Arabinoxylans from the Distillers Grains and Solubles Co-Products of Ethanol Production: Extraction, Characterisation and Hydrolysis to Oligosaccharides
Highlights
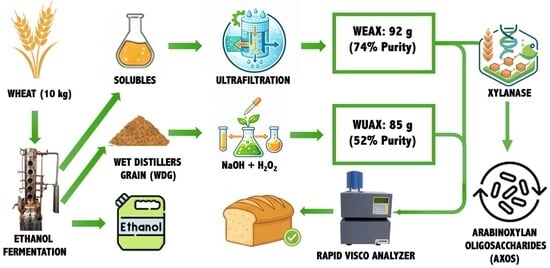
- Extraction of arabinoxylans (AX) from the Wet Distillers Grains (WDG) and Solubles can be integrated within ethanol biorefineries to create new products and revenue streams.
- Arabinoxylan oligosaccharides (AXOS), which have prebiotic functionality for animal feed and human health applications, can also be produced via enzymatic conversion of the water-extractable AX (WEAX) with xylanases.
- WEAX from the Solubles fraction was more readily extracted than from the WDG fraction, and was a more suitable feedstock for hydrolysis to oligosaccharides.
- Arabinoxylans co-produced within biorefineries from a range of agricultural and biorefinery wastes could have a global market of > £1 billion per annum, significantly impacting biorefinery economics and enhancing their commercial viability and contributions to sustainability.
- Extracting AX from Solubles and converting to AXOS is easier than from WDG, and offers a pragmatic route to producing initial AX products from a biorefinery, from which a wider portfolio of products could grow.
Abstract
1. Introduction
2. Materials and Methods
2.1. Pilot-Scale Production of Ethanol, WDG and Solubles
2.2. AX Extraction
2.3. Characterisation of AX Extracts
2.4. Production of Arabinoxylan Oligosaccharides
2.5. Effects of AX on Pasting Viscosity Characteristics of Starch
3. Results and Discussion
3.1. Yields and Mass Balances Following Ethanolic Fermentation
3.2. AX Extraction and Characterisation
3.3. Production of XOS
3.4. Effect of AX on Pasting Viscosity Characteristics of Flour
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AX | Arabinoxylans |
| AXOS | Arabinoxylan oligosaccharides |
| XOS | Xylo-oligosaccharides |
| DDGS | Distillers Dried Grains with Solubles |
| WDG | Wet Distillers Grain |
| WEAX | Water-extractable arabinoxylan |
| WUAX | Water-unextractable arabinoxylan |
| RVA | Rapid Visco Analyser |
| BSG | Brewer’s Spent Grain |
| SEC-MALLS | Size exclusion chromatography with multi-angle laser light scattering |
| HPAEC-PAD | High performance anion exchange chromatography with pulsed amperometric detection |
| HPLC | High performance liquid chromatography |
References
- Guerreiro, C.D.A.; Andrade, L.A.D.; Fernández-Lainez, C.; Fraga, L.N.; López-Velázquez, G.; Marques, T.M.; Prado, S.B.R.; Brummer, R.J.; Nascimento, J.R.O.; Castro-Alves, V. Bioactive Arabinoxylan Oligomers via Colonic Fermentation and Enzymatic Catalysis: Evidence of Interaction with Toll-like Receptors from in Vitro, in Silico and Functional Analysis. Carbohydr. Polym. 2025, 352, 123175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hou, Y.; Liu, T.; Li, Z.; Luo, S.; Liu, C.; Zhang, G.; Chen, T. Strategic Alteration of Arabinoxylan Feruloylation Enables Selective Shaping of the Human Gut Microbiota. Food Hydrocoll. 2025, 160, 110818. [Google Scholar] [CrossRef]
- Huang, Z.; Yang, X.; Liu, M.; Yin, L.; Jia, X. Effect of Glycoside Hydrolase-Mediated Wheat Arabinoxylan Hydrolysate on Gut Microbiota and Metabolite Profiles. Carbohydr. Polym. 2025, 351, 123064. [Google Scholar] [CrossRef] [PubMed]
- Naibaho, J.; Setiawan, R.D.; Korzeniowska, M. Biological Properties of Bioactive Compounds from Brewers’ Spent Grain: Current Trends, Challenges, and Perspectives. Curr. Opin. Food Sci. 2025, 62, 101268. [Google Scholar] [CrossRef]
