Methane Emissions from a Grassland-Wetland Complex in the Southern Peruvian Andes
Abstract
1. Introduction
2. Materials and Methods
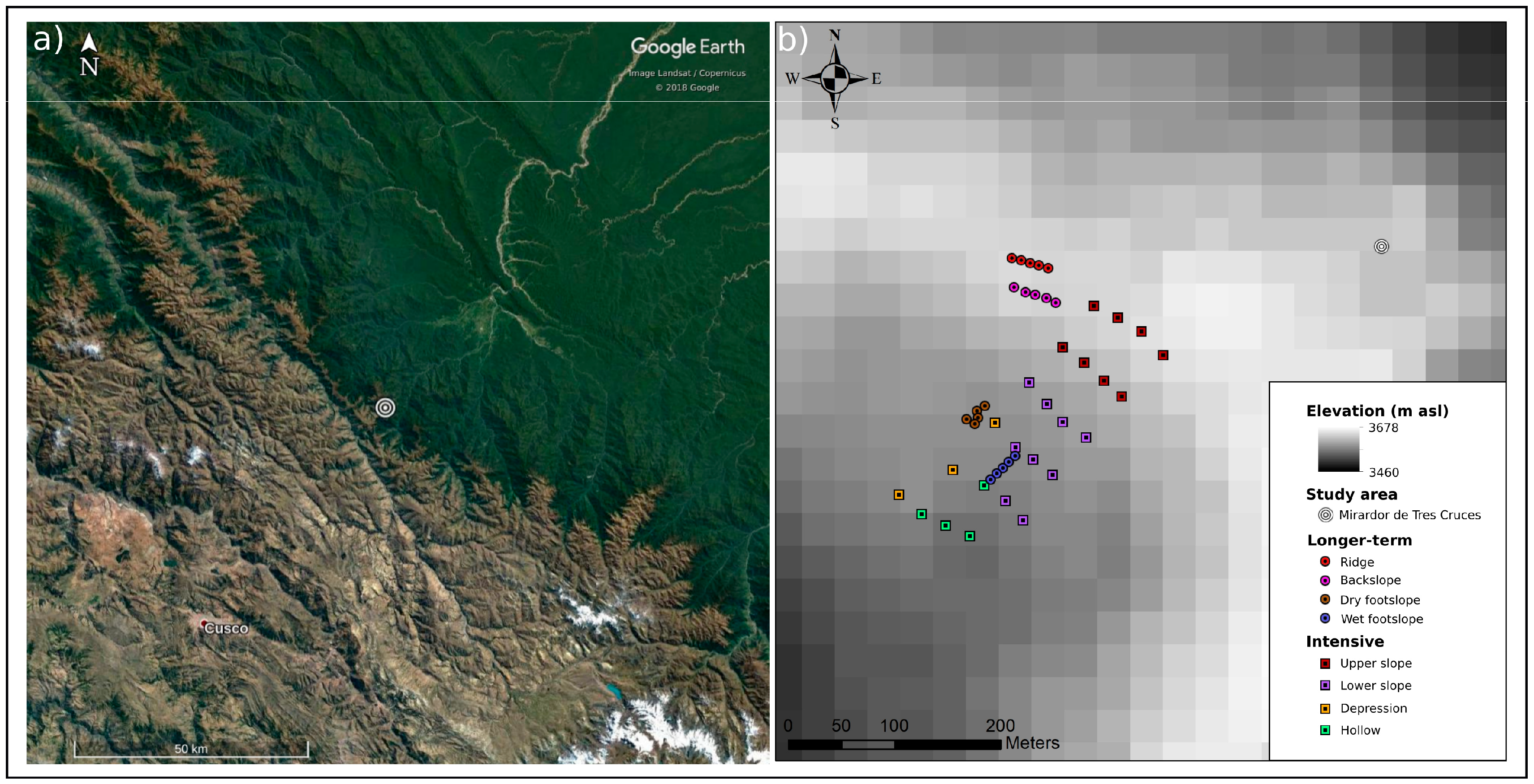
2.1. Study Sites
2.2. Sampling Approach
2.3. Soil–Atmosphere Gas Exchange and Environmental Conditions
2.4. Site Characterisation
2.5. Laboratory Incubations
2.6. Data Processing and Statistics
3. Results
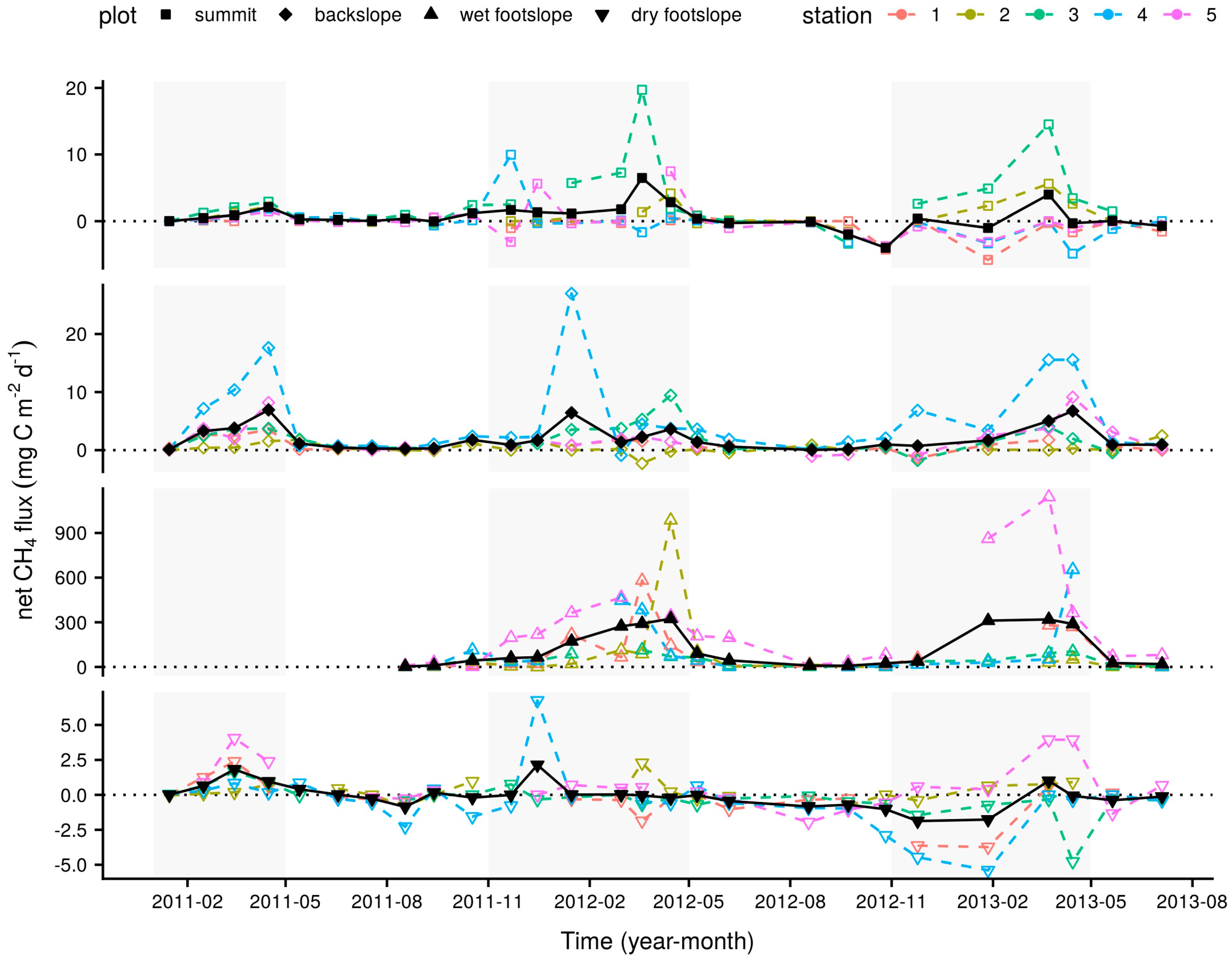
3.1. Longer-Term Measurements
3.2. Intensive Measurements
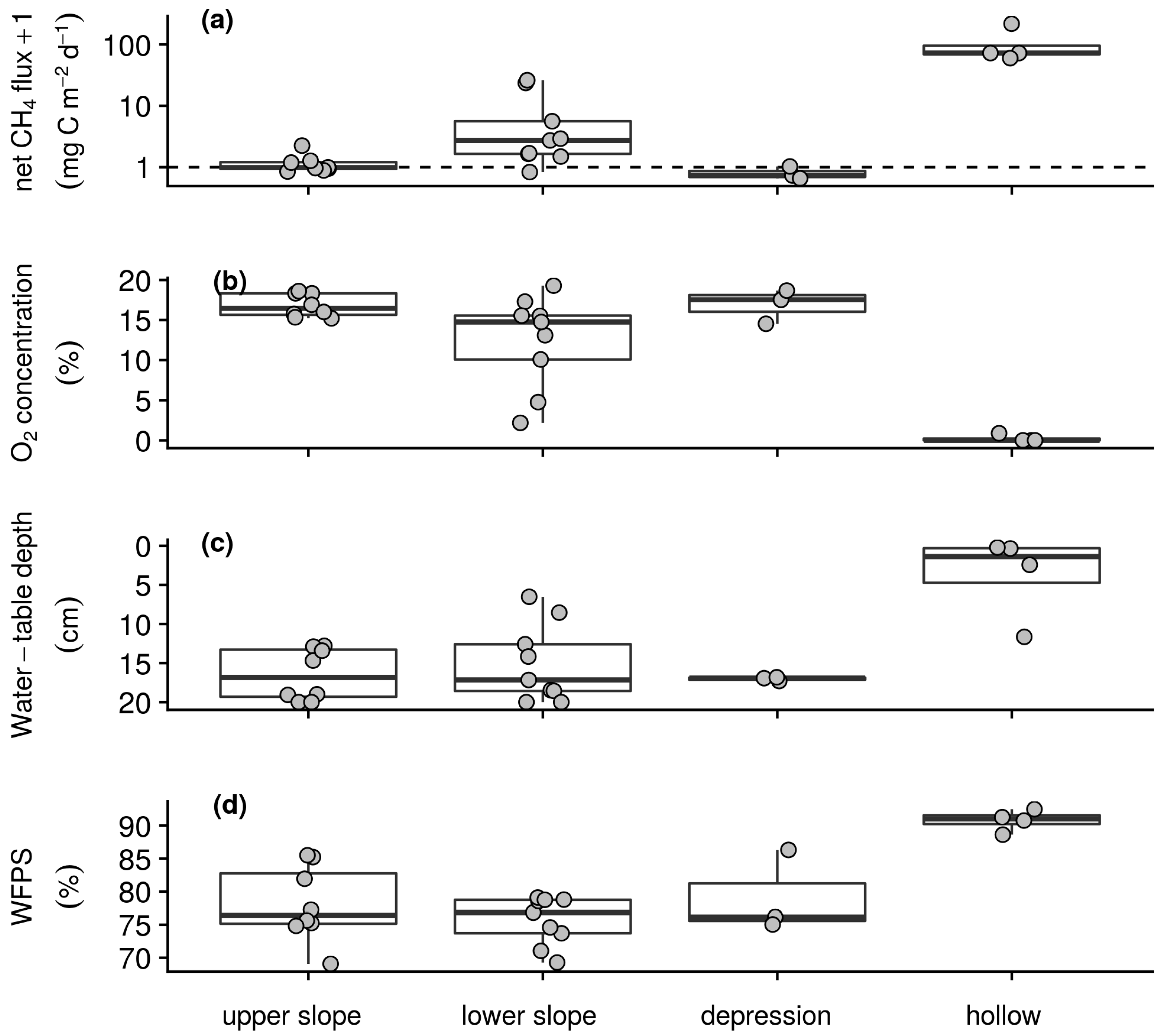
3.2.1. Site Characteristics
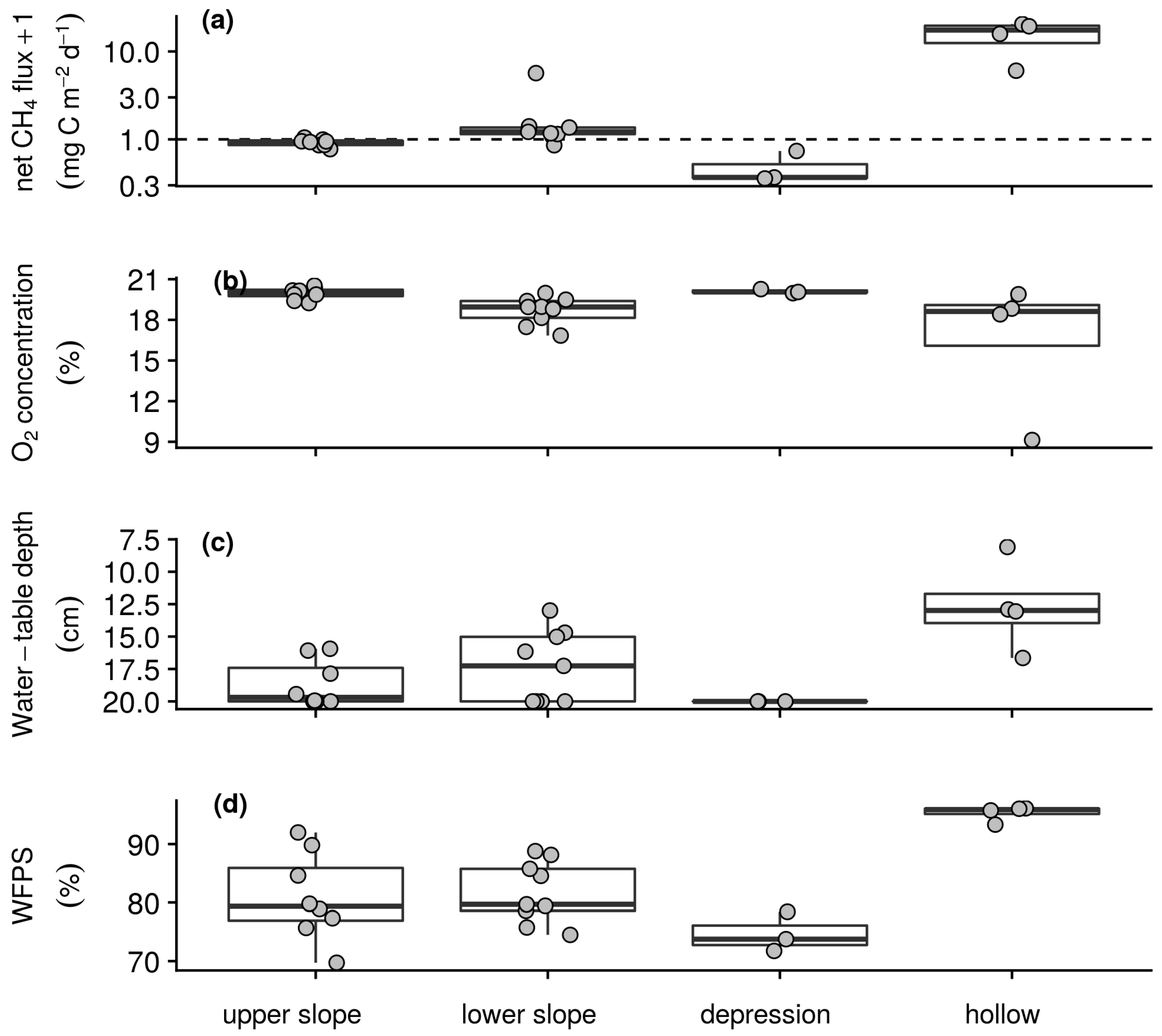
3.2.2. Soil–Atmosphere Gas Exchange and Environmental Conditions
3.3. Relationships between Soil–Atmosphere Gas Exchanges and Environmental Conditions
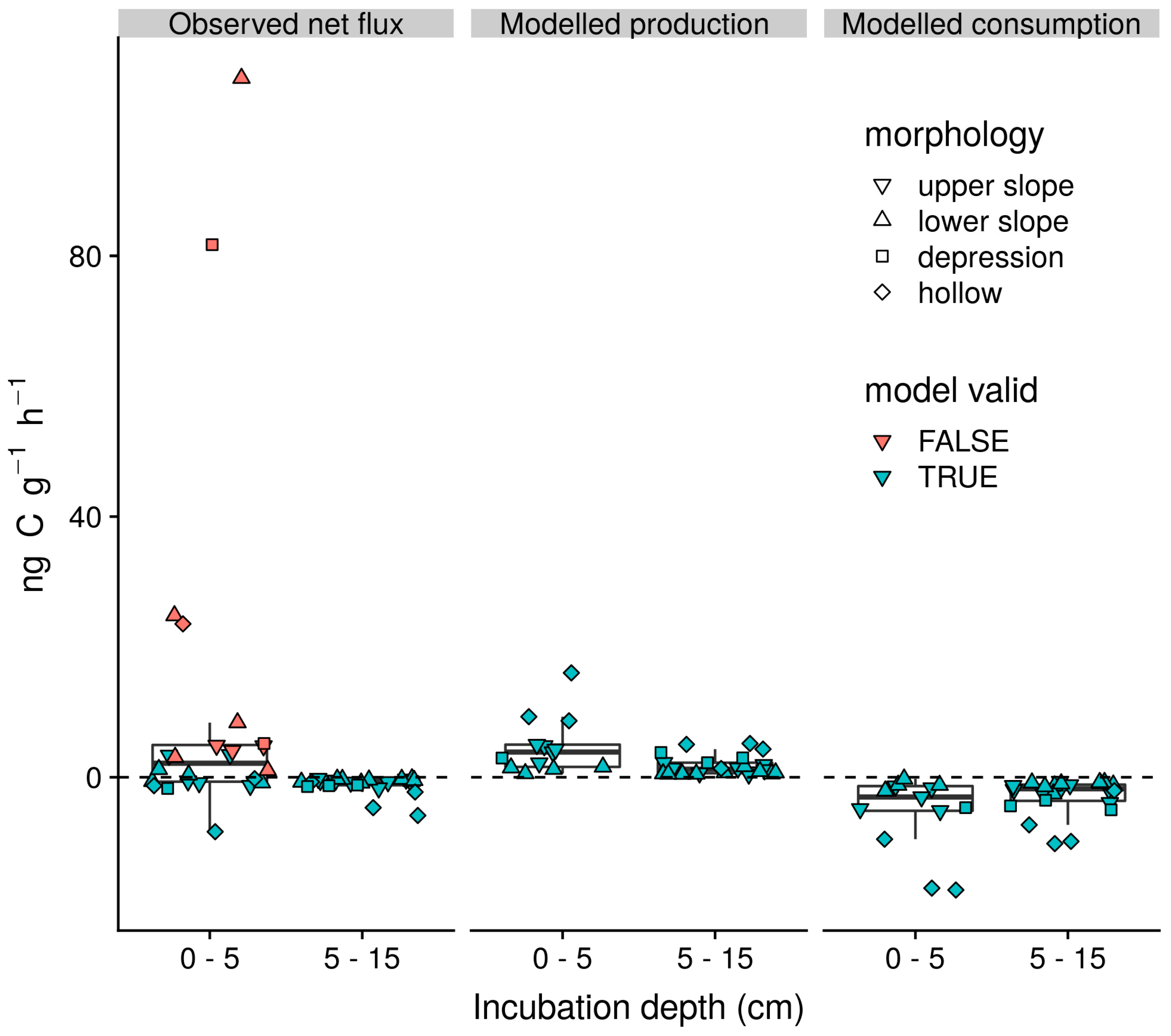
3.4. Gross Rates of Production and Consumption
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Frankenberg, C.; Meirink, J.F.; Weele, M.V.; Platt, U.; Wagner, T. Assessing methane emissions from global space-borne observations. Science 2005, 308, 1010–1014. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, P.; Frankenberg, C.; Meirink, J.F.; Krol, M.; Villani, M.G.; Houweling, S.; Dentener, F.; Dlugokencky, E.J.; Miller, J.B.; Gatti, L.V.; et al. Inverse modeling of global and regional CH4 emissions using SCIAMACHY satellite retrievals. J. Geophys. Res. Atmos. 2009, 114. [Google Scholar] [CrossRef]
- Frankenberg, C.; Aben, I.; Bergamaschi, P.; Dlugokencky, E.J.; Hees, R.V.; Houweling, S.; Meer, P.V.D.; Snel, R.; Tol, P. Global column-averaged methane mixing ratios from 2003 to 2009 as derived from SCIAMACHY: Trends and variability. J. Geophys. Res. Atmos. 2011, 116. [Google Scholar] [CrossRef]
- Mikaloff Fletcher, S.E.; Tans, P.P.; Bruhwiler, L.M.; Miller, J.B.; Heimann, M. CH4 sources estimated from atmospheric observations of CH4 and its 13C/12C isotopic ratios: 1. Inverse modeling of source processes. Glob. Biogeochem. Cycles 2004, 18. [Google Scholar] [CrossRef]
- Mikaloff Fletcher, S.E.; Tans, P.P.; Bruhwiler, L.M.; Miller, J.B.; Heimann, M. CH4 sources estimated from atmospheric observations of CH4 and its 13C/12C isotopic ratios: 2. Inverse modeling of CH4 fluxes from geographical regions. Glob. Biogeochem. Cycles 2004, 18. [Google Scholar] [CrossRef]
- Bloom, A.A.; Palmer, P.I.; Fraser, A.; Reay, D.S.; Frankenberg, C. Large-scale controls of methanogenesis inferred from methane and gravity spaceborne data. Science 2010, 327, 322–325. [Google Scholar] [CrossRef]
- Melack, J.M.; Hess, L.L.; Gastil, M.; Forsberg, B.R.; Hamilton, S.K.; Lima, I.B.; Novo, E.M. Regionalization of methane emissions in the Amazon Basin with microwave remote sensing. Glob. Chang. Biol. 2004, 10, 530–544. [Google Scholar] [CrossRef]
- Ringeval, B.; de Noblet-Ducoudré, N.; Ciais, P.; Bousquet, P.; Prigent, C.; Papa, F.; Rossow, W.B. An attempt to quantify the impact of changes in wetland extent on methane emissions on the seasonal and interannual time scales. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef]
