Ce2O3/BiVO4 Embedded in rGO as Photocatalyst for the Degradation of Methyl Orange under Visible Light Irradiation
Abstract
1. Introduction
2. Materials
2.1. Methods
2.1.1. Synthesis of Graphene Oxide
2.1.2. Preparation of Ce2O3/BiVO4 Nanoparticles
2.1.3. Preparation of Ce2O3/BiVO4 in rGO
2.2. Characterizations
3. Results and Discussion
3.1. XRD Analysis
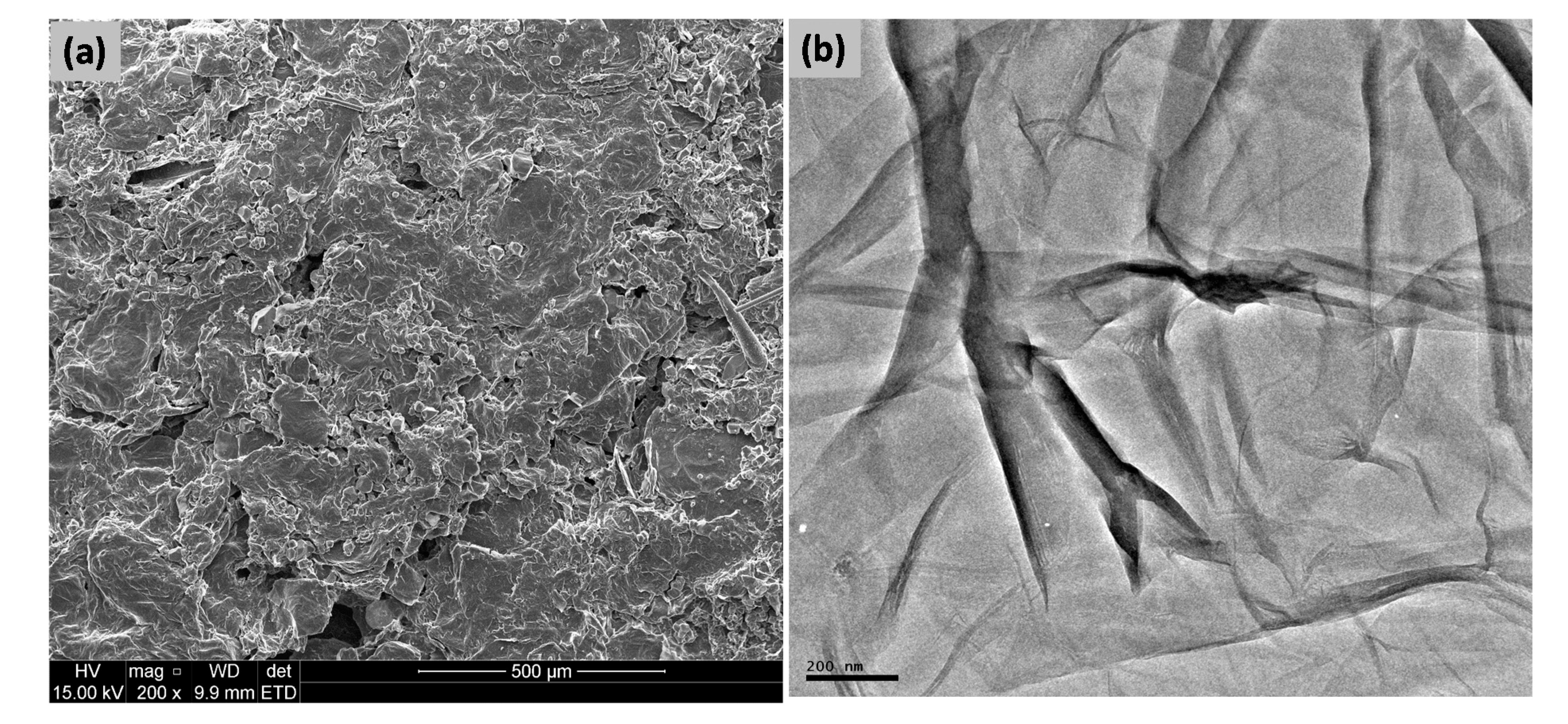
3.2. Morphology Studies
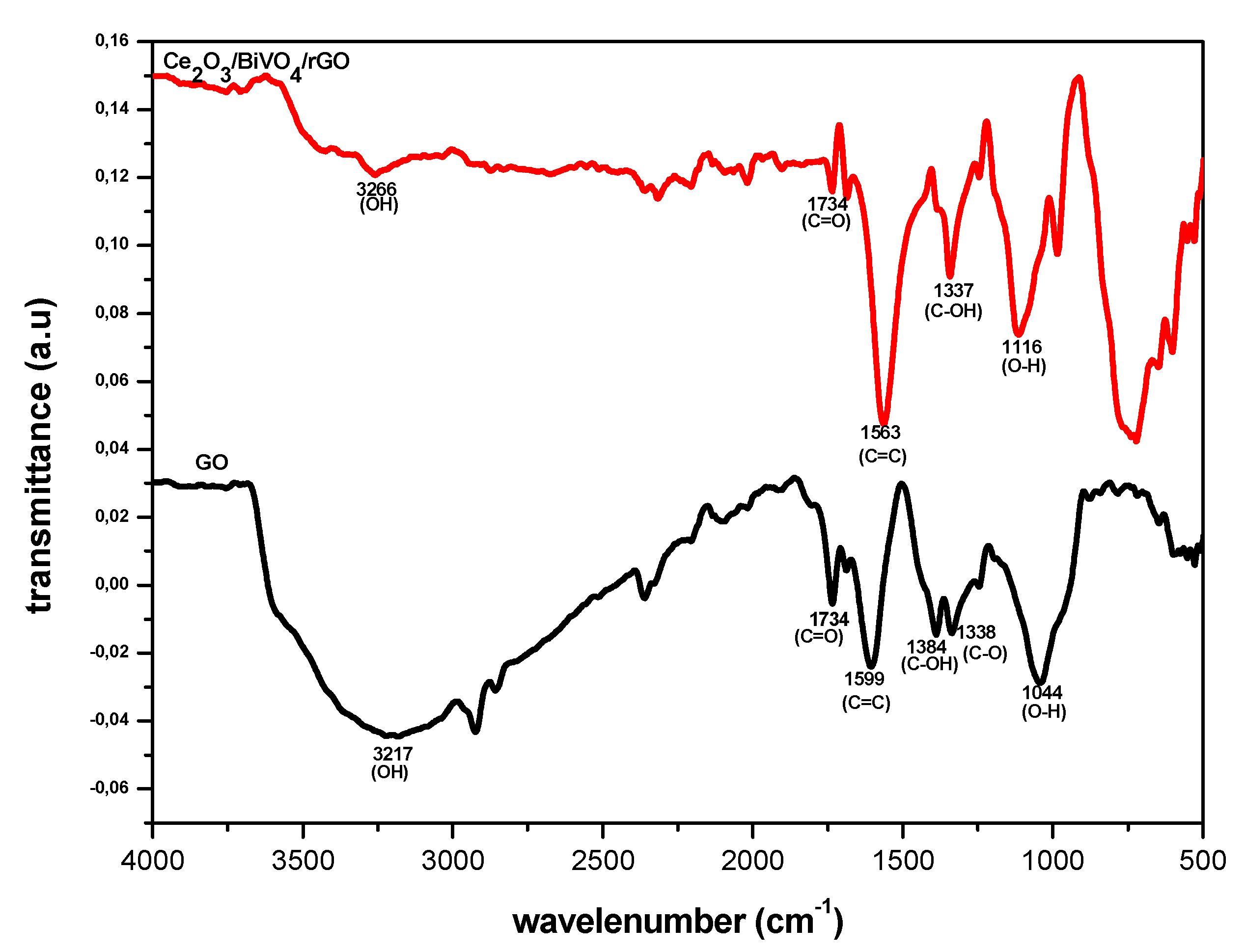
3.3. FTIR Spectroscopy
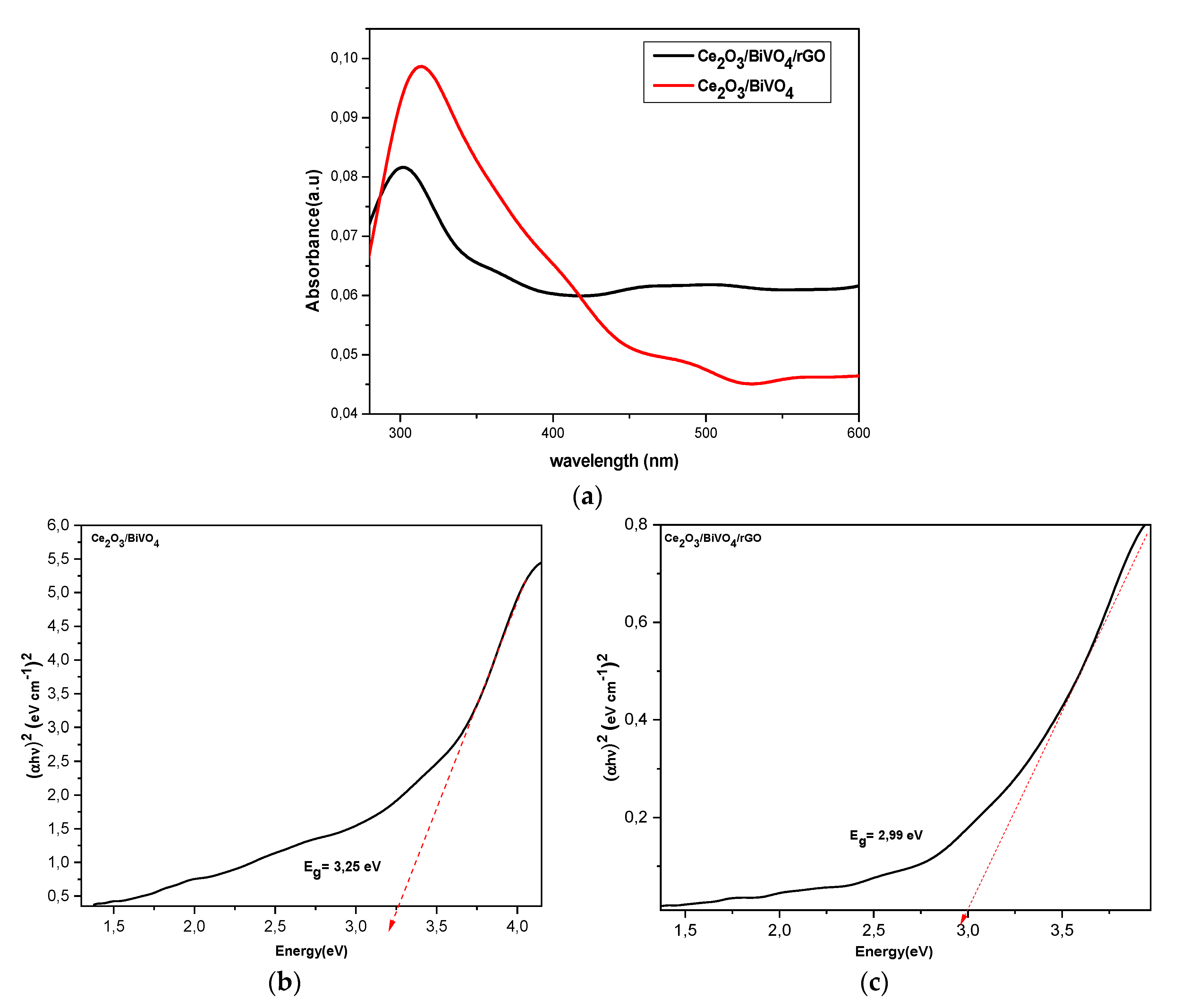
3.4. Optical Spectroscopy
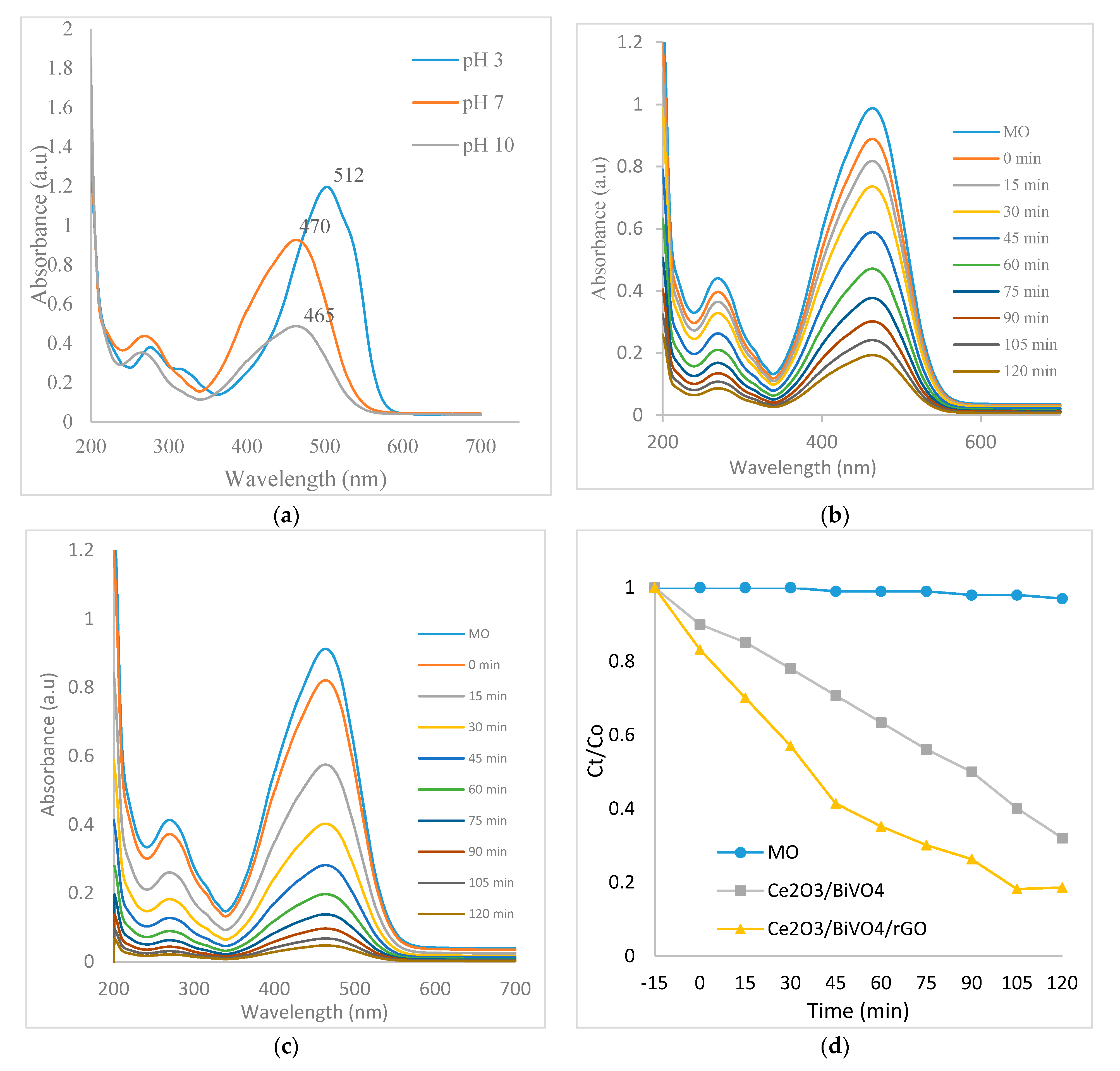
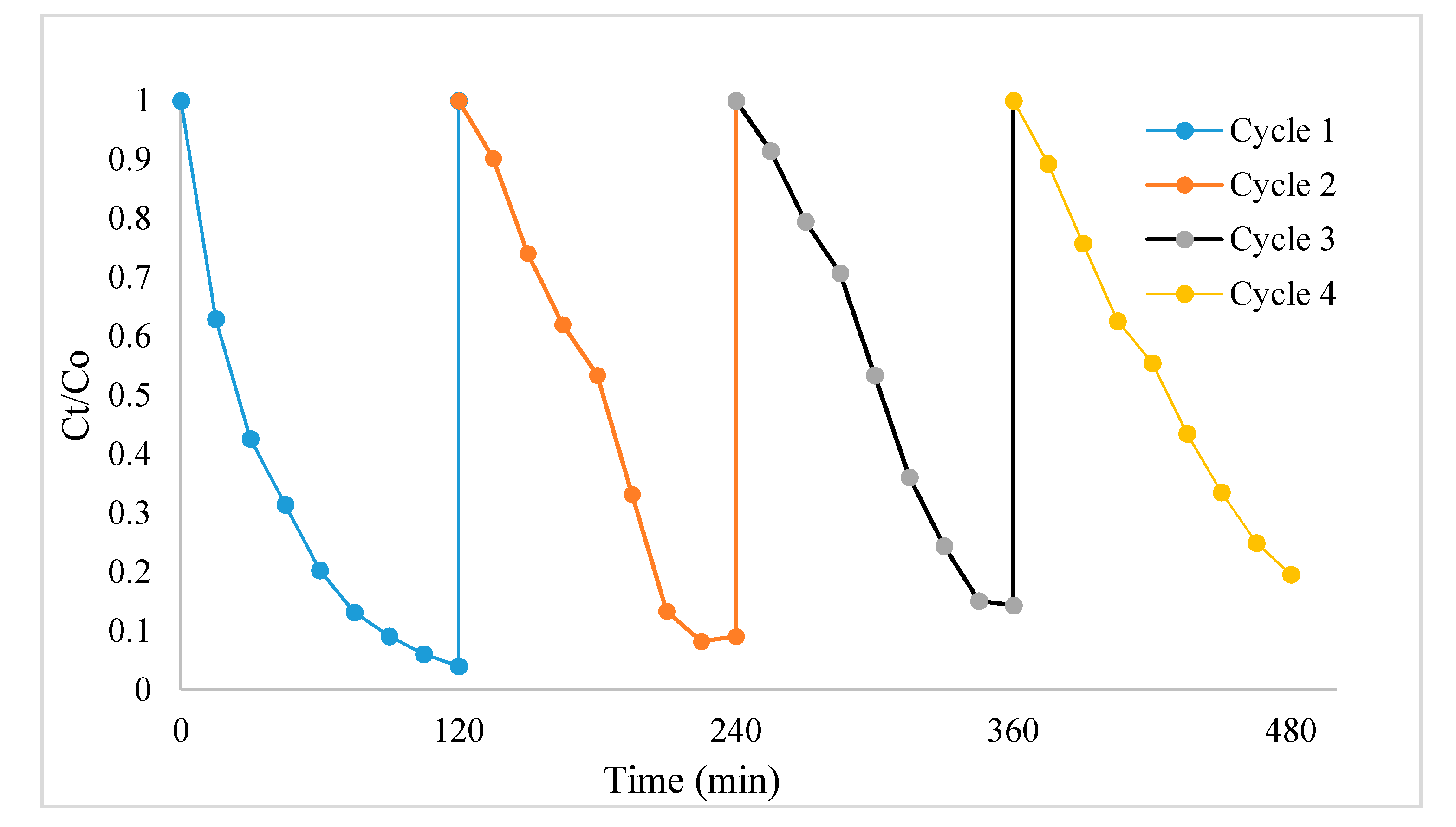
3.5. Photocatalytic Activity
3.5.1. Effect of Initial Dye Concentration
3.5.2. Effect of Catalysts Loading
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sivakumar, V.; Suresh, R.; Giribabu, K.; Narayanan, V. BiVO4 nanoparticles: Preparation, characterization and photocatalytic activity. Cogent Chem. 2015, 1, 1074647. [Google Scholar] [CrossRef]
- Xu, W.; Liu, Z.; Fang, J.; Zhou, G.; Hong, X.; Wu, S.; Zhu, X.; Chen, Y.; Cen, C. CTAB-Assisted Hydrothermal Synthesis of Bi2Sn2O7 Photocatalyst and Its Highly Efficient Degradation of Organic Dye under Visible-Light Irradiation. Int. J. Photoenergy 2013, 2013, 394079. [Google Scholar] [CrossRef]
- Lin, B.; Li, H.; An, H.; Hao, W.B.; Wei, J.J.; Dai, Y.Z.; Ma, C.S.; Yang, G.D. Preparation of 2D/2D g-C3N4 nanosheet@ZnIn2S4 nanoleaf heterojunctions with well-designed high-speed charge transfer nanochannels towards high efficiency photocatalytic hydrogen evolution. Appl. Catal. B Environ. 2018, 220, 542–552. [Google Scholar] [CrossRef]
- Yang, K.; Li, X.; Yu, C.; Zeng, D.; Chen, F.; Zhang, K.; Huang, W.; Ji, H. Review on heterophase/homophase junctions for efficient photocatalysis: The case of phase transition construction. Chin. J. Catal. 2019, 40, 796–818. [Google Scholar] [CrossRef]
