Study of the Synthetic Approach Influence in Ni/CeO2-Based Catalysts for Methane Dry Reforming
Abstract
1. Introduction
2. Materials and Methods
2.1. Incipient Wetness Impregnation
2.2. Coprecipitation
2.3. Nitrates Combustion
2.4. Characterization
2.5. Catalytic Tests
3. Results and Discussion
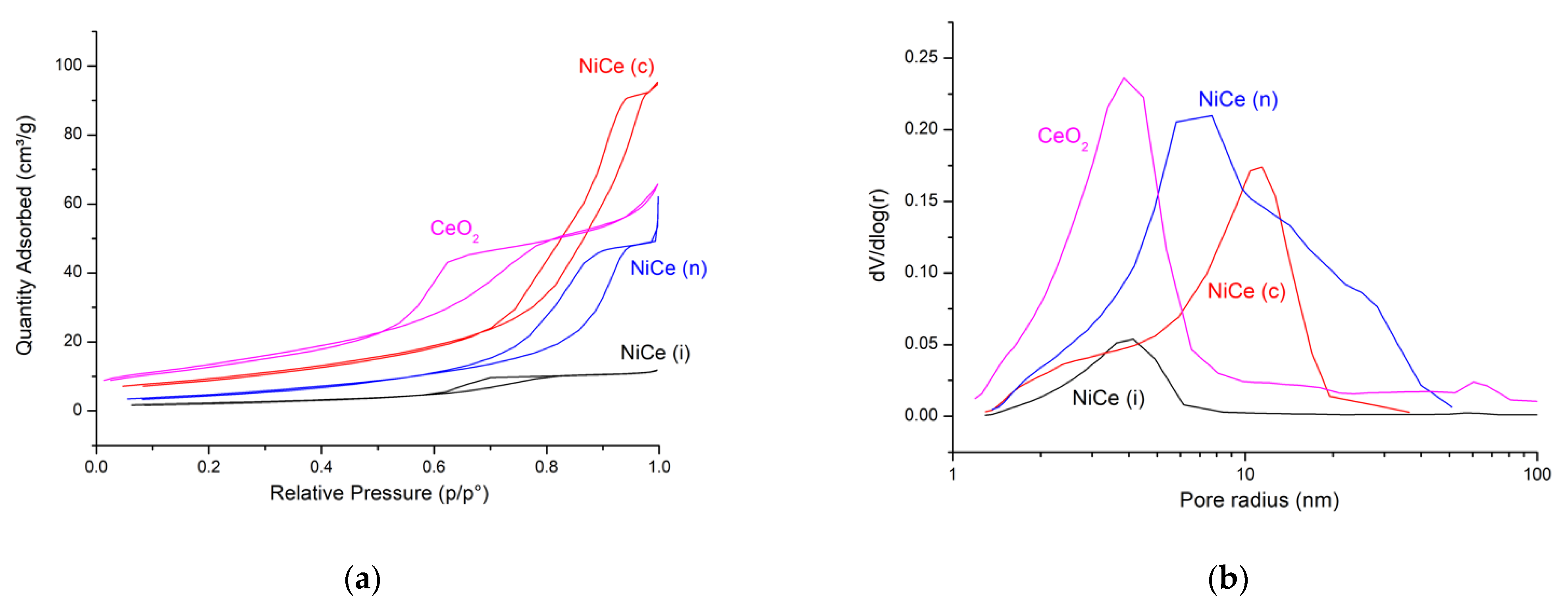
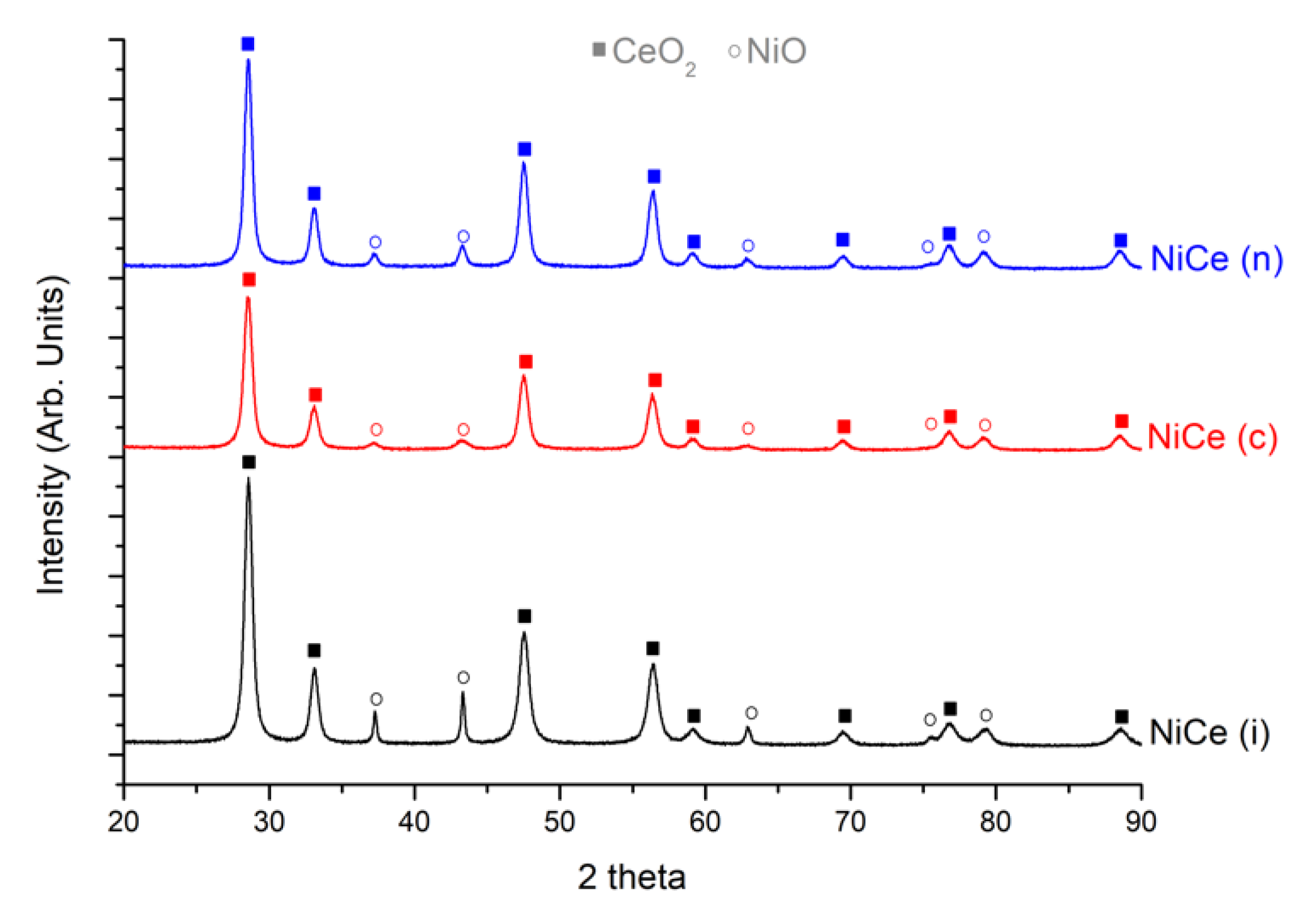
3.1. Characterizations
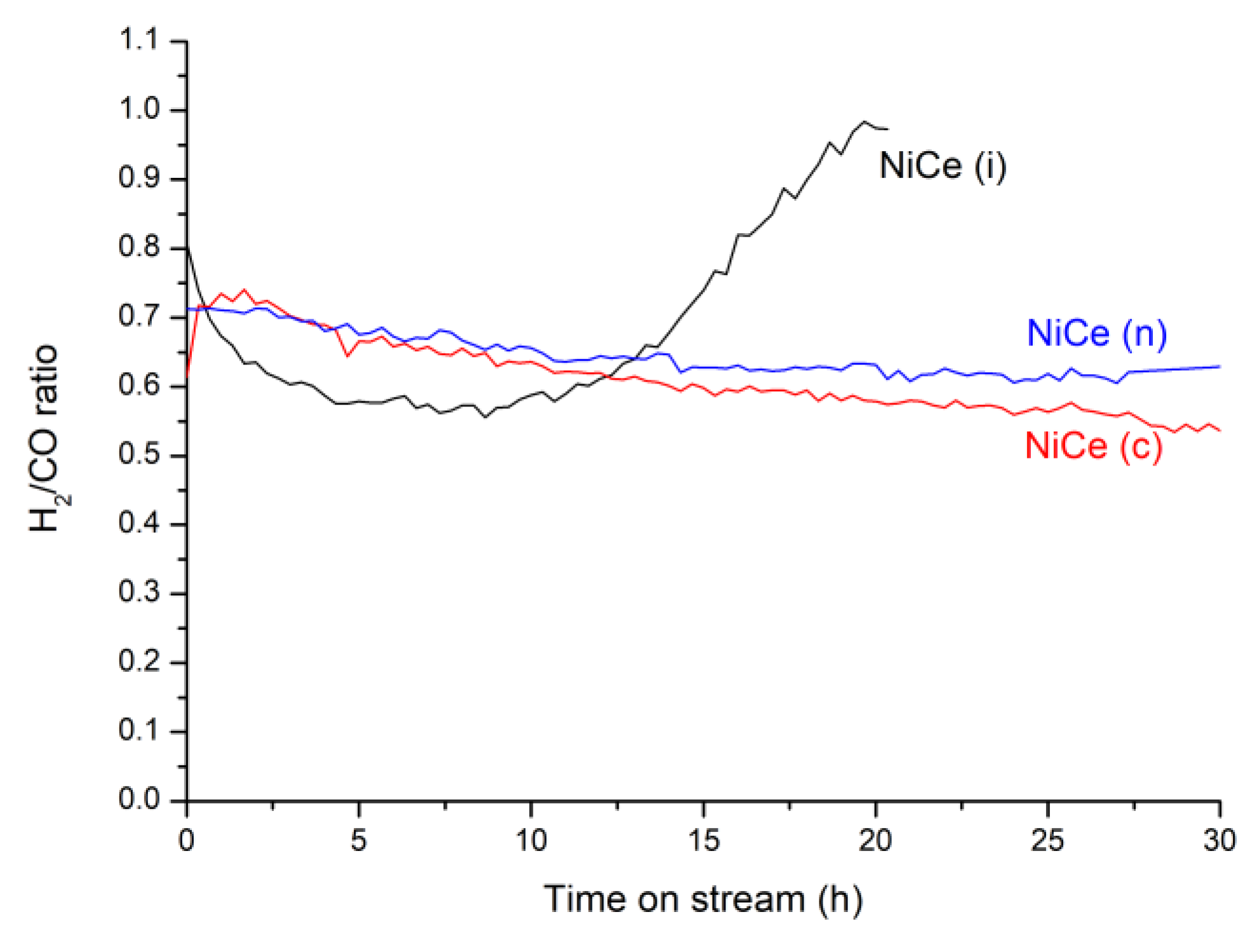
3.2. Catalytic Tests
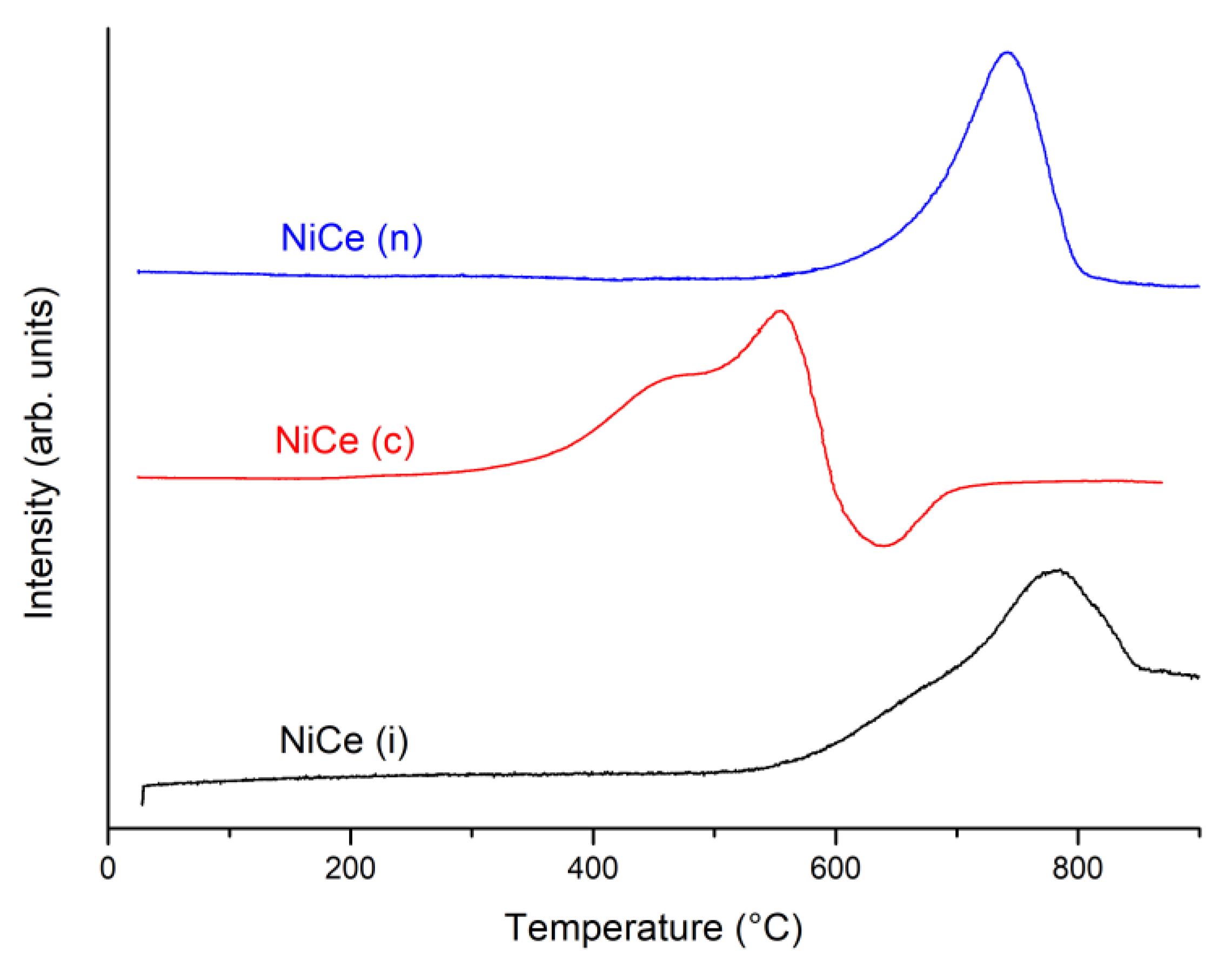
3.3. Characterization of Spent Catalyst
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thompson, L.G. Climate Change: The Evidence and Our Options. Behav. Anal. 2010, 33, 153–170. [Google Scholar] [CrossRef] [PubMed]
- AR6 Climate Change 2022: Impacts, Adaptation and Vulnerability—IPCC. Available online: https://www.ipcc.ch/report/sixth-assessment-report-working-group-ii/ (accessed on 26 April 2022).
- Naik, S.N.; Goud, V.V.; Rout, P.K.; Dalai, A.K. Production of First and Second Generation Biofuels: A Comprehensive Review. Renew. Sust. Energ. Rev. 2010, 14, 578–597. [Google Scholar] [CrossRef]
- Yang, L.; Ge, X.; Wan, C.; Yu, F.; Li, Y. Progress and Perspectives in Converting Biogas to Transportation Fuels. Renew. Sust. Energ. Rev. 2014, 40, 1133–1152. [Google Scholar] [CrossRef]
- Boyano, A.; Morosuk, T.; Blanco-Marigorta, A.M.; Tsatsaronis, G. Conventional and Advanced Exergoenvironmental Analysis of a Steam Methane Reforming Reactor for Hydrogen Production. J. Clean. Prod. 2012, 20, 152–160. [Google Scholar] [CrossRef]
- Rostrup-Nielsen, J.R.; Sehested, J.; Nørskov, J.K. Hydrogen and Synthesis Gas by Steam- and CO2 Reforming. Adv. Catal. 2002, 47, 65–139. [Google Scholar] [CrossRef]
