The Protection of Building Materials of Historical Monuments with Nanoparticle Suspensions
Abstract
:1. Introduction
2. Experimental
3. Results and Discussion
3.1. CaCO3 Particles/Suspensions
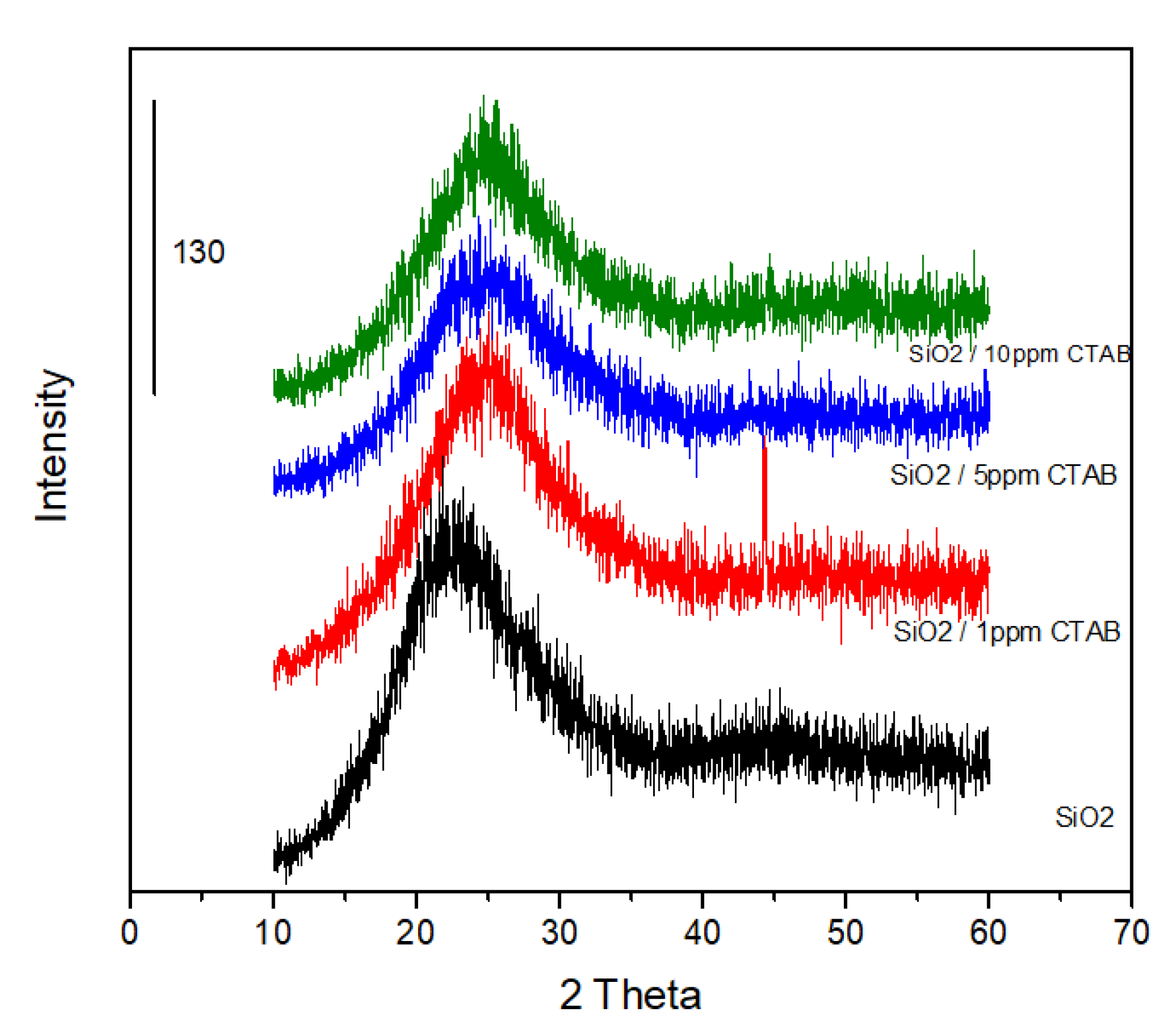
3.2. Suspensions of Amorphous SiO2 Particles
3.3. Deposition of Nanoparticles in Pentelic Marble and Dissolution of Specimens (without and with Treatment with Suspensions)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spathis, P.; Triantafyllidis, K.; Prochaska, C.; Karapanagiotis, I.; Pavlidou, E.; Stefanidou, M. Characterization and properties of silicate and nanocomposite coatings for the protection of dolomite marble against weathering. In International Symposium on the Conservation of Monuments in the Mediterranean Basin; Springer: Cham, Switzerland, 2018; pp. 287–294. [Google Scholar] [CrossRef]
- Baglioni, P.; Chelazzi, D.; Giorgi, R.; Poggi, G. Colloid and materials science for the conservation of cultural heritage: Cleaning, consolidation, and deacidification. Langmuir 2013, 29, 5110–5122. [Google Scholar] [CrossRef]
- Sadat-Shojai, M.; Ershad-Langroudi, A. Polymeric coatings for protection of historic monuments: Opportunities and chal-lenges. J. Appl. Polym. Sci. 2009, 112, 2535–2551. [Google Scholar] [CrossRef]
- Baglioni, P.; Carretti, E.; Chelazzi, D. Nanomaterials in art conservation. Nat. Nanotechnol. 2015, 10, 287–290. [Google Scholar] [CrossRef]
- De Ferri, L.; Lottici, P.P.; Lorenzi, A.; Montenero, A.; Salvioli-Mariani, E. Study of silica nanoparticles—Polysiloxane hydrophobic treatments for stone-based monument protection. J. C. Herit. 2011, 12, 356–363. [Google Scholar] [CrossRef]
- Ksinopoulou, E.; Bakolas, A.; Moropoulou, A. Modification of Si-based consolidants by the addition of colloidal nanopar-ticles: Application in porous stones. J. Nano Res. 2014, 27, 143–152. [Google Scholar] [CrossRef]
- Kapridaki, C.; Maravelaki-Kalaitzaki, P. TiO2–SiO2–PDMS nano-composite hydrophobic coating with self-cleaning properties for marble protection. Prog. Org. Coat. 2013, 76, 400–410. [Google Scholar] [CrossRef]
- Kapridaki, C.; Maravelaki-Kalaitzaki, P. TiO2-SiO2-PDMS nanocomposites with self-cleaning properties for stone protection and consolidation. Geol. Soc. Lond. Spec. Publ. 2015, 416, 285–292. [Google Scholar] [CrossRef]
- Manoudis, P.; Papadopoulou, S.; Karapanagiotis, I.; Tsakalof, A.; Zuburtikudis, I.; Panayiotou, C. Polymer-silica nanoparticles composite films as protective coatings for stone-based monuments. J. Phys. Conf. Ser. 2007, 61, 1361–1365. [Google Scholar] [CrossRef]
