Chain Propagation Mechanism of Fischer–Tropsch Synthesis: Experimental Evidence by Aldehyde, Alcohol and Alkene Addition
Abstract
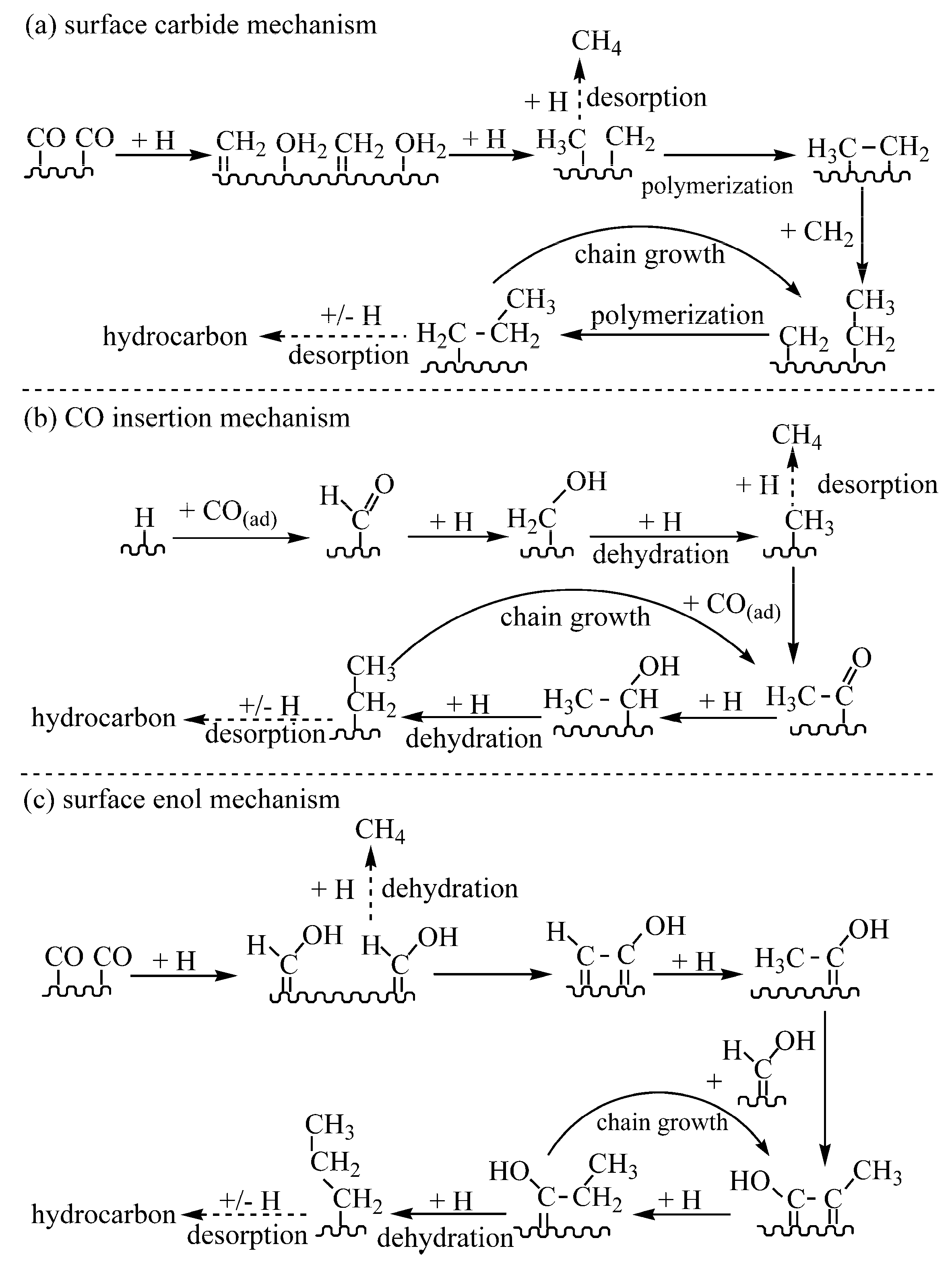
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results and Discussion
3.1. Mechanism Investigation with 1-Alkene as Single Carbon Source
3.2. Alcohol Selectivity by Co-Feeding 1-Alkene in Normal FTS Reaction
3.3. Mechanism Investigation with Aldehyde or Alcohol as Single Carbon Source
3.4. The Effects of Alkenes and Alcohols on FTS Product Distribution
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Davis, B.H. Overview of reactors for liquid phase Fischer–Tropsch synthesis. Catal. Today 2002, 71, 249–300. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, J.; Liu, Z.; Guo, Q.; Ye, M.; Wang, G.; Gao, J.; Wang, J.; Shu, Z.; Ge, W.; et al. Progress in coal chemical technologies of China. Rev. Chem. Eng. 2019, 36, 21–66. [Google Scholar] [CrossRef]
- Biloen, P.; Helle, J.N.; Sachtler, W.M.H. Incorporation of surface carbon into hydrocarbons during Fischer-Tropsch synthesis: Mechanistic implications. J. Catal. 1979, 58, 95–107. [Google Scholar] [CrossRef]
- Pichler, H. Twenty-five Years of Synthesis of Gasoline by Catalytic Conversion of Carbon Monoxide and Hydrogen. Adv. Catal. 1952, 4, 271–341. [Google Scholar] [CrossRef]
- Kummer, J.T.; Emmett, P.H. Fischer-Tropsch Synthesis Mechanism Studies. The Addition of Radioactive Alcohols to the Synthesis Gas. J. Am. Chem. Soc. 1953, 75, 5177–5183. [Google Scholar] [CrossRef]
- Kollár, M.; De Stefanis, A.; Solt, H.E.; Mihályi, M.R.; Valyon, J.; Tomlinson, A.A. The mechanism of the Fischer–Tropsch reaction over supported cobalt catalysts. J. Mol. Catal. A Chem. 2010, 333, 37–45. [Google Scholar] [CrossRef]
- Inderwildi, O.R.; Jenkins, S.J.; King, D.A. Mechanistic Studies of Hydrocarbon Combustion and Synthesis on Noble Metals. Angew. Chem. Int. Ed. 2008, 47, 5253–5255. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Jansen, A.P.J.; van Santen, R.A. Direct versus Hydrogen-Assisted CO Dissociation. J. Am. Chem. Soc. 2009, 131, 12874–12875. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.-J.; Huo, C.-F.; Liu, X.-W.; Zhao, X.-H.; Li, Y.-W.; Wang, J.; Jiao, H. Density Functional Theory Study on Surface CxHy Formation from CO Activation on Fe3C(100). J. Phys. Chem. C 2010, 114, 21585–21592. [Google Scholar] [CrossRef]
