Negative Chronotropic Effects of Class I Antiarrhythmic Drugs on Guinea Pig Right Atria: Correlation with L-Type Ca2+ Channel Blockade
Abstract
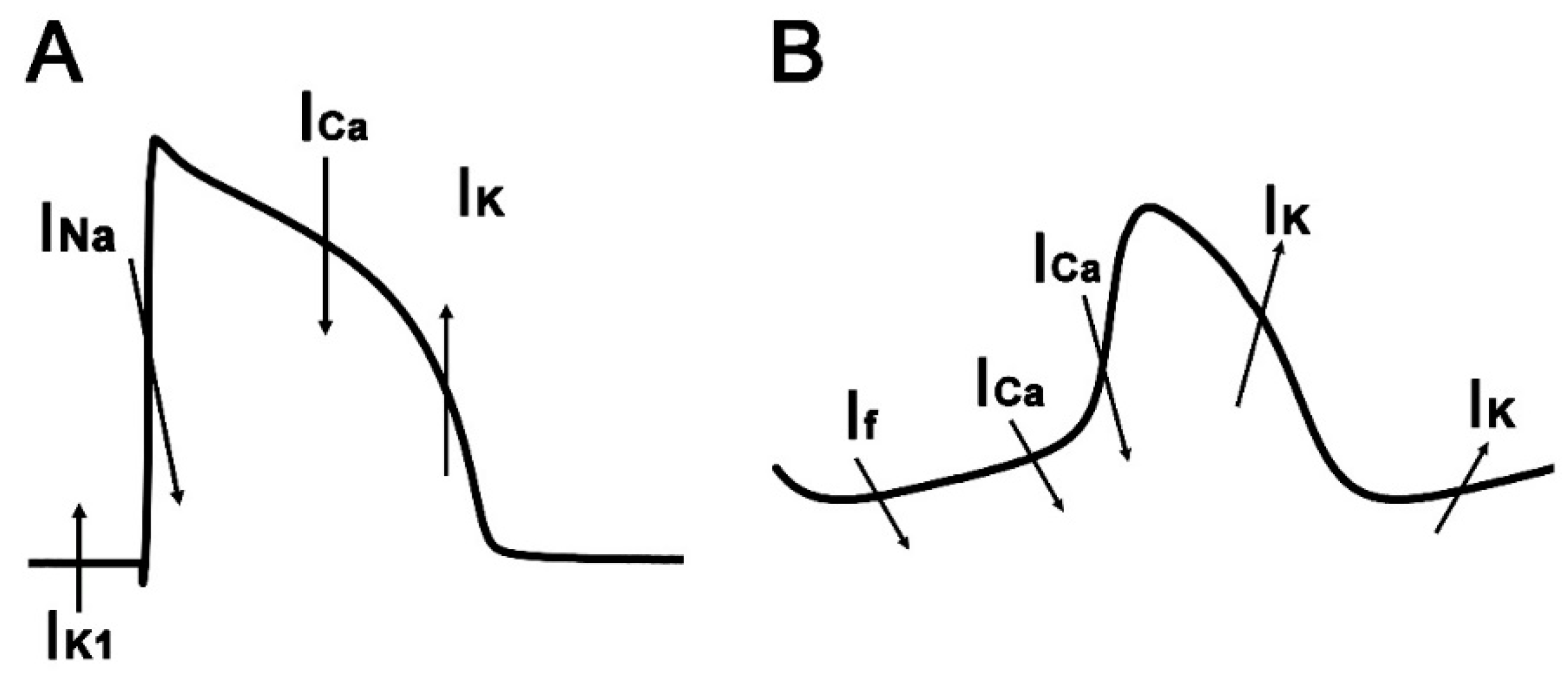
1. Introduction
2. Materials and Methods
2.1. Measurement of Beating Rate
2.2. Drugs and Chemicals
2.3. Statistics
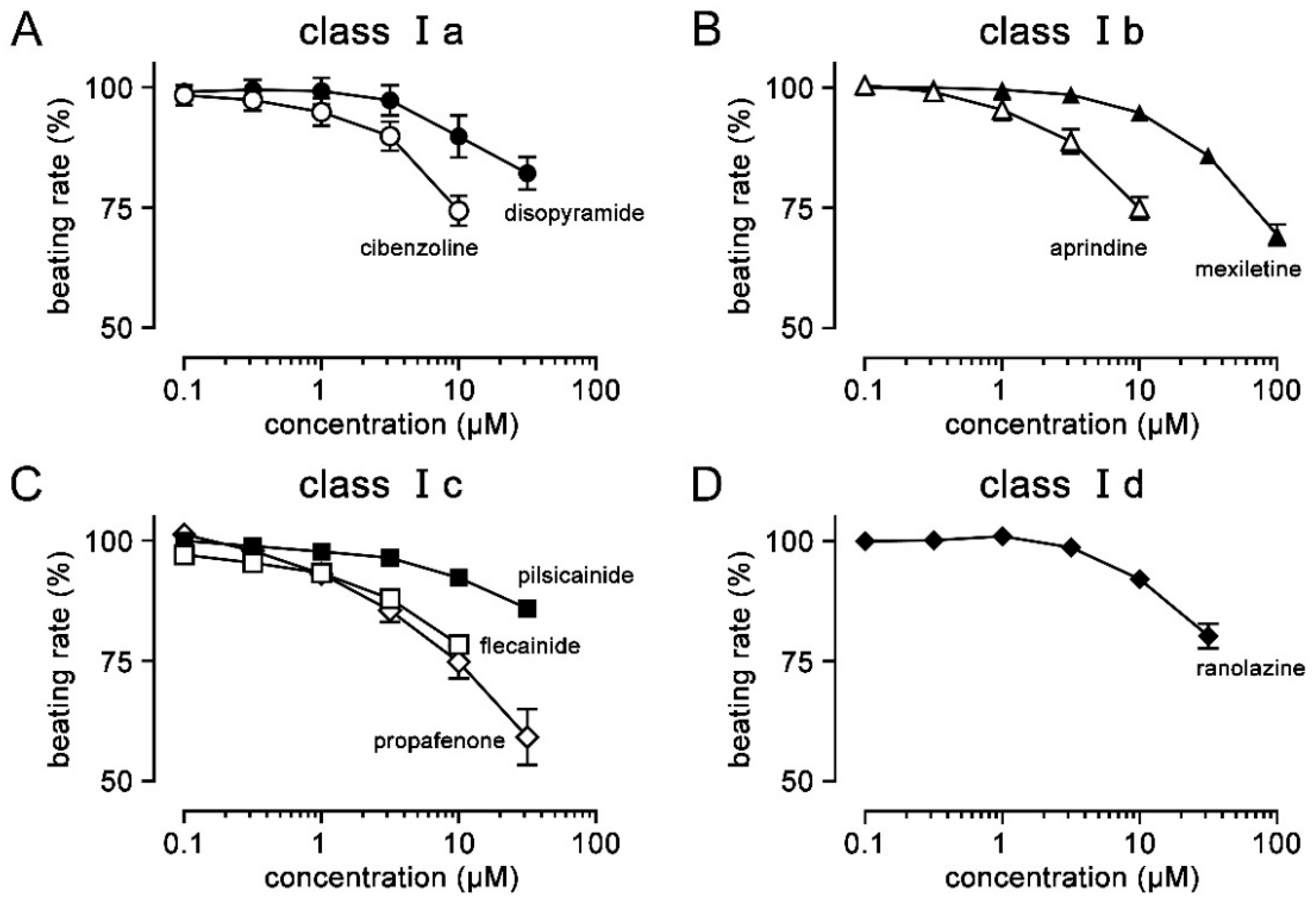
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vaughan Williams, E.M. Classification of antiarrhythmic drugs. Pharmacol. Ther. 1975, 1, 115–138. [Google Scholar] [CrossRef]
- Lei, M.; Wu, L.; Terrar, D.A.; Huang, C.L. Modernized Classification of Cardiac Antiarrhythmic Drugs. Circulation 2018, 138, 1879–1896. [Google Scholar] [CrossRef] [PubMed]
- The Japanese Circulation Society and Japanese Heart Rhythm Society Joint Working Group. JCS/JHRS 2020 Guideline on Pharmacotherapy of Cardiac Arrhythmias. Circ. J. 2022, 86, 1790–1924. [Google Scholar] [CrossRef]
- Roden, D.M. Pharmacology and toxicology of Nav1.5-class I anti-arrhythmic drugs. Card. Electrophysiol. Clin. 2014, 6, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.; Nattel, S. The past, present, and potential future of sodium channel block as an atrial fibrillation suppressing strategy. J. Cardiovas. Pharmacol. 2015, 66, 432–440. [Google Scholar] [CrossRef]
- Hiiro, H.; Hashimoto, T.; Mizoguchi, M.; Kaneko, M.; Deguchi, N.; Takahashi, Y.; Hamaguchi, S.; Namekata, I.; Tanaka, H. Negative inotropic effects of class I antiarrhythmics on guinea pig ventricular myocardium: Correlation with L-type Ca2+ channel blockade. Biol. Pharm. Bull. 2023, 46, 133–137. [Google Scholar] [CrossRef]
- Namekata, I.; Hiiro, H.; Odaka, R.; Saito, T.; Hamaguchi, S.; Tsukamoto, T.; Ishikawa, R.; Katayama, Y.; Kondo, Y.; Tanaka, H. Inhibitory effect of a late sodium current blocker, NCC-3902, on the automaticity of the guinea pig pulmonary vein myocardium. Biol. Pharm. Bull. 2022, 45, 1644–1652. [Google Scholar] [CrossRef]
