1. Introduction
The good balance of biocompatibility, mechanical properties and natural degradation in physiological settings makes Mg alloys strong candidates for biodegradable implant materials [
1,
2,
3,
4,
5]. These characteristics render Mg-based materials especially appealing for temporary load-bearing uses, like orthopedic implants, where gradual load shifts to healing tissues is beneficial [
6,
7,
8]. Nevertheless, the broad use of Mg alloys in biomedical fields is seen to be limited by their galvanic corrosion causing elevated corrosion rates and the resulting decline in mechanical integrity over time for some alloying systems, along with their susceptibility to pitting corrosion [
9,
10,
11,
12,
13].
Among the different Mg alloys, rare-earth [RE]-containing systems like WE43 and Mg10Gd based alloys and Mg-Zn-Ca alloys, such as ZX10, have attracted interest due to their enhanced corrosion resistance, mainly supported by a less porous and uniform corrosion layer, and mechanical properties [
14,
15,
16,
17,
18,
19]. For the Mg10Gd based system modified with Nd and La [
15] and also for Mg-Y-Nd-Gd-Dy in [
16], it could be shown that large corrosion pits lead to a high pitting factor when the corrosion rate is low and the protective corrosion layer is only very locally discontinuous. The pitting factor is one parameter that serves to define the degree of pitting corrosion [
10,
11,
20,
21]. Nonetheless, the corrosion behavior of these alloys in simulated body fluids and their impact on structural integrity during mechanical loading are still not fully comprehended, especially regarding localized corrosion mechanisms. For Mg alloys, which are highly susceptible to pitting corrosion due to their non-uniform corrosion layer, this topic remains current in research studies [
22,
23,
24].
The corrosion behavior, particularly the corrosion morphology, of WE43, Mg10Gd, and ZX10 demonstrates their unique chemical compositions and microstructural features, which affect their functionality in physiological environments [
24,
25,
26,
27]. Mg10Gd and WE43, both RE-based alloys, show higher susceptibility to pitting corrosion than ZX10. Localized corrosion occurrences are especially severe, acting as stress concentrators that strongly diminish residual mechanical strength and encourage early crack formation [
10,
13]. In contrast, ZX10 exhibits a more uniform corrosion pattern with fewer and smaller pits, leading to more stable mechanical performance over time, even though it has a comparable initial strength to the other alloys [
24,
28]. The RE elements in WE43 and Mg10Gd aid in grain refinement and enhance the mechanical properties; however, they also have a tendency to create intermetallic phases that may function as galvanic couples, encouraging localized attack [
15,
29,
30]. ZX10, while not as highly alloyed, seems to gain from a corrosion process that prevents severe pitting, thereby maintaining its load-bearing ability for an extended period [
24,
28]. These findings emphasize the essential importance of corrosion morphology—rather than simply the overall corrosion rate—in influencing the effective lifespan of degradable Mg-based implants.
As previously noted, the relation between corrosion morphology and mechanical performance is essential for guaranteeing the safe and reliable operation of Mg-based implants during their degradation period. Conventional mass loss values (gravimetric analysis) [
3] provide average corrosion rates but do not account for localized damage, which can act as stress concentrators and result in early failure. Aside from the pitting factor, whose calculation can be challenging [
31], the aspect ratio also serves to define the degree of pitting corrosion [
32,
33,
34,
35]. Aspect ratios of values under 1 indicate that the pit is less deep than it is wide (a/2c, a being the depth and 2c being the width), and narrower, deep pits behave more critically [
20,
32]. For a narrow (bullet-shape) pit with an aspect ratio of 1, the Mises stress normalized to nominal stress distributions results in a stress concentration factor of 2.5, and the maximum stress occurs at the bottom of the pit, not near the mouth of the pit [
35].