- Ojeda, M.; Nabar, R.; Nilekar, A.U.; Ishikawa, A.; Mavrikakis, M.; Iglesia, E. CO activation pathways and the mechanism of Fischer–Tropsch synthesis. J. Catal. 2010, 272, 287–297. [Google Scholar] [CrossRef]
- Zhou, L.-P.; Hao, X.; Gao, J.-H.; Yang, Y.; Wu, B.-S.; Xu, J.; Xu, Y.-Y.; Li, Y.-W. Studies and Discriminations of the Kinetic Models for the Iron-Based Fischer−Tropsch Catalytic Reaction in a Recycle Slurry Reactor. Energy Fuels 2011, 25, 52–59. [Google Scholar] [CrossRef]
- Brady, R.C.; Pettit, R. Reactions of diazomethane on transition-metal surfaces and their relationship to the mechanism of the Fischer-Tropsch reaction. J. Am. Chem. Soc. 1980, 102, 6181–6182. [Google Scholar] [CrossRef]
- Hussain, S.T. Addition of 2-Percent Ethene, Propene and Acetylene in the Co-Hydrogenation Experiment for the Determination of Chain Growth Intermediate in the Fisher Tropsch Synthesis on Ru Alumina-Supported Catalyst. J. Chem. Soc. Pak. 1993, 15, 97–99. [Google Scholar]
- Quyoum, R.; Berdini, V.; Turner, M.L.; Long, H.C.; Maitlis, P.M. Mechanistic Studies of Methylene Chain Propagation in the Fischer–Tropsch Synthesis. J. Catal. 1998, 173, 355–365. [Google Scholar] [CrossRef]
- Bell, A.T. Catalytic Synthesis of Hydrocarbons over Group VIII Metals. A Discussion of the Reaction Mechanism. Catal. Rev. 1981, 23, 203–232. [Google Scholar] [CrossRef] [Green Version]
- Eidus, Y.T. The mechanism of the Fischer–Tropsch reaction and the initiated hydropolymerisation of alkenes, from radiochemical and kinetic data. Russ. Chem. Rev. 1967, 36, 338. [Google Scholar] [CrossRef]
- Botes, F.G.; Breman, B.B. Development and Testing of a New Macro Kinetic Expression for the Iron-Based Low-Temperature Fischer−Tropsch Reaction. Ind. Eng. Chem. Res. 2006, 45, 7415–7426. [Google Scholar] [CrossRef]
- Van Der Laan, G.P.; Beenackers, A.A. Intrinsic kinetics of the gas–solid Fischer–Tropsch and water gas shift reactions over a precipitated iron catalyst. Appl. Catal. A 2000, 193, 39–53. [Google Scholar] [CrossRef]
- van Steen, E.; Schulz, H. Polymerisation kinetics of the Fischer–Tropsch CO hydrogenation using iron and cobalt based catalysts. Appl. Catal. A 1999, 186, 309–320. [Google Scholar] [CrossRef]
- Fernandes, F.A.N. Polymerization Kinetics of Fischer-Tropsch Reaction on Iron Based Catalysts and Product Grade Optimization. Chem. Eng. Technol. 2005, 28, 930–938. [Google Scholar] [CrossRef]
