From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach
Abstract
1. Introduction
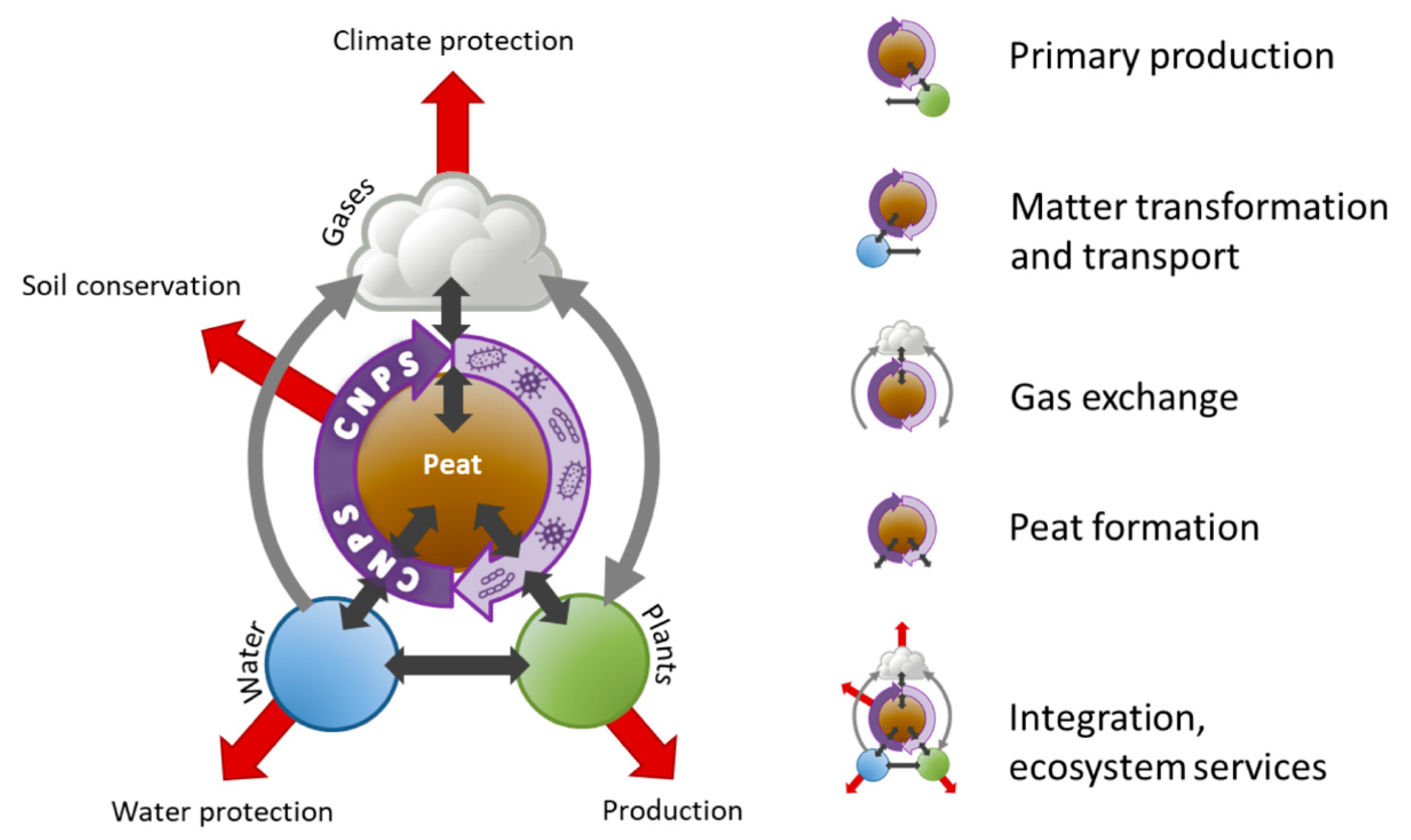
2. The WETSCAPES Approach
- Primary production: How does the water level influence the production of above- and below-ground herbaceous and tree biomass? (plant ecology, ecosystem dynamics, soil physics, soil science, paleoecology)
- Matter transformation and transport: How are nutrients and water transported through the peat body? (soil physics, soil science, microbiology)
- Gas exchange: Which (microbial) processes lead to production and uptake of GHG and what are the driving factors? (microbiology, landscape ecology and grassland sciences for measuring GHG fluxes, soil science)
- Peat formation: How does the interplay of production and decomposition of above- and below-ground herbaceous and tree biomass drive peat growth? (plant ecology, ecosystem dynamics, paleoecology, soil physics, soil science, microbiology)
- Integration, ecosystem services: Can we develop indicators for ecosystem services of rewetted, agriculturally used peatlands? (input from all of the above, landscape ecology, paleoecology, geoinformatics, remote sensing)
Interacting Research Areas in the WETSCAPES Approach
3. The WETSCAPES Research Infrastructure and Methods
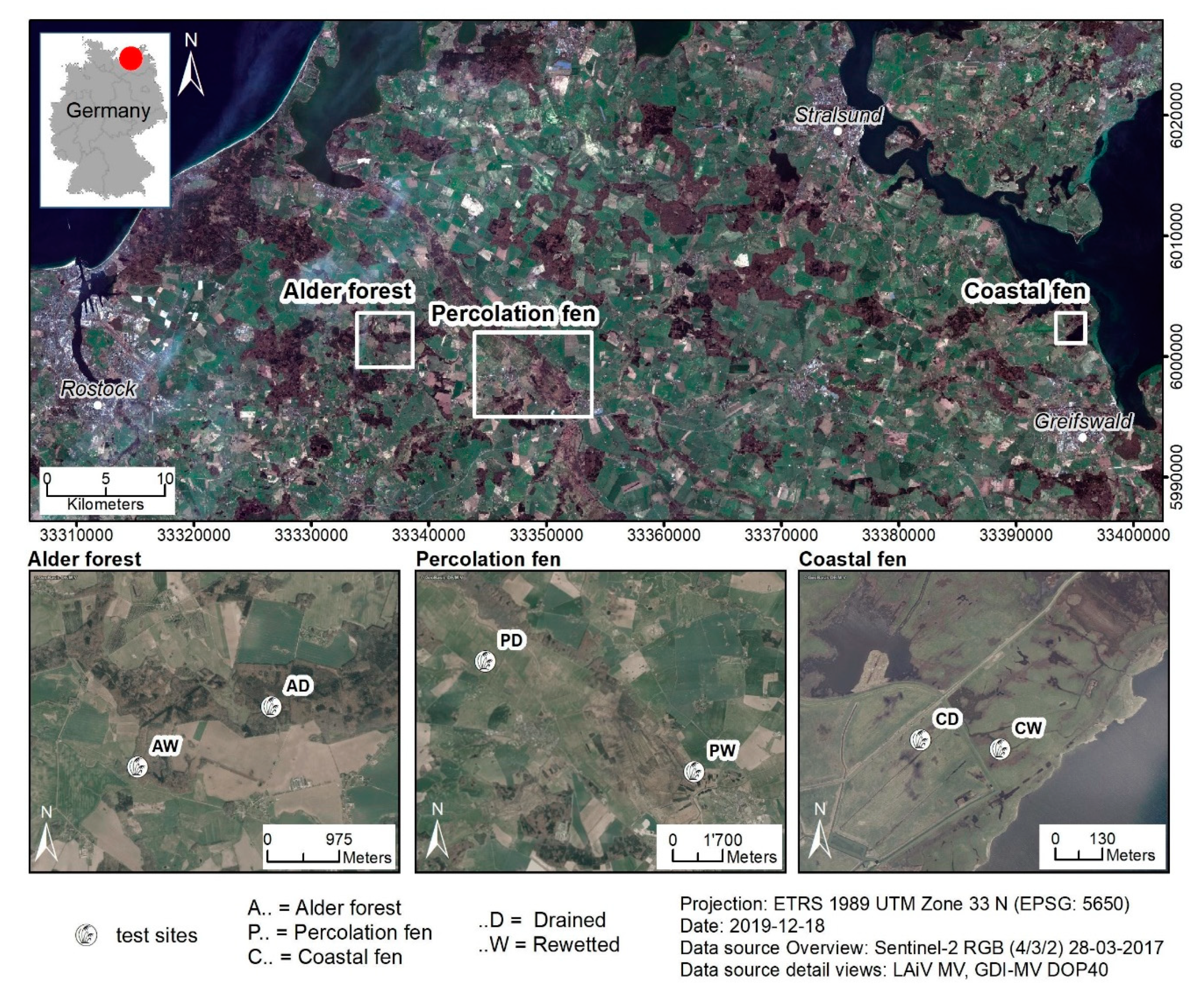
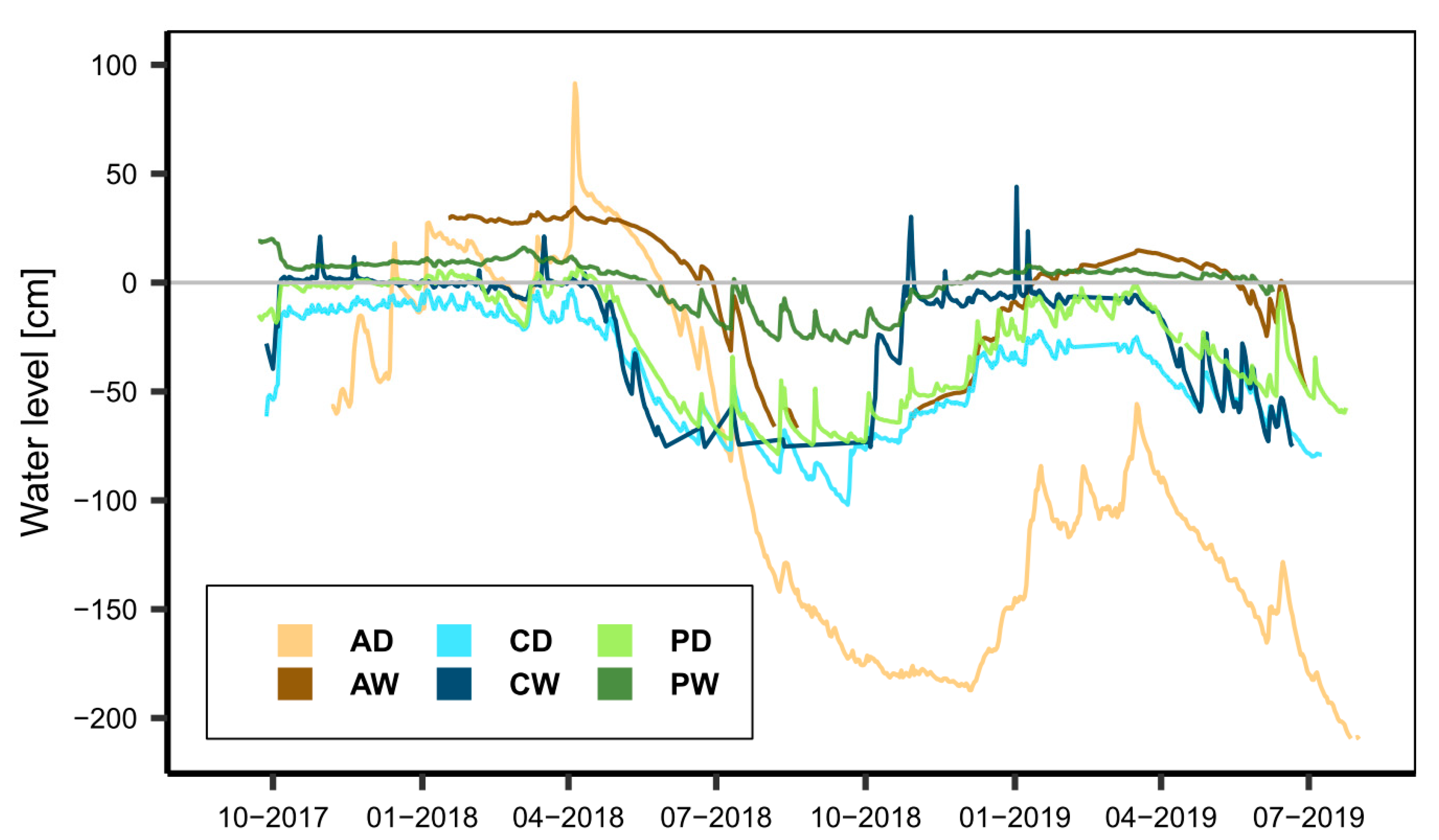
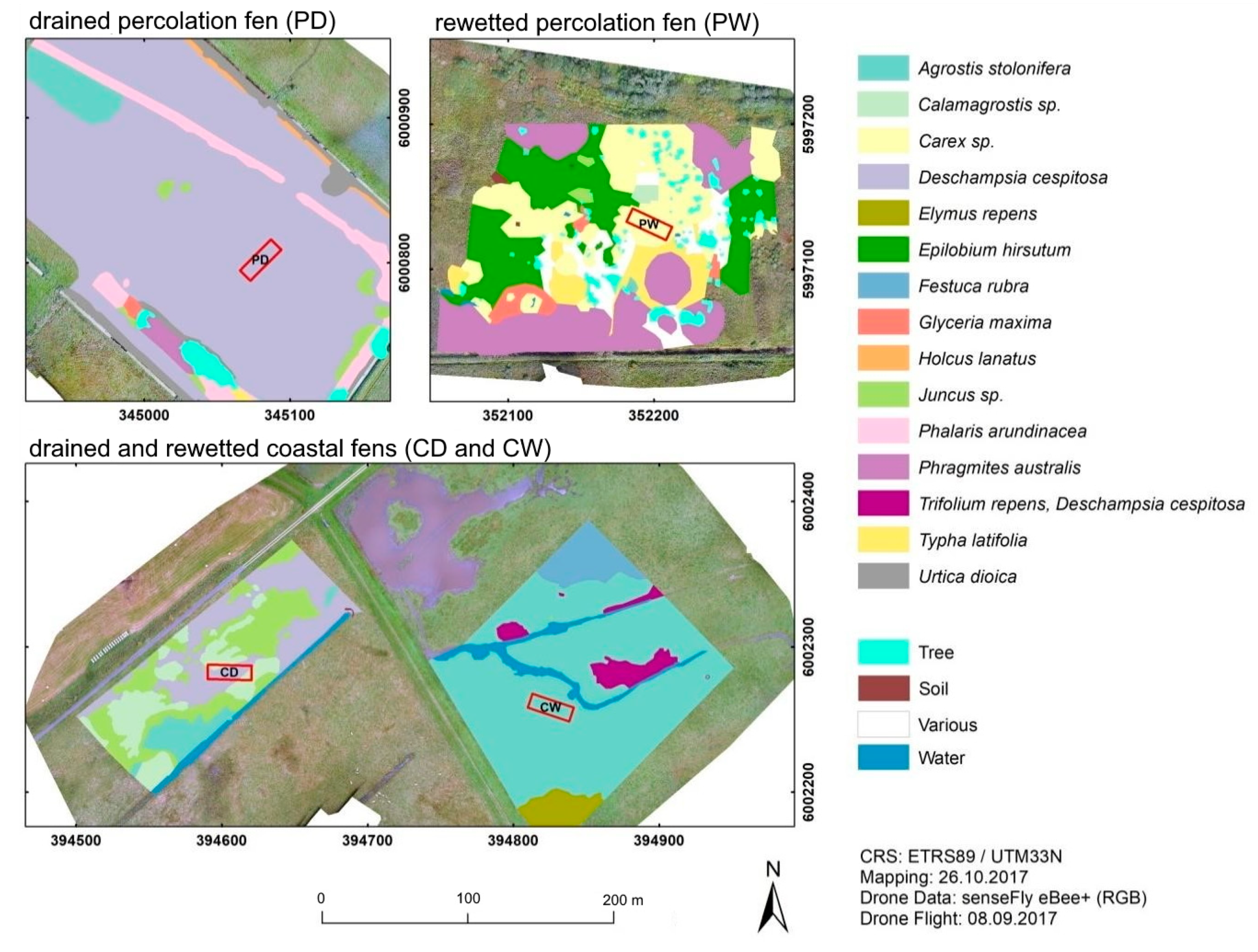
3.1. Study Sites
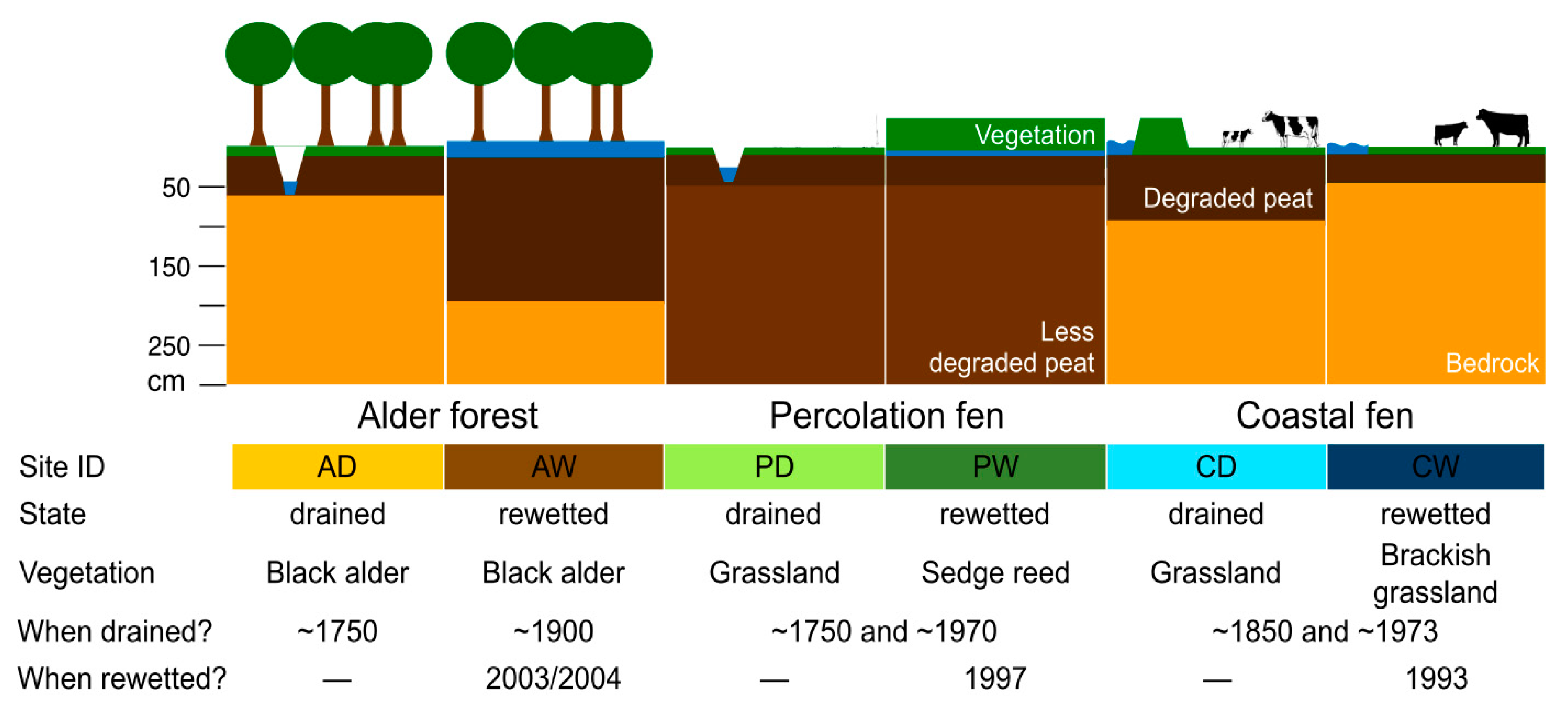
3.1.1. Alder Forest
3.1.2. Percolation Fen
3.1.3. Coastal Fen
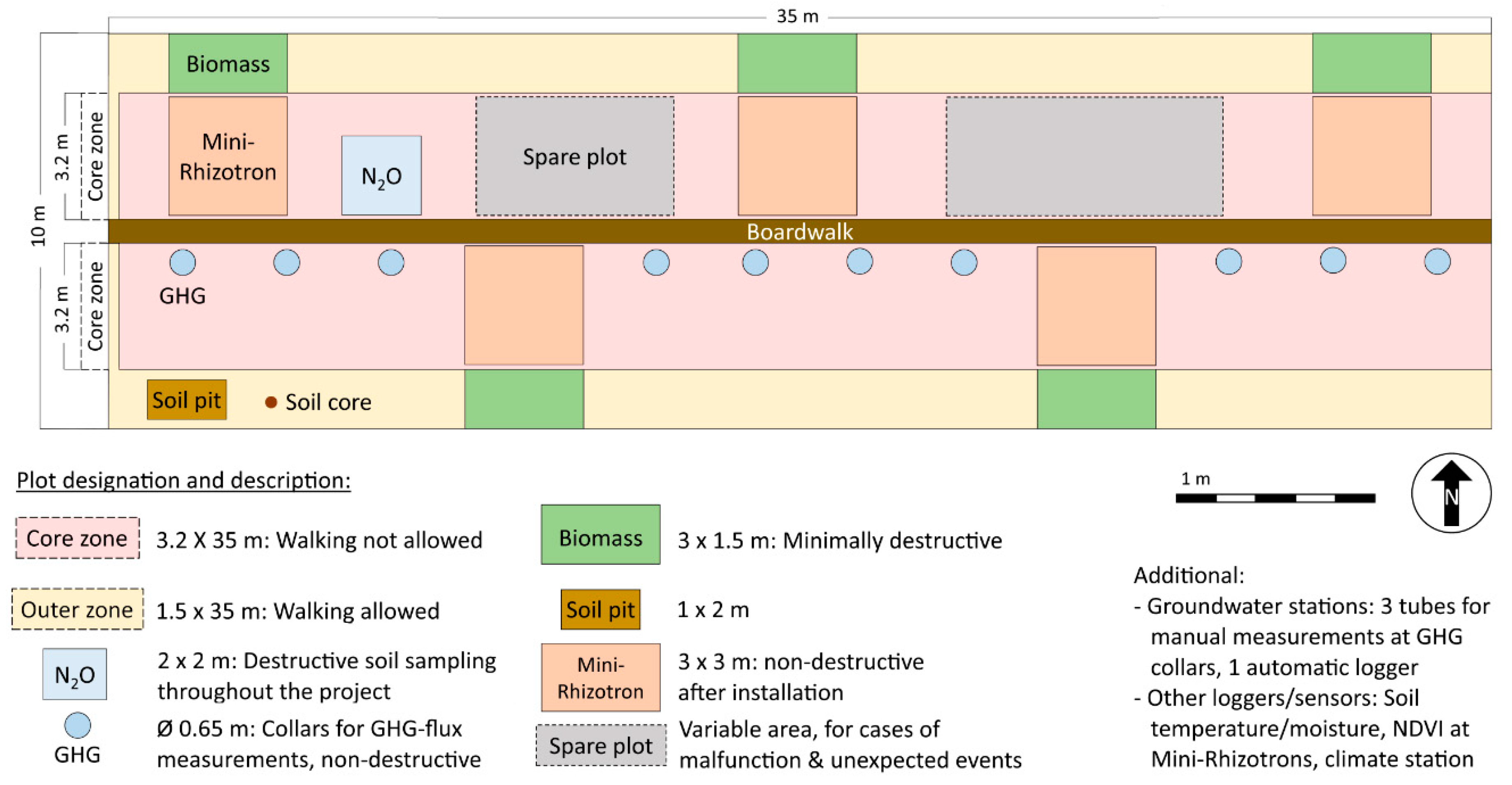
3.2. Sampling Design and Field Setup
3.3. Methodological Approaches
3.4. Interdisciplinary Data Analysis
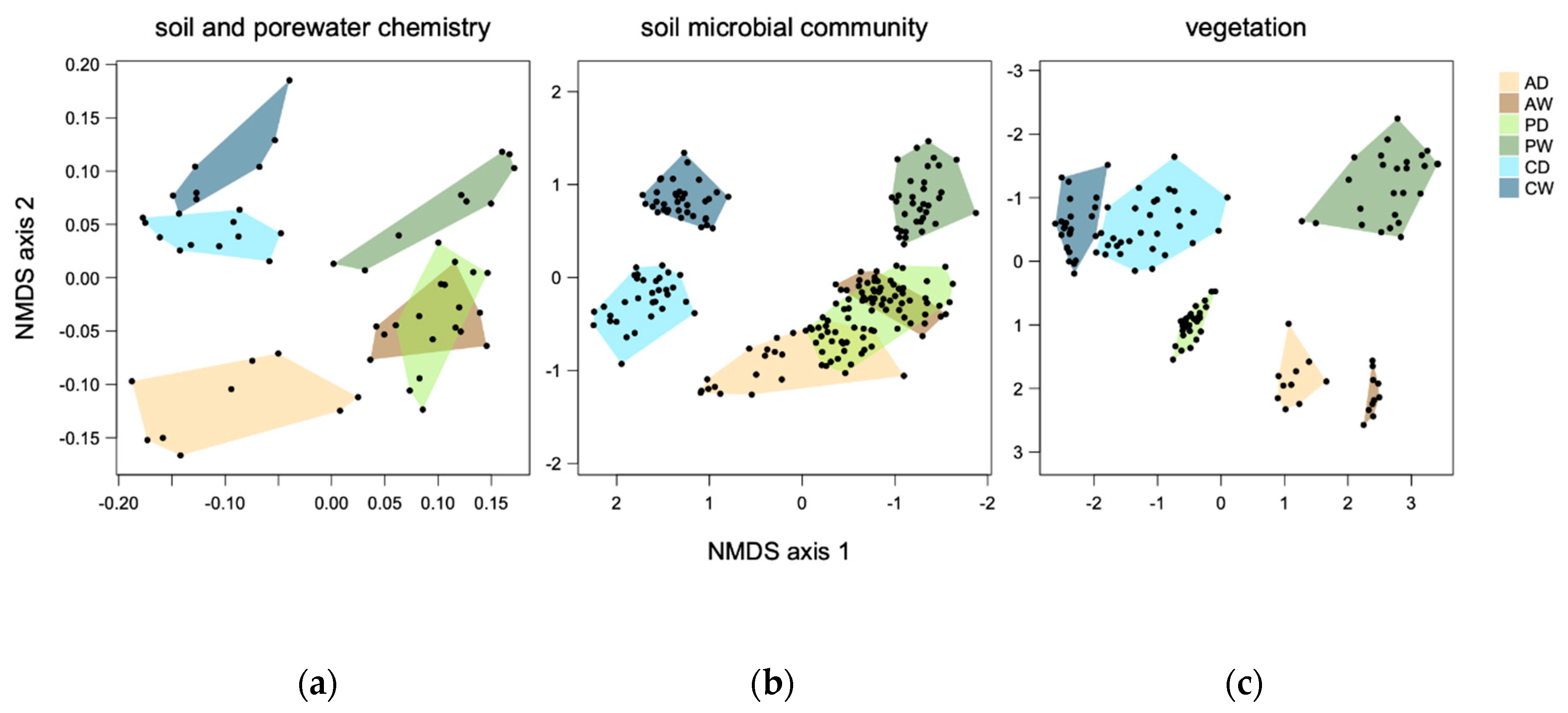
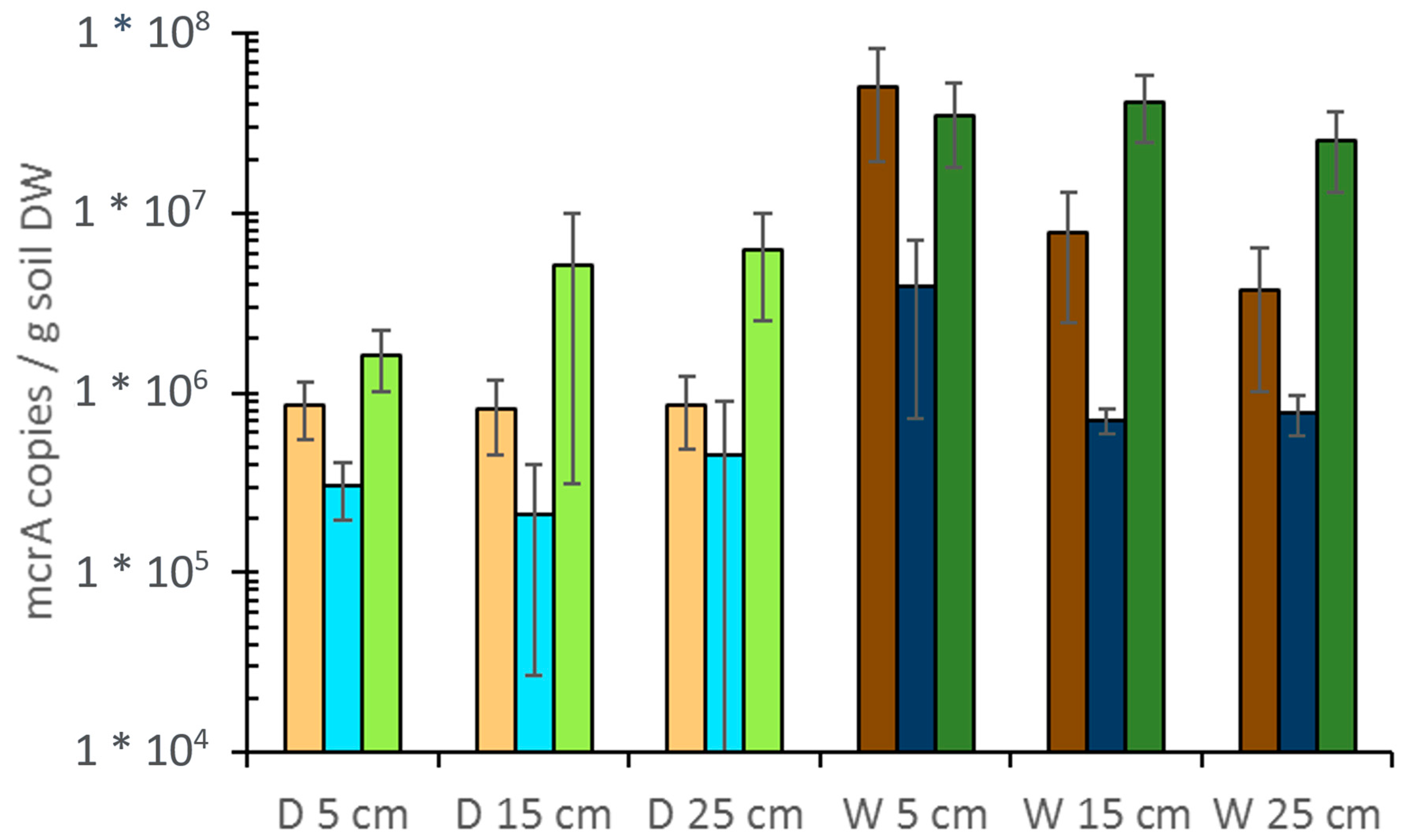
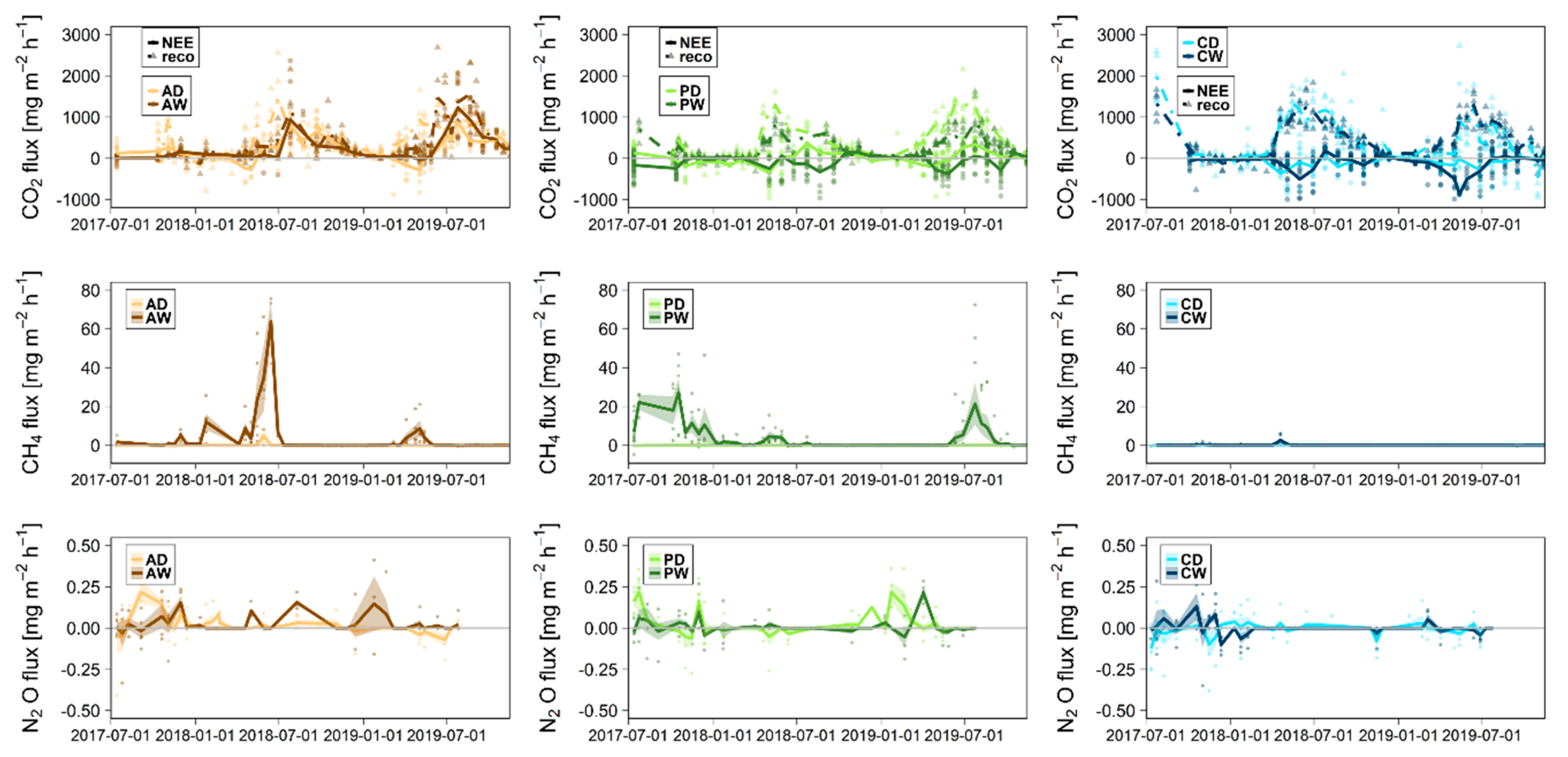
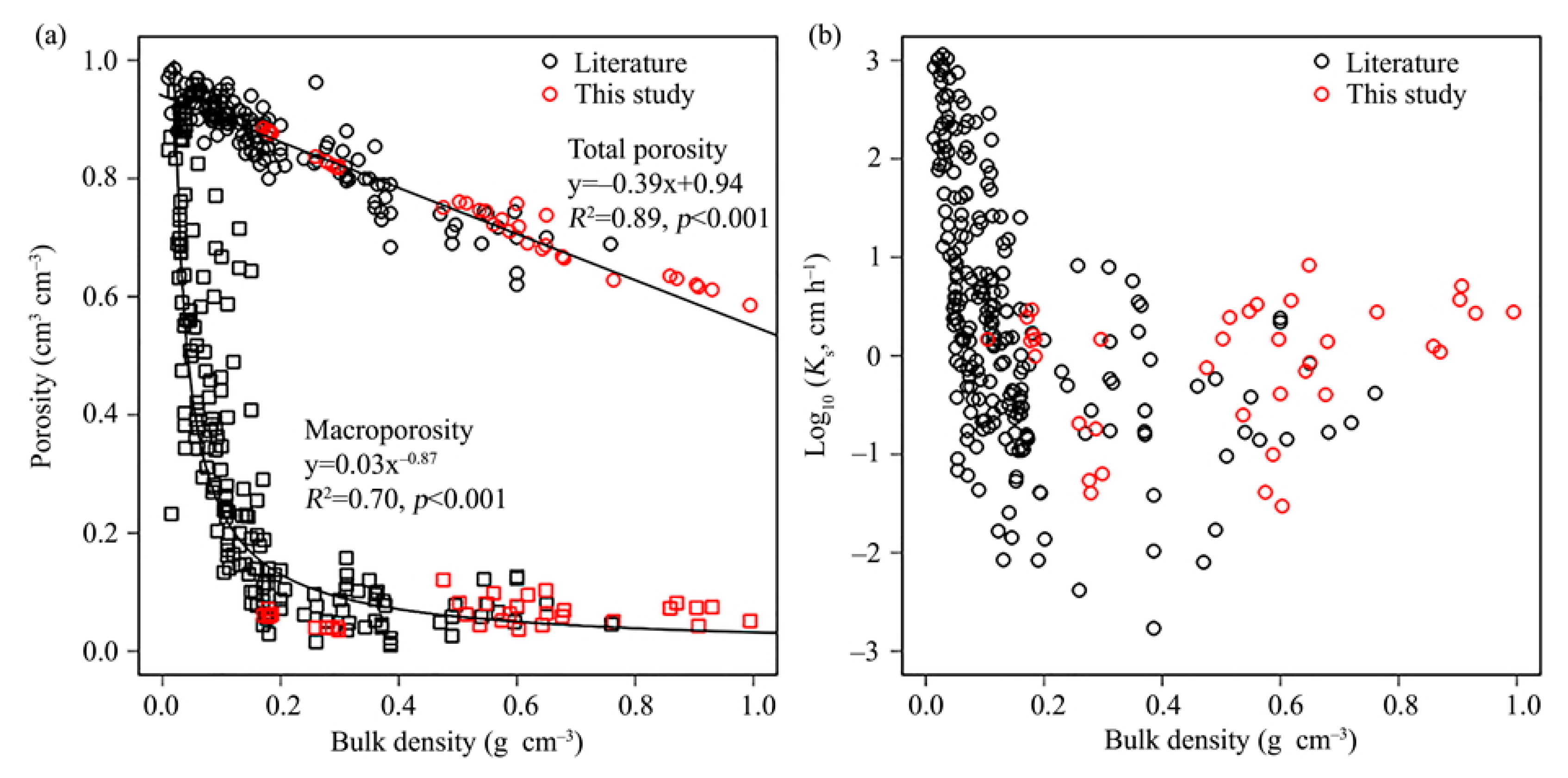
4. First Integrative Results
5. Discussion and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joosten, H. The global peatland CO2 picture. In Peatland Status and Drainage Associated Emissions in All Countries of the World; Wetlands International: Wageningen, The Netherlands, 2009. [Google Scholar]
- Joosten, H.; Sirin, A.; Couwenberg, J.; Laine, J.; Smith, P. The role of peatlands in climate regulation. In Peatland Restoration and Ecosystem Services: Science, Policy and Practice; Bonn, A., Allott, T., Evans, M., Joosten, H., Stoneman, R., Eds.; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Tanneberger, F.; Tegetmeyer, C.; Busse, S.; Barthelmes, A.; Shumka, S.; Moles Mariné, A.; Jenderedjian, K.; Steiner, G.M.; Essl, F.; Etzold, J.; et al. The peatland map of Europe. Mires Peat 2017, 19, 1–17. [Google Scholar]
- Poulin, M.; Rochefort, L.; Desrochers, A. Conservation of bog plant species assemblages: Assessing the role of natural remnants in mined sites. Appl. Veg. Sci. 1999, 2, 169–180. [Google Scholar] [CrossRef]
- Le Quéré, C.; Andrew, R.M.; Friedlingstein, P.; Sitch, S.; Pongratz, J.; Manning, A.C.; Korsbakken, J.I.; Peters, G.P.; Canadell, J.G.; Jackson, R.B.; et al. Global carbon budget 2017. Earth Syst. Sci. Data 2018, 10, 405–448. [Google Scholar] [CrossRef]