- Kruk, M.; Lalowski, P.; Płecha, M.; Ponder, A.; Rudzka, A.; Zielińska, D.; Trząskowska, M. Prebiotic Potential of Spent Brewery Grain—In Vitro Study. Food Chem. 2025, 463, 141254. [Google Scholar] [CrossRef]
- Shrivastava, M.; Goyal, A. Unveiling the Structural and Functional Perspectives of a Bifunctional α-l-Arabinofuranosidase/Endo-β-1,4-Xylanase (BoGH43_35) from Bacteroides Ovatus. Arch. Biochem. Biophys. 2025, 764, 110232. [Google Scholar] [CrossRef]
- Kulathunga, J.; Islam, S. Wheat Arabinoxylans: Insight into Structure-Function Relationships. Carbohydr. Polym. 2025, 348, 122933. [Google Scholar] [CrossRef]
- Rezette, L.; Kansou, K.; Della Valle, G.; Le Gall, S.; Saulnier, L. The Role of Wheat Flour Minor Components in Predicting Water Absorption. Food Chem. 2025, 463, 141232. [Google Scholar] [CrossRef]
- Bilal, M.; Li, D.; Xie, C.; Yang, R.; Gu, Z.; Jiang, D.; Xu, X.; Wang, P. Recent Advances of Wheat Bran Arabinoxylan Exploitation as the Functional Dough Additive. Food Chem. 2025, 463, 141146. [Google Scholar] [CrossRef]
- Wan, J.; Wei, Y.; Zhang, Y.; Lan, H.; Li, X.; Wang, Y.; Luo, D.; Feng, J.; Bai, Z.; Guo, J. Innovative Use of Wastewater-Extracted Arabinoxylans: Elevating Steamed Bread Texture and Structure. Food Chem. 2025, 466, 142120. [Google Scholar] [CrossRef]
- Falade, E.O.; Kouamé, K.J.E.P.; Zhu, Y.; Zheng, Y.; Ye, X. A Review: Examining the Effects of Modern Extraction Techniques on Functional and Structural Properties of Cellulose and Hemicellulose in Brewer’s Spent Grain Dietary Fiber. Carbohydr. Polym. 2025, 348, 122883. [Google Scholar] [CrossRef] [PubMed]
- He, H.J.; Qiao, J.; Liu, Y.; Guo, Q.; Ou, X.; Wang, X. Isolation, Structural, Functional, and Bioactive Properties of Cereal Arabinoxylan—A Critical Review. J. Agric. Food Chem. 2021, 69, 15437–15457. [Google Scholar] [CrossRef] [PubMed]
- Zannini, E.; Bravo Núñez, Á.; Sahin, A.W.; Arendt, E.K.; Aiello, F.; Carullo, G.; Zannini, E.; Bravo Núñez, Á.; Sahin, A.W.; Arendt, E.K. Arabinoxylans as Functional Food Ingredients: A Review. Foods 2022, 11, 1026. [Google Scholar] [CrossRef] [PubMed]
- Pietiäinen, S.; Jimenez-Quero, A.; Moldin, A.; Ström, A.; Katina, K.; Langton, M. Feruloylation and Hydrolysis of Arabinoxylan Extracted from Wheat Bran: Effect on Bread Quality and Shelf-Life. J. Cereal Sci. 2024, 117, 103920. [Google Scholar] [CrossRef]
- Solomou, K.; Alyassin, M.; Angelis-Dimakis, A.; Campbell, G.M. Arabinoxylans: A New Class of Food Ingredients Arising from Synergies with Biorefining, and Illustrating the Nature of Biorefinery Engineering. Food Bioprod. Process. 2022, 132, 83–98. [Google Scholar] [CrossRef]
- Pang, J.; Zhang, Y.; Song, X.; Tong, X.; Wu, S.; Hu, Y.; Zhong, Y.; Qiao, Y. Molecular Characterization, Rheological Properties, and Prebiotic Properties of Arabinoxylans from Waxy and Sweet Corn Cobs. Cereal Chem. 2024, 101, 493–507. [Google Scholar] [CrossRef]
- Pérez-Flores, J.G.; García-Curiel, L.; Pérez-Escalante, E.; Contreras-López, E.; Olloqui, E.J. Arabinoxylans Matrixes as a Potential Material for Drug Delivery Systems Development—A Bibliometric Analysis and Literature Review. Heliyon 2024, 10, e25445. [Google Scholar] [CrossRef]
- UK Flour Millers Statistics. Available online: https://www.ukflourmillers.org/statistics (accessed on 19 February 2025).
