Abstract
Wildland fire is a major producer of aerosols from combustion of vegetation and soils, but little is known about the abundance and composition of smoke’s biological content. Bioaerosols, or aerosols derived from biological sources, may be a significant component of the aerosol load vectored in wildland fire smoke. If bioaerosols are injected into the upper troposphere via high-intensity wildland fires and transported across continents, there may be consequences for the ecosystems they reach. Such transport would also alter the concept of a wildfire’s perimeter and the disturbance domain of its impact. Recent research has revealed that viable microorganisms are directly aerosolized during biomass combustion, but sampling systems and methodology for quantifying this phenomenon are poorly developed. Using a series of prescribed fires in frequently burned forest ecosystems, we report the results of employing a small rotary-wing unmanned aircraft system (UAS) to concurrently sample aerosolized bacteria and fungi, particulate matter, and micrometeorology in smoke plumes versus background conditions. Airborne impaction-based bioaerosol sampling indicated that microbial composition differed between background air and smoke, with seven unique organisms in smoke vs. three in background air. The air temperature was negatively correlated with the number of fungal colony-forming units detected. Our results demonstrate the utility of a UAS-based sampling platform for active sampling of viable aerosolized microbes in smoke arising from wildland fires. This methodology can be extended to sample viable microbes in a wide variety of emissions sampling pursuits, especially those in hazardous and inaccessible environments.
Keywords:
bioaerosol; smoke; biomass burning; emissions; microorganisms; microbe; drone; prescribed fire; biological diversity 1. Introduction
Approximately 5130 Tg of forest, grassland, and agricultural biomass burns annually, producing ~38.5 Tg of fine particulate matter emissions globally each year [1]. Airborne particles containing microorganisms or their derived organic compounds (e.g., endotoxins, microbial fragments, metabolites), or bioaerosols, have been shown to comprise up to 25% of global emissions of particulate matter (PM) up to 2.5 µm in size (PM2.5) and 50% of the carbonaceous component in air pollution studies [2]. While biomass fuel-level temperatures can exceed typical biological thresholds for survival even in grassland fires (e.g., over 700 °C; [3]), air turbulence, discontinuity of fuels, and fine-scale variation in meteorological factors often result in a range of fire intensities, temperatures, and combustion efficiencies [4]. This variability is an important driver of diverse biotic responses [5,6], including those of microbes, known to tolerate a wide range of temperatures [7], and burn conditions [8]. The proportion of bioaerosols which survive pyrolysis and combustion-driven aerosolization processes has yet to be determined. If living, microbial components of biomass bioaerosols (e.g., fungi and bacteria) may be pathogenic, beneficial, or have neutral effects by impacting microbial communities and their roles in both atmospheric and terrestrial environments [9]. Aerial transport of viable microbes is widely recognized for its impact on microbial biodiversity [10], allergens and diseases affecting human health [11,12,13], forest and crop pathogens [14], and atmospheric condensation nuclei [15].
High-buoyancy smoke plumes have the potential for long-range transport of bioaerosols, which may be associated with particles, as previous studies in continental dust transport have shown [16,17]. The limited work addressing the degree to which atmospheric particulate matter correlates with bioaerosols suggests that organism type and environmental conditions are highly influential [2,18,19]. The direct relationship between bioaerosols and wildland fire smoke has received minimal scientific attention [20,21,22]. Biomass burning indicators (e.g., levoglucosan and K+) have been examined for their correlation with indicators of fungal presence in particulate matter (e.g., arabitol, mannitol, and endotoxins), and positive associations have been documented in two air pollution studies [23,24]. In Portugal, fungal spore abundance in background air was highest 10 days following known wildfire events, suggesting either aerosolization or disturbance-induced stimulation of spore production [21]. Recent research addressing these relationships has indicated direct aerosolization of diverse, viable microbes by wildland fire smoke and biomass burning under field and controlled laboratory conditions [22]. Together, these studies suggest that the biological component in smoke emissions from wildland fire is significant and additional advancement of “pyroaerobiology” is warranted.