- Bloom, A.A.; Palmer, P.I.; Fraser, A.; Reay, D.S. Seasonal variability of tropical wetland methane emissions: The role of the methanogen-available carbon pool. Biogeosciences 2012, 9, 2821–2830. [Google Scholar] [CrossRef]
- Pangala, S.R.; Moore, S.; Hornibrook, E.R.C.; Gauci, V. Trees are major conduits for methane egress from tropical forested wetlands. New Phytol. 2013, 197, 524–531. [Google Scholar] [CrossRef]
- Pangala, S.R.; Enrich-Prast, A.; Basso, L.S.; Peixoto, R.B.; Bastviken, D.; Hornibrook, E.R.C.; Gatti, L.V.; Marotta, H.; Calazans, L.S.B.; Sakuragui, C.M.; et al. Large emissions from floodplain trees close the Amazon methane budget. Nature 2017, 552, 230. [Google Scholar] [CrossRef] [PubMed]
- Teh, Y.A.; Silver, W.L.; Conrad, M.E. Oxygen effects on methane production and oxidation in humid tropical forest soils. Glob. Chang. Biol. 2005, 11, 1283–1297. [Google Scholar] [CrossRef]
- Spahni, R.; Wania, R.; Neef, L.; van Weele, M.; Pison, I.; Bousquet, P.; Frankenberg, C.; Foster, P.N.; Joos, F.; Prentice, I.C.; et al. Constraining global methane emissions and uptake by ecosystems. Biogeosciences 2011, 8, 1643–1665. [Google Scholar] [CrossRef]
- Hall, S.J.; McDowell, W.H.; Silver, W.L. When wet gets wetter: Decoupling of moisture, redox biogeochemistry, and greenhouse gas fluxes in a humid tropical forest soil. Ecosystems 2013, 16, 576–589. [Google Scholar] [CrossRef]
- Keppler, F.; Hamilton, J.T.; Braß, M.; Röckmann, T. Methane emissions from terrestrial plants under aerobic conditions. Nature 2006, 439, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Bloom, A.A.; Lee-Taylor, J.; Madronich, S.; Messenger, D.J.; Palmer, P.I.; Reay, D.S.; McLeod, A.R. Global methane emission estimates from ultraviolet irradiation of terrestrial plant foliage. New Phytol. 2010, 187, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Martinson, G.O.; Werner, F.A.; Scherber, C.; Conrad, R.; Corre, M.D.; Flessa, H.; Wolf, K.; Klose, M.; Gradstein, S.R.; Veldkamp, E. Methane emissions from tank bromeliads in neotropical forests. Nat. Geosci. 2010, 3, 766–769. [Google Scholar] [CrossRef]
- Wania, R.; Jolleys, M.; Buytaert, W. A previously neglected methane source from the Andean paramo? iLEAPS Newslett. 2009, 7, 58–59. [Google Scholar]
- Teh, Y.A.; Diem, T.; Jones, S.; Huaraca Quispe, L.P.; Baggs, E.; Morley, N.; Richards, M.; Smith, P.; Meir, P. Methane and nitrous oxide fluxes across an elevation gradient in the tropical Peruvian Andes. Biogeosciences 2014, 11, 2325–2339. [Google Scholar] [CrossRef]
- Veber, G.; Kull, A.; Villa, J.A.; Maddison, M.; Paal, J.; Oja, T.; Iturraspe, R.; Pärn, J.; Teemusk, A.; Mander, Ü. Greenhouse gas emissions in natural and managed peatlands of America: Case studies along a latitudinal gradient. Ecol. Eng. 2017. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Fonseca, G.A.D.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Tovar, C.; Arnillas, C.A.; Cuesta, F.; Buytaert, W. Diverging responses of tropical Andean biomes under future climate conditions. PLoS ONE 2013, 8, e63634. [Google Scholar] [CrossRef] [PubMed]