- Aldoasri, M.A.; Darwish, S.; Adam, M.; Elmarzugi, N.; Ahmed, S. Performance of Clay, SiO2, Ca(OH)2 and CaCO3-polymeric nanocomposites for conservation and preservation of limestone artworks. Preprints 2018. [Google Scholar] [CrossRef] [Green Version]
- Aldoasri, M.A.; Darwish, S.S.; Adam, M.A.; Elmarzugi, N.A.; Ahmed, S.M. Enhancing the durability of calcareous stone monuments of Ancient Egypt using CaCO3 nanoparticles. Sustainability 2017, 9, 1392. [Google Scholar] [CrossRef] [Green Version]
- Burgos-Cara, A.; Rodríguez-Navarro, C.; Ortega-Huertas, M.; Ruiz-Agudo, E. Bioinspired alkoxysilane conservation treatments for building materials based on amorphous calcium carbonate and oxalate nanoparticles. ACS Appl. Nano Mater. 2019, 2, 4954–4967. [Google Scholar] [CrossRef]
- Manoudis, P.N.; Karapanagiotis, I.; Tsakalof, A.; Zuburtikudis, I.; Kolinkeová, B.; Panayiotou, C. Superhydrophobic films for the protection of outdoor cultural heritage assets. Appl. Phys. A 2009, 97, 351–360. [Google Scholar] [CrossRef]
- Giorgi, R.; Ambrosi, M.; Toccafondi, N.; Baglioni, P. Nanoparticles for cultural heritage conservation: Calcium and bariumhydroxide nanoparticles for wall painting consolidation. Chem. Eur. J. 2010, 16, 9374–9382. [Google Scholar] [CrossRef] [PubMed]
- Baglioni, M.; Poggi, G.; Chelazzi, D.; Baglioni, P. Advanced materials in cultural heritage conservation. Molecules 2021, 26, 3967. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Navarro, C.; Ruiz-Agudo, E. Nanolimes: From synthesis to application. Pure Appl. Chem. 2018, 90, 523–550. [Google Scholar] [CrossRef]
- Dei, L.; Salvadori, B. Nanotechnology in cultural heritage conservation: Nanometric slaked lime saves architectonic and artistic surfaces from decay. J. C. Herit. 2006, 7, 110–115. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Kudłacz, K.; Cizer, O.; Ruiz-Agudo, E. Formation of amorphous calcium carbonate and its transformation into mesostructured calcite. CrystEngComm. 2015, 17, 58–72. [Google Scholar] [CrossRef]
- Sierra-Fernandez, A.; Gomez-Villalba, L.S.; Rabanal, M.E.; Fort, R. New nanomaterials for applications in conservation and res-toration of stony materials: A review. Mater. Constr. 2017, 67, 3–17. [Google Scholar] [CrossRef]
- Kanellopoulou, D.G. Physico-Chemical Investigation of the Deterioration of Building Materials of Historic Monuments and Protection Methods. Ph.D. Thesis, University of Patras, Patra, Greece, 2012; p. 151. [Google Scholar]
- Koga, N.; Nakagoe, Y.; Tanaka, H. Crystallization of amorphous calcium carbonate. Thermochim. Acta 1998, 318, 239–244. [Google Scholar] [CrossRef]
- Faatz, M.; Gröhn, F.; Wegner, G. Amorphous calcium carbonate: Synthesis and potential intermediate in biomineralization. Adv. Mater. 2004, 16, 996–1000. [Google Scholar] [CrossRef]