Currently, there exist no established methods for sampling living microbes in smoke plumes. General methods for sampling bioaerosols fall under three broad categories: passive sampling from stationary platforms, volumetric sampling from stationary platforms, and either mechanism of sampling from mobile platforms. The origins of aerobiological sampling began with passive sampling from stationary platforms [25]. Then and today, the presence/absence of microbiota is assessed by exposing tape or microscope slides with an adhesive surface, or petri-dishes with growth medium, to the air to allow for gravity- or wind-driven impaction of aerosolized or settling microbiota (e.g., [20,26]). Active volumetric sampling enables the assessment of total concentrations of target organisms that can be interpreted for their potential repercussions for human health [25], crop disease infection potential [27], or for comparisons across time and space [28]. Devices commonly used to sample bioaerosols while accounting for the volume of air sampled are diverse and include filters, impactors, impingers, and cyclones—each with benefits and limitations [27,29].
As interest in the meteorological and ecological implications of airborne microbes has expanded from near-ground to the lower troposphere [16], the need to sample microbes at various heights above ground level has hastened the experimentation and development of mobile platform systems. There are multiple limiting factors in the mobilization of aerobiological sampling systems. Active sampling devices require pulling a measurable rate of airflow through a device using what is often a heavy external pump. The speed at which particles are impinged or impact onto a surface can be used to partition or select for microbes of specific aerodynamic diameter ranges, as well as to correlate response variables to known sampled volumes [29]. Although piloted aircraft are useful for conducting aerial sampling and are not practically limited by payload weight constraints, their cost can be prohibitive for repeated sampling, and limited maneuverability may lead to misalignment between the sampling fetch and the extent of the targeted air mass. Piloted flights are typically limited to higher altitudes more distant from the source of smoke, thereby sampling air that has already been diluted by background aerosols. Flights at lower altitudes within a smoke plume are logistically challenging and can present safety risks for data collection.
Unmanned aircraft systems (UAS) capable of hovering (e.g., rotary-wing aircraft) present an advantage for targeted sampling of visually or chemically identifiable air masses, such as point pollution sources or smoke columns, because they can be intentionally positioned within the mass [30,31]. In this case, active pump-based sampling is required to sample known volumes of air; however, small UAS and commonly used pump and sampling apparatus have not been available until recently due to technical constraints including weight and availability. Concerns about rotor downwash have also been expressed, although the positioning of sampling devices can minimize or mitigate undesired effects of rotor downwash [30,31]. Finally, apprehension about positioning UAS and instruments in smoke plumes and columns from even low-intensity fires may have posed challenges to their deployment. These perceived challenges, in addition to identifying a commercially available, lightweight pump which can be programmed to sample once the aircraft has reached the air mass of interest, may have discouraged the development or application of UAS-based aerobiological sampling for wildland fires.
This work, therefore, serves as a proof of concept for employing bioaerosol sampling devices as part of a multi-component payload to concurrently quantify environmental factors and smoke’s aerosolized living organisms on a mobile platform. Environmental characteristics likely affected by smoke and known to influence bioaerosols include relative humidity (RH), temperature, pollutants, and pollutant gases [32]. Co-locating real-time environmental and aerobiological sampling systems enables an examination of potential influences on microbial community composition and viability. The maneuverability and hovering capability of a rotary-wing unmanned aircraft provide the flexibility needed for targeted air mass sampling, reducing (but not limiting) the influence of entrained background air and promoting sampling when particulate matter sensors indicate the presence of smoke. Our coupled PM-RH-temperature and bioaerosol impactor payload (Pyroaerobiology Package, or “PAB-Pac”) on the mobile platform also helps monitor for any potential contamination of background samples, so that evaluations of background versus smoke microbes are less confounded by mixing.
Using a series of prescribed burns in frequently burned forested ecosystems, we report the results of using a UAS to concurrently sample aerosolized microbes, particulate matter, and micrometeorology in smoke plumes versus background conditions. Impaction-based bioaerosol sampling, followed by culture-based genus identification, is used to estimate microbial abundance and composition. Because culture-based methods are inherently limited, our results do not indicate a complete characterization of the microbial community. Rather, we demonstrate comparisons between smoke-impacted and background air within the confines of the culture-based approach. Our primary objective is (1) to demonstrate the utility of the mobile sampling platform for capturing viable aerosolized microbial organisms. Secondary objectives are (2) to describe captured, viable microbial communities in background and smoke air masses, and (3) to assess relationships between aerobiological content and environmental characteristics. This methodology can be extended to a wide variety of aerobiological sampling pursuits across a range of otherwise hard-to-access air masses.