- Josse, C.; Cuesta, F.; Navarro, G.; Barrena, V.; Cabrera, E.; Chacon-Moreno, E.; Ferreira, W.; Peralvo, M.; Saito, J.; Tovar, A. Ecosistemas de los Andes del norte y centro Bolivia, Colombia, Ecuador, Peru y Venezuela. In Secretaría General de la Comunidad Andina, Programa Regional ECOBONA-Intercooperation, CONDESAN-Proyecto Páramo Andino, Programa BioAndes, EcoCiencia, NatureServe, IAvH, LTAUNALM, ICAE-ULA, CDC-UNALM, and RUMBOL SRL; Secretaria General de la Comidad Andina and Partners: Lima, Peru, 2009. [Google Scholar]
- Josse, C.; Cuesta, F.; Navarro, G.; Barrena, V.; Cabrera, E.; Chacon-Moreno, E.; Ferreira, W.; Peralvo, M.; Saito, J.; Tovar, A. Mapa de ecosistemas de los Andes del norte y centro, Bolivia, Colombia, Ecuador, Peru y Venezuela. In Secretaría General de la Comunidad Andina, Programa Regional ECOBONA-Intercooperation, CONDESAN-Proyecto Páramo Andino, Programa BioAndes, EcoCiencia, NatureServe, IAvH, LTAUNALM, ICAE-ULA, CDC-UNALM, and RUMBOL SRL; Secretaria General de la Comidad Andina and Partners: Lima, Peru, 2009. [Google Scholar]
- Josse, C.; Cuesta, F.; Navarro, G.; Barrena, V.; Becerra, M.T.; Cabrera, E.; Chacón-Moreno, E.; Ferreira, W.; Peralvo, M.; Saito, J.; et al. Physical geography and ecosystems in the tropical Andes. In Climate Change and Biodiversity in the Tropical Andes, Inter-American Institute for Global Change Research (IAI) and Scientific Committee on Problems of the Environment (SCOPE); 2011; pp. 152–169. Available online: http://www.iai.int/cambio-climatico-y-biodiversidad-en-los-andes-tropicales/ (accessed on 1 June 2018).
- Luteyn, J.L.; Churchil, S.P.; Griffin, D., III; Gradstein, S.R.; Sipman, H.J.M.; Gavilanes, A. Páramos: A Checklist of Plant Diversity, Geographical Distribution, and Botanical Literature; New York Botanical Garden Press: New York, NY, USA, 1999. [Google Scholar]
- Miller, D.C.; Birkeland, P.W. Soil catena variation along an alpine climatic transect, northern Peruvian Andes. Geoderma 1992, 55, 211–223. [Google Scholar] [CrossRef]
- Hofstede, R.G. The effects of grazing and burning on soil and plant nutrient concentrations in Colombian páramo grasslands. Plant Soil 1995, 173, 111–132. [Google Scholar] [CrossRef]
- Zimmermann, M.; Meir, P.; Silman, M.R.; Fedders, A.; Gibbon, A.; Malhi, Y.; Urrego, D.H.; Bush, M.B.; Feeley, K.J.; Garcia, K.C.; et al. No Differences in Soil Carbon Stocks Across the Tree Line in the Peruvian Andes. Ecosystems 2010, 13, 62–74. [Google Scholar] [CrossRef]
- Turetsky, M.R.; Kotowska, A.; Bubier, J.; Dise, N.B.; Crill, P.; Hornibrook, E.R.; Minkkinen, K.; Moore, T.R.; Myers-Smith, I.H.; Nykänen, H.; et al. A synthesis of methane emissions from 71 northern, temperate, and subtropical wetlands. Glob. Chang. Biol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Waddington, J.M.; Roulet, N.T. Atmosphere-wetland carbon exchanges: Scale dependency of carbon dioxide and methane exchange on the developmental topography of a peatland. Glob. Biogeochem. Cycles 1996, 10, 233–245. [Google Scholar] [CrossRef]
- Conrad, R. Soil microorganisms as controllers of atmospheric trace gases (hydrogen, carbon monoxide, methane, carbonyl sulfide, nitrous oxide, and nitric oxide). Microbiol. Rev. 1996, 60, 609–640. [Google Scholar]