- Minutillo, M.; Perna, A.; Sorce, A. Green Hydrogen Production Plants via Biogas Steam and Autothermal Reforming Processes: Energy and Exergy Analyses. Appl. Energy 2020, 277, 115452. [Google Scholar] [CrossRef]
- Wittich, K.; Krämer, M.; Bottke, N.; Schunk, S.A. Catalytic Dry Reforming of Methane: Insights from Model Systems. ChemCatChem. 2020, 12, 2130–2147. [Google Scholar] [CrossRef]
- Pizzolitto, C.; Pupulin, E.; Menegazzo, F.; Ghedini, E.; di Michele, A.; Mattarelli, M.; Cruciani, G.; Signoretto, M. Nickel Based Catalysts for Methane Dry Reforming: Effect of Supports on Catalytic Activity and Stability. Int. J. Hydrogen Energy 2019, 44, 28065–28076. [Google Scholar] [CrossRef]
- de Araújo Moreira, T.G.; de Carvalho Filho, J.F.S.; Carvalho, Y.; de Almeida, J.M.A.R.; Nothaft Romano, P.; Falabella Sousa-Aguiar, E. Highly Stable Low Noble Metal Content Rhodium-Based Catalyst for the Dry Reforming of Methane. Fuel 2021, 287, 119536. [Google Scholar] [CrossRef]
- Menegazzo, F.; Signoretto, M.; Pinna, F.; Canton, P.; Pernicone, N. Optimization of Bimetallic Dry Reforming Catalysts by Temperature Programmed Reaction. Appl. Catal. A Gen. 2012, 439–440, 80–87. [Google Scholar] [CrossRef]
- Marinho, A.L.A.; Toniolo, F.S.; Noronha, F.B.; Epron, F.; Duprez, D.; Bion, N. Highly Active and Stable Ni Dispersed on Mesoporous CeO2-Al2O3 Catalysts for Production of Syngas by Dry Reforming of Methane. Appl. Catal. B Environ. 2021, 281, 119459. [Google Scholar] [CrossRef]
- Melchionna, M.; Trovarelli, A.; Fornasiero, P. Synthesis and Properties of Cerium Oxide-Based Materials. In Cerium Oxide (CeO₂): Synthesis, Properties and Applications, 1st ed.; Scirè, S., Palmisano, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 13–43. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, Z.; Chen, X.; Rui, N.; Betancourt, L.E.; Lin, L.; Xu, W.; Sun, C.J.; Abeykoon, A.M.M.; Rodriguez, J.A.; et al. Effects of Zr Doping into Ceria for the Dry Reforming of Methane over Ni/CeZrO2 Catalysts: In Situ Studies with XRD, XAFS, and AP-XPS. ACS Catal. 2020, 10, 3274–3284. [Google Scholar] [CrossRef]
