Effects of Wildfire and the Presence of the Invasive Paulownia tomentosa on the Regeneration of Native Tree Species in North-Central Appalachia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Field Methods
2.2. Analysis of Stem Density Data
2.3. Analysis of Height Data
3. Results
3.1. Fire Effects on Paulownia
3.2. Fire and Paulownia Effects on the Number of Regenerating Stems
3.3. Fire and Paulownia Effects on Average Height of Regenerating Stems
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clewley, G.D.; Eschen, R.; Shaw, R.H.; Wright, D.J. The effectiveness of classical biological control of invasive plants. J. Appl. Ecol. 2012, 49, 1287–1295. [Google Scholar] [CrossRef]
- Mack, R.N.; Simberloff, D.; Lonsdale, W.M.; Evans, H.; Clout, M.; Bazzaz, F.A. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 2000, 10, 689–710. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Dantonio, C.M.; Loope, L.L.; Rejmanek, M.; Westbrooks, R. Introduced species: A significant component of human-caused global change. New Zeal. J. Ecol. 1997, 21, 1–16. Available online: https://www.jstor.org/stable/24054520 (accessed on 6 August 2021).
- Vila, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarosik, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pysek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef]
- Bezemer, T.M.; Harvey, J.A.; Cronin, J.T. Response of native insect communities to invasive plants. Annu. Rev. Entomol. 2014, 59, 119–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blossey, B. Before, during and after: The need for long-term monitoring in invasive plant species management. Biol. Invasions 1999, 1, 301–311. [Google Scholar] [CrossRef]
- Zedler, J.B.; Kercher, S. Causes and consequences of invasive plants in wetlands: Opportunities, opportunists, and out-comes. Crit. Rev. Plant Sci. 2004, 23, 431–452. [Google Scholar] [CrossRef]
- Crawley, M.J.; Harvey, P.H.; Purvis, A. Comparative ecology of the native and alien floras of the British Isles. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1996, 351, 1251–1259. [Google Scholar] [CrossRef]
- Pimentel, D.; Zuniga, R.; Morrison, D. Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol. Econ. 2005, 52, 273–288. [Google Scholar] [CrossRef]
- Essl, F. From ornamental to detrimental? The incipient invasion of Central Europe by Paulownia tomentosa. Preslia 2007, 79, 377–389. [Google Scholar]
- Tang, R.C.; Carpenter, S.B.; Wittwer, R.F.; Graves, D.H. Paulownia—A crop tree for wood products and reclamation of surface-mined land. South J. Appl. For. 1980, 4, 19–24. [Google Scholar] [CrossRef]
- Preston, D. Paulownia: A miracle tree or passing fancy? Am. For. 1983, 89, 15–19, 47–52. [Google Scholar]
- Rejmanek, M.; Richardson, D.M. What attributes make some plant species more invasive? Ecology 1996, 77, 1655–1661. [Google Scholar] [CrossRef]
- Goodwin, B.J.; McAllister, A.J.; Fahrig, L. Predicting invasiveness of plant species based on biological information. Conserv. Biol. 1999, 13, 422–426. [Google Scholar] [CrossRef] [Green Version]
- Herron, P.M.; Martine, C.T.; Latimer, A.M.; Leicht-Young, S.A. Invasive plants and their ecological strategies: Prediction and explanation of woody plant invasion in New England. Divers. Distrib. 2007, 13, 633–644. [Google Scholar] [CrossRef]
- Martin, P.H.; Canham, C.D.; Marks, P.L. Why forests appear resistant to exotic plant invasions: Intentional introductions, stand dynamics, and the role of shade tolerance. Front Ecol. Environ. 2009, 7, 142–149. [Google Scholar] [CrossRef]
- Millsaps, V. The structure and development of the seed Paulownia tomentosa. J. Elisha Mitch. Sci. Soc. 1936, 52, 56–75. [Google Scholar]
- Carpenter, S.B.; Immel, M.J.; Smith, N.D. Effect of photoperiod on the growth and photosynthetic capacity of Paulownia seedlings. Castanea 1983, 48, 13–18. [Google Scholar]
- Kuppinger, D.M. Post-Fire Vegetation Dynamics and the Invasion of Paulownia tomentosa in the Southern Appalachians. Ph.D. Thesis, Univ. of North Carolina at Chapel Hill, Chapel Hill, NC, USA, 2008. [Google Scholar]
- Hu, S.Y. The economic botany of the Paulownias. Econ. Bot. 1961, 15, 11–27. [Google Scholar] [CrossRef]
- Langdon, K.R.; Johnson, K.D. Additional notes on invasiveness of Paulownia tomentosa in natural areas. Nat. Area J. 1994, 14, 139–140. [Google Scholar]
- Beckjord, P.R.; McIntosh, M.S. Paulownia tomentosa: Effects of fertilization and coppicing in plantation establishment. South J. Appl. For. 1983, 7, 81–85. [Google Scholar] [CrossRef]