- Bonn, A.; Allott, T.; Evans, M.; Joosten, H.; Stoneman, R. Peatland Restoration and Ecosystem Services: Science, Policy and Practice; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Couwenberg, J.; Dommain, R.; Joosten, H. Greenhouse gas fluxes from tropical peatlands in south-east Asia. Glob. Chang. Biol. 2010, 16, 1715–1732. [Google Scholar] [CrossRef]
- Clymo, R.S. Peat; Elsevier: Amsterdam, The Netherlands, 1983. [Google Scholar]
- Eggelsmann, R.; Blankenburg, J. Zur Entwicklung der Heidemoore am Nordhümmling in Niedersachsen. Available online: https://e-docs.geo-leo.de/handle/11858/7548 (accessed on 6 January 2020).
- Evans, C.D.; Chapman, P.J.; Clark, J.M.; Monteith, D.T.; Cresser, M.S. Alternative explanations for rising dissolved organic carbon export from organic soils. Glob. Chang. Biol. 2006, 12, 2044–2053. [Google Scholar] [CrossRef]
- Turetsky, M.R.; Donahue, W.F.; Benscoter, B.W. Experimental drying intensifies burning and carbon losses in a northern peatland. Nat. Commun. 2011, 2, 514. [Google Scholar] [CrossRef]
- Erkens, G.; van der Meulen, M.J.; Middelkoop, H. Double trouble: Subsidence and CO2 respiration due to 1,000 years of dutch coastal peatlands cultivation. Hydrogeol. J. 2016, 24, 551–568. [Google Scholar] [CrossRef]
- Hooijer, A.; Vernimmen, R.; Mawdsley, N.; Page, S.; Mulyadi, D.; Visser, M. Assessment of Impacts of Plantation Drainage on the Kampar Peninsula Peatland, Riau. Available online: https://www.deltares.nl/app/uploads/2015/12/Plantation-Impacts-Kampar-Peatland-DELTARES-2015.pdf (accessed on 10 March 2020).
- Hooijer, A.; Vernimmen, R.; Visser, M.; Mawdsley, N. Flooding Projections from Elevation and Subsidence Models for Oil Palm Plantations in the Rajang Delta Peatlands, Sarawak, Malaysia. Available online: https://www.preventionweb.net/files/45060_45060rajangdeltapeatlandsubsidencef.pdf (accessed on 10 March 2020).
- Schmatzler, B.; Schmatzler, E. Moorland. Moorlandschaften in Niedersachsen Nach Industriellem Torfabbau; Industrieverband Garten e.V.: Ratingen, Germany, 2010. [Google Scholar]
- Quinty, F.; Rochefort, L. Peatland Restoration Guide, Canadian Sphagnum Peat Moss Association: St. Albert, AB, Canada, 2003.
- Strack, M. Peatlands and Climate Change; International Peatland Society: Jyväskylä, Finnland, 2008. [Google Scholar]
- Scholz, M.; Trepel, M. Water quality characteristics of vegetated groundwater-fed ditches in a riparian peatland. Sci. Total Environ. 2004, 332, 109–122. [Google Scholar] [CrossRef]
- Chimner, R.A.; Cooper, D.J.; Wurster, F.C.; Rochefort, L. An overview of peatland restoration in North America: Where are we after 25 years? Restor. Ecol. 2017, 25, 283–292. [Google Scholar] [CrossRef]
- Andersen, R.; Farrell, C.; Graf, M.; Muller, F.; Calvar, E.; Frankard, P.; Caporn, S.; Anderson, P. An overview of the progress and challenges of peatland restoration in Western Europe. Restor. Ecol. 2017, 25, 271–282. [Google Scholar] [CrossRef]
