The Use of Soundscapes to Monitor Fish Communities: Meaningful Graphical Representations Differ with Acoustic Environment
Abstract
:1. Introduction
2. Materials and Methods
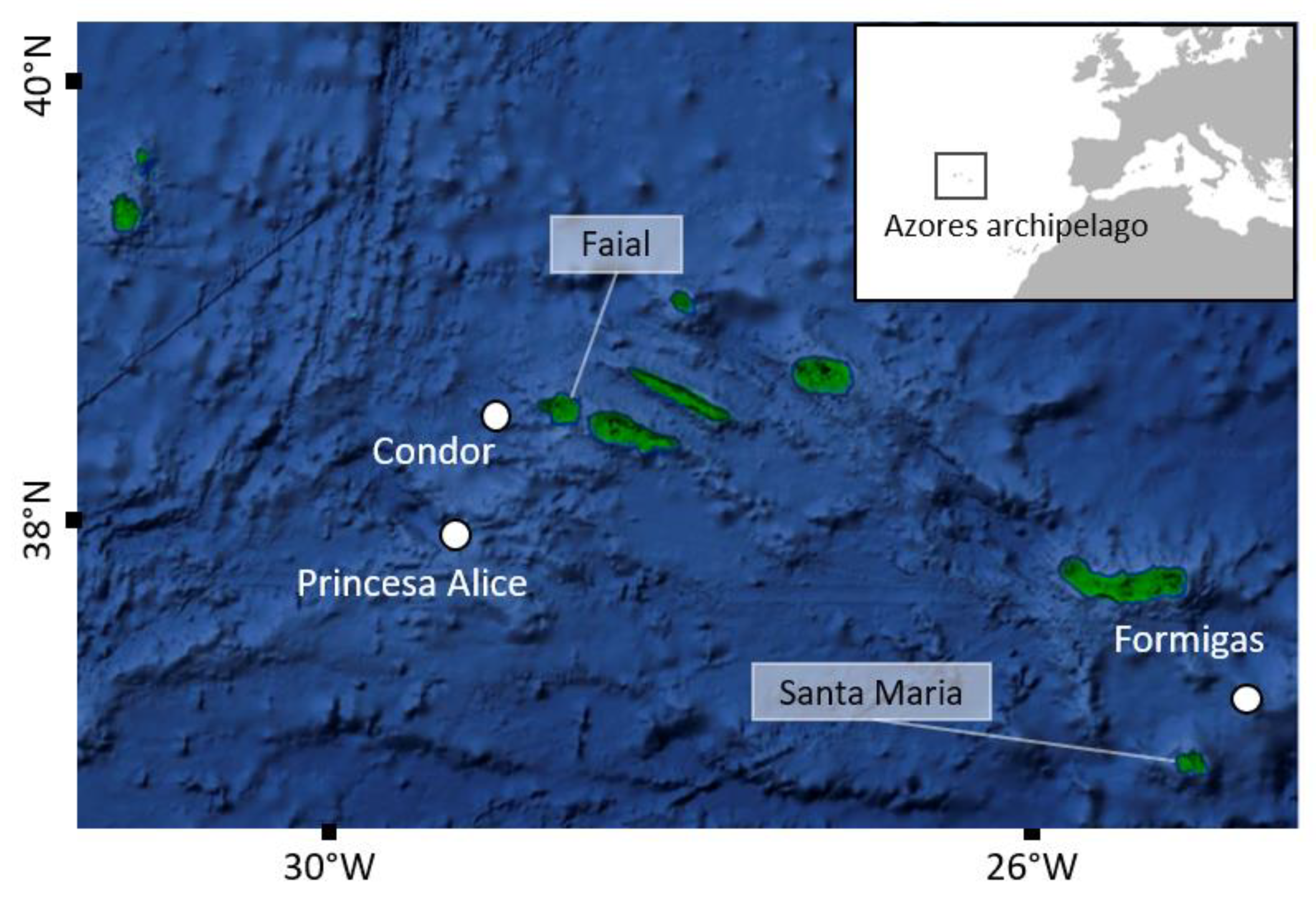
2.1. Study Site
2.2. Acoustic Recordings
2.3. Data Analysis
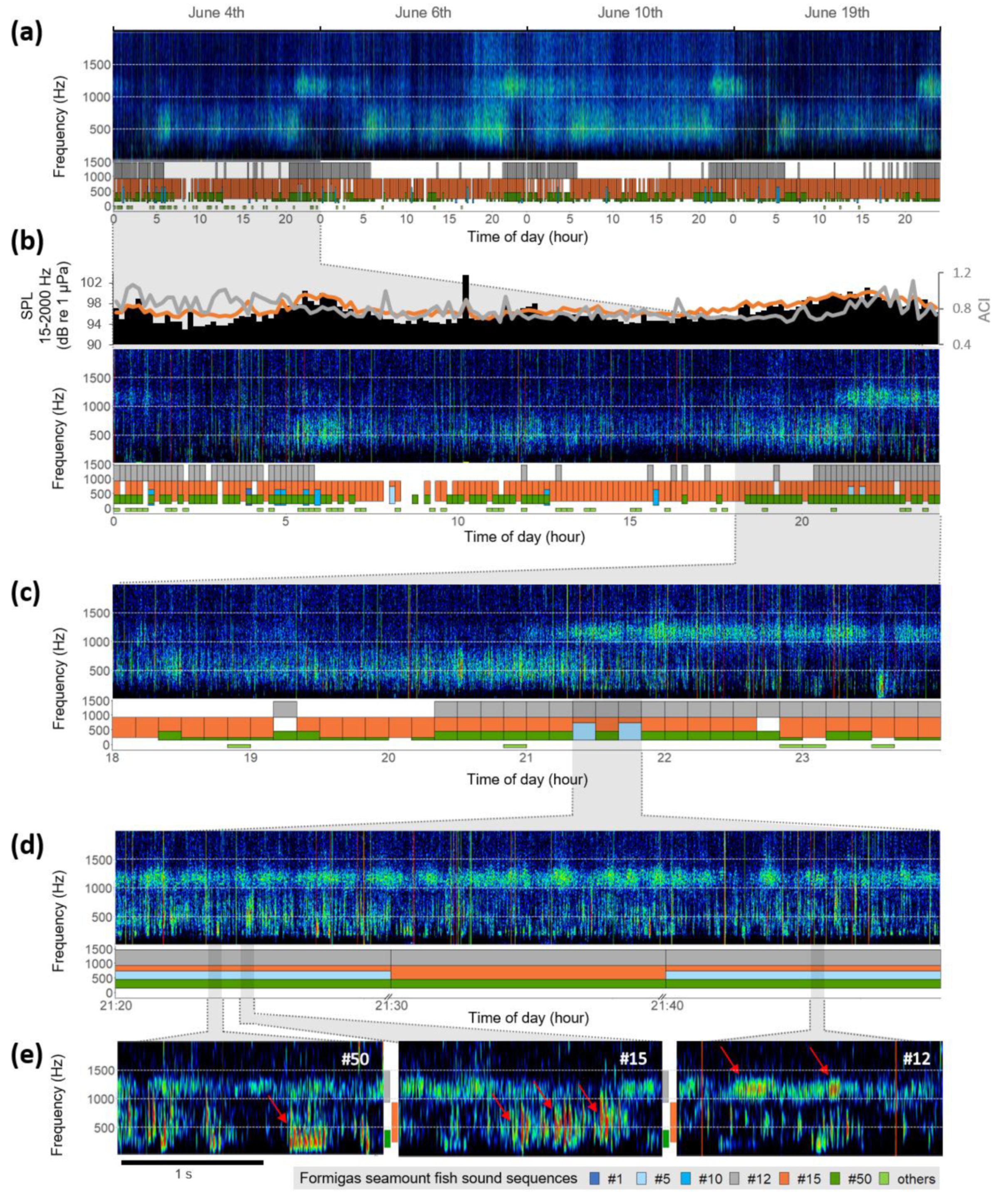
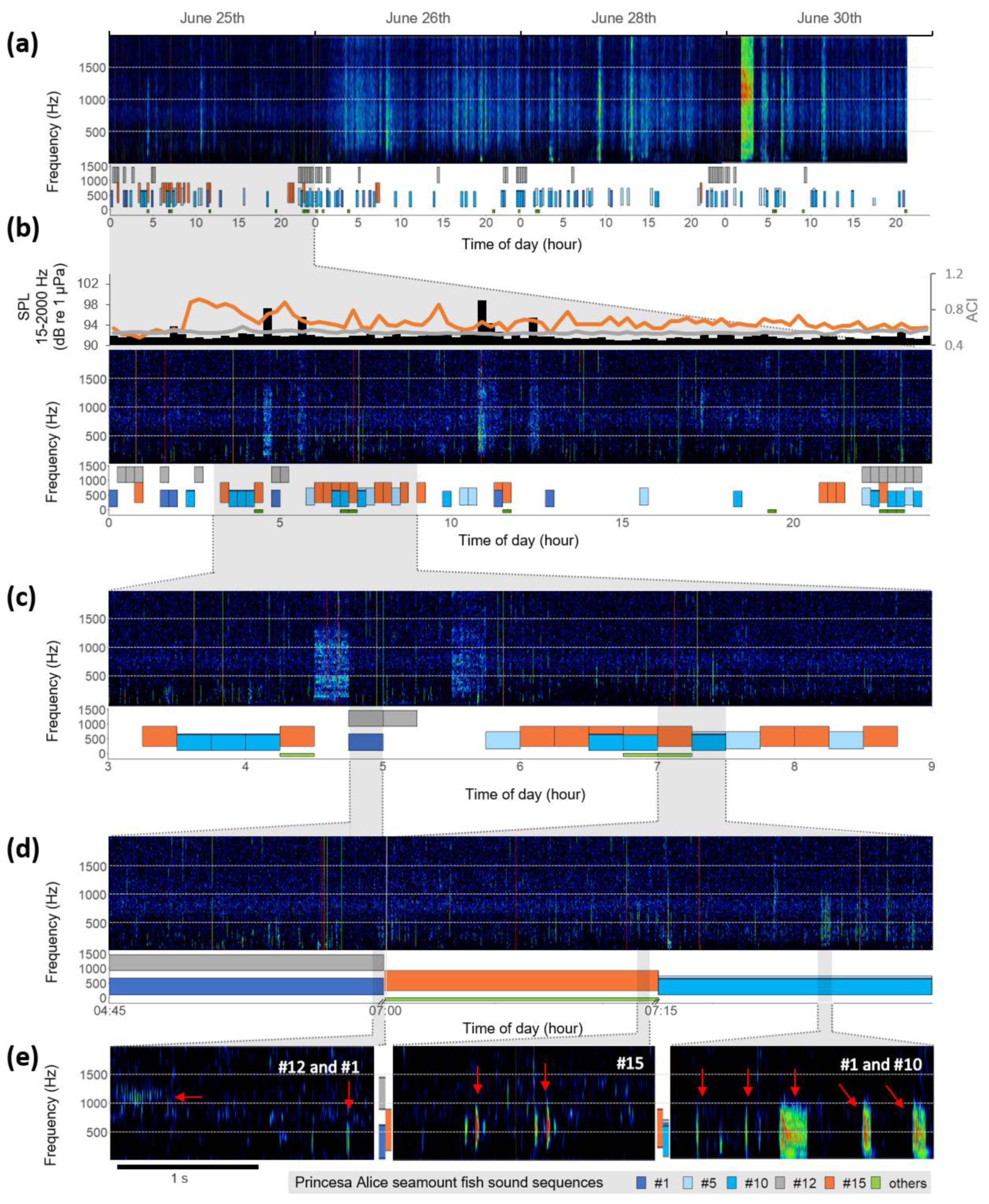
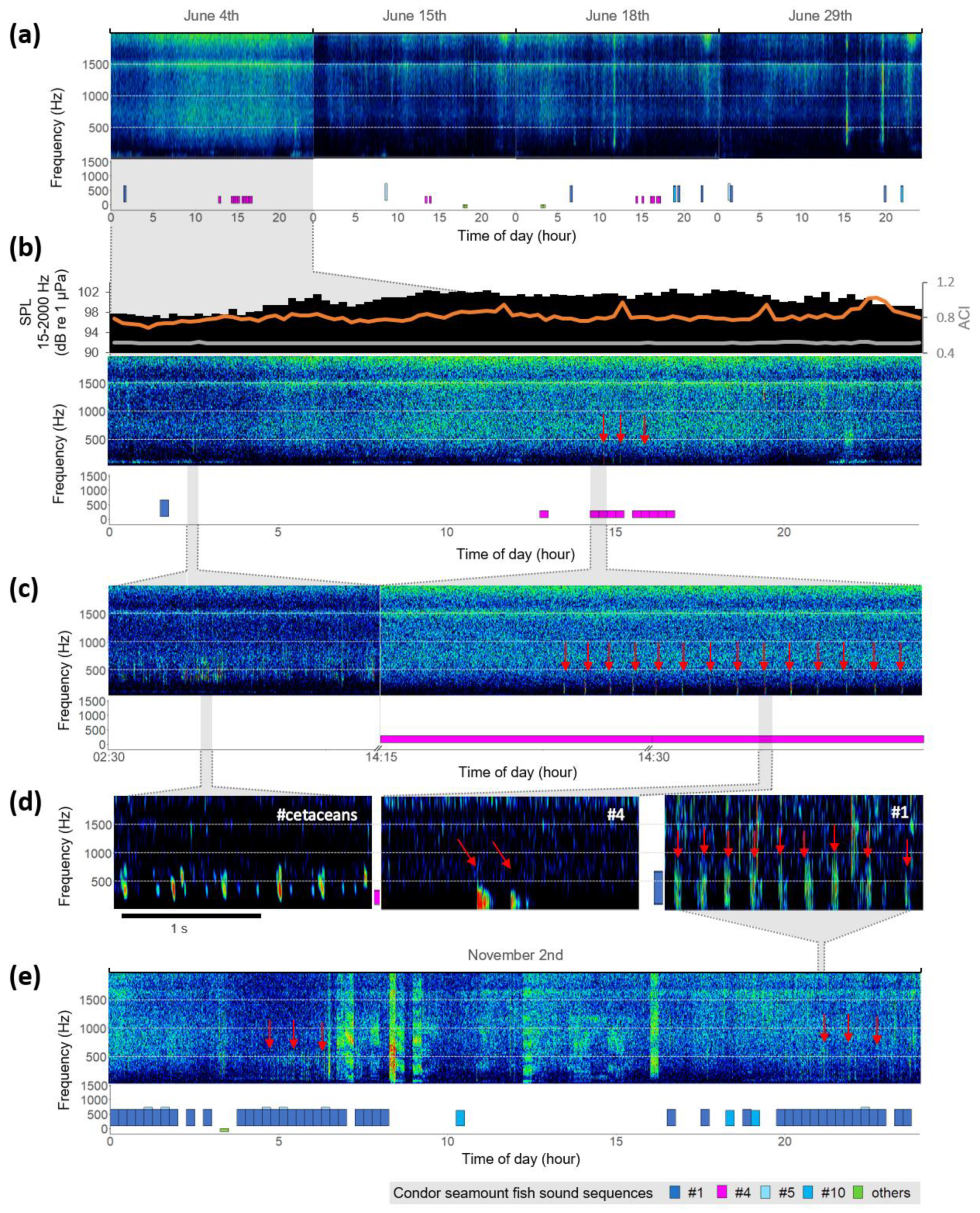
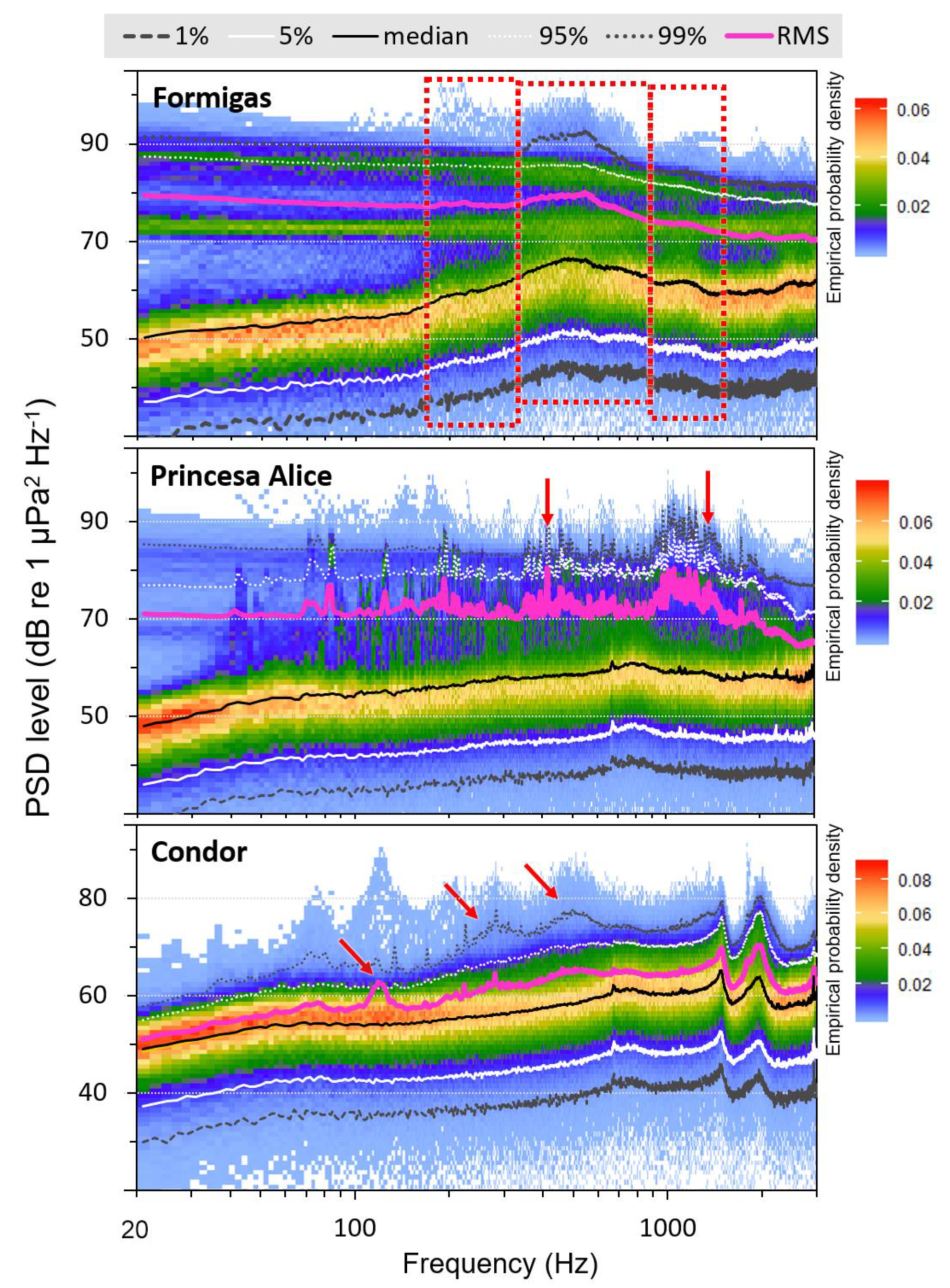
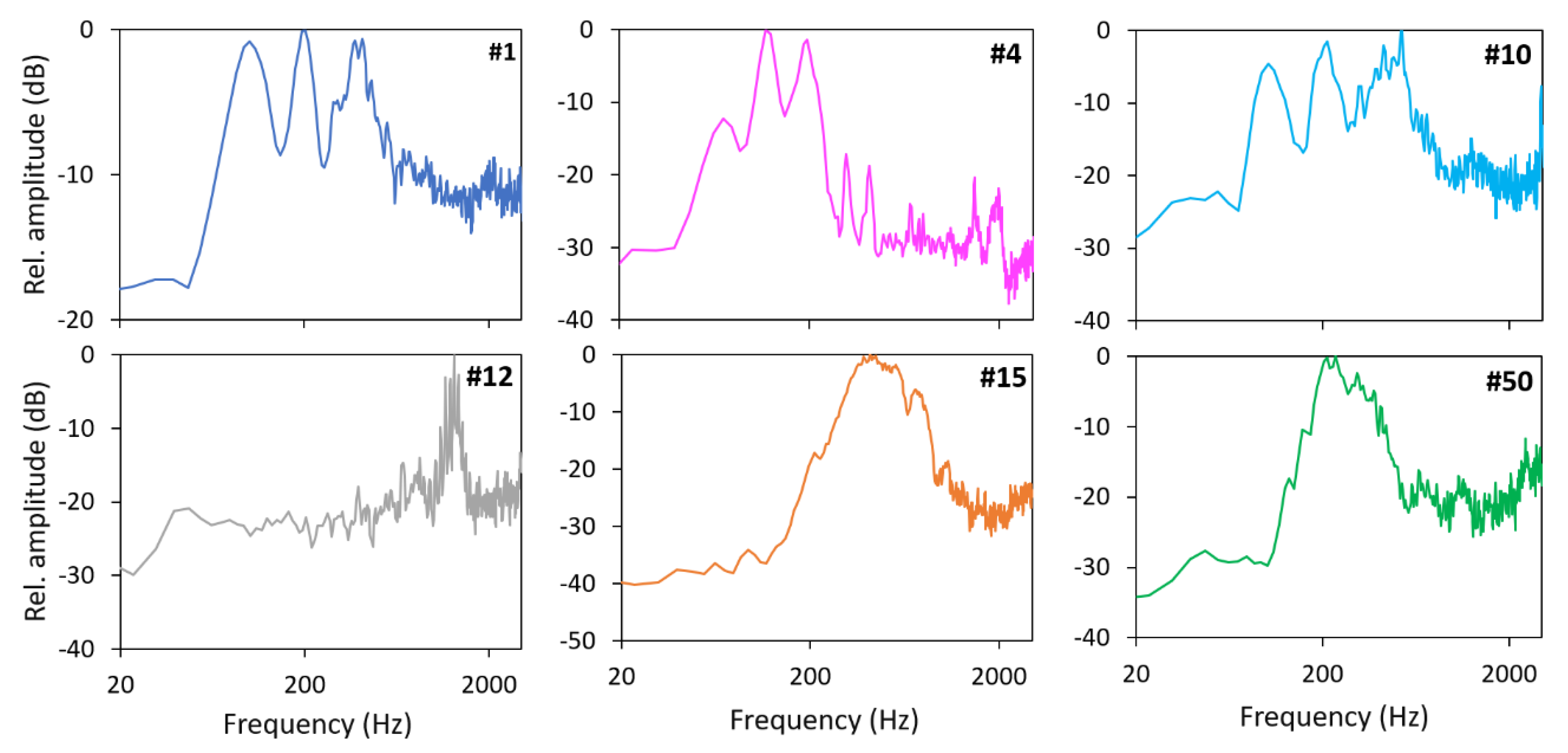
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mann, D.A.; Lobel, P.S. Propagation of damselfish (Pomacentridae) courtship sounds. J. Acoust. Soc. Am. 1997, 101, 3783–3791. [Google Scholar] [CrossRef]
- Buscaino, G.; Filiciotto, F.; Gristina, M.; Bellante, A.; Buffa, G.; Di Stefano, V.; Maccarrone, V.; Tranchida, G.; Buscaino, C.; Mazzola, S. Acoustic behaviour of the European spiny lobster Palinurus elephas. Mar. Ecol. Prog. Ser. 2011, 441, 177–184. [Google Scholar] [CrossRef] [Green Version]
- Popper, A.N.; Hawkins, A.D. An overview of fish bioacoustics and the impacts of anthropogenic sounds on fishes. J. Fish Biol. 2019, 94, 692–713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popper, A.N.; Fay, R.R.; Platt, C.; Sand, O. Sound detection mechanisms and capabilities of teleost fishes. In Sensory Processing in Aquatic Environments; Collin, S.P., Marshall, N.J., Eds.; Springer: New York, NY, USA, 2003; pp. 1–53. [Google Scholar]