- Namekata, I.; Jitsukata, K.; Fukuda, A.; Odaka, R.; Hamaguchi, S.; Tanaka, H. Intracellular Ca2+-mediated mechanisms for the pacemaker depolarization of the mouse and guinea pig sinus node tissue. Biomolecules 2022, 12, 377. [Google Scholar] [CrossRef]
- Takada, M.; Goto, T.; Kotake, T.; Saito, M.; Kawato, N.; Nakai, M.; Gunji, T.; Shibakawa, M. Appropriate Dosing of Antiarrhythmic Drugs in Japan Requires Therapeutic Drug Monitoring*. J. Clin. Pharm. Ther. 2005, 30, 5–12. [Google Scholar] [CrossRef]
- Kodama, I.; Ogawa, S.; Inoue, H.; Kasanuki, H.; Kato, T.; Mitamura, H.; Hiraoka, M.; Sugimoto, T. Profiles of aprindine, cibenzoline, pilsicainide and pirmenol in the framework of the Sicilian Gambit. The Guideline Committee for Clinical Use of Antiarrhythmic Drugs in Japan (Working Group of Arrhythmias of the Japanese Society of Electrocardiology). Jpn. Circ. J. 1999, 63, 1–12. [Google Scholar] [CrossRef]
- Hiramatsu, M.; Wu, L.M.; Hirano, Y.; Kawano, S.; Furukawa, T.; Hiraoka, M. Block of HERG current expressed in HEK293 cells by the Na-channel blocker cibenzoline. Heart Vessels. 2004, 19, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Tamura, A.; Ogura, T.; Umemura, H.; Reien, Y.; Kishimoto, T.; Nagai, T.; Komuro, I.; Miyazaki, M.; Nakaya, H. Effects of antiarrhythmic drugs on the hyperpolarization-activated cyclic nucleotide-gated channel current. J. Pharmacol. Sci. 2009, 110, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Satoh, H. Comparative actions of cibenzoline and disopyramide on I(Kr) and I(Ks) currents in rat sino-atrial nodal cells. Eur. J. Pharmacol. 2000, 407, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Harchi, A.E.; Zhang, Y.H.; Hussein, L.; Dempsey, C.E.; Hancox, J.C. Molecular determinants of hERG potassium channel inhibition by disopyramide. J. Mol. Cell. Cardiol. 2012, 52, 185–190. [Google Scholar] [CrossRef]
- Yoshida, H.; Horie, M.; Otani, H.; Takano, M.; Tsuji, K.; Kubota, T.; Fukunami, M.; Sasayama, S. Characterization of a novel missense mutation in the pore of HERG in a patient with long QT syndrome. J. Cardiovas. Electrophysiol. 1999, 10, 1262–1270. [Google Scholar] [CrossRef] [PubMed]