- Zinder, S.H. Physiological ecology of methanogens. In Methanogenesis; Springer: Boston, MA, USA, 1993; pp. 128–206. [Google Scholar]
- Conrad, R. Contribution of hydrogen to methane production and control of hydrogen concentrations in methanogenic soils and sediments. FEMS Microbiol. Ecol. 1999, 28, 193–202. [Google Scholar] [CrossRef]
- Le Mer, J.; Roger, P. Production, oxidation, emission and consumption of methane by soils: A review. Eur. J. Soil Biol. 2001, 37, 25–50. [Google Scholar] [CrossRef]
- Achtnich, C.; Bak, F.; Conrad, R. Competition for electron donors among nitrate reducers, ferric iron reducers, sulfate reducers, and methanogens in anoxic paddy soil. Biol. Fertility Soils 1995, 19, 65–72. [Google Scholar] [CrossRef]
- Chidthaisong, A.; Conrad, R. Turnover of glucose and acetate coupled to reduction of nitrate, ferric iron and sulfate and to methanogenesis in anoxic rice field soil. FEMS Microbiol. Ecol. 2000, 31, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Hanson, R.S.; Hanson, T.E. Methanotrophic bacteria. Microbiol. Rev. 1996, 60, 439–471. [Google Scholar] [PubMed]
- Bender, M.; Conrad, R. Kinetics of methane oxidation in oxic soils exposed to ambient air or high methane mixing ratios. FEMS Microbiol. Lett. 1992, 101, 261–269. [Google Scholar] [CrossRef]
- Reay, D.S.; Radajewski, S.; Murrell, J.C.; McNamara, N.; Nedwell, D.B. Effects of land-use on the activity and diversity of methane oxidizing bacteria in forest soils. Soil Biol. Biochem. 2001, 33, 1613–1623. [Google Scholar] [CrossRef]
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, J.; Chhabra, A.; DeFries, R.; Galloway, J.; Heimann, M.; et al. Carbon and Other Biogeochemical Cycles. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; pp. 465–570. ISBN 978-1-107-66182-0. [Google Scholar]
- Whiting, G.J.; Chanton, J.P. Primary production control of methane emission from wetlands. Nature 1993, 364, 794–795. [Google Scholar] [CrossRef]
- Watanabe, A.; Takeda, T.; Kimura, M. Evaluation of origins of CH4 carbon emitted from rice paddies. J. Geophys. Res. Atmos. 1999, 104, 23623–23629. [Google Scholar] [CrossRef]
- Frenzel, P.; Karofeld, E. Methane emission from a hollow-ridge complex in a raised bog: The role of methane production and oxidation. Biogeochemistry 2000, 51, 91–112. [Google Scholar] [CrossRef]
- Hornibrook, E.R.C.; Bowes, H.L.; Culbert, A.; Gallego-Sala, A.V. Methanotrophy potential versus methane supply by pore water diffusion in peatlands. Biogeosciences 2009, 6, 1491–1504. [Google Scholar] [CrossRef]
- Schimel, J.P. Plant transport and methane production as controls on methane flux from arctic wet meadow tundra. Biogeochemistry 1995, 28, 183–200. [Google Scholar] [CrossRef]
- Shannon, R.D.; White, J.R.; Lawson, J.E.; Gilmour, B.S. Methane efflux from emergent vegetation in peatlands. J. Ecol. 1996, 239–246. [Google Scholar] [CrossRef]
- Terazawa, K.; Ishizuka, S.; Sakata, T.; Yamada, K.; Takahashi, M. Methane emissions from stems of Fraxinus mandshurica var. japonica trees in a floodplain forest. Soil Biol. Biochem. 2007, 39, 2689–2692. [Google Scholar] [CrossRef]