- Wilson, D.; Blain, D.; Couwenberg, J.; Evans, C.D.; Murdiyarso, D.; Page, S.E.; Renou-Wilson, F.; Rieley, J.O.; Sirin, A.; Strack, M.; et al. Greenhouse gas emission factors associated with rewetting of organic soils. Mires Peat 2016, 17, 1–28. [Google Scholar]
- Wilson, D.; Farrell, C.A.; Fallon, D.; Moser, G.; Müller, C.; Renou-Wilson, F. Multiyear greenhouse gas balances at a rewetted temperate peatland. Glob. Chang. Biol. 2016, 22, 4080–4095. [Google Scholar] [CrossRef]
- Günther, A.; Huth, V.; Jurasinski, G.; Glatzel, S. The effect of biomass harvesting on greenhouse gas emissions from a rewetted temperate fen. GCB Bioenergy 2015, 7, 1092–1106. [Google Scholar] [CrossRef]
- Herbst, M.; Friborg, T.; Schelde, K.; Jensen, R.; Ringgaard, R.; Vasquez, V.; Thomsen, A.G.; Soegaard, H. Climate and site management as driving factors for the atmospheric greenhouse gas exchange of a restored wetland. Biogeosciences 2013, 10, 39–52. [Google Scholar] [CrossRef]
- Leifeld, J.; Menichetti, L. The underappreciated potential of peatlands in global climate change mitigation strategies. Nat. Commun. 2018, 9, 1071. [Google Scholar] [CrossRef]
- Bonn, A.; Reed, M.S.; Evans, C.D.; Joosten, H.; Bain, C.; Farmer, J.; Emmer, I.; Couwenberg, J.; Moxey, A.; Artz, R.; et al. Investing in nature: Developing ecosystem service markets for peatland restoration. Ecosyst. Serv. 2014, 9, 54–65. [Google Scholar] [CrossRef]
- Moen, A.; Joosten, H.; Tanneberger, F. Mire diversity in Europe: Mire regionality. In Mires an Peatlands of Europe; Joosten, H.T.F., Moen, A., Eds.; Schweizerbart Science Publishers: Stuttgart, Germany, 2017. [Google Scholar]
- Cabezas, A.; Pallasch, M.; Schönfelder, I.; Gelbrecht, J.; Zak, D. Carbon, nitrogen, and phosphorus accumulation in novel ecosystems: Shallow lakes in degraded fen areas. Ecol. Eng. 2014, 66, 63–71. [Google Scholar] [CrossRef]
- Knox, S.H.; Sturtevant, C.; Matthes, J.H.; Koteen, L.; Verfaillie, J.; Baldocchi, D. Agricultural peatland restoration: Effects of land-use change on greenhouse gas (CO2 and CH4) fluxes in the Sacramento-San Joaquin delta. Glob. Chang. Biol. 2015, 21, 750–765. [Google Scholar] [CrossRef]
- Lamers, L.P.M.; Vile, M.A.; Grootjans, A.P.; Acreman, M.C.; van Diggelen, R.; Evans, M.G.; Richardson, C.J.; Rochefort, L.; Kooijman, A.M.; Roelofs, J.G.M.; et al. Ecological restoration of rich fens in Europe and North America: From trial and error to an evidence-based approach. Biol. Rev. 2015, 90, 182–203. [Google Scholar] [CrossRef]
- Timmermann, T.; Margóczi, K.; Takács, G.; Vegelin, K. Restoration of peat-forming vegetation by rewetting species-poor fen grasslands. Appl. Veg. Sci. 2006, 9, 241–250. [Google Scholar] [CrossRef]
- Urbanová, Z.; Picek, T.; Hájek, T.; Bufková, I.; Tuittila, E.S. Impact of drainage and restoration on vegetation and carbon gas dynamics in Central European peatlands, extended abstract no. 214. In Proceedings of the 14th International Peat Congress, Stockholm, Sweden, 3–8 June 2012. [Google Scholar]