- Chien, A.C.; Ye, N.J.; Huang, C.W.; Tseng, I.H. Studies of Nickel/Samarium-Doped Ceria for Catalytic Partial Oxidation of Methane and Effect of Oxygen Vacancy. Catalysts 2021, 11, 731–743. [Google Scholar] [CrossRef]
- Gurav, H.R.; Dama, S.; Samuel, V.; Chilukuri, S. Influence of Preparation Method on Activity and Stability of Ni Catalysts Supported on Gd Doped Ceria in Dry Reforming of Methane. J. CO2 Util. 2017, 20, 357–367. [Google Scholar] [CrossRef]
- Odedairo, T.; Ma, J.; Chen, J.; Wang, S.; Zhu, Z. Influences of Doping Cr/Fe/Ta on the Performance of Ni/CeO2 Catalyst under Microwave Irradiation in Dry Reforming of CH4. J. Solid State Chem. 2016, 233, 166–177. [Google Scholar] [CrossRef]
- Lara-García, H.A.; Araiza, D.G.; Méndez-Galván, M.; Tehuacanero-Cuapa, S.; Gómez-Cortés, A.; Díaz, G. Dry Reforming of Methane over Nickel Supported on Nd–Ceria: Enhancement of the Catalytic Properties and Coke Resistance. RSC Adv. 2020, 10, 33059–33070. [Google Scholar] [CrossRef]
- Mitchell, S.; Michels, N.L.; Pérez-Ramírez, J. From Powder to Technical Body: The Undervalued Science of Catalyst Scale Up. Chem. Soc. Rev. 2013, 42, 6094–6112. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 2002, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 2002, 73, 373–380. [Google Scholar] [CrossRef]
- Manzoli, M.; Menegazzo, F.; Signoretto, M.; Cruciani, G.; Pinna, F. Effects of Synthetic Parameters on the Catalytic Performance of Au/CeO2 for Furfural Oxidative Esterification. J. Catal. 2015, 330, 465–473. [Google Scholar] [CrossRef]
