar] [CrossRef]
- Mjejri, I.; Etteyeb, N.; Sediri, F. Mesoporous vanadium oxide nanostructures: Hydrothermal synthesis, optical and electrochemical properties. Ceram. Int. 2014, 40, 1387–1397. [Google Scholar] [CrossRef]
- Li, G.; Chao, K.; Peng, H.; Chen, K.; Zhang, Z. Low-Valent Vanadium Oxide Nanostructures with Controlled Crystal Structures and Morphologies. Inorg. Chem. 2007, 46, 5787–5790. [Google Scholar] [CrossRef]
- Livage, J. Hydrothermal Synthesis of Nanostructured Vanadium Oxides. Materials 2010, 3, 4175–4195. [Google Scholar] [CrossRef]
- Subba Reddy, C.V.; Mho, S.-i.; Kalluru, R.R.; Williams, Q.L. Hydrothermal synthesis of hydrated vanadium oxide nanobelts using poly (ethylene oxide) as a template. J. Power Sources 2008, 179, 854–857. [Google Scholar] [CrossRef]
- Grigorieva, A.V.; Goodilin, E.A.; Anikina, A.V.; Kolesnik, I.V.; Tretyakov, Y.D. Surfactants in the formation of vanadium oxide nanotubes. Mendeleev. Commun. 2008, 18, 71–72. [Google Scholar] [CrossRef]
- Chandrappa, G.T.; Steunou, N.; Cassaignon, S.; Bauvais, C.; Livage, J. Hydrothermal synthesis of vanadium oxide nanotubes from V2O5 gels. Catal. Today 2003, 78, 85–89. [Google Scholar] [CrossRef]
- Li, H.-Y.; Qiu, X.; Dong, M.; Li, X.; Zhang, Y.; Xie, B. Tuned hydrothermal synthesis of vanadium dioxide nanotubes. Ceram. Int. 2015, 41, 13967–13973. [Google Scholar] [CrossRef]
- Vo, T.N.; Kim, H.; Hur, J.; Choi, W.; Kim, I.T. Surfactant-assisted ammonium vanadium oxide as a superior cathode for calcium-ion batteries. J. Mater. Chem. A 2018, 6, 22645–22654. [Google Scholar] [CrossRef]
- Wang, X.; Liu, L.; Jacobson, A.J. Electrochemical–hydrothermal synthesis and structure determination of a novel layered mixed-valence oxide: BaV7O16•nH2O. Chem. Commun. 1998, 9, 1009–1010. [Google Scholar] [CrossRef]
- Petkov, V.; Zavalij, P.Y.; Lutta, S.; Whittingham, M.S.; Parvanov, V.; Shastri, S. Structure beyond Bragg: Study of V2O5 nanotubes. Phys. Rev. B 2004, 69, 085410. [Google Scholar] [CrossRef]
- Hellmann, I.; Täschner, C.; Klingeler, R.; Leonhardt, A.; Büchner, B.; Knupfer, M. Structure and electronic properties of Li-doped vanadium oxide nanotubes. J. Chem. Phys. 2008, 128, 224701. [Google Scholar] [CrossRef] [PubMed]
- McNulty, D.; Buckley, D.N.; O’Dwyer, C. Synthesis and electrochemical properties of vanadium oxide materials and structures as Li-ion battery positive electrodes. J. Power Sources 2014, 267, 831–873. [Google Scholar] [CrossRef]
- Krumeich, F.; Muhr, H.J.; Niederberger, M.; Bieri, F.; Schnyder, B.; Nesper, R. Morphology and Topochemical Reactions of Novel Vanadium Oxide Nanotubes. J. Am. Chem. Soc. 1999, 121, 8324–8331. [Google Scholar] [CrossRef]
- Nordlinder, S.; Lindgren, J.; Gustafsson, T.; Edström, K. The Structure and Electrochemical Performance of Na +, K+, and Ca2+ Vanadium Oxide Nanotubes. J. Electrochem. Soc. 2003, 150, E280. [Google Scholar] [CrossRef]
- Liu, A.; Ichihara, M.; Honma, I.; Zhou, H. Vanadium-oxide nanotubes: Synthesis and template-related electrochemical properties. Electrochem. Commun. 2007, 9, 1766–1771. [Google Scholar] [CrossRef]
- Huang, C.; Liu, X.; Kong, L.; Zhou, H.; Liu, Y.; Qiu, J.; Wang, Y. Hydrothermal synthesis of vanadium oxide nanotubes by a facile route. Rare Met. 2006, 25, 88–93. [Google Scholar] [CrossRef]
- Navas, D.; Fuentes, S.; Castro-Alvarez, A.; Chavez-Angel, E. Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials. Gels 2021, 7, 275. [Google Scholar] [CrossRef] [PubMed]