- Statista Global Sugar Cane Production 2023|Statista. Available online: https://www.statista.com/statistics/249604/sugar-cane-production-worldwide/ (accessed on 23 October 2025).
- Mordor Intelligence Distiller’s Dried Grains with Solubles (DDGS) Feed Market Analysis|Industry Report, Size & Forecast. Available online: https://www.mordorintelligence.com/industry-reports/ddgs-feed-market (accessed on 23 October 2025).
- Liu, K. Particle Size Distribution of Distillers Dried Grains with Solubles (DDGS) and Relationships to Compositional and Color Properties. Bioresour. Technol. 2008, 99, 8421–8428. [Google Scholar] [CrossRef]
- Kim, Y.; Mosier, N.S.; Hendrickson, R.; Ezeji, T.; Blaschek, H.; Dien, B.; Cotta, M.; Dale, B.; Ladisch, M.R. Composition of Corn Dry-Grind Ethanol by-Products: DDGS, Wet Cake, and Thin Stillage. Bioresour. Technol. 2008, 99, 5165–5176. [Google Scholar] [CrossRef]
- Cromwell, G.L.; Herkelman, K.L.; Stahly, T.S. Physical, Chemical, and Nutritional Characteristics of Distillers Dried Grains with Solubles for Chicks and Pigs. J. Anim. Sci. 1993, 71, 679–686. [Google Scholar] [CrossRef][Green Version]
- Immerzeel, P.; Falck, P.; Galbe, M.; Adlercreutz, P.; Nordberg Karlsson, E.; Stålbrand, H. Extraction of Water-Soluble Xylan from Wheat Bran and Utilization of Enzymatically Produced Xylooligosaccharides by Lactobacillus, Bifidobacterium and Weissella Spp. LWT-Food Sci. Technol. 2014, 56, 321–327. [Google Scholar] [CrossRef]
- Mendis, M.; Simsek, S. Arabinoxylans and Human Health. Food Hydrocoll. 2014, 42, 239–243. [Google Scholar] [CrossRef]
- Broekaert, W.F.; Courtin, C.M.; Verbeke, K.; Van de Wiele, T.; Verstraete, W.; Delcour, J.A. Prebiotic and Other Health-Related Effects of Cereal-Derived Arabinoxylans, Arabinoxylan-Oligosaccharides, and Xylooligosaccharides. Crit. Rev. Food Sci. Nutr. 2011, 51, 178–194. [Google Scholar] [CrossRef] [PubMed]
- Van Craeyveld, V.; Swennen, K.; Dornez, E.; Van de Wiele, T.; Marzorati, M.; Verstraete, W.; Delaedt, Y.; Onagbesan, O.; Decuypere, E.; Buyse, J.; et al. Structurally Different Wheat-Derived Arabinoxylooligosaccharides Have Different Prebiotic and Fermentation Properties in Rats. J. Nutr. 2008, 138, 2348–2355. [Google Scholar] [CrossRef]
- Šimić, A.; González-Ortiz, G.; Mansbridge, S.C.; Rose, S.P.; Bedford, M.R.; Yovchev, D.; Pirgozliev, V.R. Broiler Chicken Response to Xylanase and Fermentable Xylooligosaccharide Supplementation. Poult. Sci. 2023, 102, 103000. [Google Scholar] [CrossRef]
- Davies, C.; González-Ortiz, G.; Rinttilä, T.; Apajalahti, J.; Alyassin, M.; Bedford, M.R. Stimbiotic Supplementation and Xylose-Rich Carbohydrates Modulate Broiler’s Capacity to Ferment Fibre. Front. Microbiol. 2023, 14, 1301727. [Google Scholar] [CrossRef]
- Nadi, P. Xylooligosaccharide Market Size, Share, Trends, Forecast|2032. Available online: https://www.marketresearchfuture.com/reports/xylooligosaccharide-market-12084 (accessed on 19 February 2025).