- Menegazzo, F.; Burti, P.; Signoretto, M.; Manzoli, M.; Vankova, S.; Boccuzzi, F.; Pinna, F.; Strukul, G. Effect of the Addition of Au in Zirconia and Ceria Supported Pd Catalysts for the Direct Synthesis of Hydrogen Peroxide. J. Catal. 2008, 257, 369–381. [Google Scholar] [CrossRef]
- Menegazzo, F.; Pizzolitto, C.; Ghedini, E.; di Michele, A.; Cruciani, G.; Signoretto, M. Development of La Doped Ni/CeO2 for CH4/CO2 Reforming. J. Carbon Res. 2018, 4, 60–76. [Google Scholar] [CrossRef]
- Sepehri, S.; Rezaei, M.; Wang, Y.; Younesi, A.; Arandiyan, H. The Evaluation of Autothermal Methane Reforming for Hydrogen Production over Ni/CeO2 Catalysts. Int. J. Hydrogen Energy 2018, 43, 22340–22346. [Google Scholar] [CrossRef]
- Khatri, J.; Fakeeha, A.H.; Kasim, S.O.; Lanre, M.S.; Abasaeed, A.E.; Ibrahim, A.A.; Kumar, R.; Al-Fatesh, A.S. Ceria Promoted Phosphate-Zirconia Supported Ni Catalyst for Hydrogen Rich Syngas Production through Dry Reforming of Methane. Int. J. Energy Res. 2021, 45, 19289–19302. [Google Scholar] [CrossRef]
- Pizzolitto, C.; Menegazzo, F.; Ghedini, E.; Innocenti, G.; di Michele, A.; Cruciani, G.; Cavani, F.; Signoretto, M. Increase of Ceria Redox Ability by Lanthanum Addition on Ni Based Catalysts for Hydrogen Production. ACS Sustain. Chem. Eng. 2018, 6, 13867–13876. [Google Scholar] [CrossRef]
- Lamonier, C.; Ponchel, A.; D’Huysser, A.; Jalowiecki-Duhamel, L. Studies of the Cerium-Metal–Oxygen–Hydrogen System (Metal=Cu, Ni). Catal. Today 1999, 50, 247–259. [Google Scholar] [CrossRef]
- Li, L.; Jiang, B.; Tang, D.; Zheng, Z.; Zhao, C. Hydrogen Production from Chemical Looping Reforming of Ethanol Using Ni/CeO2 Nanorod Oxygen Carrier. Catalysts 2018, 8, 257. [Google Scholar] [CrossRef]