- Perera, S.D.; Patel, B.; Bonso, J.; Grunewald, M.; Ferraris, J.P.; Balkus, K.J., Jr. Vanadium Oxide Nanotube Spherical Clusters Prepared on Carbon Fabrics for Energy Storage Applications. ACS Appl. Mater. Interfaces 2011, 3, 4512–4517. [Google Scholar] [CrossRef] [PubMed]
- Navas, D.; Donoso, J.P.; Magon, C.; Sotomayor-Torres, C.M.; Moreno, M.; Lozano, H.; Benavente, E.; González, G. Ammonium hexadeca-oxo-heptavanadate microsquares. A new member in the family of the V7O16 mixed-valence nanostructures. New J. Chem. 2019, 43, 17548–17556. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, J.; Wang, H.; Wei, X.; Zhang, J.; Chen, H.; Liu, S.; Wei, S.; Lu, X. Hydrothermal synthesis of ammonium vanadate [(NH4)2V7O16•3.6 H2O] as a promising zinc-ion cathode: Experimental and theoretical study of its storage. Electrochim. Acta 2022, 404, 139785. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Lavayen, V.; Fuenzalida, D.; Newcomb, S.B.; Santa Ana, M.A.; Benavente, E.; González, G.; Sotomayor Torres, C.M. Six-fold rotationally symmetric vanadium oxide nanostructures by a morphotropic phase transition. Phys. Status Solidi (b) 2007, 244, 4157–4160. [Google Scholar] [CrossRef]
- Shao, J.; Li, X.; Qu, Q.; Zheng, H. One-step hydrothermal synthesis of hexangular starfruit-like vanadium oxide for high power aqueous supercapacitors. J. Power Sources 2012, 219, 253–257. [Google Scholar] [CrossRef]
- Saleta, M.E.; Aurelio, G.; Bardelli, F.; Sánchez, R.D.; Malta, M.; Torresi, R.M. The local environment of Co2+ ions intercalated in vanadium oxide/hexadecylamine nanotubes. J. Phys. Condens. Matter 2012, 24, 435302. [Google Scholar] [CrossRef]
- Won Lee, K.; Kweon, H.; Lee, C.E. Electron spin resonance of defect-rich vanadium oxide nanotubes. J. Appl. Phys. 2009, 106, 044313. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Navas, D.; Lavayen, V.; Benavente, E.; Santa Ana, M.A.; González, G.; Newcomb, S.B.; Sotomayor Torres, C.M. Nano-Urchin: The Formation and Structure of High-Density Spherical Clusters of Vanadium Oxide Nanotubes. Chem. Mater. 2006, 18, 3016–3022. [Google Scholar] [CrossRef]
- Roppolo, M.; Jacobs, C.B.; Upreti, S.; Chernova, N.A.; Whittingham, M.S. Synthesis and characterization of layered and scrolled amine-templated vanadium oxides. J. Mater. Sci. 2008, 43, 4742–4748. [Google Scholar] [CrossRef]
- Wörle, M.; Krumeich, F.; Bieri, F.; Muhr, H.-J.; Nesper, R. Flexible V7O16 Layers as the Common Structural Element of Vanadium Oxide Nanotubes and a New Crystalline Vanadate. Z. Für Anorg. Allg. Chem. 2002, 628, 2778–2784. [Google Scholar] [CrossRef]
- Ma, Y.; Ji, S.; Zhou, H.; Zhang, S.; Li, R.; Zhu, J.; Li, W.; Guo, H.; Jin, P. Synthesis of novel ammonium vanadium bronze (NH4)0.6V2O5 and its application in Li-ion battery. RSC Adv. 2015, 5, 90888–90894. [Google Scholar] [CrossRef]
- Ma, Y.; Wu, M.; Jin, X.; Shu, R.; Hu, C.; Xu, T.; Li, J.; Meng, X.; Cao, X. (NH4)2V7O16 Microbricks as a Novel Anode for Aqueous Lithium-Ion Battery with Good Cyclability. Chem.—A Eur. J. 2021, 27, 12341–12351. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.W.; Bu, H.; Hyoung, J.; Hong, S.-T. Ammonium Vanadium Bronze, (NH4)2V7O16, as a New Lithium Intercalation Host Material. Inorg. Chem. 2020, 59, 4320–4327. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Shu, R.; Xu, T.; Li, J.; Zhu, D.; Jin, X.; Wu, M.; Cao, X. Self-assembled (NH4)2V7O16 hierarchical structures with improved electrochemical performance for aqueous Li-ion batteries. New J. Chem. 2021, 45, 20099–20102. [Google Scholar] [CrossRef]