- Bender, M.; Conrad, R. Methane oxidation activity in various soils and freshwater sediments: Occurrence, characteristics, vertical profiles, and distribution on grain size fractions. J. Geophys. Res. Atmos. 1994, 99, 16531–16540. [Google Scholar] [CrossRef]
- Teh, Y.A.; Silver, W.L.; Conrad, M.E.; Borglin, S.E.; Carlson, C.M. Carbon isotope fractionation by methane-oxidizing bacteria in tropical rain forest soils. J. Geophys. Res. 2006, 111, G02001. [Google Scholar] [CrossRef]
- Smith, K.A.; Dobbie, K.E.; Ball, B.C.; Bakken, L.R.; Sitaula, B.K.; Hansen, S.; Brumme, R.; Borken, W.; Christensen, S.; Priemé, A.; et al. Oxidation of atmospheric methane in Northern European soils, comparison with other ecosystems, and uncertainties in the global terrestrial sink. Glob. Chang. Biol. 2000, 6, 791–803. [Google Scholar] [CrossRef]
- Verchot, L.V.; Davidson, E.A.; Cattânio, J.H.; Ackerman, I.L. Land-use change and biogeochemical controls of methane fluxes in soils of eastern Amazonia. Ecosystems 2000, 3, 41–56. [Google Scholar] [CrossRef]
- Bodelier, P.L.E.; Laanbroek, H.J. Nitrogen as a regulatory factor of methane oxidation in soils and sediments. FEMS Microbiol. Ecol. 2004, 47, 265–277. [Google Scholar] [CrossRef]
- Kolb, S. The quest for atmospheric methane oxidizers in forest soils. Environ. Microbiol. Rep. 2009, 1, 336–346. [Google Scholar] [CrossRef]
- Peters, V.; Conrad, R. Sequential reduction processes and initiation of methane production upon flooding of oxic upland soils. Soil Biol. Biochem. 1996, 28, 371–382. [Google Scholar] [CrossRef]
- Angel, R.; Claus, P.; Conrad, R. Methanogenic archaea are globally ubiquitous in aerated soils and become active under wet anoxic conditions. ISME J. 2012, 6, 847–862. [Google Scholar] [CrossRef]
- Von Fischer, J.C.; Hedin, L.O. Separating methane production and consumption with a field-based isotope pool dilution technique. Glob. Biogeochem. Cycles 2002, 16. [Google Scholar] [CrossRef]
- Silver, W.L.; Lugo, A.E.; Keller, M. Soil oxygen availability and biogeochemistry along rainfall and topographic gradients in upland wet tropical forest soils. Biogeochemistry 1999, 44, 301–328. [Google Scholar] [CrossRef]
- Yang, W.H.; McNicol, G.; Teh, Y.A.; Estera-Molina, K.; Wood, T.E.; Silver, W.L. Evaluating the Classical Versus an Emerging Conceptual Model of Peatland Methane Dynamics. Glob. Biogeochem. Cycles 2017, 31, 1435–1453. [Google Scholar] [CrossRef]
- Angle, J.C.; Morin, T.H.; Solden, L.M.; Narrowe, A.B.; Smith, G.J.; Borton, M.A.; Rey-Sanchez, C.; Daly, R.A.; Mirfenderesgi, G.; Hoyt, D.W.; et al. Methanogenesis in oxygenated soils is a substantial fraction of wetland methane emissions. Nat. Commun. 2017, 8, 1567. [Google Scholar] [CrossRef]
- Von Fischer, J.C.; Hedin, L.O. Controls on soil methane fluxes: Tests of biophysical mechanisms using stable isotope tracers. Glob. Biogeochem. Cycles 2007, 21. [Google Scholar] [CrossRef]
- Sexstone, A.J.; Revsbech, N.P.; Parkin, T.B.; Tiedje, J.M. Direct measurement of oxygen profiles and denitrification rates in soil aggregates. Soil Sci. Soc. Am. J. 1985, 49, 645–651. [Google Scholar] [CrossRef]