- Matus, E.V.; Shlyakhtina, A.S.; Sukhova, O.B.; Ismagilov, I.Z.; Ushakov, V.A.; Yashnik, S.A.; Nikitin, A.P.; Bharali, P.; Kerzhentsev, M.A.; Ismagilov, Z.R. Effect of Preparation Methods on the Physicochemical and Functional Properties of Ni/CeO2 Catalysts. Kinet. Catal. 2019, 60, 221–230. [Google Scholar] [CrossRef]
- Vita, A.; Italiano, C.; Fabiano, C.; Laganà, M.; Pino, L. Influence of Ce-Precursor and Fuel on Structure and Catalytic Activity of Combustion Synthesized Ni/CeO2 Catalysts for Biogas Oxidative Steam Reforming. Mater. Chem. Phys. 2015, 163, 337–347. [Google Scholar] [CrossRef]
- Palneedi, H.; Mangam, V.; Das, S.; Das, K. Effect of Fuel-to-Nitrate Ratio on the Powder Characteristics of Nanosized CeO2 Synthesized by Mixed Fuel Combustion Method. J. Alloys Compd. 2011, 509, 9912–9918. [Google Scholar] [CrossRef]
- Zhao, K.; Hou, X.; Norton, M.G.; Ha, S. Application of a NiMo–Ce0.5Zr0.5O2-Δ Catalyst for Solid Oxide Fuel Cells Running on Gasoline. J. Power Source 2019, 435, 226732. [Google Scholar] [CrossRef]
- Odedairo, T.; Chen, J.; Zhu, Z. Metal–Support Interface of a Novel Ni–CeO2 Catalyst for Dry Reforming of Methane. Catal. Commun. 2013, 31, 25–31. [Google Scholar] [CrossRef]
- Chein, R.; Yang, Z. Experimental Study on Dry Reforming of Biogas for Syngas Production over Ni-Based Catalysts. ACS Omega 2019, 4, 20911–20922. [Google Scholar] [CrossRef] [PubMed]
- Yahi, N.; Menad, S.; Rodríguez-Ramos, I. Dry Reforming of Methane over Ni/CeO2 Catalysts Prepared by Three Different Methods. Green Process. Synth. 2015, 4, 479–486. [Google Scholar] [CrossRef]
- Kambolis, A.; Matralis, H.; Trovarelli, A.; Papadopoulou, C. Ni/CeO2-ZrO2 Catalysts for the Dry Reforming of Methane. Appl. Catal. A Gen. 2010, 377, 16–26. [Google Scholar] [CrossRef]