- González-Ortiz, G.; dos Santos, T.T.; Bedford, M.R. Evaluation of Xylanase and a Fermentable Xylo-Oligosaccharide on Performance and Ileal Digestibility of Broiler Chickens Fed Energy and Amino Acid Deficient Diets. Anim. Nutr. 2021, 7, 488–495. [Google Scholar] [CrossRef]
- Cho, H.M.; González-Ortiz, G.; Melo-Durán, D.; Heo, J.M.; Cordero, G.; Bedford, M.R.; Kim, J.C. Stimbiotic Supplementation Improved Performance and Reduced Inflammatory Response via Stimulating Fiber Fermenting Microbiome in Weaner Pigs Housed in a Poor Sanitary Environment and Fed an Antibiotic-Free Low Zinc Oxide Diet. PLoS ONE 2020, 15, e0240264. [Google Scholar] [CrossRef]
- González-Ortiz, G.; Bedford, M.R.; Bach Knudsen, K.-E.; Courtin, C.M.; Classen, H.L. The Value of Fibre: Engaging the Second Brain for Animal Nutrition; Wageningen Academic Publishers: Leiden, The Netherlands, 2019; ISBN 9086863426. [Google Scholar]
- Misailidis, N.; Campbell, G.M.; Du, C.; Sadhukhan, J.; Mustafa, M.; Mateos-Salvador, F.; Weightman, R.M. Evaluating the Feasibility of Commercial Arabinoxylan Production in the Context of a Wheat Biorefinery Principally Producing Ethanol: Part 2. Process Simulation and Economic Analysis. Chem. Eng. Res. Des. 2009, 87, 1239–1250. [Google Scholar] [CrossRef]
- Martinez-Hernandez, E.; Tibessart, A.; Campbell, G.M. Conceptual Design of Integrated Production of Arabinoxylan Products Using Bioethanol Pinch Analysis. Food Bioprod. Process. 2018, 112, 1–8. [Google Scholar] [CrossRef]
- Hollmann, J.; Lindhauer, M. Pilot-Scale Isolation of Glucuronoarabinoxylans from Wheat Bran. Carbohydr. Polym. 2005, 59, 225–230. [Google Scholar] [CrossRef]
- Maes, C.; Delcour, J.A. Structural Characterisation of Water-Extractable and Water-Unextractable Arabinoxylans in Wheat Bran. J. Cereal Sci. 2002, 35, 315–326. [Google Scholar] [CrossRef]
- Swennen, K.; Courtin, C.M.; Delcour, J.A. Non-Digestible Oligosaccharides with Prebiotic Properties. Crit. Rev. Food Sci. Nutr. 2006, 46, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Swennen, K.; Courtin, C.M.; Van Der Bruggen, B.; Vandecasteele, C.; Delcour, J.A. Ultrafiltration and Ethanol Precipitation for Isolation of Arabinoxylooligosaccharides with Different Structures. Carbohydr. Polym. 2005, 62, 283–292. [Google Scholar] [CrossRef]
- Peng, F.; Peng, P.; Xu, F.; Sun, R.-C. Fractional Purification and Bioconversion of Hemicelluloses. Biotechnol. Adv. 2012, 30, 879–903. [Google Scholar] [CrossRef]
- Peng, F.; Ren, J.L.; Xu, F.; Bian, J.; Peng, P.; Sun, R.C. Fractional Study of Alkali-Soluble Hemicelluloses Obtained by Graded Ethanol Precipitation from Sugar Cane Bagasse. J. Agric. Food Chem. 2010, 58, 1768–1776. [Google Scholar] [CrossRef]
- Zhang, Z.; Smith, C.; Li, W. Extraction and Modification Technology of Arabinoxylans from Cereal By-Products: A Critical Review. Food Res. Int. 2014, 65, 423–436. [Google Scholar] [CrossRef]