- Kuppinger, D.M.; Jenkins, M.A.; White, P.S. Predicting the post-fire establishment and persistence of an invasive tree species across a complex landscape. Biol. Invasions 2010, 12, 3473–3484. [Google Scholar] [CrossRef]
- Keeley, J.E. Fire management impacts on invasive plants in the western United States. Conserv. Biol. 2006, 20, 375–384. [Google Scholar] [CrossRef]
- Pysek, P.; Jarosik, V.; Hulme, P.E.; Pergl, J.; Hejda, M.; Schaffner, U.; Vila, M. A global assessment of invasive plant impacts on resident species, communities and ecosystems: The interaction of impact measures, invading species’ traits and environment. Glob. Chang. Biol. 2012, 18, 1725–1737. [Google Scholar] [CrossRef]
- Hagan, D.L.; Waldrop, T.A.; Reilly, M.; Shearman, T.M. Impacts of repeated wildfire on long-unburned plant communities of the southern Appalachian Mountains. Int. J. Wildland Fire 2015, 24, 911–920. [Google Scholar] [CrossRef]
- Lovenshimer, J.B.; Madritch, M.D. Plant community effects and genetic diversity of post-fire princess tree (Paulownia tomentosa) invasions. Invasive Plant Sci. Manag. 2017, 10, 125–135. [Google Scholar] [CrossRef]
- Sutherland, E.K.; Hutchinson, T.F. Characteristics of Mixed-Oak Forest Ecosystems in Southern Ohio Prior to the Reintroduction of Fire; USDA For. Serv., Gen. Tech. Rep. NE-299; Northeastern Forest Experiment Station: Newtown Square, PA, USA, 2003; 159p. [Google Scholar]
- NIFC; Fire Statistics. National Interagency Fire Center, 2021. Available online: https://www.nifc.gov/fire-information/statistics (accessed on 6 August 2021).
- Chongpinitchai, A.R.; Williams, R.A. The response of the invasive princess tree (Paulownia tomentosa) to wildland fire and other disturbances in an Appalachian hardwood forest. Glob. Ecol. Conserv. 2021, 29, e01734. [Google Scholar] [CrossRef]
- Innes, R.J. Paulownia Tomentosa. In: Fire Effects Information System, U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory. 2009. Available online: https://www.fs.fed.us/database/feis/plants/tree/pautom/all.html (accessed on 18 June 2021).
- Longbrake, A.C.W. Ecology and Invasive Potential of Paulownia tomentosa (Scrophulariaceae) in a Hardwood Forest Landscape. Ph.D. Thesis, Ohio Univ., Athens, OH, USA, 2001. [Google Scholar]
- Alexander, H.D.; Arthur, M.A.; Loftis, D.L.; Green, S.R. Survival and growth of upland oak and co-occurring competitor seedlings following single and repeated prescribed fires. For. Ecol. Manag. 2008, 256, 1021–1030. [Google Scholar] [CrossRef]
- Fei, S.; Steiner, K.C. Rapid capture of growing space by red maple. Can. J. For. Res. 2009, 39, 1444–1452. [Google Scholar] [CrossRef]
- Green, S.R.; Arthur, M.A.; Blankenship, B.A. Oak and red maple seedling survival and growth following periodic pre-scribed fire on xeric ridgetops on the Cumberland Plateau. For. Ecol. Manag. 2010, 259, 2256–2266. [Google Scholar] [CrossRef]
- Groninger, J.W.; Long, M.A. Oak ecosystem management considerations for central hardwoods stands arising from silvicultural clearcutting. North J. Appl. For. 2008, 25, 173–179. [Google Scholar] [CrossRef] [Green Version]
- Hutnik, R.J.; Yawney, H.W. Red maple (Acer rubrum L.). In Silvics of forest trees of the United States; Handbook 271; Fowells, H.A., Ed.; U.S. Department of Agriculture: Washington, DC, USA, 1965; pp. 57–62. [Google Scholar]
- Royse, J.; Arthur, M.A.; Schorgendorfer, A.; Loftis, D.L. Establishment and growth of oak (Quercus alba, Quercus prinus) seedlings in burned and fire-excluded upland forests on the Cumberland Plateau. For. Ecol. Manag. 2010, 260, 502–510. [Google Scholar] [CrossRef]
- Schweitzer, C.J.; Dey, D.C. Forest structure, composition, and tree diversity response to a gradient of regeneration harvests in the mid-Cumberland Plateau escarpment region, USA. For. Ecol. Manag. 2010, 262, 1729–1741. [Google Scholar] [CrossRef]
- Xin, Y.; Williams, R.A. Effects of burn season on large seedlings of oak and other hardwood regeneration three years after shelterwood harvest. For. Stud. Metsanduslikud Uurim. 2019, 71, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Iverson, L.R.; Hutchinson, T.F.; Prasad, A.M.; Peters, M.P. Thinning, fire, and oak regeneration across a heterogeneous landscape in the eastern U.S.: 7-year results. For. Ecol. Manag. 2008, 255, 3035–3050. [Google Scholar] [CrossRef] [Green Version]