- Rood, S.C.; Ahmet, H.B.; Gomez-Ramon, A.; Torrente-Murciano, L.; Reina, T.R.; Eslava, S. Enhanced Ceria Nanoflakes Using Graphene Oxide as a Sacrificial Template for CO Oxidation and Dry Reforming of Methane. Appl. Catal. B Environ. 2019, 242, 358–368. [Google Scholar] [CrossRef]
- Ay, H.; Üner, D. Dry Reforming of Methane over CeO2 Supported Ni, Co and Ni-Co Catalysts. Appl. Catal. B Environ. 2015, 179, 128–138. [Google Scholar] [CrossRef]
- Gonzalez-Delacruz, V.M.; Ternero, F.; Pereñíguez, R.; Caballero, A.; Holgado, J.P. Study of Nanostructured Ni/CeO2 Catalysts Prepared by Combustion Synthesis in Dry Reforming of Methane. Appl. Catal. A Gen. 2010, 384, 1–9. [Google Scholar] [CrossRef]
- Guerrero-Caballero, J.; Kane, T.; Haidar, N.; Jalowiecki-Duhamel, L.; Löfberg, A. Ni, Co, Fe Supported on Ceria and Zr Doped Ceria as Oxygen Carriers for Chemical Looping Dry Reforming of Methane. Catal. 2019, 333, 251–258. [Google Scholar] [CrossRef]
- Grabchenko, M.; Pantaleo, G.; Puleo, F.; Kharlamova, T.S.; Zaikovskii, V.I.; Vodyankina, O.; Liotta, L.F. Design of Ni-Based Catalysts Supported over Binary La-Ce Oxides: Influence of La/Ce Ratio on the Catalytic Performances in DRM. Catal. Today 2021, 382, 71–81. [Google Scholar] [CrossRef]
- Marinho, A.L.A.; Rabelo-Neto, R.C.; Epron, F.; Bion, N.; Toniolo, F.S.; Noronha, F.B. Embedded Ni Nanoparticles in CeZrO2 as Stable Catalyst for Dry Reforming of Methane. Appl. Catal. B Environ. 2020, 268, 118387. [Google Scholar] [CrossRef]
- Luisetto, I.; Tuti, S.; di Bartolomeo, E. Co and Ni Supported on CeO2 as Selective Bimetallic Catalyst for Dry Reforming of Methane. Int. J. Hydrogen Energy 2012, 37, 15992–15999. [Google Scholar] [CrossRef]