- Yonemizu, S.; Masuda, K.; Kurata, Y.; Notsu, T.; Higashi, Y.; Fukumura, K.; Li, P.; Ninomiya, H.; Miake, J.; Tsuneto, M.; et al. Inhibitory effects of class I antiarrhythmic agents on Na+ and Ca2+ currents of human iPS cell-derived cardiomyocytes. Regen. Ther. 2019, 10, 104–111. [Google Scholar] [CrossRef]
- Ono, K.; Kiyosue, T.; Arita, M. Comparison of the inhibitory effects of mexiletine and lidocaine on the calcium current of single ventricular cells. Life Sci. 1986, 39, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Gualdani, R.; Tadini-Buonisegni, F.; Roselli, M.; Defrenza, I.; Contino, M.; Colabufo, N.A.; Lentini, G. Inhibition of hERG potassium channel by the antiarrhythmic agent mexiletine and its metabolite m-hydroxymexiletine. Pharmacol. Res. Perspect. 2015, 3, e00160. [Google Scholar] [CrossRef]
- Heath, B.M.; Cui, Y.; Worton, S.; Lawton, B.; Ward, G.; Ballini, E.; Doe, C.P.; Ellis, C.; Patel, B.A.; McMahon, N.C. Translation of flecainide- and mexiletine-induced cardiac sodium channel inhibition and ventricular conduction slowing from nonclinical models to clinical. J. Pharmacol. Toxicol. Methods 2011, 63, 258–268. [Google Scholar] [CrossRef]
- Kramer, J.; Obejero-Paz, C.A.; Myatt, G.; Kuryshev, Y.A.; Bruening-Wright, A.; Verducci, J.S.; Brown, A.M. MICE models: Superior to the HERG model in predicting Torsade de Pointes. Sci. Rep. 2013, 3, 2100. [Google Scholar] [CrossRef]
- Melgari, D.; Zhang, Y.; El Harchi, A.; Dempsey, C.E.; Hancox, J.C. Molecular basis of hERG potassium channel blockade by the class Ic antiarrhythmic flecainide. J. Mol. Cell. Cardiol. 2015, 86, 42–53. [Google Scholar] [CrossRef]
- Inomata, N.; Ishihara, T.; Akaike, N. SUN 1165: A new antiarrhythmic Na current blocker in ventricular myocytes of guinea-pig. Comp. Biochem. Physiol. C Comp. Pharmacol. Toxicol. 1987, 87, 237–243. [Google Scholar] [CrossRef]