2. Materials and Methods
2.1. Site Description
The study was conducted over lands managed by the Tall Timbers Research Station (TTRS), located approximately 30 km north of Tallahassee, FL, USA, in Leon County (30°39′24.1″ N, 84°12′32.3″ W). Sampling took place prior to and during two prescribed burns in forests which had previously been cultivated for corn and cotton in the early 1800s, but now represent native pine forests of the region. Soils in these sites are fine-loamy, kaolinitic, thermic Typic Kandiudults of the Orangeburg and Faceville series. The overstory is dominated by low-density loblolly (Pinus taeda L.), longleaf pine (P. palustris P. Mill), and shortleaf pine (P. echinata P. Mill.) with a mixed grass, herbaceous, and broad-leaved understory. The burn units have been maintained with prescribed fire on a three-year cycle on average for the last decade, so that fuel load and forest floor build-up are minimal (i.e., the organic, or “O” horizon of the soil is less than 1 cm deep on average).
2.2. Prescribed Burns
Prescribed broadcast burns were conducted as part of a demonstration by the TTRS-based Prescribed Fire Science Consortium, with the objective of maintaining the natural fire return interval for the sites while providing opportunities for fire managers and scientists to co-produce research. The first of the two prescribed burns, which was 10 ha in area, was ignited on 19 April 2018 at 11:15 EDT in a location near the TTRS compound. To reduce the potential impact of personnel within the burn unit, we sampled only above the spreading fire where personnel were not present and where visible smoke was rising above the forest canopy. The study design involved sampling where the presence of visible smoke covered an area: (1) large enough so that the UAS would be entirely within the smoke plume, (2) above free-burning vegetation (not being ignited by personnel), and (3) over the interior of the units. Two UAS-based samples were collected, but sampling opportunities were limited due to low-intensity fire behavior, which did not produce visible smoke above the tree canopy. The second 40-ha burn on 20 April, in a site located approximately 15 km from the first site, was ignited at 13:30 EDT, and the burn duration and smoke production was sufficient for four additional smoke samples to be taken above the canopy where no ground-level disturbance was observed (e.g., downwind of the ignition lines). Sampling locations were above the burn unit interiors and avoided sampling over fire lines. All burns were conducted using a combination of strip head firing and flanking fire, producing flame lengths between 0.2 and 1.5 m in length and rates of spread averaging below 0.7 m s−1 based on ocular estimates. Combustion sources were limited to the understory grasses and shrubs, and to litter and duff on the forest floor (soil O horizon).
2.3. Aerobiological Sampling
In order to demonstrate that this new sampling system had the capacity to collect and process living microbes aerosolized by smoke, we focused on culturable fungal and bacterial organisms that could be successfully sampled [29]. To capture these microbes in smoke, we designed a portable, lightweight aluminum frame to secure the sampling device and the sampling pump directly below the fuselage of the UAS, where downwash from the rotors would be minimized (Figure 1) [30]. The battery-powered compensating Leland Legacy sampling pump (SKC Inc., Covington, GA, USA) was chosen as the most low-cost, low-weight, adequate-flow pre-manufactured pump available with a delayed-start programming capability. The SKC Inc. Biostage 200 single stage impactor (meeting NIOSH Method 0800 and 0801 requirements) with 200 holes of 0.25 mm diameter was connected to the Leland Legacy, which pulled air through a small inlet to impact on a 90 mm petri-dish with either trypticase-soy agar (TSA) or malt extract agar (MEA). The TSA is a commonly used medium for bacteria while MEA is used for a wide range of taxa including fungi and bacteria. Individual petri plates with alternating media were paired as close together in time and positioned where real-time particulate matter readings and smoke conditions were similar so that organisms captured on both media could be exposed to similar conditions. The impactor and all connecting tubing were cleaned with 80% ethanol in a sterilized mobile glovebox in between samples to prevent contamination. Blanks were collected both in the field and within the glovebox. The sampling inlet was covered until immediately prior to lift off. The impactor was positioned directly below the body of the UAS to reduce down wash effects (Figure 1: see [31] for further discussion).