- Martinez-Hernandez, E.; Sadhukhan, J.; Campbell, G.M. Integration of Bioethanol as an In-Process Material in Biorefineries Using Mass Pinch Analysis. Appl. Energy 2013, 104, 517–526. [Google Scholar] [CrossRef]
- Klopfenstein, T.J.; Erickson, G.E.; Bremer, V.R. BOARD-INVITED REVIEW: Use of Distillers by-Products in the Beef Cattle Feeding Industry1. J. Anim. Sci. 2008, 86, 1223–1231. [Google Scholar] [CrossRef]
- Kingsly, A.R.P.; Ileleji, K.E.; Clementson, C.L.; Garcia, A.; Maier, D.E.; Stroshine, R.L.; Radcliff, S. The Effect of Process Variables during Drying on the Physical and Chemical Characteristics of Corn Dried Distillers Grains with Solubles (DDGS)—Plant Scale Experiments. Bioresour. Technol. 2010, 101, 193–199. [Google Scholar] [CrossRef]
- Jensen, H.G.; Björnsson, A.H. By-Products from Ethanol Production—The Forgotten Part of the Equation Possibilities and Challenges; University of Copenhagen: Copenhagen, Denmark, 2012. [Google Scholar]
- Coelho, E.; Rocha, M.A.M.; Moreira, A.S.P.; Domingues, M.R.M.; Coimbra, M.A. Revisiting the Structural Features of Arabinoxylans from Brewers’ Spent Grain. Carbohydr. Polym. 2016, 139, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Laine, C.; Kemppainen, K.; Kuutti, L.; Varhimo, A.; Asikainen, S.; Grönroos, A.; Määttänen, M.; Buchert, J.; Harlin, A. Extraction of Xylan from Wood Pulp and Brewer’s Spent Grain. Ind. Crops Prod. 2015, 70, 231–237. [Google Scholar] [CrossRef]
- Mandalari, G.; Faulds, C.B.; Sancho, A.I.; Saija, A.; Bisignano, G.; LoCurto, R.; Waldron, K.W. Fractionation and Characterisation of Arabinoxylans from Brewers’ Spent Grain and Wheat Bran. J. Cereal Sci. 2005, 42, 205–212. [Google Scholar] [CrossRef]
- Chatzifragkou, A.; Charalampopoulos, D. Distiller’s Dried Grains with Solubles (DDGS) and Intermediate Products as Starting Materials in Biorefinery Strategies. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Woodhead Publishing: Cambridge, UK, 2018; pp. 63–86. [Google Scholar] [CrossRef]
- De Vries, S.; Pustjens, A.M.; Kabel, M.A.; Salazar-Villanea, S.; Hendriks, W.H.; Gerrits, W.J.J. Processing Technologies and Cell Wall Degrading Enzymes to Improve Nutritional Value of Dried Distillers Grain with Solubles for Animal Feed: An in Vitro Digestion Study. J. Agric. Food Chem. 2013, 61, 8821–8828. [Google Scholar] [CrossRef]
- Schroeder, J.W. NDUS Extension Service; NDSU: Fargo, ND, USA, 2012; pp. 1–8. [Google Scholar]
- Pedersen, M.B.; Dalsgaard, S.; Knudsen, K.E.B.; Yu, S.; Lærke, H.N. Compositional Profile and Variation of Distillers Dried Grains with Solubles from Various Origins with Focus on Non-Starch Polysaccharides. Anim. Feed. Sci. Technol. 2014, 197, 130–141. [Google Scholar] [CrossRef]