- Kabachii, Y.A.; Golub, A.S.; Kochev, S.Y.; Lenenko, N.D.; Abramchuk, S.S.; Antipin, M.Y.; Valetsky, P.M.; Stein, B.D.; Mahmoud, W.E.; Al-Ghamdi, A.A.; et al. Multifunctional Nanohybrids by Self-Assembly of Monodisperse Iron Oxide Nanoparticles and Nanolamellar MoS2 Plates. Chem. Mater. 2013, 25, 2434–2440. [Google Scholar] [CrossRef]
- Yuzik-Klimova, E.Y.; Kuchkina, N.V.; Sorokina, S.A. Magnetically recoverable catalysts based on polyphenylenepyridyl dendrons and dendrimers. RSC Adv. 2014, 4, 23271–23280. [Google Scholar] [CrossRef]
- Mahmoud, W.E. Solar blind avalanche photodetector based on the cation exchange growth of β-Ga2O3/SnO2 bilayer heterostructure thin film. Sol. Energy Mater. Sol. Cells 2016, 152, 65–72. [Google Scholar] [CrossRef]
- Al-Hazmi, F.S.; Al-Harbi, G.H.; Beall, G.W. Synthesis and structure of high quality graphene prepared via solvothermal exfoliation of intercalated graphite flakes. Superlattices Microstruct. 2015, 86, 270–274. [Google Scholar] [CrossRef]
- Sitaaraman, S.R.; Shanmugapriyan, M.I.; Varunkumar, K. Synthesis of heterojunction tungsten oxide (WO3) and Bismuth vanadate (BiVO4) photoanodes by spin coating method for solar water splitting applications. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Lai, B.-R.; Lin, L.-Y.; Xiao, B.-C.; Chen, Y.-S. Facile synthesis of bismuth vanadate/bismuth oxide heterojunction for enhancing visible light-responsive photoelectrochemical performance. J. Taiwan Inst. Chem. Eng. 2019, 100, 178–185. [Google Scholar] [CrossRef]
- Claudino, C.H.; Kuznetsova, M.; Rodrigues, B.S.; Chen, C.; Wang, Z.; Sardela, M.; Souza, J.S. Facile one-pot microwave-assisted synthesis of tungsten-doped BiVO4/WO3 heterojunctions with enhanced photocatalytic activity. Mater. Res. Bull. 2020, 125, 110783. [Google Scholar] [CrossRef]