- Knief, C.; Kolb, S.; Bodelier, P.L.E.; Lipski, A.; Dunfield, P.F. The active methanotrophic community in hydromorphic soils changes in response to changing methane concentration. Environ. Microbiol. 2005, 8, 321–333. [Google Scholar] [CrossRef]
- Yang, W.H.; Silver, W.L. Net soil–atmosphere fluxes mask patterns in gross production and consumption of nitrous oxide and methane in a managed ecosystem. Biogeosciences 2016, 13, 1705–1715. [Google Scholar] [CrossRef]
- Gibbon, A.; Silman, M.R.; Malhi, Y.; Fisher, J.B.; Meir, P.; Zimmermann, M.; Dargie, G.C.; Farfan, W.R.; Garcia, K.C. Ecosystem carbon storage across the grassland–forest transition in the high Andes of Manu National Park, Peru. Ecosystems 2010, 13, 1097–1111. [Google Scholar] [CrossRef]
- Oliveras, I.; Anderson, L.O.; Malhi, Y. Application of remote sensing to understanding fire regimes and biomass burning emissions of the tropical Andes. Glob. Biogeochem. Cycles 2014, 28, 480–496. [Google Scholar] [CrossRef]
- Oliveras, I.; Eynden, M.; Malhi, Y.; Cahuana, N.; Menor, C.; Zamora, F.; Haugaasen, T. Grass allometry and estimation of above-ground biomass in tropical alpine tussock grasslands. Austral Ecol. 2014, 39, 408–415. [Google Scholar] [CrossRef]
- Girardin, C.A.J.; Malhi, Y.; Aragao, L.; Mamani, M.; Huasco, W.H.; Durand, L.; Feeley, K.J.; Rapp, J.; SILVA-ESPEJO, J.; Silman, M.; et al. Net primary productivity allocation and cycling of carbon along a tropical forest elevational transect in the Peruvian Andes. Glob. Chang. Biol. 2010, 16, 3176–3192. [Google Scholar] [CrossRef]
- Jones, S.P.; Diem, T.; Huaraca Quispe, L.P.; Cahuana, A.J.; Reay, D.S.; Meir, P.; Teh, Y.A. Drivers of atmospheric methane uptake by montane forest soils in the southern Peruvian Andes. Biogeosciences 2016, 13, 4151–4165. [Google Scholar] [CrossRef]
- Miller, B.A.; Schaetzl, R.J. Digital Classification of Hillslope Position. Soil Sci. Soc. Am. J. 2015, 79, 132–145. [Google Scholar] [CrossRef]
- Varner, R.K.; Keller, M.; Robertson, J.R.; Dias, J.D.; Silva, H.; Crill, P.M.; McGroddy, M.; Silver, W.L. Experimentally induced root mortality increased nitrous oxide emission from tropical forest soils. Geophys. Res. Lett. 2003, 30, 1144. [Google Scholar] [CrossRef]
- Sparks, D.L.; Page, A.L.; Helmke, P.A.; Loeppert, R.H.; Soltanpour, P.N.; Tabatabai, M.A.; Johnston, C.T.; Sumner, M.E. Methods of Soil Analysis. Part 3-Chemical Methods; Soil Science Society of America Inc.: Madison, WI, USA, 1996. [Google Scholar]
- Klute, A. Methods of Soil Analysis: Part 1—Physical and Mineralogical Methods; SSSA Book Series; Soil Science Society of America, American Society of Agronomy: Madison, WI, USA, 1986; ISBN 978-0-89118-864-3. [Google Scholar]
- Yang, W.H.; Teh, Y.A.; Silver, W.L. A test of a field-based 15N–nitrous oxide pool dilution technique to measure gross N2O production in soil. Glob. Chang. Biol. 2011, 17, 3577–3588. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Statistics for Biology and Health; Springer: New York, NY, USA, 2009; ISBN 978-0-387-87458-6. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models; 2014; Available online: https://CRAN.R-project.org/package=nlme (accessed on 1 June 2018).