Advanced characterization methods like micro-computed tomography (µCT) and digital image correlation (DIC) provide the ability to illustrate and correlate corrosion morphology, internal damage progression, and cross-sectional areas to stress distributions in corroded samples, thus facilitating a deeper insight into the failure mechanisms involved [
12,
36,
37,
38,
39]. The studies applying DIC show a robust non-invasive optical method for measuring full-field surface strain and deformation in materials subjected to mechanical load. DIC facilitates the detailed mapping of localized strain concentrations, especially near defects caused by corrosion defects like pits. These strain concentrations suggest heightened stress intensities, which may serve as precursors to crack formation and eventual failure, referred as “pit-to-crack-transition”, while conventional mechanical testing provides average stress–strain data. DIC provides true-strain and true-strain, providing an essential understanding of how corrosion morphology—particularly pit depth, shape, and spatial distribution—affects mechanical performance. The “pit-to-crack transition” is often discussed for corrosion fatigue and stress corrosion cracking [
40,
41,
42]. Pits are discussed as stress increasers due to their notch effect, where the stress concentration depends on the pit geometry, and as crack initiation sides. In Mg alloys, where pitting corrosion prevails as a major degradation mechanism, the use of DIC aids in pinpointing mechanically significant areas and enables the connection between localized surface damage and fracture initiation. This ability is particularly beneficial for evaluating the structural strength of biodegradable Mg implants, where it is crucial to sustain adequate mechanical support throughout the degradation process.
Based on the preliminary results [
43], this research seeks to further analyze the corrosion characteristics and remaining tensile strength of three extruded Mg alloys—WE43, Mg10Gd, and ZX10—after submersion in Hanks’ Balanced Salt Solution (HBSS) at body temperature (37 °C) for different exposure times. Through the integration of gravimetric analysis, µCT imaging, mechanical testing, and DIC, we assess the impact of various corrosion morphologies on crack initiation and fracture behavior. Specifically, we want to evaluate the relation between localized pitting and mechanical failure to understand the significant damage characteristics affecting strength reduction. The first results indicated that, at earlier stages, failure is affected by pitting corrosion, while at extended corrosion periods, where the full surface is degraded, the smallest cross section becomes the weakest point. If another area is severely corroded, a failure is anticipated in that location as well. If corrosion pits begin to overlap rather than growing deeper, the alloy may transition from localized corrosion to a more uniform type, leading to reduced stresses from the corrosion morphology. We aim to identify alloys behaving like this and enhance the overall comprehension of damage progression in biodegradable Mg alloys to guide the creation of safer and more dependable Mg-based biomedical devices.
2. Materials and Methods
In this study, materials with different susceptibilities to pitting corrosion were investigated: Mg10Gd (with 10 wt.% Gd), WE43 (containing 3.5 wt.% Y, 2.3 wt.% Nd, and 0.5 wt.% Zr), and ZX10 (with 0.94 wt.% Zn and 0.15 wt.% Ca). Cast billets with a diameter of 49 mm and a length of 150 mm were homogenized at 400 °C for 16 h followed by air cooling. Prior to hot extrusion, the billets were heated for 60 min and subsequently extruded into round bars with a diameter of 8 mm using an automatic extrusion press of Müller Engineering (Sand/Todtenweis, Germany). Mg10Gd and WE43 were extruded at a temperature of 350 °C with a ram speed of 0.6 mm/s, whereas ZX10 was extruded at 300 °C at a ram speed of 1.3 mm/s. The samples were metallographically prepared by grinding with SiC paper to a grit size of 4000, followed by polishing with 3 and 1 µm water-free diamond paste and 0.25 µm colloidal silica suspension. The samples were then cleaned with ethanol and dried with hot air. The microstructure was developed by etching in a solution of 4.2 g picric acid, 10 mL acetic acid, 10 mL distilled water, and 70 mL ethanol for a few seconds only. A LEICA DMi8 optical microscope (Leica Microsystems GmbH, Wetzlar, Germany) was used to evaluate the microstructure. IMAGIC IMS software 16.4.1.6 was used to take grain size measurements, applying the line intercept technique.
Corrosion, with a focus on the corrosion morphology of the gauge length, of at least 10 tensile samples each alloy was assessed by immersion tests in HBSS at 37 °C (3 samples 500 mL) in intervals for up to two weeks (mostly one sample per time point, sometimes two). These immersion tests at various time points were conducted to induce different degrees of corrosion morphology and, consequently, variations in residual strength. Additionally, cylindrical pins of each alloy with a height of 15 mm and a diameter of 6.5 mm were used to determine the corrosion rate for 168 h (1 pin in 330 mL). For each alloy, three pins were tested. A µCT system Bruker Skyscan 2214 (Bruker, Billerica, MA, USA) was employed to investigate the corrosion morphology after corrosion and before tensile testing, as well as after tensile testing. The scanning parameters for the computed tomography scans were voltage 80 kV, current 150 μA, voxel size up to 6 µm and rotation step 0.4°. NRECON software 2.0.0.5 was used to transform the longitudinal projection images into transversal sectional images. DATAVIEWER analysis software 1.5.6.5 was used for slice-by-slice inspection of the transversal images, and CTVOX analysis software 3.3.0.0 was applied for realistic visualization of volumes with different density ranges by 3D rendering.
