Gamification Concepts to Promote and Maintain Therapy Adherence in Children with Growth Hormone Deficiency †
Abstract
:1. Introduction
2. Overview of Gamification Strategies for Health
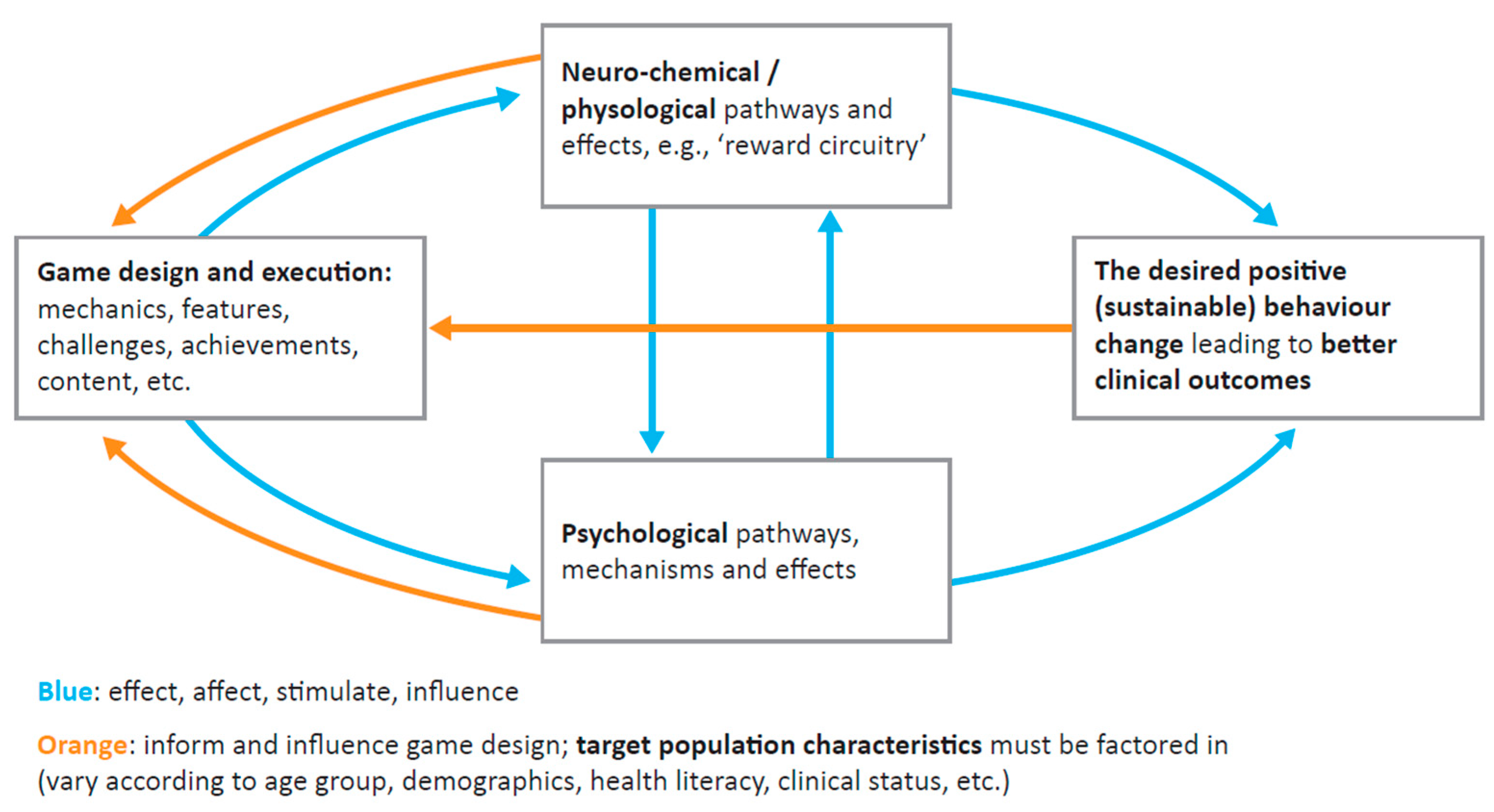
3. How Could Gamification Contribute to Improved Adherence to GH Therapy?
4. Limitations of mHealth and Gamification
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| CI | confidence interval |
| CMHD | common mental health disorders |
| DMN | default mode network |
| fMRI | functional magnetic resonance imaging |
| GH | growth hormone |
| mHealth | mobile health |
| sMRI | structural magnetic resonance imaging |
| VR | virtual reality |
References
- Stanley, T. Diagnosis of growth hormone deficiency in childhood. Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 47–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemp, S.; Pediatric Growth Hormone Deficiency. Medscape. Available online: http://emedicine.medscape.com/article/923688-overview (accessed on 5 January 2017).