- Jacobs, C.; Roppolo, M.; Butterworth, K.; Ban, C.; Chernova, N.A.; Whittingham, M.S. Magnetic Properties of Vanadium Oxide Nanotubes, Nanourchins, and Nanorods. MRS Online Proc. Libr. 2007, 988. [Google Scholar] [CrossRef]
- Lavayen, V.; O’Dwyer, C.; Cárdenas, G.; González, G.; Sotomayor Torres, C.M. Towards thiol functionalization of vanadium pentoxide nanotubes using gold nanoparticles. Mater. Res. Bull. 2007, 42, 674–685. [Google Scholar] [CrossRef] [Green Version]
- Saleta, M.E.; Curiale, J.; Troiani, H.E.; Ribeiro Guevara, S.; Sánchez, R.D.; Malta, M.; Torresi, R.M. Influence of Ni doping on vanadium oxide/hexadecylamine multiwall nanotubes. Phys. B Condens. Matter. 2007, 398, 333–336. [Google Scholar] [CrossRef]
- Zhang, K.; Gao, G.; Sun, W.; Liang, X.; Liu, Y.; Wu, G. Large interlayer spacing vanadium oxide nanotubes as cathodes for high performance sodium ion batteries. RSC Adv. 2018, 8, 22053–22061. [Google Scholar] [CrossRef] [Green Version]
- Azita Saliman, H.R.A.; Ketabi, S. Synthesis and Characterization of Zr-Doped Vanadium Oxide Nanotubes. Am. J. Chem. Eng. 2018, 6, 49–53. [Google Scholar] [CrossRef]
- Krusin-Elbaum, L.; Newns, D.M.; Zeng, H.; Derycke, V.; Sun, J.Z.; Sandstrom, R. Room-temperature ferromagnetic nanotubes controlled by electron or hole doping. Nature 2004, 431, 672–676. [Google Scholar] [CrossRef] [PubMed]
- Popa, A.I.; Vavilova, E.; Arango, Y.C.; Kataev, V.; Täschner, C.; Klauss, H.H.; Maeter, H.; Luetkens, H.; Büchner, B.; Klingeler, R. High-temperature ferromagnetism of Li-doped vanadium oxide nanotubes. EPL 2009, 88, 57002. [Google Scholar] [CrossRef] [Green Version]
- Saleta, M.E.; Tobia, D.; Figueroa, S.J.A.; López, C.A.; Granada, M.; Sánchez, R.D.; Malta, M.; Torresi, R.M. Aging effect on vanadium oxide hybrid nanotubes. J. Phys. Condens. Matter 2019, 31, 505701. [Google Scholar] [CrossRef] [PubMed]









| Vanadium Oxide Morphology | V5+ Precursor | Surfactant | Lattice |
|---|---|---|---|
| Nanotubes | VO(OCH(CH3)2)3 [58,59] | CnH(2n+1)NH2 | V7O162− |
| V2O5 and V2O5•1,5 H2O [60,61] | CnH(2n)(NH2)2 | V7O162− | |
| VOCl3 [15] NH4VO3 [16] | 3 < n < 20 | V7O162− | |
| Urchins | VO(OCH(CH3)2)3 [62] | 1-hexadecylamine | V7O162− |
| V2O5 [63] | 1-dodecylamine | V7O162− | |
| Squares Crosses | NH4VO3 [64,65] | 1-hexadecylamine | V7O162− |
| NH4VO3 [64] | 1-hexadecylamine | VO2 | |
| Six-fold cogs Star fruits | V2O5•1,5 H2O [66] | 1-dodecanethiol | V6O11 |
| NH4VO3 [67] | 123 Pluronic | V7O162− |
| Lattice | V4+ % | Morphology |
|---|---|---|
| V7O162− | 46 to 75% | VOx-NTs, NU and MSQ |
| VO2 V6O11 | 100% 66.7% | VOx-MC and VOx-NSF VOx-NC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navas, D. Hydrothermal Synthesis of Vanadium Oxide Microstructures with Mixed Oxidation States. Reactions 2023, 4, 1-25. https://doi.org/10.3390/reactions4010001
Navas D. Hydrothermal Synthesis of Vanadium Oxide Microstructures with Mixed Oxidation States. Reactions. 2023; 4(1):1-25. https://doi.org/10.3390/reactions4010001
Chicago/Turabian StyleNavas, Daniel. 2023. "Hydrothermal Synthesis of Vanadium Oxide Microstructures with Mixed Oxidation States" Reactions 4, no. 1: 1-25. https://doi.org/10.3390/reactions4010001