A method had to be developed to describe or classify the extent of corrosion at the fracture region, especially that causing a crack. The following criteria were used to classify crack initiation mechanisms, considering the smallest remaining cross section, the deepest corrosion pit, and the identified critical region; see
Figure 1 for mechanism with corresponding images and description. The “smallest cross section” criterion reveals predominantly uniform corrosion, with no individual pit detected as critical. Quantitative evaluation of the smallest cross section is based on the object area along the gauge length derived from the µCT analysis. “The deepest pit” as a feature is clearly based on the pit with the highest depth and evaluated by the heat map and the 2D-µCT slides. When the smallest cross section includes the deepest pit, this is assigned to the criterion “deepest pit”. A “critical region” arises when the crack initiation or fracture region is neither at the deepest pit nor at the smallest cross section. This cross section might or might not show critical pits, which can made responsible for crack initiation. When the smallest cross section has one or more critical pits clearly initiating the crack, but not being the deepest pit, this section is also assigned to the “critical region”. And when samples fail without visible pits or any corrosion attack, it is assumed, that lattice defects, like second phases or casting defects as well as other manufacturing defects are responsible for the fracture in this “critical region”.
Tensile tests were performed using two machines: an INSTRON E10000 mechanical testing machine (INSTRON, Norwood, MA, USA) for initial strength and ductility measurements and a TIRA Test 2705 (TIRA GmbH, Schalkau, Deutschland) for residual strength and ductility evaluation. The latter was also used in combination with digital image correlation (DIC) to identify regions of stress concentration caused by localized corrosion. An ARAMIS Adjustable SRX system by Zeiss (Carl Zeiss IQS Deutschland GmbH, Oberkochen, Germany) with the software “GOM Correlate Pro 2021” was implemented. The tensile specimens had a gauge length of 9 mm and a diameter of 4 mm. For residual strength and ductility testing, the tensile samples were coated with wax, leaving only the gauge length exposed to the electrolyte. For DIC measurements, only the side of the gauge length facing the cameras was left uncoated. This ensured that the critical corrosion region developed within the observed area and not on the opposite side of the gauge length, which would not have been captured during measurement.
3. Results and Discussion
Among the investigated alloys, Mg10Gd exhibits the highest ultimate tensile strength of 238.1 ± 0.5 MPa. WE43 and ZX10 show slightly lower but comparable strengths of 218.4 ± 5.0 MPa and 221.2 ± 6.9 MPa, respectively. WE43 has the smallest grain size at 6.6 ± 0.9 μm, followed by Mg10Gd with 9.6 ± 1.8 μm and ZX10 with 13.0 ± 0.4 μm. All alloys exhibit a fully recrystallized microstructure, see
Figure 2. Due to its lower alloying element content, ZX10 shows no visible precipitates under light microscopy (
Figure 2c,f), whereas precipitates aligned along the extrusion direction are observed in the Mg–RE alloys (
Figure 2a,b,d,e). While the corrosion rates of Mg10Gd increase over time, ZX10 exhibits a decreasing corrosion rate. After two weeks, Mg10Gd shows a corrosion rate of 1.9 mm/a, whereas ZX10 reaches 0.45 mm/a. The corrosion rate of WE43 increases up to six days, reaching 1.8 mm/a—followed by reducing to 0.78 mm/a at two weeks.
Figure 3 presents a WE43 tensile sample after 1 day of corrosion exposure. Different viewpoints, obtained by rotating the corroded gauge length of the tensile specimen, reveal the presence of local (pitting) corrosion, and the fractured sample shows the location of failure (
Figure 3a). The surface map of the corrosion morphology (
Figure 3b) enables identification of the deepest pit, located at a z-position of 12.8 mm; see the corresponding cross section in
Figure 3c. The curve of the cross-sectional object area in
Figure 3c indicates the smallest cross section at a z-position of 12.3 mm. In this sample, the smallest cross section does not coincide with the deepest pit. Additional local minima in the cross-sectional area are observed at z-positions of 11.7 mm, 15.2 mm, and 16.7 mm. The 2D-µCT images in
Figure 3d present the cross section of the smallest object areas (smallest cross section) and the one with the deepest pit. The fractured corroded sample in
Figure 3a allows, in correlation with the corroded sample before tensile test, the evaluation of the fracture line, marked in light red in the surface map in
Figure 3b. With a small amount of necking, a ductile fracture without a high shear deformation can be seen. The curve of the object area was extended by a red marking (
Figure 3c), indicating the fracture area—this clarifies whether the deepest corrosion pit and/or the smallest cross section is located in this fracture area or not.