- Fisher, B.G.; Acerini, C.L. Understanding the growth hormone therapy adherence paradigm: A systematic review. Horm. Res. Paediatr. 2013, 79, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Hughes, I.P.; Choong, C.; Rath, S.; Atkinson, H.; Cotterill, A.; Cutfield, W.; Hofman, P.; Harris, M. Early cessation and non-response are important and possibly related problems in growth hormone therapy: An OZGROW analysis. Growth Horm. IGF Res. 2016, 29, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Cutfield, W.S.; Derraik, J.G.; Gunn, A.J.; Reid, K.; Delany, T.; Robinson, E.; Hofman, P.L. Non-compliance with growth hormone treatment in children is common and impairs linear growth. PLoS ONE 2011, 6, e16223. [Google Scholar] [CrossRef] [PubMed]
- Growth Hormone Research Society. Consensus guidelines for the diagnosis and treatment of Growth Hormone (GH) deficiency in childhood and adolescence: Summary statement of the GH Research Society. J. Clin. Endocrinol. Metab. 2000, 85, 3990–3993. [Google Scholar]
- Francischinelli, A.G.B.; Almeida, F.D.A.; Fernandes, D.M.S.O. Routine use of therapeutic play in the care of hospitalized children: Nurses’ perceptions. Acta Paul. Enferm. 2012, 25, 18–23. [Google Scholar] [CrossRef]
- Li, W.; Chung, J.O.; Ho, K.Y.; Kwok, B.M.C. Play interventions to reduce anxiety and negative emotions in hospitalized children. BMC Pediatr. 2016, 16, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- GameTrack Digest: Quarter1 2016. Available online: http://www.isfe.eu/sites/isfe.eu/files/attachments/gametrack_european_summary_data_2016_q1.pdf (accessed on 5 January 2017).
- Kabali, H.K.; Irigoyen, M.M.; Nunez-Davis, R.; Budacki, J.G.; Mohanty, S.H.; Leister, K.P.; Bonner, R.L. Exposure and use of mobile media devices by young children. Pediatrics 2015, 136, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Holtz, B.; Lauckner, C. Diabetes management via mobile phones: A systematic review. Telemed. J. E Health 2012, 18, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Sardi, L.; Idri, A.; Fernández-Alemán, J.L. A systematic review of gamification in e-Health. J. Biomed. Inform. 2017, 71, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Deterding, S.; Miguel, S.; Nacke, L.; O’Hara, K.; Dixon, D. Gamification: Using game-design elements in non-gaming contexts. In Proceedings of the CHI‘11 Extended Abstracts on Human Factors in Computing Systems, Vancouver, BC, Canada, 7–12 May 2011; pp. 2425–2428. [Google Scholar]
- Kamel Boulos, M.N.; Gammon, S.; Dixon, M.C.; MacRury, S.M.; Fergusson, M.J.; Miranda, R.F.; Mourinho, B.T.; Yang, S.P. Digital games for type 1 and type 2 diabetes: Underpinning theory with three illustrative examples. JMIR Serious Games 2015, 3, e3. [Google Scholar] [CrossRef] [PubMed]
- Charlier, N.; Zupancic, N.; Fieuws, S.; Denhaerynck, K.; Zaman, B.; Moons, P. Serious games for improving knowledge and self-management in young people with chronic conditions: A systematic review and meta-analysis. J. Am. Med. Inform. Assoc. 2015, 23, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Stokes, B. Video games have changed: Time to consider “serious games”. Dev. Educ. J. 2005, 11, 108. [Google Scholar]
- Miller, A.S.; Cafazzo, J.A.; Seto, E. A game plan: Gamification design principles in mHealth applications for chronic disease management. Health Inform. J. 2014, 22, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Von Bargen, T.; Zientz, C.; Haux, R. Gamification for mHealth—A review of playful mobile healthcare. Stud. Health Technol. Inform. 2014, 202, 225–228. [Google Scholar] [PubMed]
- Cugelman, B. Gamification: What it is and why it matters to digital health behavior change developers. JMIR Serious Games 2013, 1, e3. [Google Scholar] [CrossRef] [PubMed]
- Koepp, M.J.; Gunn, R.N.; Lawrence, A.D.; Cunningham, V.J.; Dagher, A.; Jones, T.; Brooks, D.J.; Bench, C.J.; Grasby, P.M. Evidence for striatal dopamine release during a video game. Nature 1998, 393, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Votinov, M.; Pripfl, J.; Windischberger, C.; Sailer, U.; Lamm, C. Better you lose than I do: Neural networks involved in winning and losing in a real time strictly competitive game. Sci. Rep. 2015, 5, 11017. [Google Scholar] [CrossRef] [PubMed]
- Beck, S.M.; Locke, H.S.; Savine, A.C.; Jimura, K.; Braver, T.S. Primary and secondary rewards differentially modulate neural activity dynamics during working memory. PLoS ONE 2010, 5, e9251. [Google Scholar] [CrossRef] [PubMed]
- Howard-Jones, P.A.; Jay, T.; Mason, A.; Jones, H. Gamification of learning deactivates the default mode network. Front. Psychol. 2016, 6, 1891. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.M.; Deci, E.L. Intrinsic and extrinsic motivations: Classic definitions and new directions. Contemp. Educ. Psychol. 2000, 25, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, S. A user-centered theoretical framework for meaningful gamification. Presented at the Games + Learning + Society 8.0, Madison, WI, USA, 13–15 June 2012. [Google Scholar]
- Chou, Y. Octalysis: Complete Gamification Framework. Available online: http://www.yukaichou.com/ (accessed on 9 January 2017).