- Zuur, A.F.; Ieno, E.N.; Smith, G.M. Analysing Ecological Data; Statistics for Biology and Health; Springer: New York, NY, USA, 2007; Volume 680, ISBN 978-0-387-45972-1. [Google Scholar]
- Wolf, K.; Flessa, H.; Veldkamp, E. Atmospheric methane uptake by tropical montane forest soils and the contribution of organic layers. Biogeochemistry 2012, 111, 469–483. [Google Scholar] [CrossRef]
- Palm, C.A.; Alegre, J.C.; Arevalo, L.; Mutuo, P.K.; Mosier, A.R.; Coe, R. Nitrous oxide and methane fluxes in six different land use systems in the Peruvian Amazon. Glob. Biogeochem. Cycles 2002, 16. [Google Scholar] [CrossRef]
- Teh, Y.A.; Murphy, W.A.; Berrio, J.-C.; Boom, A.; Page, S.E. Seasonal variability in methane and nitrous oxide fluxes from tropical peatlands in the western Amazon basin. Biogeosciences 2017, 14, 3669–3683. [Google Scholar] [CrossRef]
- Winton, R.S.; Flanagan, N.; Richardson, C.J. Neotropical peatland methane emissions along a vegetation and biogeochemical gradient. PLoS ONE 2017, 12, e0187019. [Google Scholar] [CrossRef]
- McNamara, N.P.; Plant, T.; Oakley, S.; Ward, S.; Wood, C.; Ostle, N. Gully hotspot contribution to landscape methane and carbon dioxide fluxes in a northern peatland. Sci. Total Environ. 2008, 404, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Teh, Y.A.; Silver, W.L.; Sonnentag, O.; Detto, M.; Kelly, M.; Baldocchi, D.D. Large greenhouse gas emissions from a temperate peatland pasture. Ecosystems 2011, 14, 311–325. [Google Scholar] [CrossRef]
- Teh, Y.A.; Dubinsky, E.A.; Silver, W.L.; Carlson, C.M. Suppression of methanogenesis by dissimilatory iron (III)-reducing bacteria in tropical rain forest soils: Implications for ecosystem methane flux. Glob. Chang. Biol. 2008, 14, 413–422. [Google Scholar] [CrossRef]
- Dubinsky, E.A.; Silver, W.L.; Firestone, M.K. Tropical forest soil microbial communities couple iron and carbon biogeochemistry. Ecology 2010, 91, 2604–2612. [Google Scholar] [CrossRef] [PubMed]
| Plot | Season | Net CH4 Flux (mg C m−2 d−1) | Net CO2 Flux (g C m−2 d−1) | O2 Concentration (%) | VWC (%) | Soil Temperature (°C) |
|---|---|---|---|---|---|---|
| summit | dry | −0.4 (0.36) a | 1.0 (0.15) a | 17.6 (0.81) a | 72.3 (0.74) a | 9.3 (0.33) a |
| summit | wet | 1.6 (0.51) b | 1.2 (0.14) a | 15.5 (0.93) a | 72.7 (0.61) a | 11.6 (0.24) b |
| backslope | dry | 0.7 (0.15) a | 1.3 (0.13) a | 15.3 (1.10) a | 74.0 (0.61) a | 8.9 (0.42) a |
| backslope | wet | 3.2 (0.62) b | 1.7 (0.34) a | 11.7 (2.06) a | 74.0 (0.34) a | 11.7 (0.28) b |
| wet footslope | dry | 27.8 (8.30) a | 2.1 (0.30) a | 9.7 (2.69) a | 76.2 (0.24) a | 9.5 (0.38) a |
| wet footslope | wet | 214.2 (37.46) b | 1.9 (0.21) a | 3.8 (3.82) a | 75.6 (0.31) a | 12.1 (0.36) b |
| dry footslope | dry | −0.3 (0.12) a | 2.3 (0.35) a | 15.7 (1.06) a | 72.6 (0.77) a | 9.6 (0.33) a |
| dry footslope | wet | 0.2 (0.30) a | 1.9 (0.36) a | 9.6 (1.41) b | 74.3 (0.35) a | 11.7 (0.29) b |
| Plot | Net CO2 Flux | O2 Concentration | VWC | Soil Temperature |
|---|---|---|---|---|