- Wu, L.M.; Orikabe, M.; Hirano, Y.; Kawano, S.; Hiraoka, M. Effects of Na+ channel blocker, pilsicainide, on HERG current expressed in HEK-293 cells. J. Cardiovasc. Pharmacol. 2003, 42, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Siddoway, L.A.; Roden, D.M.; Woosley, R.L. Clinical Pharmacology of Propafenone: Pharmacokinetics, Metabolism and Concentration-Response Relations. Am. J. Cardiol. 1984, 54, 9D–12D. [Google Scholar] [CrossRef]
- Antzelevitch, C.; Nesterenko, V.; Shryock, J.C.; Rajamani, S.; Song, Y.; Belardinelli, L. The role of late I Na in development of cardiac arrhythmias. Handb. Exp. Pharmacol. 2014, 221, 137–168. [Google Scholar] [CrossRef] [PubMed]
- Delgado, C.; Tamargo, J.; Henzel, D.; Lorente, P. Effects of propafenone on calcium current in guinea-pig ventricular myocytes. Br. J. Pharmacol. 1993, 108, 721–727. [Google Scholar] [CrossRef]
- Arias, C.; González, T.; Moreno, I.; Caballero, R.; Delpón, E.; Tamargo, J.; Valenzuela, C. Effects of propafenone and its metabolite, 5-hydroxypropafenone, on HERG channels. Cardiovas. Res. 2003, 57, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Antzelevitch, C.; Burashnikov, A.; Sicouri, S.; Belardinelli, L. Electrophysiological Basis for the Antiarrhythmic Actions of Ranolazine. Heart Rhythm. 2011, 8, 1281. [Google Scholar] [CrossRef]
- Antzelevitch, C.; Belardinelli, L.; Zygmunt, A.C.; Burashnikov, A.; Di Diego, J.M.; Fish, J.M.; Cordeiro, J.M.; Thomas, G. Electrophysiological effects of ranolazine, a novel antianginal agent with antiarrhythmic properties. Circulation 2004, 110, 904–910. [Google Scholar] [CrossRef]
- Satoh, H. Sino-atrial nodal cells of mammalian hearts: Ionic currents and gene expression of pacemaker ionic channels. J. Smooth Muscle Res. 2003, 39, 175–193. [Google Scholar] [CrossRef]
- Kodama, I.; Nikmaram, M.R.; Boyett, M.R.; Suzuki, R.; Honjo, H.; Owen, J.M. Regional differences in the role of the Ca2+ and Na+ currents in pacemaker activity in the sinoatrial node. Am. J. Physiol. 1997, 272, H2793–H2806. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, H.; Nathan, R.D.; Shimura, T. A TTX-sensitive transient Na+ current recorded in morphologically identified primary pacemaker cells. J. Nippon. Med. Sch. 1999, 66, 350–352. [Google Scholar] [CrossRef]