- Larkin, M. Want to Create an Educational Game? Here’s What You Need to Know? Available online: https://www.elsevier.com/connect/story/product-development/apps-and-technology/want-to-create-an-educational-game-heres-what-you-need-to-know (accessed on 9 January 2017).
- Haverkamp, F.; Johansson, L.; Dumas, H.; Langham, S.; Tauber, M.; Veimo, D.; Chiarelli, F. Observations of nonadherence to recombinant human growth hormone therapy in clinical practice. Clin. Ther. 2008, 30, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Andela, C.D.; Biermasz, N.R.; Kaptein, A.A.; Pereira, A.M.; Tiemensma, J. More concerns and stronger beliefs about the necessity of medication in patients with acromegaly are associated with negative illness perceptions and impairment in quality of life. Growth Horm. IGF Res. 2015, 25, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Ayogo Health Inc. Monster Manor. Google Play/iTunes. Available online: https://play.google.com/store/apps/details?id=com.ayogohealth.monstermanor or https://itunes.apple.com/gb/app/monstermanor/id719080981?mt=8 (accessed on 9 January 2017).
- Oxford Academic Health Science Network. Child’s Play: Encouraging 5–10 Years Olds with Type 1 Diabetes to Log Blood Glucose Readings. Available online: http://www.oxfordahsn.org/wp-content/uploads/2015/07/monster_manor_website.pdf (accessed on 9 January 2017).
- Garde, A.; Umedaly, A.; Abulnaga, S.M.; Junker, A.; Chanoine, J.P.; Johnson, M.; Ansermino, J.M.; Dumont, G.A. Evaluation of a novel mobile exergame in a school-based environment. Cyberpsychol. Behav. Soc. Netw. 2016, 19, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Timpel, P.; Cesena, F.H.Y.; da Silva Costa, C.; Soldatelli, M.D.; Gois, E., Jr.; Castrillon, E.; Díaz, L.J.J.; Repetto, G.M.; Hagos, F.; Castillo Yermenos, R.E.; et al. Efficacy of gamification-based smartphone application for weight loss in overweight and obese adolescents: Study protocol for a phase II randomized controlled trial. Ther. Adv. Endocrinol. Metab. 2018, 9, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Abe, S.; Okumura, A.; Mukae, T.; Nakazawa, T.; Niijima, S.I.; Yamashiro, Y.; Shimizu, T. Depressive tendency in children with growth hormone deficiency. J. Paediatr. Child Health 2009, 45, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Onenli-Mungan, N.; Topaloglu, K.; Avci, A.; Ozer, G. Short stature due to growth hormone neurosecretory dysfunction in a child with major depressive disorder. Indian Pediatr. 2004, 41, 83–86. [Google Scholar] [PubMed]
- Quek, Y.H.; Tam, W.W.S.; Zhang, M.W.B.; Ho, R.C.M. Exploring the association between childhood and adolescent obesity and depression: A meta-analysis. Obes. Rev. 2017, 18, 742–754. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.W.B.; Ying, J.B.; Song, G.; Ho, R.C.M. A review of gamification approaches in commercial cognitive bias modification gaming applications. Technol. Health Care 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Van Dongen, N.; Kaptein, A.A. Parents’ views on growth hormone treatment for their children: Psychosocial issues. Patient Prefer. Adher. 2012, 6, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Schiff, W.B.; Holtz, K.D.; Peterson, N.; Rakusan, T. Effect of an intervention to reduce procedural pain and distress for children with HIV infection. J. Pediatr. Psychol. 2001, 26, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Pham, Q.; Khatib, Y.; Stansfeld, S.; Fox, S.; Green, T. Feasibility and efficacy of an mHealth Game for managing anxiety: “Flowy” randomized controlled pilot trial and design evaluation. Games Health J. 2016, 5, 50–67. [Google Scholar] [CrossRef] [PubMed]
- Playlab London. Google Play. 1 December 2015. Flowy Beta. Available online: https://play.google.com/store/apps/details?id=com.playlab.flowyfree&hl=en_GB (accessed on 9 January 2017).