Figure 3e shows the geometry of the deepest pit in the transverse and longitudinal micrographs, and their simplified black-and-white segmentation is seen in
Figure 3f. According to the ASTM guide for the examination and evaluation of pitting corrosion [
20], the deepest pit is undercutting/subsurface-shaped and has a depth of 700 µm. The smallest cross section, also shown in
Figure 3d, also contains undercutting subsurface corrosion pits, however of smaller size and more spread around the surface. The two simplified black-and-white segmentations of the cross section in
Figure 3f enable the measurement of the distances of the pit depth in the cross section a
CS, its corresponding width 2c
CS, as well as the pit depth in the longitudinal section a
LS and its corresponding width 2c
LS, used to calculate the aspect ratio of the corrosion pit. Based on the cross section of the deepest pit, an aspect ratio of 1.2 is determined. Although this value is only slightly higher than 1, no other pits show a similar aspect ratio. In addition, the geometry of the pit shows a higher material corrosion underneath the surface than expected from the outside, and the corrosion of the surrounding material volume is the reason, as it neighbors the smallest cross section, that this region is the most likely site of crack initiation.
Figure 4 presents the 3D-µCT analysis of Mg10Gd after 5 days of corrosion.
Figure 4a shows the corroded sample from different viewing angles, highlighting the presence of localized pronounced corrosion and the fractured sample indicating the location of failure. The corresponding surface map of the corrosion morphology is given in
Figure 4b. The deepest is highlighted in yellow and the fracture line in light red.
Figure 4c displays the object area curve, illustrating the variation in cross-sectional area as a function of the z-position along the gauge length of the corroded tensile sample. The deepest pit has a z-position of 15.3 mm and the smallest cross section of 10.0 mm. The red marked area of the object curve indicating the fractured area neither corresponds with the smallest cross section nor with the region including the deepest pit. A complementary 2D-µCT analysis is shown in
Figure 4d, where the cross-sectional slices with the deepest pit and the smallest cross section are presented. The deepest pit is clearly seen. Although it is surrounded by pronounced corrosion, almost half of the cross section is not corroded—acting as support in the load bearing situation. The fractured corroded specimen shown in
Figure 4e shows a moderate ductile fracture with shear lips. The 2D-µCT cross-sectional slices in
Figure 4f–h show the corrosion morphology in the transverse and longitudinal direction, providing information of the geometry of pits. The main shape is elliptical, and at the deepest point of the pit, subsurface corrosion is seen during the corrosion progress. The accumulation of a few critical pits, more pronounced at the bottom half of the cross sections in
Figure 4f–h, caused the crack initiation. None of these pits are deeper than the deepest pit. The cross-sectional micrographs also reveal the appearance of second phases in white by their higher density; they are cathodic and dissolve after the Mg matrix [
15]. The volume fraction in Mg10Gd is rather small, and the influence here less pronounced.
The aspect ratios were not calculated using the simplified black-and-white segmentations of the sections. However, it can be seen that the values would be below 1, as the corrosion sites with their elliptical shape are wider than they are deep. The deepest pit is also below 1 but above the aspect ratio of the critical pits due to its highest depth—however, the material around the deepest pit has the described supporting function preventing this region from initiating a crack.
Figure 5 shows the 3D µCT analysis of the Mg10Gd specimen after eight days of corrosion.
Figure 5a shows the corroded sample from different viewing angles, revealing severe corrosion throughout, and the resulting fractured tensile samples.
Figure 5b illustrates the surface map of the corrosion morphology, highlighting the region of the deepest pit in yellow at a z-position of 13.7 mm. The fractured region is marked in light red and corresponds with the red marking of the object area curve in
Figure 5c.