- Cato, D.H.; Noad, M.J.; McCauley, R.D. Passive acoustics as a key to the study of marine animals. In Sounds in the Sea: From Ocean Acoustics to Acoustical Oceanography; Medwin, H., Ed.; Cambridge University Press: Cambridge, UK, 2005; pp. 411–429. [Google Scholar]
- McWilliam, J.; Hawkins, A. A comparison of inshore marine soundscapes. J. Exp. Mar. Biol. Ecol. 2013, 446, 166–176. [Google Scholar] [CrossRef]
- Kaatz, I.M. Multiple sound producing mechanisms in teleost fishes and hypotheses regarding their behavioural significance. Bioacoustics 2002, 12, 230–233. [Google Scholar] [CrossRef]
- Vermeij, M.J.A.; Marhaver, K.L.; Huijbers, C.M.; Nagelkerken, I.; Simpson, S.D. Coral Larvae Move toward Reef Sounds. PLoS ONE 2010, 5, e10660. [Google Scholar] [CrossRef] [Green Version]
- Chapuis, L.; Bshary, R. Signalling by the cleaner shrimp Periclimenes longicarpus. Anim. Behav. 2010, 79, 645–647. [Google Scholar] [CrossRef] [Green Version]
- Lillis, A.; Eggleston, D.; Bohnenstieh, D. Estuarine soundscapes: Distinct acoustic characteristics of oyster reefs compared to soft-bottom habitats. Mar. Ecol. Prog. Ser. 2014, 505, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Harris, S.; Shears, N.; Radford, C. Ecoacoustic indices as proxies for biodiversity on temperate reefs. Methods Ecol. Evol. Br. Ecol. Soc. 2016. [Google Scholar] [CrossRef]