Figure 1.
(a) The UAS Matrice 600 equipped with custom payload assembly (“PAB-Pac”) includingan SKC 200 single-stage impactor, an SKC Leland Legacy compensating pump, and meteorological and particulate matter sensing packages; (b) the airborne UAS operated by Desert Research Institute pilots during the prescribed burn: the UAS is shown en route to the interior of the unit to sample above canopy level directly above burning fuels at the Pebble Hill site, north of Tallahassee, Florida, US.
The median cut point diameter, or D50, for the Biostage 200 is 0.6 µm at a flow rate of 14.15 L min−1. Due to backpressure difficulties sampling in heavy smoke, the in-line rotameter revealed that the Leland Legacy pump was not able to sustain a continuous pull of 14.15 L min−1 while airborne. The reduced sampling flow rate of 10–11.5 L min−1 we used as a result likely has a different D50 value: theoretically, a lower flow rate would shift the D50 curve to the right, favoring slightly larger diameter materials. This may have biased our samples in comparison to other studies, but this bias did not affect our objectives. We used the same flow rate for the background air samples so that comparisons could be made.
To help describe smoke-aerosolized microbes and those already airborne at the site, we conducted a sampling of both smoke-influenced and background air [22]. Background samples were taken at the same height, for the same duration, and over the same fuels that were later (within one hour) ignited. Background air samples were taken in the interior of the units to reduce the influence of edge effects. Two background samples were also taken at ground level prior to ignition on 20 April 2018 by suspending the sampling platform at 2 m height above ground level (AGL; Table 1). Process blanks for both handling and transport of petri-dishes were included and showed no contamination after being incubated at 25 °C for seven days. Field blanks were used both before and after the burn to test for contamination that might occur prior to the pump being turned on. Field blanks mirrored the process for loading and unloading petri-dishes on the UAS, as well as sealing and storing the dishes, but without flying or pumping air. The UAS took off and landed on a tarpaulin to reduce any effects of soil perturbation, which could result in aerosolization before the targeted air mass was reached.

Table 1.
Description of weather and sampling characteristics for six smoke and two background samples acquired on board the UAS and two background air samples acquired at 2.0 m above ground level during two burns located near Tall Timbers Research Station, Florida, USA.
2.4. Particulate Matter and Meteorology Sampling
A lightweight measurement device designed for use on UAS was employed to collect air samples and monitor particulate matter and meteorology [31]. The microcontroller-based device includes a data acquisition system that records particulate matter and meteorology sensor data at ~0.5 Hz. A remote control device was used to actuate the valve system for air sample collection and display real-time data from the measurement device to confirm that the UAS was located in the target smoke plume.
Particulate matter data were collected using a PMS5003 particulate sensor (Plantower, Beijing, China) and included mass:volume concentration (µg m−3) estimates for particles less than 1.0 µm diameter (PM1), less than 2.5 µm diameter (PM2.5), and less than 10.0 µm diameter (PM10). A detailed description of the sensor, its specifications, and its limitations can be found in [31]. Meteorology data included temperature, RH, and pressure. Temperature and humidity were measured using an SHT-15 sensor (Sensirion, Staefa ZH, Switzerland) and pressure was measured using an MPL3115A2 pressure sensor (NXP Semiconductors, Netherlands).
2.5. Microbial Cultures and Identification
Following sampling using sterile methods, all petri-dishes were sealed, stored in a cooler with ice packs, and shipped overnight with cooling packs from Florida to the lab in Idaho. Samples were then incubated at 25 °C and counts of colony-forming units (CFUs) conducted at 3, 5, and 10 days following incubation were performed by microscopy. Sub-samples of each unique organism were plated on MEA (for fungi) and TSA (for bacteria), incubated for 3 to 5 days and examined for purity, then sent to EMLab P&K Inc. (South San Francisco, CA) for microscopic identification to genus. Fungi were identified based on microscopic examination of colony characteristics on specific growth media, and if needed, using a lacto-phenol blue stain. Bacteria were identified using enzymatic properties (API), MIDI, and Biolog ID systems. In some cases, the colonies could be identified directly based on colony characteristics and Gram staining.