- Liu, Y.; Yuan, A.; Xiao, Y.; Yu, H.; Dong, X. Two-dimensional/two-dimensional Z-scheme photocatalyst of graphitic carbon nitride/bismuth vanadate for visible-light-driven photocatalytic synthesis of imines. Ceram. Int. 2020, 46, 16157–16165. [Google Scholar] [CrossRef]
- Sun, M.; Yan, T.; Zhang, Y.; He, Y.; Shao, Y.; Wei, Q.; Du, B. Rod-like Bi4O7 decorated Bi2O2CO3 plates: Facile synthesis, promoted charge separation, and highly efficient photocatalytic degradation of organic contaminants. J. Colloid Interface Sci. 2018, 514, 240–249. [Google Scholar] [CrossRef]
- Sun, M.; Wang, Y.; Shao, Y.; He, Y.; Zeng, Q.; Liang, H.; Yan, T.; Du, B. Fabrication of a novel Z-scheme g-C3N4/Bi4O7 heterojunction photocatalyst with enhanced visible light-driven activity toward organic pollutants. J. Colloid Interface Sci. 2017, 501, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Wei, W.; Zheng, L.; Liu, Y.; Wu, D.; Gu, X.; Wei, A. Enhanced visible light photoreduction of aqueous Cr(VI) by Ag/Bi4O7/g-C3N4 nanosheets ternary metal/non-metal Z-scheme heterojunction. J. Hazard. Mater. 2019, 365, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Li, S.; Ma, H.; Gao, J.; Zhu, G.; Zhang, F.; Park, J.Y.; Cha, S.; Bae, J.-S.; Liu, C. Oxygen vacancy rich Bi2O4-Bi4O7-BiO2-x composites for UV-vis-NIR activated high efficient photocatalytic degradation of bisphenol A. J. Hazard. Mater. 2020, 382, 121121. [Google Scholar] [CrossRef]
- Pujar, M.S.; Hunagund, S.M.; Barretto, D.A.; Desai, V.R.; Patil, S.; Vootla, S.K.; Sidarai, A.H. Synthesis of cerium-oxide NPs and their surface morphology effect on biological activities. Bull. Mater. Sci. 2020, 43. [Google Scholar] [CrossRef]
- Nourmohammadi, E.; Khoshdel-Sarkarizi, H.; Nedaeinia, R.; Darroudi, M.; Kazemi Oskuee, R. Cerium oxide nanoparticles: A promising tool for the treatment of fibrosarcoma in-vivo. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 109, 110533. [Google Scholar] [CrossRef]