- Van Der Laan, G.P.; Beenackers, A.A.C.M. Hydrocarbon Selectivity Model for the Gas−Solid Fischer−Tropsch Synthesis on Precipitated Iron Catalysts. Ind. Eng. Chem. Res. 1999, 38, 1277–1290. [Google Scholar] [CrossRef]
- Blyholder, G.; Neff, L.D. Infrared study of the interaction of carbon monoxide and hydrogen on silica-supported iron. J. Phys. Chem. 1962, 66, 1664–1667. [Google Scholar] [CrossRef]
- Henrici-Olivé, G.; Olivé, S. The Fischer-Tropsch Synthesis: Molecular Weight Distribution of Primary Products and Reaction Mechanism. Angew. Chem. Int. Ed. 1976, 15, 136–141. [Google Scholar] [CrossRef]
- Huff, G.A.; Satterfield, C.N. Evidence for two chain growth probabilities on iron catalysts in the Fischer-Tropsch synthesis. J. Catal. 1984, 85, 370–379. [Google Scholar] [CrossRef]
- Iglesia, E.; Reyes, S.C.; Madon, R.J. Transport-Enhanced Alpha-Olefin Readsorption Pathways in Ru-Catalyzed Hydrocarbon Synthesis. J. Catal. 1991, 129, 238–256. [Google Scholar] [CrossRef]
- Liu, X.; Hamasaki, A.; Honma, T.; Tokunaga, M. Anti-ASF distribution in Fischer-Tropsch synthesis over unsupported cobalt catalysts in a batch slurry phase reactor. Catal. Today 2011, 175, 494–503. [Google Scholar] [CrossRef]
- Bukur, D.; Mukesh, D.; Patel, S.A. Promoter effects on precipitated iron catalysts for Fischer-Tropsch synthesis. Ind. Eng. Chem. Res. 1990, 29, 194–204. [Google Scholar] [CrossRef]
- Patzlaff, J.; Liu, Y.; Graffmann, C.; Gaube, J. Studies on product distributions of iron and cobalt catalyzed Fischer–Tropsch synthesis. Appl. Catal. A 1999, 186, 109–119. [Google Scholar] [CrossRef]
- Kuipers, E.; Scheper, C.; Wilson, J.; Vinkenburg, I.; Oosterbeek, H. Non-ASF Product Distributions Due to Secondary Reactions during Fischer–Tropsch Synthesis. J. Catal. 1996, 158, 288–300. [Google Scholar] [CrossRef]
- Schulz, H.; Claeys, M. Kinetic modelling of Fischer–Tropsch product distributions. Appl. Catal. A 1999, 186, 91–107. [Google Scholar] [CrossRef]
- Kuipers, E.; Vinkenburg, I.; Oosterbeek, H. Chain Length Dependence of α-Olefin Readsorption in Fischer-Tropsch Synthesis. J. Catal. 1995, 152, 137–146. [Google Scholar] [CrossRef]
- Koenig, L.; Gaube, J. Fischer-Tropsch synthesis—Recent studies and developments. Chem. Inf. 1983, 14, 14–22. [Google Scholar] [CrossRef]
- Tau, L.-M.; Dabbagh, H.; Bao, S.; Davis, B.H. Fischer-Tropsch synthesis. Evidence for two chain growth mechanisms. Catal. Lett. 1991, 7, 127–140. [Google Scholar] [CrossRef]