- Cesario, M.R.; Souza, G.S.; Loureiro, F.J.A.; Araújo, A.J.M.; Grilo, J.P.F.; Aouad, S.; Tidahy, H.L.; Macedo, D.A.; Fagg, D.P.; Gennequin, C.; et al. Synthesis of Co–Ni and Cu–Ni Based-Catalysts for Dry Reforming of Methane as Potential Components for SOFC Anodes. Ceram. Int. 2021, 47, 33191–33201. [Google Scholar] [CrossRef]
- Majewski, A.J.; Singh, S.K.; Labhasetwar, N.K.; Steinberger-Wilckens, R. Nickel–Molybdenum Catalysts for Combined Solid Oxide Fuel Cell Internal Steam and Dry Reforming. Chem. Eng. Sci. 2021, 232, 116341. [Google Scholar] [CrossRef]
- da Fonseca, R.O.; Ponseggi, A.R.; Rabelo-Neto, R.C.; Simões, R.C.C.; Mattos, L.V.; Noronha, F.B. Controlling Carbon Formation over Ni/CeO2 Catalyst for Dry Reforming of CH4 by Tuning Ni Crystallite Size and Oxygen Vacancies of the Support. J. CO2 Util. 2022, 57, 101880. [Google Scholar] [CrossRef]
- Jang, W.J.; Shim, J.O.; Kim, H.M.; Yoo, S.Y.; Roh, H.S. A Review on Dry Reforming of Methane in Aspect of Catalytic Properties. Catal. Today 2019, 324, 15–26. [Google Scholar] [CrossRef]





| CeO2 | NiCe (i) | NiCe (c) | NiCe (n) | |
|---|---|---|---|---|
| Surface area (m2/g) | 49 | 8 | 34 | 18 |
| Pore volume (cm3/g) | 0.11 | 0.02 | 0.15 | 0.08 |
| Average pore radius (nm) | 4 | 4 | 10 | 7 |
| Effective Ni amount (wt. %) | - | 14.5 | 14.0 | 15.0 |
| Catalyst | NiO Dimensions (nm) | NiO wt. % | CeO2 wt. % |
|---|---|---|---|
| NiCe (i) | 30 | 20 | 80 |
| NiCe (c) | 7 | 21 | 79 |
| NiCe (n) | 13 | 21 | 79 |
| Catalyst | Ni wt. % | Synthetic Method | Conversion (%) | H2/CO Ratio | Temp. (°C) | Ref. | |
|---|---|---|---|---|---|---|---|
| CH4 | CO2 | ||||||
| NiCe (c) | 14 | Coprecipitation | 95 | 97 | 0.6 | 700 | This work |
| Ni/CeO2 flakes | 10 | Impregnation | 15 | 42 | 0.5 | 700 | [38] |
| Ni/CeO2 Nanoparticles | 10 | Impregnation | 18 | 45 | 0.6 | 700 | [38] |
| Ni/CeO2 | 8 | Wet impregnation | 80 | 75 | Lower than 1.0 | 700 | [39] |