- Albert, E.L.; Che Abdullah, C.A.; Shiroshaki, Y. Synthesis and characterization of graphene oxide functionalized with magnetic nanoparticle via simple emulsion method. Results Phys. 2018, 11, 944–950. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef]
- Pant, B.; Saud, P.S.; Park, M.; Park, S.-J.; Kim, H.-Y. General one-pot strategy to prepare Ag–TiO2 decorated reduced graphene oxide nanocomposites for chemical and biological disinfectant. J. Alloys Compd. 2016, 671, 51–59. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Park, S.-J.; Kim, H.-Y. One-pot synthesis of CdS sensitized TiO2 decorated reduced graphene oxide nanosheets for the hydrolysis of ammonia-borane and the effective removal of organic pollutant from water. Ceram. Int. 2016, 42, 15247–15252. [Google Scholar] [CrossRef]
- Mahmoodi, N.M. Synthesis of core-shell magnetic adsorbent nanoparticle and selectivity analysis for binary system dye removal. J. Ind. Eng. Chem. 2014, 20, 2050–2058. [Google Scholar] [CrossRef]
- Garg, N.; Bera, S.; Rastogi, L.; Ballal, A.; Balaramakrishna, M.V. Synthesis and characterization of L-asparagine stabilised gold nanoparticles: Catalyst for degradation of organic dyes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 232, 118126. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Zhou, Q. Action and mechanism of semiconductor photocatalysis on degradation of organic pollutants in water treatment: A review. Environ. Nanotechnol. Monit. Manag. 2019, 12, 100255. [Google Scholar] [CrossRef]
- Mohammadi, P.; Sheibani, H. Green synthesis of Fe3O4@SiO2-Ag magnetic nanocatalyst using safflower extract and its application as recoverable catalyst for reduction of dye pollutants in water. Appl. Organomet. Chem. 2018, 32, e4249. [Google