- Kosik, O.; Powers, S.J.; Chatzifragkou, A.; Prabhakumari, P.C.; Charalampopoulos, D.; Hess, L.; Brosnan, J.; Shewry, P.R.; Lovegrove, A. Changes in the Arabinoxylan Fraction of Wheat Grain during Alcohol Production. Food Chem. 2017, 221, 1754–1762. [Google Scholar] [CrossRef]
- Alyassin, M. Arabinoxylan Prebiotics Co-Production Within Integrated Biorefineries; University of Huddersfield: Huddersfield, UK, 2019. [Google Scholar]
- Englyst, H.N.; Quigley, M.E.; Hudson, G.J.; Cummings, J.H. Determination of Dietary Fibre as Non-Starch Polysaccharides by Gas-Liquid Chromatography. Analyst 1992, 117, 1707–1714. [Google Scholar] [CrossRef]
- Cyran, M.; Courtin, C.M.; Delcour, J.A. Structural Features of Arabinoxylans Extracted with Water at Different Temperatures from Two Rye Flours of Diverse Breadmaking Quality. J. Agric. Food Chem. 2003, 51, 4404–4416. [Google Scholar] [CrossRef]
- Horwitz, W. Protein in Grains. Method 979.09. In Official Method of Analysis of AOAC International; AOAC: Rockville, MD, USA, 2000; pp. 30–34. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Ash in Biomass Laboratory Analytical Procedure (LAP) Issue Date: 7/17/2005; National Renewable Energy Laboratory: Golden, CO, USA, 2008. [Google Scholar]
- Alyassin, M.; Campbell, G.M.; Masey O’Neill, H.; Bedford, M.R. Simultaneous Determination of Cereal Monosaccharides, Xylo- and Arabinoxylo-Oligosaccharides and Uronic Acids Using HPAEC-PAD. Food Chem. 2020, 315, 126221. [Google Scholar] [CrossRef]
- Murphy, J.D.; Power, N.M. How Can We Improve the Energy Balance of Ethanol Production from Wheat? Fuel 2008, 87, 1799–1806. [Google Scholar] [CrossRef]
- Morey, R.V.; Hatfield, D.L.; Sears, R.; Haak, D.; Tiffany, D.G.; Kaliyan, N. Fuel Properties of Biomass Feed Streams at Ethanol Plants. Appl. Eng. Agric. 2009, 25, 57–64. [Google Scholar] [CrossRef]
- Barron, C.; Bar-L’Helgouac’h, C.; Champ, M.; Saulnier, L. Arabinoxylan Content and Grain Tissue Distribution Are Good Predictors of the Dietary Fibre Content and Their Nutritional Properties in Wheat Products. Food Chem. 2020, 328, 127111. [Google Scholar] [CrossRef] [PubMed]
- Liu, K. Chemical Composition of Distillers Grains, a Review. J. Agric. Food Chem. 2011, 59, 1508–1526. [Google Scholar] [CrossRef] [PubMed]
- Jarret, G.; Cozannet, P.; Martinez, J.; Dourmad, J.Y. Effect of Different Quality Wheat Dried Distiller’s Grain Solubles (DDGS) in Pig Diets on Composition of Excreta and Methane Production from Faeces and Slurry. Livest. Sci. 2011, 140, 275–282. [Google Scholar] [CrossRef]
- Chatzifragkou, A.; Prabhakumari, P.C.; Kosik, O.; Lovegrove, A.; Shewry, P.R.; Charalampopoulos, D. Extractability and Characteristics of Proteins Deriving from Wheat DDGS. Food Chem. 2016, 198, 12–19. [Google Scholar] [CrossRef]