- Bolgan, M.; Amorim, M.C.P.; Fonseca, P.J.; Di Iorio, L.; Parmentier, E. Acoustic Complexity of vocal fish communities: A field and controlled validation. Sci. Rep. 2018, 8, 10559. [Google Scholar] [CrossRef] [Green Version]
- Miksis-Olds, J.L.; Martin, B.; Tyack, P.L. Exploring the Ocean through Soundscapes. Acoust. Today 2018, 14, 26–34. [Google Scholar]
- Erbe, C.; McCauley, R.; Gavrilov, A. Chapter 31—Characterizing Marine Soundscapes. In The Effects of Noise on Aquatic Life II, Advances in Experimental Medicine and Biology; Popper, A.N., Hawkins, A., Eds.; Springer Science+Business Media: New York, NY, USA, 2013. [Google Scholar]
- Bertucci, F.; Parmentier, E.; Lecellier, G.; Hawkins, A.D.; Lecchini, D. Acoustic indices provide information on the status of coral reefs: An example from Moorea Island in the South Pacific. Sci. Rep. 2016, 6, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bolgan, M.; Gervaise, C.; Di Iorio Lossent, J.; Lejeune, P.; Raick, X.; Parmentier, E. Fish biophony in a Mediterranean submarine canyon. J. Acoust. Soc. Am. 2020, 147, 2466. [Google Scholar] [CrossRef] [PubMed]
- Luczkovich, J.J.; Mann, D.A.; Rountree, R.A. Passive acoustics as a tool in fisheries science. Trans. Am. Fish. Soc. 2008, 137, 533–541. [Google Scholar] [CrossRef] [Green Version]
- Davies, G.E.; Baumgartner, M.F.; Van Parijs, S.M. Long-term passive acoustic recordings track the changing distribution of North Atlantic right whales (Eubalaena glacialis) from 2004 to 2014. Sci. Rep. 2017, 7, 13460. [Google Scholar] [CrossRef] [PubMed]