- Sanders, L.; Rakovic, S.; Lowe, M.; Mattick, P.A.D.; Terrar, D.A. Fundamental importance of Na+–Ca2+ exchange for the pacemaking mechanism in the guina-pig sino-atrial node. J. Physiol. 2006, 571, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Masumiya, H.; Tanaka, H.; Shigenobu, K. Effects of Ca2+ channel antagonists on sinus node: Prolongation of late phase 4 depolarization by efonidipine. Eur. J. Pharmacol. 1997, 335, 15–21. [Google Scholar] [CrossRef]
- Yamamoto, M.; Gotoh, Y.; Imaizumi, Y.; Watanabe, M. Mechanisms of long-lasting effects of benidipine on Ca current in guinea-pig ventricular cells. Br. J. Pharmacol. 1990, 100, 669–676. [Google Scholar] [CrossRef]
- Hof, R.P.; Scholtysik, G. Effects of the calcium antagonist PY 108-068 on myocardial tissues in vitro and on reflex tachycardia in vivo. J. Cardiovas. Pharmacol. 1983, 5, 176–183. [Google Scholar] [CrossRef]
- Wettver, E.; Scholtysik, G.; Schaad, A.; Himmel, H.; Ravens, U. Effects of the new class III antiarrhythmic drug E-4031 on myocardial contractility and electrophysiological parameters. J. Cardiovas. Pharmacol. 1991, 17, 480–487. [Google Scholar] [CrossRef]
- Zhou, Z.; Gong, Q.; Ye, B.; Fan, Z.; Makielski, J.C.; Robertson, G.A.; January, C.T. Properties of HERG channels stably expressed in HEK293 cells studied at physiological temperature. Biophys. J. 1998, 74, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Denyer, J.C.; Brown, H.F. Pacemaking in rabbit isolated sino-atrial node cells during Ca+ block of the hyperpolarization-activated current if. J. Physiol. 1990, 429, 401–409. [Google Scholar] [CrossRef]
- Sohn, H.G.; Vassalle, M. Cesium effects on dual pacemaker mechanisms in guinea pig sinoatrial node. J. Mol. Cell. Cardiol. 1995, 27, 563–577. [Google Scholar] [CrossRef] [PubMed]
- Melgari, D.; Brack, K.E.; Zhang, C.; Zhang, Y.; El Harchi, A.; Mitcheson, J.S.; Dempsey, C.E.; Ng, G.A.; Hancox, J.C. hERG potassium channel block by the HCN channel inhibitor bradycardic agent ivabradine. J. Am. Heart Assoc. 2015, 4, e001813. [Google Scholar] [CrossRef]