- Singh, T.K.; Jain, C.L.; Sharma, S.K.; Singh, S.S. Preparation of dispersed silica by hydrolysis of tetraethyl orthosilicate. Indian J. Eng. Mater. Sci. 1999, 6, 349–351. [Google Scholar]
- Vagenas, N.V.; Gatsouli, A.; Kontoyannis, C.G. Quantitative analysis of synthetic calcium carbonate polymorphs using FT-IR spectroscopy. Talanta 2003, 59, 831–836. [Google Scholar] [CrossRef]
- Khouzani, M.F.; Chevrier, D.M.; Güttlein, P.; Hauser, K.; Zhang, P.; Hedinc, N.; Gebauer, D. Disordered amorphous calcium carbonate from direct precipitation. CrystEngComm. 2015, 17, 4842–4849. [Google Scholar] [CrossRef] [Green Version]
- Chakrabarty, D.; Mahapatra, S. Aragonite crystals with unconventional morphologies. J. Mater. Chem. 1999, 9, 2953–2957. [Google Scholar] [CrossRef]
- Xyla, A.G.; Koutsoukos, P.G. Quantitative analysis of calcium carbonate polymorphs by infrared spectroscopy. J. Chem. Soc. Faraday Trans. 1 Phys. Chem. Condens. Phases 1989, 85, 3165–3172. [Google Scholar] [CrossRef]
- Rodriguez-Blanco, J.D.; Shaw, S.; Benning, L.G. The kinetics and mechanisms of amorphous calcium carbonate (ACC) crys-tallization to calcite, viavaterite. Nanoscale 2011, 3, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Siva, T.; Muralidharan, S.; Sathiyanarayanan, S.; Manikandan, E.; Jayachandran, M. Enhanced polymer induced precipitation of polymorphous in calcium carbonate: Calcite aragonite vaterite phases. J. Inorg. Organomet. Polym. Mater. 2017, 27, 770–778. [Google Scholar] [CrossRef]
- Khachani, M.; El Hamidi, A.; Halim, M.; Arsalane, S. Non-isothermal kinetic and thermodynamic studies of the dehydroxylation process of synthetic calcium hydroxide Ca(OH)2. J. Mater. Environ. Sci. 2014, 5, 615–624. [Google Scholar]
- Nehrke, G.; Poigner, H.; Wilhelms-Dick, D.; Brey, T.; Abele, D. Coexistence of three calcium carbonate polymorphs in the shell of the Antarctic clam Laternula elliptica. Geochem. Geophys. Geosyst. 2012, 13, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Behrens, G.; Kuhn, L.T.; Ubic, R.; Heuer, A.H. Raman spectra of vateritic calcium carbonate. Spectrosc. Lett. 1995, 28, 983–995. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Elert, K.; Ševčík, R. Amorphous and crystalline calcium carbonate phases during carbonation of nanolimes: Implications in heritage conservation. CrystEngComm 2016, 18, 6594–6607. [Google Scholar] [CrossRef]













 ) PM with ACC deposits, method CCA; (
) PM with ACC deposits, method CCA; (  ) PM with ACC deposits, method CCB; (
) PM with ACC deposits, method CCB; (  ) calcite powder.