- Blankenship, B.A.; Arthur, M.A. Stand structure over 9 years in burned and fire-excluded oak stands on the Cumberland Plateau, Kentucky. For. Ecol. Manag. 2006, 225, 134–145. [Google Scholar] [CrossRef]
- Hutchinson, T.F.; Sutherland, E.K.; Yaussy, D.A. Effects of repeated prescribed fires on the structure, composition, and regeneration of mixed oak forests in Ohio. For. Ecol. Manag. 2005, 218, 210–228. [Google Scholar] [CrossRef]
- Rouse, C. Fire effects in northeastern forests: Oak. In General Technical Report NC-105; U.S. Department of Agriculture, Forest Service, North Central Forest Experiment Station: St. Paul, MN, USA, 1986; 7p. [Google Scholar]
- Rundel, P.W. Adaptations of Mediterranean climate oaks to environmental stress. In Proceedings of the Symposium on the Ecology, Management and Utilization of California Oaks. In General Technical Report PSW-44. Berkeley; Department of Agriculture, Forest Service, Pacific Southwest Forest and Range Experiment Station: Claremont, CA, USA, 1980; pp. 43–54. [Google Scholar]
- Brose, P.H.; Van Lear, D.H. Responses of hardwood advance regeneration to seasonal prescribed fires in oak-dominated shelterwood stands. Can. J. For. Res. 1998, 28, 331–339. [Google Scholar] [CrossRef]
- Reich, P.B.; Abrams, M.D.; Ellsworth, D.S.; Kruger, E.L.; Tabone, T.J. Fire affects ecophysiology and community dynamics of central Wisconsin oak forest regeneration. Ecology 1990, 71, 2179–2190. [Google Scholar] [CrossRef]
- Elliott, K.J.; Hendrick, R.L.; Major, A.E.; Vose, J.M.; Swank, W.T. Vegetation dynamics after a prescribed fire in the southern Appalachians. For. Ecol. Manag. 1999, 114, 199–213. [Google Scholar] [CrossRef]
- Shearin, A.T.; Bruner, M.H.; Goebel, N.B. Prescribed burning stimulates natural regeneration of yellow-poplar. J. For. 1972, 70, 482–484. [Google Scholar] [CrossRef]
- Hodgkin, E.J. Effects of Fire on Undergrowth Vegetation in Upland Southern Pine Forests. Ecology 1958, 39, 36–46. [Google Scholar] [CrossRef]
- Ferguson, E.R. Effects of prescribed fires on understory stems in pine-hardwood stands in east Texas. J. For. 1961, 59, 356–359. [Google Scholar] [CrossRef]
- Drewa, P.B.; Platt, W.J.; Moser, E.B. Fire effects on resprouting of shrubs in headwaters of southeastern longleaf pine savannas. Ecology 2002, 83, 755–767. [Google Scholar] [CrossRef]
- Langdon, O.G. Some effects of prescribed fire on understory vegetation in loblolly pine stands. In Proceedings, Prescribed Fire and Wildlife in Southern Forests: Wood; Clemson University: Clemson, SC, USA, 1981; pp. 143–153. [Google Scholar]
- Garrison, G.A. Carbohydrate reserves and response to use. In Wildland Shrubs—Their Biology and Utilization; McKell, C.M., Blaisdell, J.P., Goodin, J.R., Eds.; U.S. Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station: Ogden, UT, USA, 1972; pp. 271–278. [Google Scholar]
- Hough, W.A. Carbohydrate reserves of saw-palmetto: Seasonal variation and effects of burning. For. Sci. 1968, 14, 399–405. [Google Scholar] [CrossRef]
- Volland, L.A.; Dell, J.D. Fire Effects on Pacific Northwest Forest and Range Vegetation; U.S. Department of Agriculture, Forest Service, Pacific Northwest Region: Portland, OR, USA, 1981; 23p. [Google Scholar]
- Bond, W.J.; van Wilgen, B.W. Fire and Plants; Chapman and Hall: London, UK, 1996; 272p. [Google Scholar]
- DeBano, L.F.; Neary, D.G.; Ffolliott, P.F. Fire’s Effects on Ecosystems; John Wiley & Sons: New York, NY, USA, 1998; 333p. [Google Scholar]
- Cain, M.D.; Shelton, M.G. Survival and growth of Pinus echinata and Quercus seedlings in response to simulated summer and winter prescribed burns. Can. J. For. Res. 2000, 30, 1830–1836. [Google Scholar] [CrossRef]
- McNab, W.H.; Berg, E.C.; Oprean, T.M., III. Response to prescribed burning of five year old hardwood regeneration on a mesic site in the southern Appalachian Mountains. In Proceedings of the 15th Biennial Southern Silvicultural Research Conference; U.S. Department of Agriculture, Forest Service, Southern Research Station: Asheville, NC, USA, 2013; pp. 513–517. [Google Scholar]
- Beck, D.E. Liriodendron tulipifera L. Yellow-poplar. In Silvics of North America; 2. Hardwoods. U.S.D.A., Agricul. Handb. 654; Burns, R.M., Honkala, B.H., Eds.; USDA Forest Service: Washington, DC, USA, 1990; pp. 406–416. [Google Scholar]
- Griffith, R.S. Liriodendron tulipifera. In Fire Effects Information System; U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer): Missoula, MT, USA, 1991; Available online: https://www.fs.fed.us/database/feis/plants/tree/lirtul/all.html (accessed on 6 August 2021).