- Himeno, Y.; Toyoda, F.; Satoh, H.; Amano, A.; Cha, C.Y.; Matsuura, H.; Noma, A. Minor contribution of cytosolic Ca2+ transients to the pacemaker rhythm in guinea pig sinoatrial node cells. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H251–H261. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Iwamoto, T.; Shigekawa, M.; Kimura, J. Inhibitory effect of aprindine on Na+/Ca2+ exchange current in guinea-pig cardiac ventricular myocytes. Br. J. Pharmacol. 2002, 136, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Koide, Y.; Kimura, J. Topics on the Na+/Ca2+ exchange: Pharmacological characterization of Na+/Ca2+ exchanger inhibitors. J. Pharmacol. Sci. 2006, 102, 7–16. [Google Scholar] [CrossRef]
- Yamakawa, T.; Watanabe, Y.; Watanabe, H.; Kimura, J. Inhibitory effect of cibenzoline on Na+/Ca2+ exchange current in guinea-pig cardiac ventricular myocytes. J. Pharmacol. Sci. 2012, 120, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Ono, K.; Iijima, T. Pathophysiological significance of T-type Ca2+ channels: Properties and functional roles of T-type Ca2+ channels in cardiac pacemaking. J. Pharmacol. Sci. 2005, 99, 197–204. [Google Scholar] [CrossRef]
- Ono, K.; Iijima, T. Cardiac T-type Ca2+ channels in the heart. J. Mol. Cell. Cardiol. 2010, 48, 65–70. [Google Scholar] [CrossRef]
- Tanaka, H.; Komikado, C.; Namekata, I.; Nakamura, H.; Suzuki, M.; Tsuneoka, Y.; Shigenobu, K.; Takahara, A. Species difference in the contribution of T-type Ca2+ current to cardiac pacemaking as revealed by R(−)-efonidipine. J. Pharmacol. Sci. 2008, 107, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Mitsuiye, T.; Shinagawa, Y.; Noma, A. Sustained inward current during pacemaker depolarization in mammalian sinoatrial node cells. Circ. Res. 2000, 87, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, F.; Mesirca, P.; Dubel, S.; Ding, W.G.; Striessnig, J.; Mangoni, M.E.; Matsuura, H. Cav1.3 L-type Ca2+ channel contributes to the heartbeat by generating a dihydropyridine-sensitive persistent Na+ current. Sci. Rep. 2017, 7, 7869. [Google Scholar] [CrossRef]