Scholar] [CrossRef]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Cao, X.; Gu, Y.; Tian, H.; Fang, Y.; Johnson, D.; Ren, Z.; Chen, C.; Huang, Y. Microemulsion synthesis of ms/tz-BiVO4 composites: The effect of pH on crystal structure and photocatalytic performance. Ceram. Int. 2020, 46, 20788–20797. [Google Scholar] [CrossRef]
- Moreno-Román, E.J.; Cruz-López, A.; García-Gómez, C.; Zanella, R.; Suárez-Vázquez, S.I. Evaluation of the catalytic oxidation of soot by CeOX-LaMnO3 at different O2 pressures synthesized by ultrasonic-assisted hydrothermal method. Environ. Sci. Pollut. Res. Int. 2020, 27, 15475–15487. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Liu, J.; Sun, D.D.; Ng, W. Graphene oxide-CdS composite with high photocatalytic degradation and disinfection activities under visible light irradiation. J. Hazard. Mater. 2013, 250–251, 412–420. [Google Scholar] [CrossRef]
- Yang, R.; He, J.; Niu, M.; Fan, Y.; Zhu, R. The photocatalytic activity of GO-modified BiVO4 for the degradation of phenol under visible light irradiation. Chem. Phys. Lett. 2019, 735, 136770. [Google Scholar] [CrossRef]
- Xiong, S.; Wu, T.; Fan, Z.; Zhao, D.; Du, M.; Xu, X. Preparation of a Leaf-Like BiVO4 Reduced Graphene Oxide Composite and Its Photocatalytic Activity. J. Nanomater. 2017, 2017, 3475248. [Google Scholar] [CrossRef]
- Dhandapani, P.; AlSalhi, M.S.; Karthick, R.; Chen, F.; Devanesan, S.; Kim, W.; Rajasekar, A.; Ahmed, M.; Aljaafreh, M.J.; Muhammad, A. Biological mediated synthesis of RGO-ZnO composites with enhanced photocatalytic and antibacterial activity. J. Hazard. Mater. 2020, 124661. [Google Scholar] [CrossRef]
- Ran, R.; McEvoy, J.G.; Zhang, Z. Synthesis and Optimization of Visible Light Active BiVO4 Photocatalysts for the Degradation of RhB. Int. J. Photoenergy 2015. [Google Scholar] [CrossRef]