Figure 5c shows a strong variation in the cross-sectional area as a function of the z-position along the gauge length of the heavily corroded tensile sample. The fractured region corresponds to the smallest cross section at a z-position of 16.1 mm. Complementary 2D µCT cross-sectional micrographs of the smallest cross section and of the cross section including the deepest pit are shown in
Figure 5d. It should be noted that the deepest pit is actually the deepest corrosion site formed by overlapping corrosion pits, as is the case with the smallest cross section. This makes it clear that there is no critical increase in stress concentration by the deepest pit, meaning that strongly corroded samples fail at the smallest cross section rather than at the deepest pit. The influence of individual corrosion pits in initiating the crack cannot be ruled out but cannot be clearly identified. According to
Figure 3c,
Figure 4c and
Figure 5c, the object area is getting smaller in the region of fracture with the increase in corrosion time, resulting in shallower corrosion pits. For neither WE43 nor Mg10Gd is it evident that the initial corrosion pits grow deeper through localized corrosion rather than becoming overlapped by other corrosion pits to form an overall corroded surface.
The methodology used to evaluate crack initiation sites, as illustrated in
Figure 3,
Figure 4 and
Figure 5, was applied to further selected samples as part of an extended analysis. Since most of ZX10 samples do not show any pitting or a very few pits, 3D-µCT analysis is not shown in this paper.
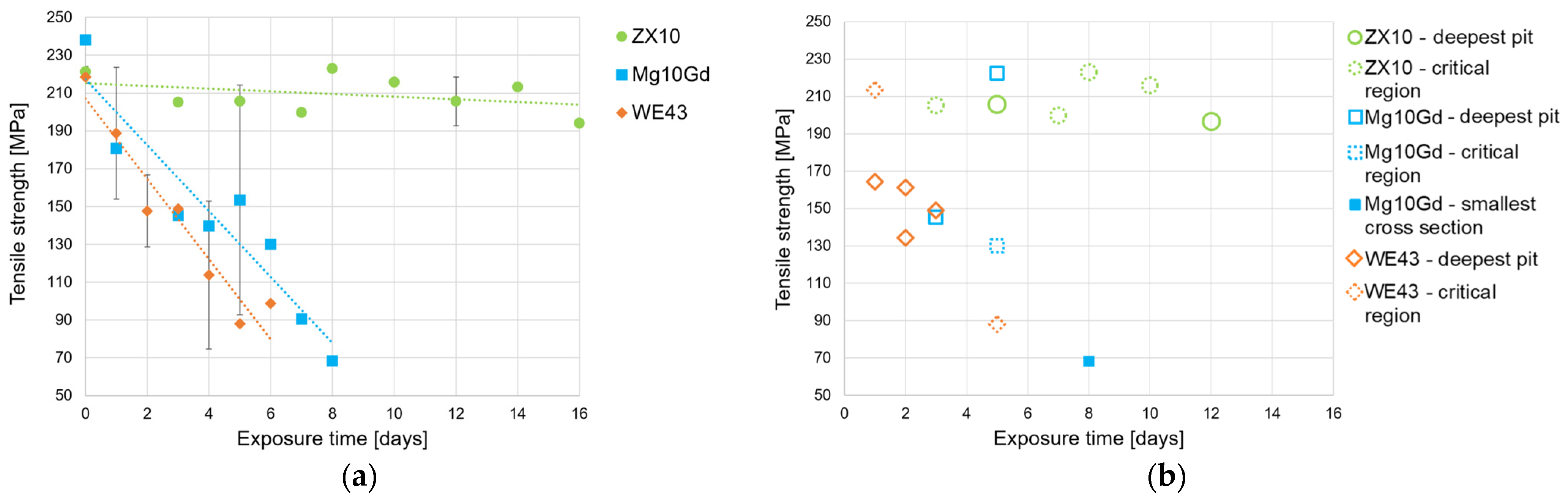
Figure 6a shows the loss of tensile strength as a function of the corrosion exposure time for the ZX10, WE43, and Mg10Gd alloys. Among these materials, ZX10 exhibits the highest retention of tensile strength up to 16 days of exposure. This behavior is consistent with the literature reports [
17,
18,
19], which attribute the superior performance of ZX10 to its low corrosion rate and limited susceptibility to pitting corrosion. The low-alloyed Mg–Zn–Ca system is known to exhibit reduced corrosion rates due to the smaller number of galvanic corrosion sites present in the microstructure [
18,
19]. In contrast, the tensile strength of Mg10Gd and WE43 decreases markedly with increasing exposure time, see
Figure 6a; it is slightly stronger for WE43, which can be explained by the higher corrosion rate up to this time point. This agrees with the findings on WE43 and ZXM100 wires in [
44].