- Belardinelli, L.; Giles, W.R.; Rajamani, S.; Karagueuzian, H.S.; Shryock, J.C. Cardiac late Na+ current: Proarrhythmic effects, roles in long QT syndromes, and pathological relationship to CaMKII and oxidative stress. Heart Rhythm. 2014, 12, 440–448. [Google Scholar] [CrossRef]
- Parikh, A.; Mantravadi, R.; Kozhevnikov, D.; Roche, M.A.; Ye, Y.; Owen, L.J.; Puglisi, J.L.; Abramson, J.J.; Salama, G. Ranolazine stabilizes cardiac ryanodine receptors: A novel mechanism for the suppression of early afterdepolarization and torsades de pointes in long QT type 2. Heart Rhythm. 2012, 9, 953–960. [Google Scholar] [CrossRef]
- Rouhana, S.; Virsolvy, A.; Fares, N.; Richard, S.; Thireau, J. Ranolazine: An old drug with emerging potential; lessons from pre-clinical and clinical investigations for possible repositioning. Pharmaceuticals 2021, 15, 31. [Google Scholar] [CrossRef]
- Irie, M.; Hiiro, H.; Hamaguchi, S.; Namekata, I.; Tanaka, H. Involvement of the persistent Na+ current in the diastolic depolarization and automaticity of the guinea pig pulmonary vein myocardium. J. Pharmacol. Sci. 2019, 141, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Namekata, I.; Tsuneoka, Y.; Tanaka, H. Electrophysiological and pharmacological properties of the pulmonary vein myocardium. Biol. Pharm. Bull. 2013, 36, 2–7. [Google Scholar] [CrossRef]
- Takahara, A.; Hagiwara, M.; Namekata, I.; Tanaka, H. Pulmonary vein myocardium as a possible pharmacological target for the treatment of atrial fibrillation. J. Pharmacol. Sci. 2014, 126, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Namekata, I.; Hiiro, H.; Odaka, R.; Saito, T.; Hamaguchi, S.; Tsukamoto, T.; Hamaguchi, S.; Ishikawa, R.; Kawai, Y.; Katayama, Y.; et al. NCC-3902, a novel late sodium current blocker, exhibit potent antiarrhythmic effect in isolated guinea pig pulmonary vein and canine rapid atrial pacing model. In Proceedings of the 95th Annual Meeting of the Japanese Pharmacological Society, Fukuoka, Japan, 7–9 March 2022. 1-LBS-02. [Google Scholar]