- Kotowski, W.; Acreman, M.; Grootjans, A.; Klimkowska, A.; Rössling, H.; Wheeler, B. Restoration of temperate fens: Matching strategies with site potential. In Peatland Restoration and Ecosystem Services: Science, Policy and Practice; Bonn, A., Allott, T., Evans, M., Joosten, H., Stoneman, R., Eds.; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Hendriks, D.M.D.; van Huissteden, J.; Dolman, A.J.; van der Molen, M.K. The full greenhouse gas balance of an abandoned peat meadow. Biogeosciences 2007, 4, 411–424. [Google Scholar] [CrossRef]
- Zeitz, J.; Velty, S. Soil properties of drained and rewetted fen soils. Journal of Plant Nutrition and Soil Science 2002, 165, 618–626. [Google Scholar] [CrossRef]
- Succow, M.; Joosten, H. Landschaftsökologische Moorkunde, 2nd ed.; Schweizerbart: Stuttgart, Germany, 2001. [Google Scholar]
- Szajdak, L.; Szatyłowicz, J. Impact of drainage on hydrophobicity of fen peat-moorsh soils. In Mires and peat; Kļaviņš, M., Ed.; University of Latvia Press: Riga, Latvia, 2010. [Google Scholar]
- Wassen, M.; Grootjans, A.P. Ecohydrology: An interdisciplinary approach for wetland management and restoration. Vegetatio 1996, 126, 1–4. [Google Scholar]
- Moore, T.R.; Bubier, J.L.; Bledzki, L. Litter decomposition in temperate peatland ecosystems: The effect of substrate and site. Ecosystems 2007, 10, 949–963. [Google Scholar] [CrossRef]
- Smiljanić, M.; Seo, J.-W.; Läänelaid, A.; van der Maaten-Theunissen, M.; Stajić, B.; Wilmking, M. Peatland pines as a proxy for water table fluctuations: Disentangling tree growth, hydrology and possible human influence. Sci. Total Environ. 2014, 500–501, 52–63. [Google Scholar] [CrossRef]
- Scharnweber, T.; Couwenberg, J.; Heinrich, I.; Wilmking, M. New insights for the interpretation of ancient bog oak chronologies? Reactions of oak (Quercus robur l.) to a sudden peatland rewetting. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2015, 417, 534–543. [Google Scholar] [CrossRef]
- Murphy, M.T.; Moore, T.R. Linking root production to aboveground plant characteristics and water table in a temperate bog. Plant Soil 2010, 336, 219–231. [Google Scholar] [CrossRef]
- Zak, D.; Roth, C.; Unger, V.; Goldhammer, T.; Fenner, N.; Freeman, C.; Jurasinski, G. Unraveling the importance of polyphenols for microbial carbon mineralization in rewetted riparian peatlands. Front. Environ. Sci. 2019, 7, 1–14. [Google Scholar] [CrossRef]
- Freeman, C.; Ostle, N.; Kang, H. An enzymic ’latch’ on a global carbon store. Nature 2001, 409, 149. [Google Scholar] [CrossRef]
- Thauer, R.K.; Shima, S. Methane as fuel for anaerobic microorganisms. Ann. N.Y. Acad. Sci. 2008, 1125, 158–170. [Google Scholar] [CrossRef]
- Welte, C.U. Revival of archaeal methane microbiology. mSystems 2018, 3. [Google Scholar] [CrossRef]
- Whalen, S.C. Biogeochemistry of methane exchange between natural wetlands and the atmosphere. Environ. Eng. Sci. 2005, 22, 73–94. [Google Scholar] [CrossRef]