- Hall, W.K.; Kokes, R.J.; Emmett, P.H. Mechanism Studies of the Fischer-Tropsch Synthesis: The Incorporation of Radioactive Ethylene, Propionaldehyde and Propanol. J. Am. Chem. Soc. 1960, 82, 1027–1037. [Google Scholar] [CrossRef]
- Snel, R.; Espinoza, R.L. Secondary reactions of primary products of the Fischer-Tropsch synthesis. 1. The Role of Ethene. J. Mol. Catal. 1987, 43, 237–247. [Google Scholar] [CrossRef]
- Snel, R.; Espinoza, R.L. Secondary reactions of primary products of the Fischer-Tropsch synthesis. 2. The Role of Propene. J. Mol. Catal. 1989, 54, 103–117. [Google Scholar] [CrossRef]
- Snel, R.; Espinoza, R.L. Secondary reactions of primary products of the Fischer-Tropsch synthesis. 3. The Role of Butene. J. Mol. Catal. 1989, 54, 119–130. [Google Scholar] [CrossRef]
- Adesina, A.; Hudgins, R.; Silveston, P. Effect of ethene addition during the Fischer-Tropsch reaction. Appl. Catal. 1990, 62, 295–308. [Google Scholar] [CrossRef]
- Jordan, D.S.; Bell, A.T. The influence of propylene on CO hydrogenation over silica-supported ruthenium. J. Catal. 1987, 107, 338–350. [Google Scholar] [CrossRef]
- Hanlon, R.T.; Satterfield, C.N. Reactions of selected 1-olefins and ethanol added during the Fischer-Tropsch synthesis. Energy Fuels 1988, 2, 196–204. [Google Scholar] [CrossRef]
- Tau, L.-M.; Dabbagh, H.A.; Chawla, B.; Davis, B.H. Fischer-Tropsch synthesis with an iron catalyst: Incorporation of ethene into higher carbon number alkanes. Catal. Lett. 1991, 7, 141–149. [Google Scholar] [CrossRef]
- Shi, B.; Davis, B.H. Fischer–Tropsch Synthesis: Evidence for Chain Initiation by Ethene and Ethanol for an Iron Catalyst. Top. Catal. 2003, 26, 157–161. [Google Scholar] [CrossRef]
- Fujimoto, K.; Fan, L.; Yoshii, K. New controlling method for product distribution in Fischer-Tropsch synthesis reaction. Top. Catal. 1995, 2, 259–266. [Google Scholar] [CrossRef]
- Shi, B.; Jacobs, G.; Sparks, D.; Davis, B.H. Fischer–Tropsch synthesis: 14C labeled 1-alkene conversion using supercritical conditions with Co/A12O3. Fuel 2005, 84, 1093–1098. [Google Scholar] [CrossRef]
- Satterfield, C.N.; Huff, G.A.; Summerhayes, R. Olefin addition in Fischer-Tropsch synthesis on an iron catalyst. J. Catal. 1983, 80, 486–490. [Google Scholar] [CrossRef]