- Clark, F.B.; Boyce, S.G. Yellow-poplar seed remains viable in the forest litter. J. For. 1964, 62, 564–567. [Google Scholar] [CrossRef]
- Clark, F.B. Measures necessary for natural regeneration of oaks, yellow-poplar, sweetgum, and black walnut. In The Silviculture of Oaks and Associated Species; Research Paper NE-144; USDA Forest Service, Northeastern Forest Experiment Station: Broomall, PA, USA, 1970; pp. 1–16. [Google Scholar]
- Brose, P.H.; Van Lear, D.H. Survival of hardwood regeneration during prescribed fires: The importance of root development and root collar location. In General Technical Report SRS-73; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2004; pp. 123–127. [Google Scholar]
- Call, L.J.; Nilsen, E.T. Analysis of spatial patterns and spatial association between the invasive tree-of-heaven (Ailanthus altissima) and the native black locust (Robinia pseudoacacia). Am. Midl. Nat. 2003, 150, 1–14. [Google Scholar] [CrossRef]
- McNabb, W.H. A preliminary model of yellow-poplar seedling establishment two years after a growing season pre-scribed fire in southern Appalachian oak stands. In Proceedings of the 18th Biennial Southern Silvicultural Research Conference, Knoxville, TN, USA, 2–5 March 2015; pp. 582–588. [Google Scholar]