- Saulnier, L.; Guillon, F.; Sado, P.-E.; Chateigner-Boutin, A.L.; Rouau, X. Plant Cell Wall Polysaccharides in Storage Organs: Xylans (Food Applications). Ref. Modul. Chem. Mol. Sci. Chem. Eng. 2013. [Google Scholar] [CrossRef]
- Shi, Y.; Yokoyama, T.; Akiyama, T.; Yashiro, M.; Matsumoto, Y. Degradation of Monosaccharides. Bioresources 2012, 7, 4085–4097. [Google Scholar] [CrossRef]
- Liu, D.; Tang, W.; Yin, J.Y.; Nie, S.P.; Xie, M.Y. Monosaccharide Composition Analysis of Polysaccharides from Natural Sources: Hydrolysis Condition and Detection Method Development. Food Hydrocoll. 2021, 116, 106641. [Google Scholar] [CrossRef]
- Whitfield, M.B.; Chinn, M.S.; Veal, M.W. Improvement of Acid Hydrolysis Procedures for the Composition Analysis of Herbaceous Biomass. Energy Fuels 2016, 30, 8260–8269. [Google Scholar] [CrossRef]
- Garcés, D.; Díaz, E.; Ordóñez, S. Aqueous Phase Conversion of Hexoses into 5-Hydroxymethylfurfural and Levulinic Acid in the Presence of Hydrochloric Acid: Mechanism and Kinetics. Ind. Eng. Chem. Res. 2017, 56, 5221–5230. [Google Scholar] [CrossRef]
- Nguyen, H.S.H.; Heinonen, J.; Laatikainen, M.; Sainio, T. Evolution of the Molar Mass Distribution of Oat β-Glucan during Acid Catalyzed Hydrolysis in Aqueous Solution. Chem. Eng. J. 2020, 382, 122863. [Google Scholar] [CrossRef]
- Willför, S.; Pranovich, A.; Tamminen, T.; Puls, J.; Laine, C.; Suurnäkki, A.; Saake, B.; Uotila, K.; Simolin, H.; Hemming, J.; et al. Carbohydrate Analysis of Plant Materials with Uronic Acid-Containing Polysaccharides-A Comparison between Different Hydrolysis and Subsequent Chromatographic Analytical Techniques. Ind. Crops Prod. 2009, 29, 571–580. [Google Scholar] [CrossRef]
- Dale, T.; Bedford, M.R.; King, J.; Tucker, G.A.; Brameld, J.M.; Parr, T. The Effects of Exogenous Fibrolytic Enzymes on the in Vitro Release of Xylooligosaccharides and Monosaccharides Varies across Six Varieties of Wheat. Anim. Feed Sci. Technol. 2023, 295, 115526. [Google Scholar] [CrossRef]
- Solomou, K.; Čukelj Mustač, N.; Alyassin, M.; Campbell, G.M. Using the Chopin Mixolab to Model the Effects of Arabinoxylan Ingredients on Breadmaking. Part 2: Predicting the Effects of Combinations of Different Arabinoxylans. Food Bioprod. Process. 2025, 150, 432–451. [Google Scholar] [CrossRef]
- Campbell, G.M.; Solomou, K.; O’Byrne, K.J.; Spencer, K.L. Using the Chopin Mixolab to Model the Effects of Arabinoxylan Ingredients on Breadmaking. Part 1: Modelling Combined Effects of AX and Water Adjustment on Mixolab Parameters. Food Bioprod. Process. 2025, 150, 416–431. [Google Scholar] [CrossRef]
- Courtin, C.M.; Delcour, J.A. Arabinoxylans and Endoxylanases in Wheat Flour Bread-Making. J. Cereal Sci. 2002, 35, 225–243. [Google Scholar] [CrossRef]
- Ayala-Soto, F.E.; Serna-Saldívar, S.O.; Welti-Chanes, J. Effect of Arabinoxylans and Laccase on Batter Rheology and Quality of Yeast-Leavened Gluten-Free Breads. J. Cereal Sci. 2017, 73, 10–17. [Google Scholar] [CrossRef]