) calcite powder.
 ) PM with ACC deposits, method CCA; (
) PM with ACC deposits, method CCA; (  ) PM with ACC deposits, method CCB; (
) PM with ACC deposits, method CCB; (  ) calcite powder.
) calcite powder.
 ) PM with AmSiO2 deposits, method AmSiA; (
) PM with AmSiO2 deposits, method AmSiA; (  ) PM with AmSiO2 deposits, method AmSiB.
) PM with AmSiO2 deposits, method AmSiB.
 ) PM with AmSiO2 deposits, method AmSiA; (
) PM with AmSiO2 deposits, method AmSiA; (  ) PM with AmSiO2 deposits, method AmSiB.
) PM with AmSiO2 deposits, method AmSiB.
| Parameter/Preparation | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Concentration of DMC (M) | 0.1 | 0.1 | 0.1 | 0.1 | 0.05 |
| Concentration of CaCl2·2H2O (M) | 0.05 | 0.05 | 0.05 | 0.05 | 0.01 |
| Concentration of ΝaOH (M) | 0.3 | 0.3 | 0.3 | 0.3 | 0.1 |
| Concentration of CTAB (ppm) | - | 1 | 10 | 100 | - |
| Material | BET Specific Surface Area (m2/g) | Pore Volume (cm3/g) | % w/w CaCO3/SiO2 in Material |
|---|---|---|---|
| Powdered Pentelic marble | 8.4 | 0.029 | |
| CaCO3 (ACC) | 4.7 | 0.006 | |
| CCA method of CaCO3 deposition | 7.8 | 0.012 | 2.9 |
| CCB method of CaCO3 deposition | 7.9 | 0.015 | 2.8 |
| AmSiO2 | 32.5 | 0.103 | |
| AmSiA method of SiO2 deposition | 23.7 | 0.110 | 13.9 |
| AmSiB method of SiO2 deposition | 22.0 | 0.065 | 13.7 |
| Material | Relative Undersaturation, σ | Dissolution Rate of CaCO3 /×10−8 mol·m−2·s−1 |
|---|---|---|
| Powdered PM | 0.89 | 1.4 |
| CaCO3 (ACC) | 3.4 | |
| CCA | 1.3 | |
| CCB | 2.0 | |
| AmSiO2 | N/A | |
| AmSiA | 0.3 | |
| AmSiB | 0.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavlakou, E.I.; Agrafiotis, A.G.; Tsolaki, T.G.; Lemonia, C.; Zouvani, E.; Paraskeva, C.A.; Koutsoukos, P.G. The Protection of Building Materials of Historical Monuments with Nanoparticle Suspensions. Heritage 2021, 4, 3970-3986. https://doi.org/10.3390/heritage4040218
Pavlakou EI, Agrafiotis AG, Tsolaki TG, Lemonia C, Zouvani E, Paraskeva CA, Koutsoukos PG. The Protection of Building Materials of Historical Monuments with Nanoparticle Suspensions. Heritage. 2021; 4(4):3970-3986. https://doi.org/10.3390/heritage4040218
Chicago/Turabian StylePavlakou, Efstathia I., Anastasios G. Agrafiotis, Theokleiti G. Tsolaki, Christine Lemonia, Emily Zouvani, Christakis A. Paraskeva, and Petros G. Koutsoukos. 2021. "The Protection of Building Materials of Historical Monuments with Nanoparticle Suspensions" Heritage 4, no. 4: 3970-3986. https://doi.org/10.3390/heritage4040218
APA StylePavlakou, E. I., Agrafiotis, A. G., Tsolaki, T. G., Lemonia, C., Zouvani, E., Paraskeva, C. A., & Koutsoukos, P. G. (2021). The Protection of Building Materials of Historical Monuments with Nanoparticle Suspensions. Heritage, 4(4), 3970-3986. https://doi.org/10.3390/heritage4040218