| Ni/CeO2 | 13 | Combustion synthesis | 90 | - | 0.95 | 750 | [40] |
| Ni/CeO2 | 9.5 | Wet Impregnation | 65 | 65 | 2 | 700 | [41] |
| Ni/commercial CeO2 | 7.5 | Wet impregnation | 45 | 45 | 2 | 700 | [41] |
| CeNi0.3Oy | 7.5 | Coprecipitation | 5 | 25 | 2 | 700 | [41] |
| Ni/CeO2 | 10 | Wetness Impregnation | 35 | 65 | 0.65 | 700 | [42] |
| Ni/@CeO2 | 10 | Self-assembly | 55 | 65 | 0.8 | 800 | [43] |
| Ni/CeO2 | 10 | Incipient wetness impregnation | 60 | 70 | 0.8 | 800 | [43] |
| Ni/CeO2 | 7.5 | Surfactant assisted coprecipitation | 70 | 75 | 0.8 | 700 | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pizzolato, M.; Da Pian, G.; Ghedini, E.; Di Michele, A.; Menegazzo, F.; Cruciani, G.; Signoretto, M. Study of the Synthetic Approach Influence in Ni/CeO2-Based Catalysts for Methane Dry Reforming. Reactions 2022, 3, 634-647. https://doi.org/10.3390/reactions3040043
Pizzolato M, Da Pian G, Ghedini E, Di Michele A, Menegazzo F, Cruciani G, Signoretto M. Study of the Synthetic Approach Influence in Ni/CeO2-Based Catalysts for Methane Dry Reforming. Reactions. 2022; 3(4):634-647. https://doi.org/10.3390/reactions3040043
Chicago/Turabian StylePizzolato, Marco, Giulia Da Pian, Elena Ghedini, Alessandro Di Michele, Federica Menegazzo, Giuseppe Cruciani, and Michela Signoretto. 2022. "Study of the Synthetic Approach Influence in Ni/CeO2-Based Catalysts for Methane Dry Reforming" Reactions 3, no. 4: 634-647. https://doi.org/10.3390/reactions3040043
APA StylePizzolato, M., Da Pian, G., Ghedini, E., Di Michele, A., Menegazzo, F., Cruciani, G., & Signoretto, M. (2022). Study of the Synthetic Approach Influence in Ni/CeO2-Based Catalysts for Methane Dry Reforming. Reactions, 3(4), 634-647. https://doi.org/10.3390/reactions3040043