- Caiger, P.E.; Dean, M.J.; DeAngelis, A.I.; Hatch, L.T.; Rice, A.N.; Stanley, J.A.; Tholke, C.; Zemeckis, D.R.; Van Parijs, S.M. A decade of monitoring Atlantic cod Gadus morhua spawning aggregations in Massachusetts Bay using passive acoustics. Mar. Ecol. Prog. Ser. 2020, 635, 89–103. [Google Scholar] [CrossRef] [Green Version]
- Rice, A.N.; Soldevilla, M.S.; Quinlan, J.A. Nocturnal patterns in fish chorusing off the coasts of Georgia and eastern Florida. Bull. Mar. Sci. 2017, 93, 2. [Google Scholar] [CrossRef]
- Parmentier, E.; Di Iorio, L.; Picciulin, M.; Malavasi, S.; Lagardère, J.-P.; Bertucci, F. Consistency of spatiotemporal sound features supports the use of passive acoustics for long-term monitoring. Anim. Conserv. 2017. [Google Scholar] [CrossRef] [Green Version]
- Ricci, S.W.; Eggleston, D.B.; Bohnenstiehl, D.R. Use of passive acoustic monitoring to characterize fish spawning behavior and habitat use within a complex mosaic of estuarine habitats. Bull. Mar. Sci. 2017, 93, 439–453. [Google Scholar] [CrossRef]
- Carriço, R.; Silva, M.A.; Menezes, G.M.; Fonseca, P.J.; Amorim, M.C.P. Characterization of the acoustic community of vocal fishes in the Azores. PeerJ 2019, 7, e7772. [Google Scholar] [CrossRef] [Green Version]
- Harris, S.A.; Radford, C.A. Marine Soundscape Ecology. Available online: https://pdfs.semanticscholar.org/578c/dc57de0179040d652f7deefbabe9d9fe7399.pdf (accessed on 10 April 2020).