- Kolb, T.E.; Steiner, K.C.; McCormick, L.H.; Bowersox, T.W. Growth response of northern red oak and yellow-poplar seedlings to light, soil moisture, and nutrients in relation to ecological strategy. For. Ecol. Manag. 1990, 38, 65–78. [Google Scholar] [CrossRef]
- Schwartz, M.W.; Heim, J.R. Effects of a prescribed fire on degraded forest vegetation. Nat. Areas J. 1996, 16, 184–191. [Google Scholar]
- Brooks, M.L.; D’Antonio, C.M.; Richardson, D.M.; Grace, J.B.; Keeley, J.E.; DiTomaso, J.M.; Hobbs, R.J.; Pellant, M.; Pyke, D. Effects of invasive alien plants on fire regimes. BioScience 2004, 54, 677–688. [Google Scholar] [CrossRef] [Green Version]
- Canham, C.D. Different responses to gaps among shade-tolerant tree species. Ecology 1989, 70, 548–550. [Google Scholar] [CrossRef]
- Allendorf, F.W.; Lundquist, L.L. Introduction: Population biology, evolution, and control of invasive species. Conserv. Biol. 2003, 17, 24–30. Available online: https://www.jstor.org/stable/3095269 (accessed on 1 August 2021). [CrossRef] [Green Version]
- Sakai, A.K.; Allendorf, F.W.; Holt, J.S.; Lodge, D.M.; Molofsky, J.; With, K.A.; Baughman, S.; Cabin, R.J.; Cohen, J.E.; Ellstrand, N.C.; et al. The population biology of invasive species. Annu. Rev. Ecol. Syst. 2001, 32, 305–332. [Google Scholar] [CrossRef] [Green Version]