- Yan, W.; Yin, L.; Zhang, M.; Zhang, M.; Jia, X. Gelatinization, Retrogradation and Gel Properties of Wheat Starch–Wheat Bran Arabinoxylan Complexes. Gels 2021, 7, 200. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, M.; Zhang, M.; Yadav, M.P.; Jia, X.; Yin, L. Effect of Wheat Bran Arabinoxylan on the Gelatinization and Long-Term Retrogradation Behavior of Wheat Starch. Carbohydr. Polym. 2022, 291, 119581. [Google Scholar] [CrossRef]
- Gudmundsson, M.; Eliasson, A.; Bengtsson, S.; Aman, P. The Effects of Water Soluble Arabinoxylan on Gelatinization and Retrogradation of Starch. Starch-Stärke 1991, 43, 5–10. [Google Scholar]






| AX % | Protein % | Ash % | |
|---|---|---|---|
| Wheat (8.75 kg db) | 5.3 ± 0.0 | 12.8 ± 0.1 | 1.8 ± 0.01 |
| WDG (2.10 kg db) | 14.7 ± 0.8 | 39.2 ± 0.2 | 2.2 ± 0.0 |
| Solubles (1.33 kg db) | 10.0 ± 0.4 | 37.4 ± 0.1 | 8.5 ± 0.4 |
| AX | Protein | Ash | |
|---|---|---|---|
| Wheat (8.75 kg db) | 464 g | 1120 g | 158 g |
| WDG (2.10 kg db) | 309 g | 823 g | 46 g |
| Solubles (1.33 kg db) | 133 g | 497 g | 113 g |
| Mass balance | 95.3% | 117.9% | 100.6% |
| WEAX | WUAX | |
|---|---|---|
| Average Mw (kDa) | 70 | 110 |
| AX purity % | 74.24 ± 0.13 | 51.62 ± 0.09 |
| A:X ratio | 0.49 | 1.18 |
| Total crude protein % | 5.8 ± 0.21 | 9.8 ± 0.31 |
| Ash % | 0.72 ± 0.10 | 4.54 ± 0.10 |
| Moisture % | 8.90 ± 0.49 | 14.15 ± 0.47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Alyassin, M.; Kaderi, S.I.; Campbell, G.M.; Masey O’Neill, H.; Bedford, M.R. Arabinoxylans from the Distillers Grains and Solubles Co-Products of Ethanol Production: Extraction, Characterisation and Hydrolysis to Oligosaccharides. Clean Technol. 2026, 8, 24. https://doi.org/10.3390/cleantechnol8010024
Alyassin M, Kaderi SI, Campbell GM, Masey O’Neill H, Bedford MR. Arabinoxylans from the Distillers Grains and Solubles Co-Products of Ethanol Production: Extraction, Characterisation and Hydrolysis to Oligosaccharides. Clean Technologies. 2026; 8(1):24. https://doi.org/10.3390/cleantechnol8010024
Chicago/Turabian StyleAlyassin, Mohammad, Saffa Izzati Kaderi, Grant M. Campbell, Helen Masey O’Neill, and Michael R. Bedford. 2026. "Arabinoxylans from the Distillers Grains and Solubles Co-Products of Ethanol Production: Extraction, Characterisation and Hydrolysis to Oligosaccharides" Clean Technologies 8, no. 1: 24. https://doi.org/10.3390/cleantechnol8010024
APA StyleAlyassin, M., Kaderi, S. I., Campbell, G. M., Masey O’Neill, H., & Bedford, M. R. (2026). Arabinoxylans from the Distillers Grains and Solubles Co-Products of Ethanol Production: Extraction, Characterisation and Hydrolysis to Oligosaccharides. Clean Technologies, 8(1), 24. https://doi.org/10.3390/cleantechnol8010024