- Jones, T.; Moore, T.; Choo, J. The impact of virtual reality on chronic pain. PLoS ONE 2016, 11, e0167523. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, K.; Ahn, S.J.; Moore, J.; Brown, S.; Robertson, T.P.; Marable, A.; Basu, A. Mixed reality virtual pets to reduce childhood obesity. IEEE Trans. Vis. Comput. Graph. 2014, 20, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.J.; Johnsen, K.; Moore, J.; Brown, S.; Biersmith, M.; Ball, C. Using virtual pets to increase fruit and vegetable consumption in children: A technology-assisted social cognitive theory approach. Cyberpsychol. Behav. Soc. Netw. 2016, 19, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.J.; Johnsen, K.; Robertson, T.; Moore, J.; Brown, S.; Marable, A.; Basu, A. Using virtual pets to promote physical activity in children: An application of the youth physical activity promotion model. J. Health Commun. 2015, 20, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, S.; Heydari, Z.; Qorbani, M.; Radfar, M. Adherence to growth hormone therapy in children and its potential barriers. J. Pediatr. Endocrinol. Metab. 2018, 31, 13–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doak, L.G.; Doak, C.C. (Eds.) Principles for Clear Health Communication, 2nd ed.; Pfizer: New York, NY, USA, 2004; Available online: https://www.pfizer.com/files/health/PfizerPrinciples.pdf (accessed on 23 August 2018).
| Intervention * | ||||
|---|---|---|---|---|
| Outcome | Baseline | Week 2 | Week 4 | p † |
| Panic (PDSS-SR) | ||||
| Mean (SD) | 16.90 (5.24) | 15.42 (5.42) | 15.35 (5.78) | 0.011 |
| n | 31 | 32 | 31 | |
| Hyperventilation (Nijmegen) | ||||
| Mean (SD) | 24.45 (9.79) | 22.45 (10.61) | 21.74 (11.27) | 0.016 |
| n | 31 | 32 | 31 | |
| Quality of life (Q-LES-Q-SF) | ||||
| Mean (SD) | 32.11 (7.32) | 35.67 (5.08) | 35.89 (7.71) | 0.005 |
| n | 31 | 19 | 18 | |
| Educate |
Providing a source of information and guidance that is understandable and that resonates with the child:
|
| Reduce anxiety and stress over administration |
Providing mechanisms to distract and relax:
|
| Feedback |
Providing praise and feedback to the child, and information for healthcare professionals to better guide management:
|
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radovick, S.; Hershkovitz, E.; Kalisvaart, A.; Koning, M.; Paridaens, K.; Kamel Boulos, M.N. Gamification Concepts to Promote and Maintain Therapy Adherence in Children with Growth Hormone Deficiency. J 2018, 1, 71-80. https://doi.org/10.3390/j1010008
Radovick S, Hershkovitz E, Kalisvaart A, Koning M, Paridaens K, Kamel Boulos MN. Gamification Concepts to Promote and Maintain Therapy Adherence in Children with Growth Hormone Deficiency. J. 2018; 1(1):71-80. https://doi.org/10.3390/j1010008
Chicago/Turabian StyleRadovick, Sally, Eli Hershkovitz, Aline Kalisvaart, Marco Koning, Kristine Paridaens, and Maged N. Kamel Boulos. 2018. "Gamification Concepts to Promote and Maintain Therapy Adherence in Children with Growth Hormone Deficiency" J 1, no. 1: 71-80. https://doi.org/10.3390/j1010008
APA StyleRadovick, S., Hershkovitz, E., Kalisvaart, A., Koning, M., Paridaens, K., & Kamel Boulos, M. N. (2018). Gamification Concepts to Promote and Maintain Therapy Adherence in Children with Growth Hormone Deficiency. J, 1(1), 71-80. https://doi.org/10.3390/j1010008