- Rebbeck, J. Fire management and woody invasive plants in oak ecosystems. In Proceedings of the 4th Fire in Eastern Oak Forests Conference, Springfield, MO, USA, 17–19 May 2011; pp. 142–155. [Google Scholar]
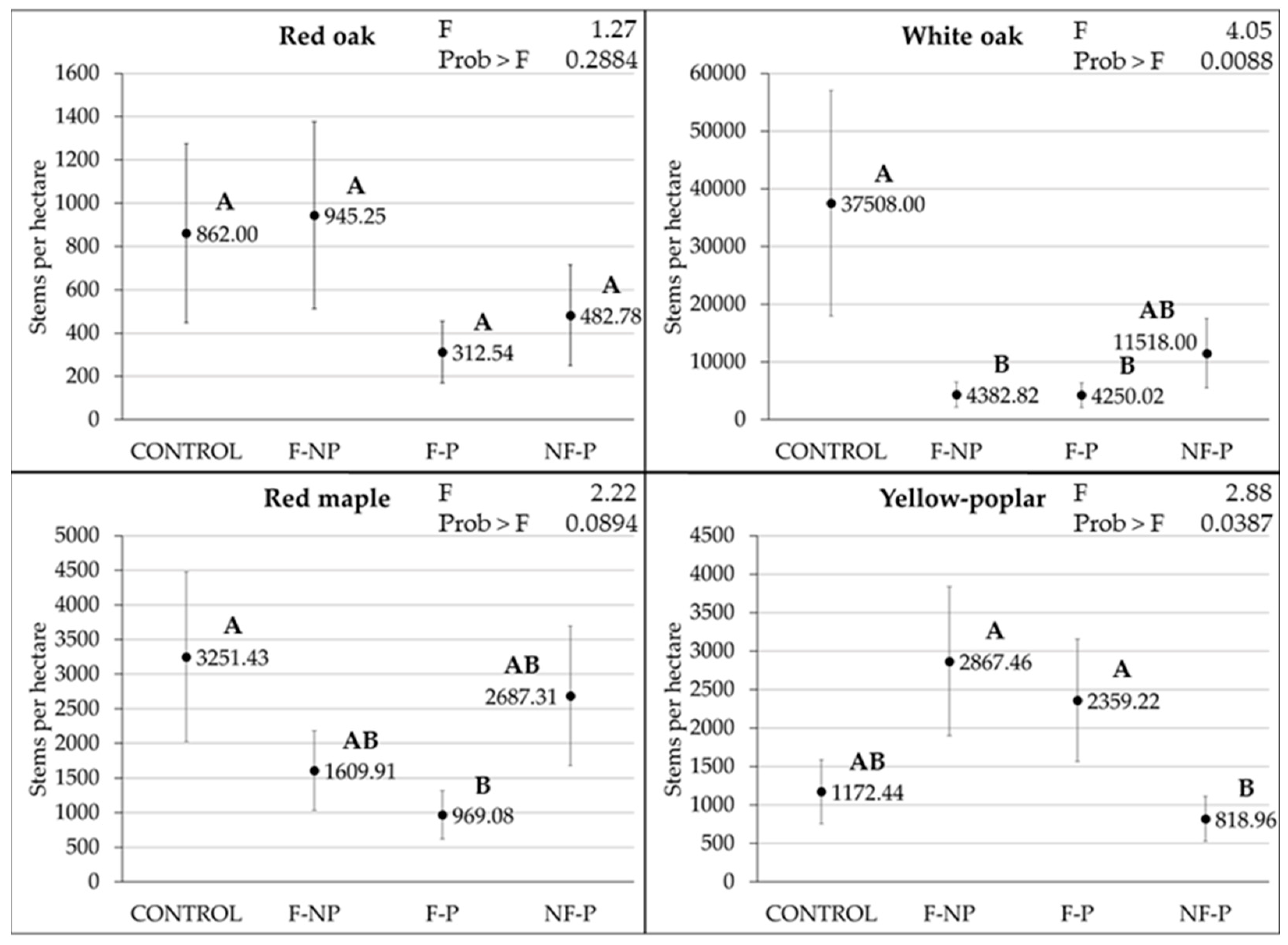
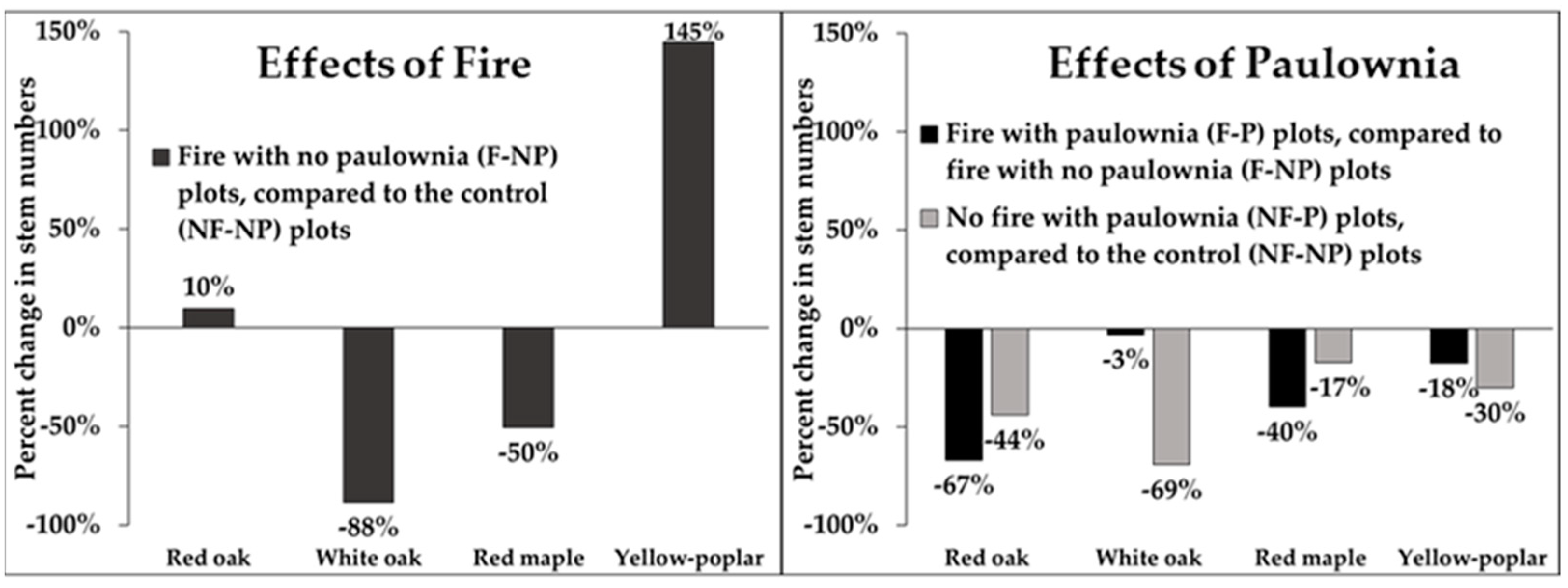
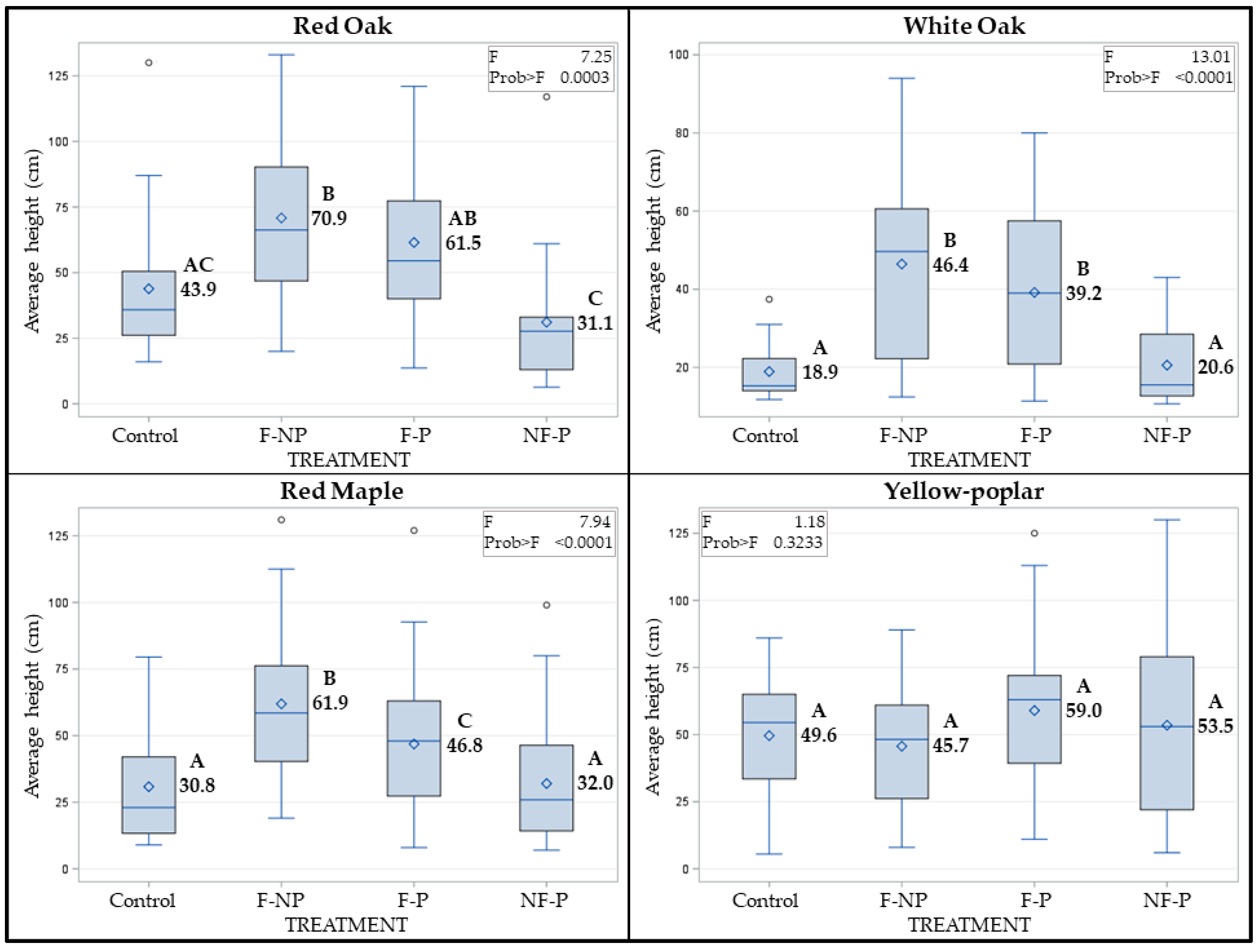
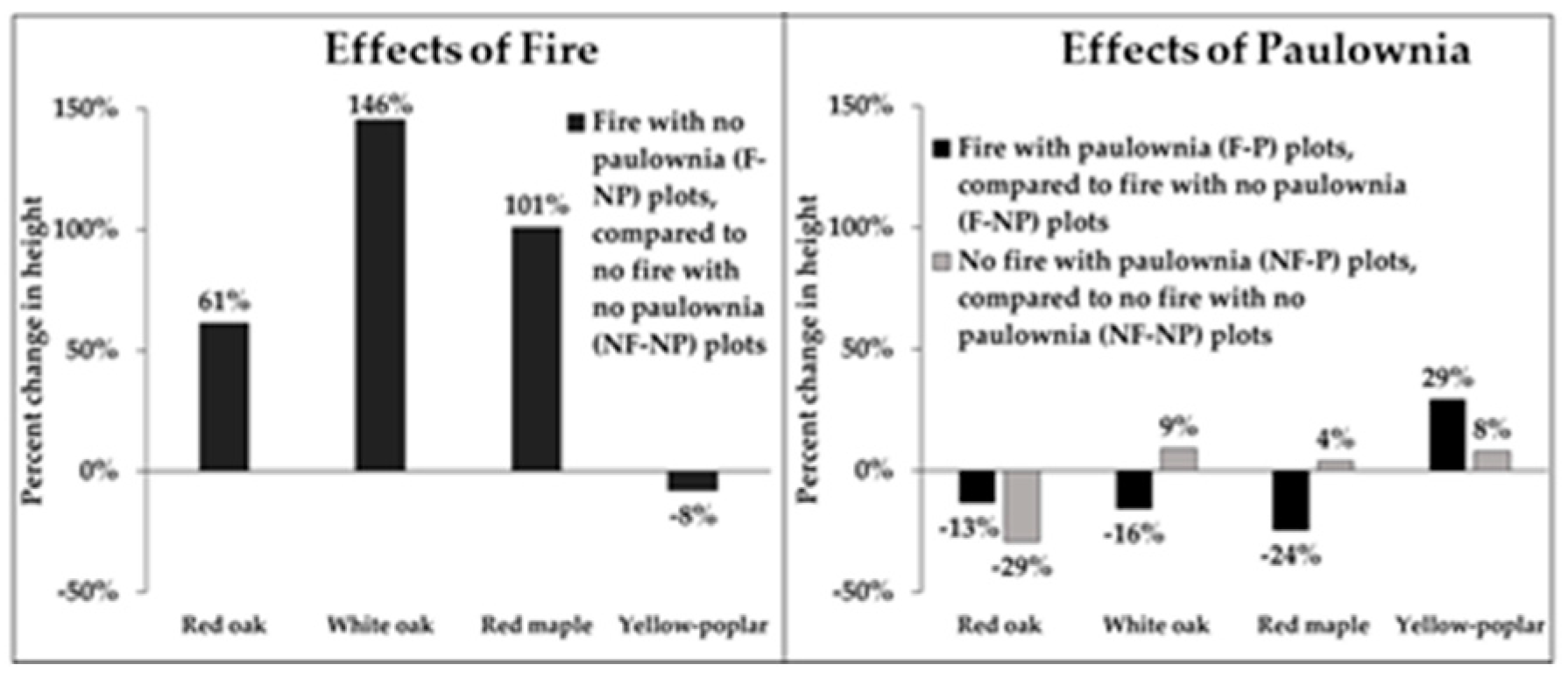
| Attribute | Effects | Mean | Std Error | F Value | Prob > F |
|---|---|---|---|---|---|
| Stems per hectare | Fire | 648.44 A | 38.4414 | 2.31 | 0.1297 |
| No Fire | 568.96 A | 35.4401 | |||
| Average height (cm) | Fire | 223.15 A | 15.4134 | 105.73 | <0.0001 |
| No Fire | 797.11 B | 56.1248 |
| Model Effects | Num DF | Den DF | F Value | Pr > F |
|---|---|---|---|---|
| Species | 3 | 472 | 29.62 | <0.0001 |
| Fire | 1 | 472 | 3.73 | 0.0540 |
| Paulownia | 1 | 472 | 6.05 | 0.0143 |
| Fire X Paulownia | 1 | 472 | 0.08 | 0.7814 |
| Species X Fire | 3 | 472 | 6.62 | 0.0002 |
| Species X Paulownia | 3 | 472 | 0.38 | 0.7705 |
| Species X Fire X Paulownia | 3 | 472 | 0.78 | 0.5031 |
| Species | Treatment Effects | Num DF | Den DF | F Value | Pr > F |
|---|---|---|---|---|---|
| Red oak | Fire | 1 | 118 | 0.13 | 0.7150 |
| Paulownia | 1 | 118 | 3.25 | 0.0741 | |
| Fire X Paulownia | 1 | 118 | 0.32 | 0.5745 | |
| White oak | Fire | 1 | 118 | 9.58 | 0.0025 |