Figure 6b highlights, for selected samples, the mechanism responsible for failure according to
Figure 1,
Figure 3,
Figure 4 and
Figure 5. Crack initiation is attributed either to the deepest pit (so far, eight samples) or to the location of the smallest cross section (so far, one sample), and the samples are labeled accordingly. In other cases, the region is designated as a critical region (so far, seven samples). A concise quantitative breakdown can be prepared once all samples have been analyzed. These critical regions warrant particular attention, as they involve pits with high pitting factors or high aspect ratios (i.e., narrow and deep pits). Additionally, the accumulation of pits or the presence of second-phase particles causing micro-galvanic corrosion [
15,
16,
24] may also contribute to crack initiation. The ZX10 sample failures are mainly classified as “critical region”. In these cases, failure is caused by mechanical overloading during tensile testing rather than corrosion, as no localized corrosion features were observed. Microstructural defects might have caused crack initiation—it is not impossible that second phases caused dissolution of surrounding matrix or weakened this region by its own dissolution [
18,
19]. In contrast, failure in WE43 is clearly influenced by pitting corrosion. With the deepest pit of a depth of approximately 700 µm in the 1-day sample, shown in
Figure 3, the material has retained only 75% of its original strength. In this case, the WE43 sample does not show significant corrosion after 1 day; it remains at 98%. With ongoing corrosion (2 days to 5 days), WE43 maintains just 40.3% of its initial strength, with failure again linked to corrosion pits—specifically those that reach a critical combination of size, shape, and distribution. Similarly, the fracture in Mg10Gd is strongly governed by pitting corrosion. After 3 days, cracking begins at the deepest pit, while by day 5, failure occurs at critical pits near the smallest cross section. By that corrosion progress, see
Figure 4, 55% of its initial strength is reached. Local deep corrosion after 5 days leads to 93% residual strength. These results show the variation in corrosion attack, and the strength loss is rather high at shorter time points—making a prediction rather challenging. By day 8, Mg10Gd is extensively corroded and fails at the smallest cross section, with a residual strength of 29%. Due to pit overlapping, single pits no longer reach an individual critical size or shape.
In addition to mapping the corrosion morphology to indicate the region of crack initiation using µCT and careful examination of the fracture features, stress mapping using DIC also supports the identification of the crack initiation mechanism qualitatively.
Figure 7 shows the surface morphology (see µCT data in
Figure 7a,c) and stress field (measured area of 9 × 4 mm
2 in
Figure 7b,d) of two WE43 tensile samples, demonstrating stress increases due to the localized corrosion of a weakly corroded tensile sample after five days (
Figure 7a,b), and of a tensile sample exhibiting significant pitting corrosion after eight days (
Figure 7c,d). Evaluating the stress map and comparing the highest and lowest stress values provides information about the geometrical influence of topography, especially corrosion pits, and the material’s ability to transform mechanical load into plastic deformation before crack initiation. In the case of the weakly corroded sample, see
Figure 7a,b, a peak stress of 313 MPa was measured in relation to 220 MPa in the uncorroded region, giving a stress increase factor of 1.42. By contrast, a pronounced pit with an approximate depth of 930 µm leads to a local stress of 305 MPa, whereas areas unaffected by corrosion exhibit stresses of only 180 MPa in the DIC heat map (
Figure 7c,d). This results in a stress concentration factor of 1.69. The analyzed pit has an elliptical shape in the longitudinal section and is undercutting shaped in the cross section. The aspect ratio in the direction of mechanical loading is 0.74. The stress concentration factor of moderate hemispherical pits (semi-elliptical) in pressure vessels is found to be approximately 2.4 at an aspect ratio of 0.75 [
33,
35]. The lower value in our study can be explained by the geometry variation of the pits and by the fact that the DIC measures at the surface of the corroded sample, more specifically at the mouth and not the pit depth [
35]. Depending on the pit aspect ratio and shape, the maximum stress occurs either at the bottom of the pit or slightly below the mouth. The crack begins at the point where the maximum stress occurs [
45].
The quality of the DIC measurement clearly depends on the black–white image quality of the surface treatment. However, it appears that the tolerable stress increase depends on the stress concentration around the stress peak location. It is clear that the significant size of the corrosion pit leads to a higher stress increase. Furthermore, the area up to the drop in stress on uncorroded material is rather small (around 180 MPa, see
Figure 7d). The adjacent material around the stress peaks of stresses above 300 MPa of the weakly corroded sample shows significantly higher stresses in the millimeter range, and almost all the gauge length shows a stress increase (
Figure 7b). This is due to the increase in dislocation density in a much larger area taking part in the plastic deformation, which significantly reduces the supporting function of the adjacent material, and a stress concentration factor of 1.42 (only) is already causing crack initiation.