- Balakos, M.; Chuang, S. Transient Response of Propionaldehyde Formation During CO/H2/C2H4 Reaction on Rh/SiO2. J. Catal. 1995, 151, 253–265. [Google Scholar] [CrossRef]
- Tau, L.M.; Dabbagh, H.A.; Davis, B.H. Fischer-Tropsch synthesis: Comparison of carbon-14 distributions when labeled alcohol is added to the synthesis gas. Energy Fuels 1991, 5, 174–179. [Google Scholar] [CrossRef]
- Tau, L.-M.; Dabbagh, H.A.; Halasz, J.; Davis, B.H. Fischer-Tropsch synthesis: Incorporation of 14C-labeled normal and isoalcohols. J. Mol. Catal. 1992, 71, 37–55. [Google Scholar] [CrossRef]
- Gnanamani, M.K.; Keogh, R.A.; Shafer, W.D.; Shi, B.; Davis, B.H. Fischer–Tropsch synthesis: Deuterium labeled ethanol tracer studies on iron catalysts. Appl. Catal. A 2010, 385, 46–51. [Google Scholar] [CrossRef]
- Yang, Y.; Xiang, H.; Zhang, R.; Zhong, B.; Li, Y. A highly active and stable Fe-Mn catalyst for slurry Fischer–Tropsch synthesis. Catal. Today 2005, 106, 170–175. [Google Scholar] [CrossRef]
- Shi, B.; O’Brien, R.J.; Bao, S.; Davis, B.H. Mechanism of the Isomerization of 1-Alkene during Iron-Catalyzed Fischer–Tropsch Synthesis. J. Catal. 2001, 199, 202–208. [Google Scholar] [CrossRef]
- Cramer, R. Olefin Coordination Compounds of Rhodium. III. The Mechanism of Olefin Isomerization. J. Am. Chem. Soc. 1966, 88, 2272–2282. [Google Scholar] [CrossRef]
- Efstathiou, A.; Chafik, T.; Bianchi, D.; Bennett, C. A Transient Kinetic Study of the Co/H2 Reaction on Rh/Al2O3 Using FTIR and Mass Spectroscopy. J. Catal. 1994, 148, 224–239. [Google Scholar] [CrossRef]
- Breejen, J.P.D.; Radstake, P.B.; Bezemer, G.L.; Bitter, J.H.; Frøseth, V.; Holmen, A.; De Jong, K.P. On the Origin of the Cobalt Particle Size Effects in Fischer−Tropsch Catalysis. J. Am. Chem. Soc. 2009, 131, 7197–7203. [Google Scholar] [CrossRef] [PubMed]
- Curtis, V.; Nicolaides, C.; Coville, N.; Hildebrandt, D.; Glasser, D. The effect of sulfur on supported cobalt Fischer–Tropsch catalysts. Catal. Today 1999, 49, 33–40. [Google Scholar] [CrossRef]
- Cornils, B.; Herrmann, W.A.; Rasch, M. Otto Roelen, Pioneer in Industrial Homogeneous Catalysis. Angew. Chem. Int. Ed. 1994, 33, 2144–2163. [Google Scholar] [CrossRef]
- Wang, Y.-N.; Ma, W.-P.; Lu, Y.-J.; Yang, J.; Xu, Y.-Y.; Xiang, H.-W.; Li, Y.-W.; Zhao, Y.-L.; Zhang, B.-J. Kinetics modelling of Fischer–Tropsch synthesis over an industrial Fe–Cu–K catalyst. Fuel 2003, 82, 195–213. [Google Scholar] [CrossRef]