- Larmola, T.; Tuittila, E.-S.; Tiirola, M.; Nykänen, H.; Martikainen, P.J.; Yrjälä, K.; Tuomivirta, T.; Fritze, H. The role of Sphagnum mosses in the methane cycling of a boreal mire. Ecology 2010, 91, 2356–2365. [Google Scholar] [CrossRef]
- Andersen, R.; Chapman, S.J.; Artz, R.R.E. Microbial communities in natural and disturbed peatlands: A review. Soil Biol. Biochem. 2013, 57, 979–994. [Google Scholar] [CrossRef]
- Rezanezhad, F.; Price, J.S.; Quinton, W.L.; Lennartz, B.; Milojevic, T.; Van Cappellen, P. Structure of peat soils and implications for water storage, flow and solute transport: A review update for geochemists. Chem. Geol. 2016, 429, 75–84. [Google Scholar] [CrossRef]
- Robinson, D.A.; Jones, S.B.; Lebron, I.; Reinsch, S.; Domínguez, M.T.; Smith, A.R.; Jones, D.L.; Marshall, M.R.; Emmett, B.A. Experimental evidence for drought induced alternative stable states of soil moisture. Sci. Rep. 2016, 6, 20018. [Google Scholar] [CrossRef]
- Wallor, E.; Rosskopf, N.; Zeitz, J. Hydraulic properties of drained and cultivated fen soils part i—horizon-based evaluation of van genuchten parameters considering the state of moorsh-forming process. Geoderma 2018, 313, 69–81. [Google Scholar] [CrossRef]
- Baird, A.J. Field estimation of macropore functioning and surface hydraulic conductivity in a fen peat. Hydrol. Process. 1997, 11, 287–295. [Google Scholar] [CrossRef]
- Berger, S.; Gebauer, G.; Blodau, C.; Knorr, K.-H. Peatlands in a eutrophic world—Assessing the state of a poor fen-bog transition in southern Ontario, Canada, after long term nutrient input and altered hydrological conditions. Soil Biol. Biochem. 2017, 114, 131–144. [Google Scholar] [CrossRef]
- Menberu, M.W.; Marttila, H.; Tahvanainen, T.; Kotiaho, J.S.; Hokkanen, R.; Kløve, B.; Ronkanen, A.-K. Changes in pore water quality after peatland restoration: Assessment of a large-scale, replicated before-after-control-impact study in finland. Water Resour. Res. 2017, 53, 8327–8343. [Google Scholar] [CrossRef]
- Koerselman, W.; Van Kerkhoven, M.B.; Verhoeven, J.T. Release of inorganic N, P and K in peat soils; effect of temperature, water chemistry and water level. Biogeochemistry 1993, 20, 63–81. [Google Scholar] [CrossRef]
- Craft, C.B.; Richardson, C.J. Relationships between soil nutrients and plant species composition in Everglades peatlands. J. Environ. Qual. 1997, 26, 224–232. [Google Scholar] [CrossRef]
- Zak, D.; Goldhammer, T.; Cabezas, A.; Gelbrecht, J.; Gurke, R.; Wagner, C.; Reuter, H.; Augustin, J.; Klimkowska, A.; McInnes, R. Top soil removal reduces water pollution from phosphorus and dissolved organic matter and lowers methane emissions from rewetted peatlands. J. Appl. Ecol. 2018, 55, 311–320. [Google Scholar] [CrossRef]
- Jurasinski, G.; Günther, A.B.; Huth, V.; Couwenberg, J.; Glatzel, S. Ecosystem services provided by paludiculture—greenhouse gas emissions. In Paludiculture—Productive Use of Wet Peatlands; Wichtmann, W., Schröder, C., Joosten, H., Eds.; Schweizerbart Scientific Publishers: Stuttgart, Germany, 2016. [Google Scholar]