- Putland, R.L.; Constantine, R.; Radford, C.A. Exploring spatial and temporal trends in the soundscape of an ecologically significant embayment. Sci. Rep. 2017, 7, 5713. [Google Scholar] [CrossRef] [Green Version]
- Bertucci, F.; Maratrat, K.; Berthe, C.; Besson, M.; Guerra, A.; Raick, X.; Lerouvreur, F.; Lecchini, D.; Parmentier, E. Local sonic activity reveals potential partitioning in a coral reef fish community. Oecologia 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-T.; Nowacek, D.P.; Akamatsu, T.; Wang, K.-X.; Liu, J.-C.; Duan, G.-Q.; Cao, H.-J.; Wang, D. Diversity of fish sound types in the Pearl River Estuary, China. PeerJ 2017, 5, e3924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pieretti, N.; Farina, A.; Morri, D. A new methodology to infer the singing activity of an avian community: The Acoustic Complexity Index (ACI). Ecol. Indic. 2011, 11, 868–873. [Google Scholar] [CrossRef]
- Sueur, J.; Farina, A.; Gasc, A.; Pieretti, N.; Pavoine, S. Acoustic Indices for Biodiversity Assessment and Landscape Investigation. Acta Acust. United. Acus. 2014, 100, 772–781. [Google Scholar] [CrossRef] [Green Version]
- Phillips, Y.F. Analysis and Visualization of Very-long-duration Acoustic Recordings of the Natural Environment. Ph. D. Thesis, Queensland University of Technology, Brisbane, Australia, 2018. [Google Scholar]