| Paulownia | 1 | 118 | 1.42 | 0.2355 | |
| Fire X Paulownia | 1 | 118 | 1.28 | 0.2600 | |
| Red maple | Fire | 1 | 118 | 5.53 | 0.0204 |
| Paulownia | 1 | 118 | 0.9 | 0.3439 | |
| Fire X Paulownia | 1 | 118 | 0.19 | 0.6650 | |
| Yellow-poplar | Fire | 1 | 118 | 7.97 | 0.0056 |
| Paulownia | 1 | 118 | 0.64 | 0.4249 | |
| Fire X Paulownia | 1 | 118 | 0.06 | 0.8131 |
| Source | Type I SS | Mean Square | F Value | Pr > F |
|---|---|---|---|---|
| Species | 24,637.89 | 8212.63 | 12.83 | <0.0001 |
| Fire | 29,743.34 | 29,743.34 | 46.46 | <0.0001 |
| Paulownia | 518.89 | 518.89 | 0.81 | 0.3686 |
| Fire X Paulownia | 245.39 | 245.39 | 0.38 | 0.5363 |
| Species X Fire | 9586.54 | 3195.51 | 4.99 | 0.0021 |
| Species X Paulownia | 5523.53 | 1841.18 | 2.88 | 0.0363 |
| Species X Fire X Paulownia | 2159.05 | 719.68 | 1.12 | 0.3394 |
| Species | Source | Type I SS | Mean Square | F Value | Pr > F |
|---|---|---|---|---|---|
| Red oak | Fire | 16,571.90 | 16,571.90 | 19.13 | <0.0001 |
| Paulownia | 2221.89 | 2221.89 | 2.57 | 0.1137 | |
| Fire X Paulownia | 54.01 | 54.01 | 0.06 | 0.8035 | |
| White oak | Fire | 10,548.67 | 10,548.67 | 37.14 | <0.0001 |
| Paulownia | 156.09 | 156.09 | 0.55 | 0.4608 | |
| Fire X Paulownia | 383.03 | 383.03 | 1.35 | 0.2493 | |