- Hahn, J.; Köhler, S.; Glatzel, S.; Jurasinski, G. Methane exchange in a coastal fen in the first year after flooding—A systems shift. PLoS ONE 2015, 10, e0140657. [Google Scholar] [CrossRef]
- Hahn-Schöfl, M.; Zak, D.; Minke, M.; Gelbrecht, J.; Augustin, J.; Freibauer, A. Organic sediment formed during inundation of a degraded fen grassland emits large fluxes of CH4 and CO2. Biogeosciences 2011, 8, 1539–1550. [Google Scholar] [CrossRef]
- Franz, D.; Koebsch, F.; Larmanou, E.; Augustin, J.; Sachs, T. High net CO2 and CH4 release at a eutrophic shallow lake on a formerly drained fen. Biogeosciences 2016, 13, 3051–3070. [Google Scholar] [CrossRef]
- Tiemeyer, B.; Albiac Borraz, E.; Augustin, J.; Bechtold, M.; Beetz, S.; Beyer, C.; Drösler, M.; Ebli, M.; Eickenscheidt, T.; Fiedler, S.; et al. High emissions of greenhouse gases from grasslands on peat and other organic soils. Glob. Chang. Biol. 2016, 22, 4134–4149. [Google Scholar] [CrossRef]
- Augustin, J.; Münchmeyer, U.; Russow, R. Nitrous oxide and dinitrogen losses from flooded fen peat with and without reed canarygrass (Phalaris arundinacea l.) and common reed (Phragmites australis (cav.) trin. Ex steud.) plants. In Plant Nutrition: Food Security and Sustainability of Agro-Ecosystems Through Basic and Applied Research; Horst, W.J., Schenk, M.K., Bürkert, A., Claassen, N., Flessa, H., Frommer, W.B., Goldbach, H., Olfs, H.W., Römheld, V., Sattelmacher, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Lohila, A.; Aurela, M.; Hatakka, J.; Pihlatie, M.; Minkkinen, K.; Penttilä, T.; Laurila, T. Responses of N2O fluxes to temperature, water table and N deposition in a northern boreal fen. Eur. J. Soil Sci. 2010, 61, 651–661. [Google Scholar] [CrossRef]
- Wrage-Mönnig, N.; Horn, M.A.; Well, R.; Müller, C.; Velthof, G.; Oenema, O. The role of nitrifier denitrification in the production of nitrous oxide revisited. Soil Biol. Biochem. 2018, 123, A3–A16. [Google Scholar] [CrossRef]
- Chapuis-Lardy, L.; Wrage, N.; Metay, A.; Chotte, J.-L.; Bernoux, M. Soils, a sink for N2O? A review. Glob. Chang. Biol. 2007, 13, 1–17. [Google Scholar] [CrossRef]
- Rydin, H.; Jeglum, J.K. The Biology of Peatlands, 2nd ed.; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Frolking, S.; Roulet, N.T.; Tuittila, E.; Bubier, J.L.; Quillet, A.; Talbot, J.; Richard, P.J.H. A new model of holocene peatland net primary production, decomposition, water balance, and peat accumulation. Earth Syst. Dynam. 2010, 1, 1–21. [Google Scholar] [CrossRef]
- Smith, J.; Gottschalk, P.; Bellarby, J.; Chapman, S.; Lilly, A.; Towers, W.; Bell, J.; Coleman, K.; Nayak, D.; Richards, M.; et al. Estimating changes in Scottish soil carbon stocks using Ecosse. I. Model description and uncertainties. Clim. Res. 2010, 45, 179–192. [Google Scholar] [CrossRef]
- Clymo, R.S.; Turunen, J.; Tolonen, K. Carbon accumulation in peatland. Oikos 1998, 81, 368–388. [Google Scholar] [CrossRef]
- Malak, D.A.; Hilarides, L. Available online: https://www.swos-service.eu/wp-content/uploads/2016/06/SWOS_Wetlands-delimitation-guidelines_FINAL_v1.1.pdf (accessed on 10 March 2020).