- Shandilya, P.; Mittal, D.; Soni, M.; Raizada, P.; Hosseini-Bandegharaei, A.; Saini, A.K.; Singh, P. Fabrication of fluorine doped graphene and SmVO4 based dispersed and adsorptive photocatalyst for abatement of phenolic compounds from water and bacterial disinfection. J. Clean. Prod. 2018, 203, 386–399. [Google Scholar] [CrossRef]
- Pan, X.; Zhao, Y.; Liu, S.; Korzeniewski, C.L.; Wang, S.; Fan, Z. Comparing graphene-TiO2; nanowire and graphene-TiO2 nanoparticle composite photocatalysts. ACS Appl. Mater. Interfaces 2012, 4, 3944–3950. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, P.; Ling, M.; Li, S.; Liu, P.; Zhao, H.; Zhang, S. Photocatalytic Synthesis of TiO2 and Reduced Graphene Oxide Nanocomposite for Lithium Ion Battery. ACS Appl. Mater. Interfaces 2012, 4, 3636–3642. [Google Scholar] [CrossRef] [PubMed]
- Khavar, A.H.; Moussavi, G.; Mahjou, A.R. The preparation of TiO2@rGO nanocomposite efficiently activated with UVA/LED and H2O2 for high rate oxidation of acetaminophen: Catalyst characterization and acetaminophen degradation and mineralization. Appl. Surf. Sci. 2018, 440, 963–973. [Google Scholar] [CrossRef]
- Bell, N.J.; Ng, Y.H.; Du, A.; Coster, H.; Smith, S.C.; Amal, R. Understanding the Enhancement in Photoelectrochemical Properties of Photocatalytically Prepared TiO2-Reduced Graphene Oxide Composite. J. Phys. Chem. C 2011, 115, 6004–6009. [Google Scholar] [CrossRef]
- Pant, B.; Pokharel, P.; Tiwari, A.P.; Saud, P.S.; Park, M.; Ghouri, Z.K.; Choi, S.; Park, S.-J.; Kim, H.-Y. Characterization and antibacterial properties of aminophenol grafted and Ag NPs decorated graphene nanocomposites. Ceram. Int. 2015, 41, 5656–5662. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, K. Fabrication of Ce/N co-doped TiO2/diatomite granule catalyst and its improved visible-light-driven photoactivity. J. Hazard. Mater. 2017, 324, 139–150. [Google Scholar] [CrossRef]