- Phillips, Y.F.; Towsey, M.; Roe, P. Revealing the ecological content of long-duration audio-recordings of the environment through clustering and visualization. PLoS ONE 2018, 13, e0193345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruppé, L.; Clément, G.; Herrel, A.; Ballesta, L.; Décamps, T.; Kéver, L.; Parmentier, E. Environmental constraints drive the partitioning of the soundscape in fishes. Proc. Natl. Acad. Sci. USA 2015, 112, 6092–6097. [Google Scholar] [CrossRef] [Green Version]
- Haver, S.M.; Klinck, H.; Nieukirk, S.L.; Matsumoto, H.; Dziak, R.P.; Miksis-Olds, J.L. The not-so-silent world: Measuring Arctic, Equatorial, and Antarctic soundscapes in the Atlantic Ocean. Deep Sea Res. Part I Oceanogr. Res. Pap. 2017, 122, 95–104. [Google Scholar] [CrossRef]
- Pitcher, T.; Morato, T.; Hart, P.; Clark, M.; Haggan, N.; Santos, R. Seamounts: Ecology, fisheries and conservation. Fish Aquat. Resour. Ser. 2007, 12, 1–476. [Google Scholar]
- Giacomello, E.; Menezes, G.M.; Bergstad, O.A. An integrated approach for studying seamounts: CONDOR observatory. Deep Sea Res. Part II Top. Stud. Oceanogr. 2013, 98, 1–6. [Google Scholar] [CrossRef]
- Christiansen, S.; Tempera, F. The Formigas Bank—A Potential MPA; WWF World Wildlife Fund: Washington, DC, USA, 2010; Available online: http://charlie-gibbs.org/charlie/NEA_Website/Publication/briefings/FormigasBank.pdf (accessed on 13 June 2020).