- Barthelmes, A. Vegetation dynamics and carbon sequestration in holocene alder (Alnus glutinosa) carrs in NE Germany. Ph.D. Thesis, Universität Greifswald, Greifswald, Germany, 2009. [Google Scholar]
- Ratzke, U. Der Moorstandortkatalog Mecklenburg-Vorpommern—eine Grundlage für das Moorschutzprogramm. TELMA Ber. Dtsch. Ges. Moor Torfkunde 2000, 30, 195–203. [Google Scholar]
- Kühnel, P. Die slawischen Ortsnamen in Meklenburg. Jahrbücher des Vereins für Mecklenburgische Geschichte und Altertumskunde 1881, 46, 3–168. [Google Scholar]
- Koch, M. Studying Vegetation and Vegetation Change in Low-Land Fens of North-Eastern Germany-Methods and Case Studies. Ph.D. Thesis, University of Rostock, Rostock, Germany, 2017. [Google Scholar]
- Bönsel, A. Revitalisierung von Regenmooren in Nordostdeutschland: Überblick und Perspektiven. Available online: https://e-docs.geo-leo.de/handle/11858/7288 (accessed on 6 January 2020).
- Von Post, L. Swedish geological peat survey with the results obtained so far (in Swedish). Sven. Mosskult. Tidskr. 1922, 36, 1–27. [Google Scholar]
- Krisch, H. Zur Kenntnis der Pflanzengesellschaften der Mecklenburgischen Boddenküste. Feddes Repert. 1974, 85, 115–158. [Google Scholar] [CrossRef]
- Jeschke, L. Vegetationsdynamik des Salzgraslandes im Bereich der Ostseeküste der DDR unter dem Einfluß des Menschen. Hercynia NF 1987, 24, 321–328. [Google Scholar]
- Härdtle, W. Vegetationskundliche Untersuchungen in Salzwiesen der Ostholsteinischen Ostseeküste; Arbeitsgemeinschaft Geobotanik: Kiel, Germany, 1984. [Google Scholar]
- Dijkema, K.S. Salt and brackish marshes around the baltic sea and adjacent parts of the North Sea: Their vegetation and management. Biol. Conserv. 1990, 51, 191–209. [Google Scholar] [CrossRef]
- Joosten, H.; Klerk, P.D. DAMOCLES: A dashing monolith cutter for fine sectioning of peats and sediments into large slices. Boreas 2007, 36, 76–81. [Google Scholar] [CrossRef]
- Barthelmes, A.; de Klerk, P.; Prager, A.; Theuerkauf, M.; Unterseher, M.; Joosten, H. Expanding NPP analysis to eutrophic and forested sites: Significance of NPPs in a holocene wood peat section (NE Germany). Rev. Palaeobot. Palynol. 2012, 186, 22–37. [Google Scholar] [CrossRef]
- Barthelmes, A.; Prager, A.; Joosten, H. Palaeoecological analysis of Alnus wood peats with special attention to non-pollen palynomorphs. Rev. Palaeobot. Palynol. 2006, 141, 33–51. [Google Scholar] [CrossRef]
- Prager, A.; Barthelmes, A.; Theuerkauf, M.; Joosten, H. Non-pollen palynomorphs from modern alder carrs and their potential for interpreting microfossil data from peat. Rev. Palaeobot. Palynol. 2006, 141, 7–31. [Google Scholar] [CrossRef]
- Prager, A.; Theuerkauf, M.; Couwenberg, J.; Barthelmes, A.; Aptroot, A.; Joosten, H. Pollen and non-pollen palynomorphs as tools for identifying alder carr deposits: A surface sample study from NE-Germany. Rev. Palaeobot. Palynol. 2012, 186, 38–57. [Google Scholar] [CrossRef]
- Johnson, M.G.; Tingey, D.T.; Phillips, D.L.; Storm, M.J. Advancing fine root research with minirhizotrons. Environ. Exp. Bot. 2001, 45, 263–289. [Google Scholar] [CrossRef]
- Keuskamp, J.A.; Dingemans, B.J.J.; Lehtinen, T.; Sarneel, J.M.; Hefting, M.M. Tea bag index: A novel approach to collect uniform decomposition data across ecosystems. Methods Ecol. Evol. 2013, 4, 1070–1075. [Google Scholar] [CrossRef]
- Pettorelli, N.; Vik, J.O.; Mysterud, A.; Gaillard, J.-M.; Tucker, C.J.; Stenseth, N.C. Using the satellite-derived NDVI to assess ecological responses to environmental change. Trends Ecol. Evol. 2005, 20, 503–510. [Google Scholar] [CrossRef]
- Gauci, V.; Gowing, D.J.G.; Hornibrook, E.R.C.; Davis, J.M.; Dise, N.B. Woody stem methane emission in mature wetland alder trees. Atmos. Environ. 2010, 44, 2157–2160. [Google Scholar] [CrossRef]
- Poulsen, M.; Schwab, C.; Borg Jensen, B.; Engberg, R.M.; Spang, A.; Canibe, N.; Højberg, O.; Milinovich, G.; Fragner, L.; Schleper, C.; et al. Methylotrophic methanogenic thermoplasmata implicated in reduced methane emissions from bovine rumen. Nat. Commun. 2013, 4, 1428. [Google Scholar] [CrossRef]
- Söllinger, A.; Schwab, C.; Weinmaier, T.; Loy, A.; Tveit, A.T.; Schleper, C.; Urich, T. Phylogenetic and genomic analysis of Methanomassiliicoccales in wetlands and animal intestinal tracts reveals clade-specific habitat preferences. FEMS Microbiol. Ecol. 2015, 92, 1–12. [Google Scholar]
- Tveit, A.T.; Urich, T.; Frenzel, P.; Svenning, M.M. Metabolic and trophic interactions modulate methane production by arctic peat microbiota in response to warming. Proc. Natl. Acad. Sci. USA 2015, 112, E2507–E2516. [Google Scholar] [CrossRef]
- Urich, T.; Lanzén, A.; Qi, J.; Huson, D.H.; Schleper, C.; Schuster, S.C. Simultaneous assessment of soil microbial community structure and function through analysis of the meta-transcriptome. PLoS ONE 2008, 3, e2527. [Google Scholar] [CrossRef]
- Livingston, G.P.; Hutchinson, G.L. Enclosure-based measurement of trace gas exchange: Applications and sources of error. In Biogenic Trace Gases: Measuring Emissions from Soil and Water; Matson, P.A., Harriss, R.C., Eds.; Blackwell Science Ltd: Oxford, UK, 1995. [Google Scholar]
- Kool, D.M.; Van Groenigen, J.W.; Wrage, N. Source determination of nitrous oxide based on nitrogen and oxygen isotope tracing: Dealing with oxygen exchange. Methods Enzymol. 2011, 496, 139–160. [Google Scholar]
- Wrage, N.; van Groenigen, J.W.; Oenema, O.; Baggs, E.M. A novel dual-isotope labelling method for distinguishing between soil sources of N2O. Rapid Commun. Mass Spectrom. 2005, 19, 3298–3306. [Google Scholar] [CrossRef]
- Welti, N.; Siljanen, H.; Biasi, C.; Martikainen, P.J. Quantification of Nitrous Oxide (N2O) Uptake in Boreal Forest Soils by Combining Isotopic and Microbial Approaches; EGU General Assembly: Vienna, Austria, 2015. [Google Scholar]
- Koch, M.; Jurasinski, G.; Bäthge, J.; Glatzel, S. Raumzeitliche Analyse der Entwicklung von Wäldern und Forsten in Mecklenburg. In Virtuelle Forschungsumgebung für die Kulturlandschaftsforschung auf Basis von Internet-GIS-Technologie; Bill, R., Ed.; Wichmann: Offenbach am Main, Germany, 2012. [Google Scholar]
- VKLandLab. Vklandlab-Demo. Available online: http://kvwmap.geoinformatik.uni-rostock.de/VKLandLab/index.php?gast=45 (accessed on 17 January 2020).