- Gao, J.; Wu, B.; Zhou, L.; Yang, Y.; Hao, X.; Xu, J.; Xu, Y.; Cao, L.; Li, Y. Effective control of α-olefin selectivity during Fischer–Tropsch synthesis over polyethylene-glycol enwrapped porous catalyst. Catal. Commun. 2011, 12, 1466–1470. [Google Scholar] [CrossRef]











| Product Selectivity (mol%) | H2/C2H4 | |||
|---|---|---|---|---|
| 2 | 4 | 5 | 10 | |
| CH4 | 1.4 | 2.0 | 2.0 | 1.5 |
| C2H4 | 7.1 | - | - | - |
| C2H6 | 84.9 | 93.8 | 95.1 | 97.2 |
| C3+ | 6.6 | 4.2 | 2.9 | 1.3 |
| Product Selectivity (mol%) | H2/C3H6 | ||
|---|---|---|---|
| 3 | 7 | 11 | |
| CH4 | 2.0 | 3.0 | 3.6 |
| C2H4 + C2H6 | 3.0 | 3.4 | 3.6 |
| C3H6 | - | - | - |
| C3H8 | 89.3 | 90.3 | 90.5 |
| C4+ | 5.7 | 3.3 | 2.3 |
| Product Selectivity (mol%) | H2/C6H12 | H2/C8H16 | ||
|---|---|---|---|---|
| 2.1 (X = 84.7%) a | 5.1 (X = 98.6%) | 2.1 (X = 98.0%) | 5.1 (X = 99.4%) | |
| methane | 1.2 | 2.4 | 0.9 | 2.6 |
| 2-alkene b | 56.4 | 41.1 | 50.9 | 23.9 |
| n-alkane | 35.5 | 51.6 | 43.2 | 66.5 |
| Others | 6.9 | 4.9 | 5.0 | 7.0 |
| Reaction Results (mol%) | H2/C3H6O Ratio (C3H6O, 0.5 mL/h) | ||||
|---|---|---|---|---|---|
| 2.83 | 4.38 | 7.96 | 11.88 | 21.75 | |
| Conversion, C3H6O% | 66.19 | 86.52 | 97.51 | 98.16 | 99.15 |
| Selectivity, propanol% | 93.30 | 94.45 | 95.39 | 95.84 | 96.45 |
| Selectivity, hydrocarbon (C2–C4)% | 4.73 | 3.46 | 4.51 | 4.05 | 3.35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.; Gao, J.; Hao, X.; Yang, Y.; Li, Y. Chain Propagation Mechanism of Fischer–Tropsch Synthesis: Experimental Evidence by Aldehyde, Alcohol and Alkene Addition. Reactions 2021, 2, 161-174. https://doi.org/10.3390/reactions2020012
Zhou L, Gao J, Hao X, Yang Y, Li Y. Chain Propagation Mechanism of Fischer–Tropsch Synthesis: Experimental Evidence by Aldehyde, Alcohol and Alkene Addition. Reactions. 2021; 2(2):161-174. https://doi.org/10.3390/reactions2020012
Chicago/Turabian StyleZhou, Liping, Junhu Gao, Xu Hao, Yong Yang, and Yongwang Li. 2021. "Chain Propagation Mechanism of Fischer–Tropsch Synthesis: Experimental Evidence by Aldehyde, Alcohol and Alkene Addition" Reactions 2, no. 2: 161-174. https://doi.org/10.3390/reactions2020012
APA StyleZhou, L., Gao, J., Hao, X., Yang, Y., & Li, Y. (2021). Chain Propagation Mechanism of Fischer–Tropsch Synthesis: Experimental Evidence by Aldehyde, Alcohol and Alkene Addition. Reactions, 2(2), 161-174. https://doi.org/10.3390/reactions2020012