- Lammers, M.; Brainard, R.; Whitlow, W.; Mooney, T.; Wong, K. An Ecological Acoustic Recorder (EAR) for long-term monitoring of biological and anthropogenic sounds on coral reefs and other marine habitats. J. Acoust. Soc. Am. 2008, 123, 1720–1728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sousa-Lima, R.S.; Norris, T.F.; Oswald, J.N.; Fernandes, D.P. A Review and Inventory of Fixed Autonomous Recorders for Passive Acoustic Monitoring of Marine Mammals. Aquat. Mamm. 2013, 39, 216–221. [Google Scholar] [CrossRef]
- Obrist, M.K.; Pavan, G.; Sueur, J.; Riede, K.; Llusia, D.; Márquez, R. Bioacoustics approaches in biodiversity inventories. Abc Taxa 2010, 8, 68–99. [Google Scholar]
- McCauley, R.D.; Cato, D.H. Patterns of fish calling in a nearshore environment in the Great Barrier Reef. Philos. Trans. R. Soc. Lond. B 2000, 355, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Amorim, M.C.P.; Vasconcelos, R.O.; Marques, J.F.; Almada, F. Seasonal variation of sound production in the Lusitanian toadfish Halobatrachus didactylus. J. Fish Biol. 2006, 69, 1892–1899. [Google Scholar] [CrossRef] [Green Version]
- Carriço, R.; Silva, M.A.; Menezes, G.M.; Vieira, M.; Bolgan, M.; Fonseca, P.J.; Amorim, M.C.P. Temporal dynamics in diversity patterns of fish sound production in the Condor seamount (Azores, NE Atlantic). Deep-Sea Res. I. (under review).
- Farina, A.; Lattanzi, E.; Piccioli, L.; Pieretti, N. The SoundscapeMeter. Available online: http://www.iinsteco.org/research-and-technology.php (accessed on 10 April 2020).
- Sueur, J.; Pavoine, S.; Hamerlynck, O.; Duvail, S. Rapid acoustic survey for biodiversity appraisal. PLoS ONE 2008, 3, e4065. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 14 April 2020).
- Merchant, N.D.; Fristrup, K.M.; Johnson, M.P.; Tyack, P.L.; Witt, M.J.; Blondel, P. Measuring acoustic habitats. Methods Ecol. Evol. 2015, 6, 257–265. [Google Scholar] [CrossRef] [Green Version]
- Parsons, M.; Erbe, C.; McCauley, R.; McWilliam, J.; Marley, S.; Gavrilov, A.; Parnum, I. Long-term monitoring of soundscapes and deciphering a usable index: Examples of fish choruses from Australia. Acoust. Soc. Am. Proc. Meet. Acoust. 2016, 27, 010023. [Google Scholar] [CrossRef] [Green Version]
- Desiderà, E.; Guidetti, P.; Panzalis, P.; Navone, A.; Valentini-Poirrier, C.A.; Boissery, P.; Gervaise, C.; Di Iorio, L. Acoustic fish communities: Sound diversity of rocky habitats reflects fish species diversity. Mar. Ecol. Prog. Ser. 2019, 608, 183–197. [Google Scholar] [CrossRef] [Green Version]
- Kaatz, I.M.; Rice, A.N.; Loble, P.S. How Fishes Use Sound: Quiet to Loud and Simple to Complex Signaling. Ref. Model Life Sci. 2017, 1–12. [Google Scholar] [CrossRef]
- Parsons, M.J.; McCauley, R.D.; Mackie, M.C.; Siwabessy, P.J.; Duncan, A.J. In situ source levels of mulloway (Argyrosomus japonicus) calls. J. Acoust. Soc. Am. 2012, 132, 3559–3568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radford, C.A.; Ghazali, S.; Jeffs, A.G.; Montgomery, J.C. Vocalisations of the bigeye Pempheris adspersa: Characteristics, source level and active space. J. Exp. Biol. 2015, 218, 940–948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amorim, M.C.P.; Vasconcelos, R.O.; Bolgan, M.; Pedroso, S.S.; Fonseca, P.J. Acoustic communication in marine shallow waters: Testing the acoustic adaptive hypothesis in sand gobies. J. Exp. Biol. 2018, 16, 221. [Google Scholar] [CrossRef] [Green Version]
- Parsons, M.J.; Salgado-Kent, C.P.; Marley, S.A.; Gavrilov, A.N.; McCauley, R.D. Characterizing diversity and variation in fish choruses in Darwin Harbour. ICES J. Mar. Sci. 2016, 73, 2058–2074. [Google Scholar] [CrossRef] [Green Version]
- Bolgan, M.; Soularde, J.; Di Iorio, L.; Gervaise, C.; Lejeune, P.; Gobert, S.; Parmentier, E. Sea chordophones make the mysterious /Kwa/ sound: Identification of the emitter of the dominant fish sound in Mediterranean seagrass meadows. J. Exp. Biol. 2019, 222. [Google Scholar] [CrossRef] [Green Version]
- Sankupellay, M.; Towsey, M.; Truskinger, A.; Roe, P. Visual fingerprints of the acoustic environment. The use of acoustic indices to characterize natural habitats. In 2015 Big Data Visual Analytics (BDVA), Hobart, TAS, Australia, 22–25 September 2015; IEEE: Piscataway, NJ, USA, 2015. [Google Scholar]