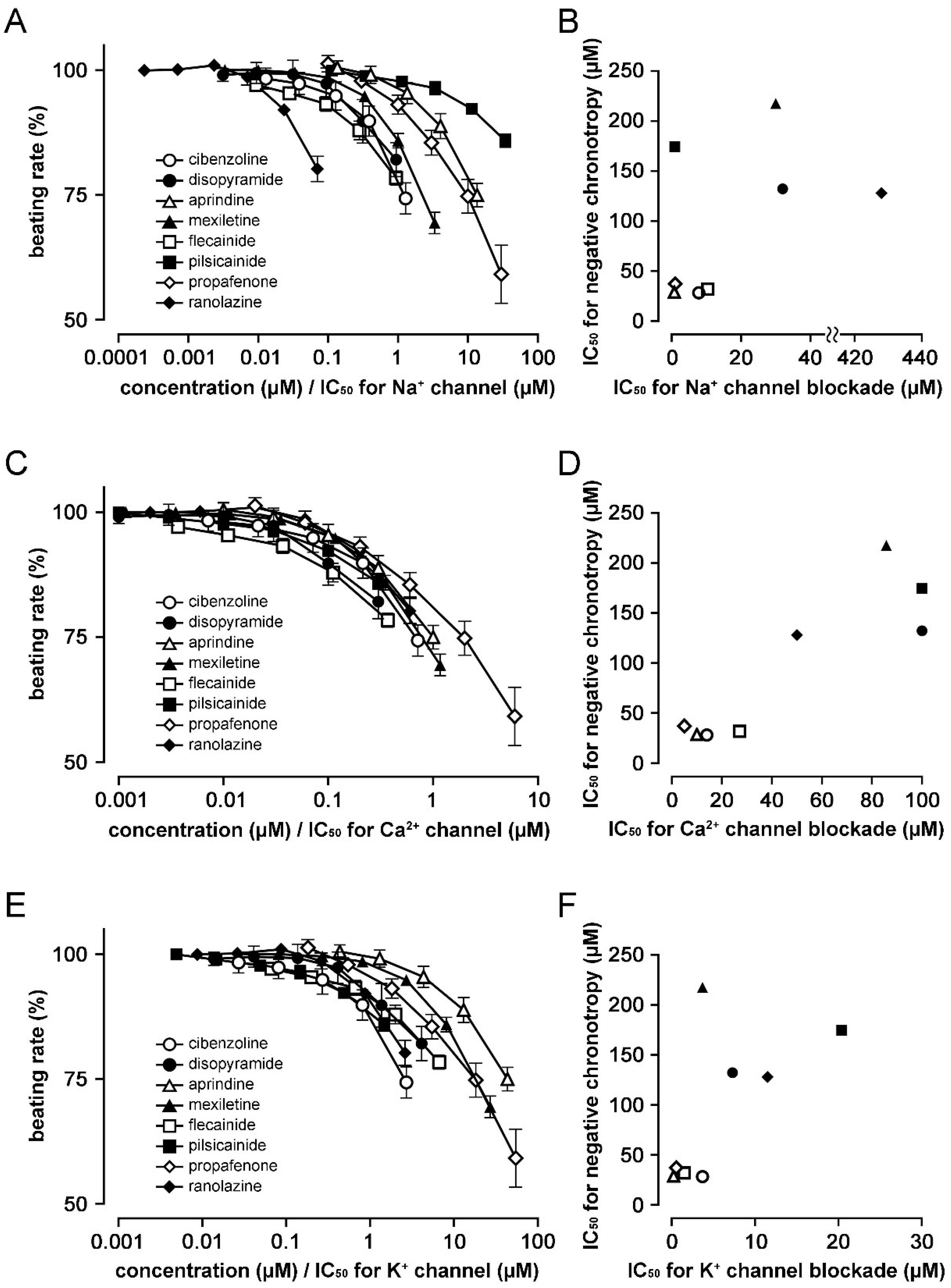
| Therapeutic Concentration Range | IC50 for Negative Chronotropy * | IC50 for Na+ Channel | IC50 for Ca2+ Channel | IC50 for K+ Channel | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ia | Cibenzoline | 0.8–3.0 | [9] | 28.3 | 7.8 | [10] | 14 | [10] | 3.7 | [11] |
| Disopyramide | 5.9–14.7 | [12] | 132.2 | 32 | [13] | 100 | [13] | 7.3 | [14] | |
| Ib | Aprindine | 0.4–2.1 | [9] | 28.8 | 0.74 | [10] | 10 | [10] | 0.23 | [15] |
| Mexiletine | 2.8–11.1 | [12] | 217.6 | 30 | [16] | 85.74 | [17] | 3.7 | [18] | |
| Ic | Flecainide | 0.48–2.4 | [12] | 32.0 | 10.4 | [19] | 27.1 | [20] | 1.5 | [21] |
| Pilsicainide | 0.73–3.67 | [9] | 174.5 | 0.88 | [16] | >100 | [22] | 20.4 | [23] | |
| Propafenone | 0.27–9.58 | [24] | 37.2 | 1 | [25] | 5 | [26] | 0.55 | [27] | |
| Id | Ranolazine | 2–8 | [28] | 128.0 | 428 | [25] | 50 | [29] | 11.5 | [29] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hiiro, H.; Otsuka, K.; Hamaguchi, S.; Namekata, I.; Tanaka, H. Negative Chronotropic Effects of Class I Antiarrhythmic Drugs on Guinea Pig Right Atria: Correlation with L-Type Ca2+ Channel Blockade. J 2023, 6, 104-114. https://doi.org/10.3390/j6010008
Hiiro H, Otsuka K, Hamaguchi S, Namekata I, Tanaka H. Negative Chronotropic Effects of Class I Antiarrhythmic Drugs on Guinea Pig Right Atria: Correlation with L-Type Ca2+ Channel Blockade. J. 2023; 6(1):104-114. https://doi.org/10.3390/j6010008
Chicago/Turabian StyleHiiro, Haruhito, Kentaro Otsuka, Shogo Hamaguchi, Iyuki Namekata, and Hikaru Tanaka. 2023. "Negative Chronotropic Effects of Class I Antiarrhythmic Drugs on Guinea Pig Right Atria: Correlation with L-Type Ca2+ Channel Blockade" J 6, no. 1: 104-114. https://doi.org/10.3390/j6010008
APA StyleHiiro, H., Otsuka, K., Hamaguchi, S., Namekata, I., & Tanaka, H. (2023). Negative Chronotropic Effects of Class I Antiarrhythmic Drugs on Guinea Pig Right Atria: Correlation with L-Type Ca2+ Channel Blockade. J, 6(1), 104-114. https://doi.org/10.3390/j6010008