- Nogueira, A.E.; Silva, G.T.S.T.; Oliveira, J.A.; Lopes, O.F.; Torres, J.A.; Carmo, M.; Ribeiro, C. CuO Decoration Controls Nb2O5 Photocatalyst Selectivity in CO2 Reduction. ACS Appl. Energy Mater. 2020, 3, 7629–7636. [Google Scholar] [CrossRef]
- Torres-Luna, J.A.; Sanabria, N.R.; Carriazo, J.G. Powders of iron(III)-doped titanium dioxide obtained by direct way from a natural ilmenite. Powder Technol. 2016, 302, 254–260. [Google Scholar] [CrossRef]
- Yan, W.; Hoekman, S.K.; Broch, A.; Coronella, C.J. Effect of Hydrothermal Carbonization Reaction Parameters on the properties of hydrochar and pellets. Environ. Prog. Sustain. Energy 2014, 33, 676–680. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, W.; Mao, H.; Lu, Y.; Lu, J.; Huang, J.; Ye, Z.; Lu, B. Electrostatic Self-Assembly of BiVO4–Reduced Graphene Oxide Nanocomposites for Highly Efficient Visible Light Photocatalytic Activities. ACS Appl. Mater. Interfaces 2014, 6, 12698–12706. [Google Scholar] [CrossRef]
- Zheng, Y.M.; Li, N.; De Zhang, W. Preparation of nanostructured microspheres of Zn-Mg-Al layered double hydroxides with high adsorption property. Colloids Surf. A Phys. Eng. Asp. 2012, 415, 195–201. [Google Scholar] [CrossRef]
- Nguyen, C.H.; Fu, C.C.; Juang, R.S. Degradation of methylene blue and methyl orange by palladium-doped TiO2 photocatalysis for water reuse: Efficiency and degradation pathways. J. Clean. Prod. 2018, 202, 413–427. [Google Scholar] [CrossRef]
- Nezamzadeh-Ejhieh, A.; Karimi-Shamsabadi, M. Comparison of photocatalytic efficiency of supported CuO onto micro and nano particles of zeolite X in photodecolorization of Methylene blue and Methyl orange aqueous mixture. Appl. Catal. A Gen. 2014, 477, 83–92. [Google Scholar] [CrossRef]
- Wei, T.Y.; Wan, C.C. Heterogeneous Photocatalytic Oxidation of Phenol with Titanium Dioxide Powders. Ind. Eng. Chem. Res. 1991, 30, 1293–1300. [Google Scholar] [CrossRef]