| Summit | 0.56 (27) ** | −0.31 (23) | 0.12 (25) | 0.54 (27) ** |
| Backslope | 0.15 (27) | −0.44 (23) * | 0.31 (26) | 0.53 (27) ** |
| Wet footslope | −0.17 (20) | −0.61 (10) | −0.06 (19) | 0.64 (20) ** |
| Dry footslope | −0.55 (27) ** | −0.23 (23) | 0.31 (26) | 0.56 (26) ** |
| Morphology | Elevation (m asl) | Slope NW–SE (°) | Slope NE–SW (°) | Soil Depth (cm) | Total Grass Biomass (g m−2) | Calama-grostis Biomass (%) | Scirpus Biomass (%) | Juncus Biomass (%) |
|---|---|---|---|---|---|---|---|---|
| upper slope (n = 8) | 3663 (6) | −2 (2.8) | −14 (2.8) | 26 (3.6) | 286.2 (219.62) | 92.8 (5.5) | 5.5 (3.8) | 1.7 (3.6) |
| lower slope (n = 9) | 3641 (5) | −2 (3.6) | −10 (1.5) | 29 (6.4) | 161.4 (91.39) | 57.4 (26.1) | 18.3 (14.0) | 24.3 (32.0) |
| depression (n = 3) | 3634 (5) | 0 (0.6) | −2 (1.5) | 67 (23.5) | 150.4 (86.12) | 59.4 (52.0) | 6.3 (10.8) | 34.4 (56.9) |
| hollow (n = 4) | 3630 (4) | −1 (0.5) | −1 (1.4) | 63 (32.9) | 76.8 (16.25) | 0.0 (0.0) | 16.4 (11.1) | 83.6 (11.1) |
| Morphology | Depth (cm) | Bulk Density (g cm−3) | Particle Density (g cm−3) | C Content (%) | C:N | pH |
|---|---|---|---|---|---|---|
| upper slope (n = 8) | 0–5 | 0.16 (0.036) | 2.07 (0.101) | 21.3 (3.68) | 14.8 (0.66) | 4.3 (0.29) |
| lower slope (n = 9) | 0–5 | 0.15 (0.062) | 2.03 (0.131) | 20.8 (4.64) | 15.11 (1.36) | 4.1 (0.10) |
| depression (n = 3) | 0–5 | 0.07 (0.013) | 1.62 (0.045) | 38.5 (2.69) | 19.95 (5.80) | 4.7 (0.50) |
| hollow (n = 4) | 0–5 | 0.03 (0.013) | 1.82 (0.099) | 40.8 (1.40) | 27.6 (7.10) | 4.5 (0.70) |
| upper slope (n = 8) | 5–15 | 0.49 (0.108) | 2.30 (0.115) | 12.6 (3.84) | 12.3 (0.22) | 3.9 (0.16) |
| lower slope (n = 9) | 5–15 | 0.47 (0.155) | 2.34 (0.142) | 11.5 (4.07) | 11.6 (0.50) | 4.1 (0.23) |
| depression (n = 3) | 5–15 | 0.16 (0.030) | 1.68 (0.065) | 35.5 (1.38) | 14.1 (1.49) | 4.0 (0.20) |
| hollow (n = 4) | 5–15 | 0.10 (0.088) | 1.75 (0.153) | 37.3 (9.07) | 23.7 (10.80) | 4.0 (0.61) |
| Campaign | Net CO2 Flux | O2 Concentration | Water-Table Depth | WFPS |
|---|---|---|---|---|
| Wet season | 0.00 | −0.88 ** | −0.45 * | 0.50 * |
| Dry season | 0.11 | −0.76 ** | −0.56 ** | 0.62 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, S.P.; Diem, T.; Arn Teh, Y.; Salinas, N.; Reay, D.S.; Meir, P. Methane Emissions from a Grassland-Wetland Complex in the Southern Peruvian Andes. Soil Syst. 2019, 3, 2. https://doi.org/10.3390/soilsystems3010002
Jones SP, Diem T, Arn Teh Y, Salinas N, Reay DS, Meir P. Methane Emissions from a Grassland-Wetland Complex in the Southern Peruvian Andes. Soil Systems. 2019; 3(1):2. https://doi.org/10.3390/soilsystems3010002
Chicago/Turabian StyleJones, Sam P., Torsten Diem, Yit Arn Teh, Norma Salinas, Dave S. Reay, and Patrick Meir. 2019. "Methane Emissions from a Grassland-Wetland Complex in the Southern Peruvian Andes" Soil Systems 3, no. 1: 2. https://doi.org/10.3390/soilsystems3010002
APA StyleJones, S. P., Diem, T., Arn Teh, Y., Salinas, N., Reay, D. S., & Meir, P. (2019). Methane Emissions from a Grassland-Wetland Complex in the Southern Peruvian Andes. Soil Systems, 3(1), 2. https://doi.org/10.3390/soilsystems3010002