- SVEA-Pommern. Karten und Texte der Schwedischen Landesaufnahme von Pommern 1692–1709. Eine GIS-Gestützte Auswahledition des Ersten Deutschen Katasters im Internet. Available online: http://www.svea-pommern.de (accessed on 17 January 2020).
- Müller-Westermeier, G. Numerische Verfahren Zur Erstellung klimatologischer Karten; Selbstverlag des Deutschen Wetterdienstes: Offenbach am Main, Germany, 1995. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5–6. Available online: https://CRAN.R-project.org/package=vegan (accessed on 17 January 2020).
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 17 January 2020).
- Faith, D.P.; Minchin, P.R.; Belbin, L. Compositional dissimilarity as a robust measure of ecological distance. Vegetatio 1987, 69, 57–68. [Google Scholar] [CrossRef]
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Schwertmann, U. Differenzierung der Eisenoxide des Bodens durch Extraktion mit Ammoniumoxalat-Lösung. Zeitschrift für Pflanzenernährung, Düngung, Bodenkunde 1964, 105, 194–202. [Google Scholar] [CrossRef]
- Negassa, W.; Acksel, A.; Eckhardt, K.-U.; Regier, T.; Leinweber, P. Soil organic matter characteristics in drained and rewetted peatlands of northern Germany: Chemical and spectroscopic analyses. Geoderma 2019, 353, 468–481. [Google Scholar] [CrossRef]
- Weil, M.; Wang, H.; Bengtsson, M.; Günther, A.; Jurasinski, G.; Couwenberg, J.; Negassa, W.; Zak, D.; Urich, T. Rewetting of three drained peatlands drives congruent compositional changes in pro-and eukaryotic microbiomes through environmental filtering. BioRxiv 2019, 848192, 1–23. [Google Scholar]
- Steinberg, L.M.; Regan, J.M. Mcra-targeted real-time quantitative pcr method to examine methanogen communities. Appl. Environ. Microbiol. 2009, 75, 4435–4442. [Google Scholar] [CrossRef]
- Schindler, U.; Behrendt, A.; Müller, L. Change of soil hydrological properties of fens as a result of soil development. J. Plant Nutr. Soil Sci. 2003, 166, 357–363. [Google Scholar] [CrossRef]
- Liu, H.; Lennartz, B. Hydraulic properties of peat soils along a bulk density gradient—A meta study. Hydrol. Process. 2019, 33, 101–114. [Google Scholar] [CrossRef]
- Liu, H.; Zak, D.; Rezanezhad, F.; Lennartz, B. Soil degradation determines release of nitrous oxide and dissolved organic carbon from peatlands. Environ. Res. Lett. 2019, 14, 094009. [Google Scholar] [CrossRef]
- Rahmstorf, S.; Coumou, D. Increase of extreme events in a warming world. Proc. Natl. Acad. Sci. USA 2011, 108, 17905–17909. [Google Scholar] [CrossRef]
- Stott, P. How climate change affects extreme weather events. Science 2016, 352, 1517–1518. [Google Scholar] [CrossRef]
- Sippel, S.; Meinshausen, N.; Fischer, E.M.; Székely, E.; Knutti, R. Climate change now detectable from any single day of weather at global scale. Nat. Clim. Chang. 2020, 10, 35–41. [Google Scholar] [CrossRef]
- Maxwell, S.L.; Butt, N.; Maron, M.; McAlpine, C.A.; Chapman, S.; Ullmann, A.; Segan, D.B.; Watson, J.E.M. Conservation implications of ecological responses to extreme weather and climate events. Divers. Distrib. 2019, 25, 613–625. [Google Scholar] [CrossRef]
- De Boeck, H.J.; Vicca, S.; Roy, J.; Nijs, I.; Milcu, A.; Kreyling, J.; Jentsch, A.; Chabbi, A.; Campioli, M.; Callaghan, T.; et al. Global change experiments: Challenges and opportunities. BioScience 2015, 65, 922–931. [Google Scholar] [CrossRef]
- Kechavarzi, C.; Dawson, Q.; Leeds-Harrison, P.B. Physical properties of low-lying agricultural peat soils in England. Geoderma 2010, 154, 196–202. [Google Scholar] [CrossRef]
- Liu, H.; Janssen, M.; Lennartz, B. Changes in flow and transport patterns in fen peat following soil degradation. Eur. J. Soil Sci. 2016, 67, 763–772. [Google Scholar] [CrossRef]
- Liu, H.; Forsmann, D.M.; Kjærgaard, C.; Saki, H.; Lennartz, B. Solute transport properties of fen peat differing in organic matter content. J. Environ. Qual. 2017, 46, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
| Pristine | Drained | Rewetted | ||
|---|---|---|---|---|
| Agricultural use | Nature conservation | Paludiculture | ||
| Water level | High | Low | High | High |
| Water level fluctuation | Low | Low | High | Medium |
| Biomass yield | None | High | None | Potentially high |
| Peat thickness | Increasing | Decreasing | Increasing | Stable? |
| Nutrient retention | High | Turns to source | Medium | Medium |
| CO2 emissions | Low or net sink | High | Low to medium | Low or net sink |
| CH4 emissions | Low | Low | High to low | High to low |
| N2O emissions | Low or net sink | Low to medium | Low? | Low? |
| Site ID | AD | AW | PD | PW | CD | CW |
|---|---|---|---|---|---|---|
| Decomposition after von Post [78] | H10 | H10 | H6–10 | H6–10 | H10 | H10 |
| Peat depth (cm) | 60 | 200 | 500 | 600 | 70 | 30 |
| pH | 4.4 ± 0.6 | 5.1 ± 0.2 | 5.3 ± 0 | 5.4 ± 0.3 | 4.2 ± 0.6 | 4.4 ± 0.5 |
| Dry bulk density (g cm−3) | 0.55 ± 0 | 0.16 ± 0 | 0.28 ± 0 | 0.19 ± 0 | 0.63 ± 0 | 0.57 ± 0 |
| NH4+-N (mg kg−1) | 23 ± 21 | 89 ± 70 | 40 ± 5 | 45 ± 18 | 11.1 ± 3.5 | 19.6 ± 7.2 |
| NO3−-N (mg kg−1)* | 11 ± 4 | 13 ± 11 | 10 ± 8 | 6 ± 5 | – | – |
| Total P (mg kg−1) | 632 ± 261 | 2072 ± 201 | 1102 ± 373 | 1059 ± 131 | 655 ± 100 | 757 ± 89 |
| Total K (mg kg−1) | 574 ± 15 | 487 ± 224 | 241 ± 137 | 511 ± 91 | 3014 ± 24 | 2285 ± 333 |
| Total Mg (mg kg−1) | 731 ± 140 | 1058 ± 40 | 790 ± 67 | 2031 ± 879 | 3162 ± 526 | 2531 ± 259 |
| Year | AD | AW | PD | PW | C | |
|---|---|---|---|---|---|---|
| Temperature anomaly [°C] | 2017 | +0.73 | +0.72 | +0.76 | +0.79 | +0.70 |
| 2018 | +1.34 | +1.36 | +1.31 | +1.33 | +1.25 | |
| 2019 | +1.58 | +1.59 | +1.63 | +1.64 | +1.72 | |
| Precipitation anomaly [mm] | 2017 | +197 | +202 | +187 | +170 | +119 |
| 2018 | −149 | −153 | −126 | −116 | −155 | |
| 2019 | +4 | −4 | +5 | −3 | −44 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jurasinski, G.; Ahmad, S.; Anadon-Rosell, A.; Berendt, J.; Beyer, F.; Bill, R.; Blume-Werry, G.; Couwenberg, J.; Günther, A.; Joosten, H.; et al. From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach. Soil Syst. 2020, 4, 14. https://doi.org/10.3390/soilsystems4010014
Jurasinski G, Ahmad S, Anadon-Rosell A, Berendt J, Beyer F, Bill R, Blume-Werry G, Couwenberg J, Günther A, Joosten H, et al. From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach. Soil Systems. 2020; 4(1):14. https://doi.org/10.3390/soilsystems4010014
Chicago/Turabian StyleJurasinski, Gerald, Sate Ahmad, Alba Anadon-Rosell, Jacqueline Berendt, Florian Beyer, Ralf Bill, Gesche Blume-Werry, John Couwenberg, Anke Günther, Hans Joosten, and et al. 2020. "From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach" Soil Systems 4, no. 1: 14. https://doi.org/10.3390/soilsystems4010014
APA StyleJurasinski, G., Ahmad, S., Anadon-Rosell, A., Berendt, J., Beyer, F., Bill, R., Blume-Werry, G., Couwenberg, J., Günther, A., Joosten, H., Koebsch, F., Köhn, D., Koldrack, N., Kreyling, J., Leinweber, P., Lennartz, B., Liu, H., Michaelis, D., Mrotzek, A., ... Wrage-Mönnig, N. (2020). From Understanding to Sustainable Use of Peatlands: The WETSCAPES Approach. Soil Systems, 4(1), 14. https://doi.org/10.3390/soilsystems4010014