- Saleh, R.; Djaja, N.F. UV light photocatalytic degradation of organic dyes with Fe-doped ZnO nanoparticles. Superlattices Microstruct. 2014, 74, 217–233. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; Fan, C.; Zong, Z.; Zhang, D.; Luo, Q.; Bi, C.; Fan, Y. A novel metal–organic frameworks assembled by one angular ligand and 5-aminoisophthalic acid: Synthesis, structure, electrochemical and photocatalytic properties. Polyhedron 2019, 168, 21–27. [Google Scholar] [CrossRef]
- Rauf, M.A.; Meetani, M.A.; Hisaindee, S. An overview on the photocatalytic degradation of azo dyes in the presence of TiO2 doped with selective transition metals. Desalination 2011, 276, 13–27. [Google Scholar] [CrossRef]
- Sun, J.; Qiao, L.; Sun, S.; Wang, G. Photocatalytic degradation of Orange G on nitrogen-doped TiO2 catalysts under visible light and sunlight irradiation. J. Hazard. Mater. 2008, 155, 312–319. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onwudiwe, D.C.; Phadi, B.M.; Oyewo, O.A. Ce2O3/BiVO4 Embedded in rGO as Photocatalyst for the Degradation of Methyl Orange under Visible Light Irradiation. J 2021, 4, 154-168. https://doi.org/10.3390/j4020013
Onwudiwe DC, Phadi BM, Oyewo OA. Ce2O3/BiVO4 Embedded in rGO as Photocatalyst for the Degradation of Methyl Orange under Visible Light Irradiation. J. 2021; 4(2):154-168. https://doi.org/10.3390/j4020013
Chicago/Turabian StyleOnwudiwe, Damian C., Boitumelo M. Phadi, and Opeyemi A. Oyewo. 2021. "Ce2O3/BiVO4 Embedded in rGO as Photocatalyst for the Degradation of Methyl Orange under Visible Light Irradiation" J 4, no. 2: 154-168. https://doi.org/10.3390/j4020013
APA StyleOnwudiwe, D. C., Phadi, B. M., & Oyewo, O. A. (2021). Ce2O3/BiVO4 Embedded in rGO as Photocatalyst for the Degradation of Methyl Orange under Visible Light Irradiation. J, 4(2), 154-168. https://doi.org/10.3390/j4020013