| Red maple | Fire | 12,408.71 | 12,408.71 | 19.51 | <0.0001 |
| Paulownia | 1221.87 | 1221.87 | 1.92 | 0.1693 | |
| Fire X Paulownia | 1517.28 | 1517.28 | 2.39 | 0.1261 | |
| Yellow-poplar | Fire | 18.01 | 18.01 | 0.02 | 0.8782 |
| Paulownia | 2216.20 | 2216.20 | 2.91 | 0.0919 | |
| Fire X Paulownia | 459.07 | 459.07 | 0.60 | 0.4400 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, R.; Wang, H. Effects of Wildfire and the Presence of the Invasive Paulownia tomentosa on the Regeneration of Native Tree Species in North-Central Appalachia. Fire 2021, 4, 60. https://doi.org/10.3390/fire4030060
Williams R, Wang H. Effects of Wildfire and the Presence of the Invasive Paulownia tomentosa on the Regeneration of Native Tree Species in North-Central Appalachia. Fire. 2021; 4(3):60. https://doi.org/10.3390/fire4030060
Chicago/Turabian StyleWilliams, Roger, and Haibin Wang. 2021. "Effects of Wildfire and the Presence of the Invasive Paulownia tomentosa on the Regeneration of Native Tree Species in North-Central Appalachia" Fire 4, no. 3: 60. https://doi.org/10.3390/fire4030060
APA StyleWilliams, R., & Wang, H. (2021). Effects of Wildfire and the Presence of the Invasive Paulownia tomentosa on the Regeneration of Native Tree Species in North-Central Appalachia. Fire, 4(3), 60. https://doi.org/10.3390/fire4030060