- Parsons, M.J.; Salgado Kent, C.P.; Recalde-Salas, A.; McCauley, R.D. Fish choruses off Port Hedland, Western Australia. Bioacoustics 2016, 26, 135–152. [Google Scholar] [CrossRef]
- Stonea, E. Separating the noise from the noise: A finding in support of the ‘niche hypothesis’, that birds are influenced by human-induced noise in natural habitats. Anthroös 2000, 13, 225–231. [Google Scholar] [CrossRef]
- Krause, B. Wild Soundscapes: Discovering the Voice of the Natural World; Wilderness Press: New Haven, CT, USA, 2002; p. 168. [Google Scholar]
- Vasconcelos, R.; Carriço, R.; Ramos, A.; Modesto, T.; Fonseca, P.J.; Amorim, C. Vocal behaviour predicts reproductive success in a teleost fish. Behav. Ecol. 2012, 23, 375–383. [Google Scholar] [CrossRef] [Green Version]
- Kaplan, M.B.; Mooney, T.A.; Partan, J.; Solow, A.R. Coral reef species assemblages are associated with ambient soundscapes. Mar. Ecol. Prog. Ser. 2015, 533, 93–107. [Google Scholar] [CrossRef] [Green Version]
- Bohnenstiehl, D.R.; Lyon, R.P.; Caretti, O.N.; Ricci, S.W.; Eggleston, D.B. Investigating the utility of ecoacoustic metrics in marine soundscapes. J. Ecoacoust. 2018, 2, R1156L. [Google Scholar] [CrossRef]
- Lindseth, A.V.; Lobel, P.S. Underwater Soundscape Monitoring and Fish Bioacoustics: A Review. Fishes 2018, 3, 36. [Google Scholar] [CrossRef] [Green Version]
- Belghith, E.H.; Rioult, F.; Bouzidi, M. Acoustic Diversity Classifier for Automated Marine Big Data Analysis. In Proceedings of the IEEE 30th International Conference on Tools with Artificial Intelligence (ICTAI), Volos, Greece, 5–7 November 2018. [Google Scholar]
- Davies, B.F.R.; Attrill, M.J.; Holmes, L.; Rees, A.; Witt, M.J.; Sheehan, E.V. Acoustic Complexity Index to assess benthic biodiversity of a partially protected area in the southwest of the UK. Ecol. Indic. 2020, 111, 106019. [Google Scholar] [CrossRef]
- Haver, S.M.; Fournet, M.E.H.; Dziak, R.P.; Gabriele, C.; Gedamke, J.; Hatch, L.T.; Haxel, J.; Heppell, S.A.; McKenna, M.F.; Mellinger, M.K.; et al. Comparing the Underwater Soundscapes of Four U.S. National Parks and Marine Sanctuaries. Front. Mar. Sci. 2019, 6, 500. [Google Scholar] [CrossRef] [Green Version]
- Malfante, M.; Mars, J.I.; Dalla Mura, M.; Gervaise, C. Automatic fish sounds classification. J. Acoust. Soc. Am. 2018, 143, 2834–2846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vieira, M.; Fonseca, P.J.; Amorim, M.C.P.; Teixeira, C.J.C. Call recognition and individual identification of fish vocalizations based on automatic speech recognition: An example with the Lusitanian toadfish. J. Acoust. Soc. Am. 2015, 138, 3941–3950. [Google Scholar] [CrossRef]
- Vieira, M.; Pereira, B.P.; Pousão-Ferreira, P.; Fonseca, P.J.; Amorim, M.C.P. Seasonal variation of captive meagre acoustic signalling: A manual and automatic recognition approach. Fishes 2019, 4, 28. [Google Scholar] [CrossRef] [Green Version]
- Monczak, A.; Ji, Y.; Soueidan, J.; Montie, E.W. Automatic detection, classification, and quantification of sciaenid fish calls in an estuarine soundscape in the Southeast United States. PLoS ONE 2019, 14, e0209914. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carriço, R.; Silva, M.A.; Vieira, M.; Afonso, P.; Menezes, G.M.; Fonseca, P.J.; Amorim, M.C.P. The Use of Soundscapes to Monitor Fish Communities: Meaningful Graphical Representations Differ with Acoustic Environment. Acoustics 2020, 2, 382-398. https://doi.org/10.3390/acoustics2020022
Carriço R, Silva MA, Vieira M, Afonso P, Menezes GM, Fonseca PJ, Amorim MCP. The Use of Soundscapes to Monitor Fish Communities: Meaningful Graphical Representations Differ with Acoustic Environment. Acoustics. 2020; 2(2):382-398. https://doi.org/10.3390/acoustics2020022
Chicago/Turabian StyleCarriço, Rita, Mónica A. Silva, Manuel Vieira, Pedro Afonso, Gui M. Menezes, Paulo J. Fonseca, and Maria Clara P. Amorim. 2020. "The Use of Soundscapes to Monitor Fish Communities: Meaningful Graphical Representations Differ with Acoustic Environment" Acoustics 2, no. 2: 382-398. https://doi.org/10.3390/acoustics2020022
APA StyleCarriço, R., Silva, M. A., Vieira, M., Afonso, P., Menezes, G. M., Fonseca, P. J., & Amorim, M. C. P. (2020). The Use of Soundscapes to Monitor Fish Communities: Meaningful Graphical Representations Differ with Acoustic Environment. Acoustics, 2(2), 382-398. https://doi.org/10.3390/acoustics2020022