The extent of the stress increase around the pronounced corrosion pitting (
Figure 7d) in the z-direction over 2 mm corresponds to the object area curve in
Figure 8a, which shows that the load-bearing cross section is also strongly reduced over approximately 2 mm, without a gradual transition. The marked part of the curve in red labels the fractures area—for the sample with the large corrosion pit in direct agreement. However, the tensile samples evaluated as examples in
Figure 3,
Figure 4,
Figure 5 and
Figure 6 show that the deepest pitting does not necessarily correspond to the smallest cross sections. For the example presented in
Figure 7c this is clearly not the case, because there is only one corrosion pit. More than one isolated deep corrosion pit will take the supporting function of the uncorroded material away.
Figure 8b,c present furthermore selected true stress–true strain curves of local data points on the deformed WE43 tensile samples in comparison to regions with average stress (green and blue), in
Figure 8b for the sample subjected to 5 days of corrosion (weak) and in
Figure 8c for the pronounced corrosion pitting after 8 days. While for the weakly corroded sample, exhibiting a relatively uniform corrosion behavior along the gauge length (
Figure 8a), the peak stress is reached at 40% elongation, the severely corroded sample shows the peak stress at 70% elongation, see
Figure 8c. This shows once more that the gauge length in the weakly corroded samples is strained overall, whereas the corrosion pit only causes very localized strain, leading to an increase in stress. The true-stress–true-strain curves of the selected data points for the weakly corroded samples are much closer together than those for the sample with pitting corrosion (see the green and blue lines in
Figure 8b,c, respectively).
Figure 8d summarizes the corresponding increase in local stress at the point of highest stress (see red lines in
Figure 7b,c) and highlights the role of corrosion-induced geometric topography changes in accelerating local failure mechanisms. As seen in
Figure 7d and
Figure 8c, the stress increase around the corrosion pit is significantly higher—at less strain. Finally,
Figure 8d compares the engineering stress–strain curves based on the full sample extension, which also illustrate the reduction in mechanical performance as the corrosion severity increases—in agreement with the average stress from the DIC stress field (different strains are based on different reference lengths).
This study demonstrates that, unlike ZX10, both REE-containing alloys WE43 and Mg10Gd exhibit pronounced pitting corrosion. Early in the corrosion process, localized pits serve as initiation sites for cracking; however, failure does not consistently originate from the deepest pit. As the corrosion progresses, individual pits merge, and fracture behavior becomes governed primarily by the regions with accumulated critical pits or with the smallest remaining cross section rather than by isolated pit depth. This indicates that, in these REE-containing alloys, pits tend to expand laterally rather than deepen significantly, resulting in overlapping corrosion sites. Such behavior is advantageous for load-bearing applications at moderate load exposure, as it mitigates the risk of sudden failure from isolated critical pits and produces a more uniform reduction in cross-sectional area. In contrast to certain Mg alloys, where a single deep pit can dominate and make residual strength unpredictable, WE43 and Mg10Gd develop a more homogeneous corrosion morphology over time. This overlapping pattern may improve the predictability of residual strength during degradation, a key factor for the safe use of bioresorbable implants. Stress mapping and heat maps of corrosion morphology agree nicely—supporting the location of critical regions and providing information on maximum stress concentration increase, whereby corrosion progresses when this level is not reached, but no mechanical failure occurs. The dissolution process must be adapted to the application.
Future work will first of all increase the sample amount to meet statistical requirements and, furthermore, focus on evaluating the criticality of individual pits in terms of their geometry, location, and depth. The focus here will be on the notch effect and the resulting increase in stress. Experimental data from DIC can be compared with theoretical approaches. In WE43 and Mg10Gd, observed pits were predominantly subsurface and undercut, with rounded geometries and no sharp features, likely contributing to lower stress concentration factors and more gradual crack initiation. While no extremely narrow or deeply penetrating pits have been identified so far, the detailed investigation of pit morphology and its effect on fracture behavior is needed. Gaining insight into the conditions under which pits transition from benign to critical will be essential for enhancing the predictability and safety of magnesium-based implant materials.