To calculate the total CFUs per unit volume of air, we first applied positive-hole correction factors as recommended by the manufacturer (SKC Inc., Eighty Four, PA, USA) for the Biostage 200 for sampling environments where the aerosol load is likely to be high [33]. We then used the known volume of air sampled to scale CFUs per petri plate to CFUs m−3. Species were identified to genus in most cases. Fungal or bacterial CFUs lacking sufficient evidence to identify to genus were classified as “non-sporulating fungi” or “unknown bacterium”.
2.6. Statistical Analyses
Data were first examined for assumptions of normality and homoscedasticity and transformed when necessary using log transformations. Median, 10th percentile, and 90th percentile values for environmental variables and PM measurements were computed from the sensor time series data for the aerobiological sampling period and tested for correlations with biological data (p-value < 0.05). Simple linear regression was used to evaluate relationships between CFUs and environmental factors including temperature, RH, and PM values. Significance was determined at the p < 0.05 level.
3. Results
3.1. Microbial Presence in Smoke and Background Air
The number of CFUs m−3 was nearly three times greater in background samples on board the UAS at 25 m AGL than those sampled from the ground at 2 m AGL (1608 vs. 523 CFUs m−3) within a 2-h mid-day period. Differences between microbial CFUs m−3 in the background and smoke samples were negligible, but there were more genera unique to smoky air than background air (Table 2). The total number of fungal organisms present in smoke was 10 in contrast to five in background air. Total numbers of bacterial species did not differ between the two air masses, although Lysinbacillus sp. and Sporolactobacillus sp. were only found in one background sample at the ground level and three unique bacteria were found only in smoke (Table 2). The most common shared species included Cladosporium sp. 1 and Bacillus sp.: Cladosporium sp. 1 was the most abundant CFU across all samples on all but one petri-dish. Penicillium sp. was found in three of the four background samples, while only one Penicillium sp. CFU was found in one smoke sample. Non-sporulating fungi were combined because distinguishing characteristics could not be identified; such fungi were found in both background and smoke samples. No fungal or bacterial colonies developed on the blanks, indicating that sterilization and processing methods were adequate to prevent contamination.

Table 2.
Total colony-forming units (CFUs m−3), median particulate matter (PM: µg m−3) values for PM size ranges below 1, 2.5, and 10 µm, and presence/absence (denoted by “x”) of distinct CFUs in each sample of smoke or background air taken during prescribed fires in northern Florida.
Based on laboratory analyses of cultured colonies, at least seven unique organisms were found only in smoke, compared to three unique organisms identified in background conditions sampled both above and below the canopy (Table 2). Cladosporium sp. was found in every sample. Seven organisms were shared across smoke and background air masses. Because non-sporulating fungi were grouped, the richness of culturable fungi is a conservative estimate.
3.2. Relationship to Environmental Factors and Particulate Matter
The average median values of PM10 were 25 µg m−3 in background conditions vs. 566 µg m−3 in smoke, with 90th percentile values reaching 30.5 vs. 645 µg m−3, respectively. Median PM10values during the background sampling period ranged from 16 to 33 µg m−3, while values ranged from 67 to 1021 µg m−3 in the smoke samples (Table 2). Smaller particulate matter in the PM2.5 class ranged from 18 to 31 µg m−3 in background and 55 to 950 µg m−3 in smoke conditions for median values covering the duration of sampling (Table 2).
Although PM measures showed different levels of aerosols across the multiple sampling iterations, bacterial, fungal, or total CFUs were not significantly related to particulate matter. In no case did the number of bacterial or fungal CFUs correlate with any size class of PM (p-value > 0.05). Particulate matter (PM) measures included only the time period during which the impactor was actively sampling. Equipment malfunction prevented PM measurements during the second and the sixth burn sampling periods.
Median RH levels ranged from 33.6% to 47.9% and were lowest at the end of the sampling period on the second day (Table 1). Because background samples were necessarily taken prior to ignition (to avoid smoke contamination), RH was generally higher during background samples, ranging from 41.6% to 44.6%. Median temperatures were lowest during background samples (19.5 to 21.5 °C) and, as expected, higher in the smoke (21.4 to 29.4 °C). Meteorological conditions showed a limited correlation with culturable microbes. Increasing temperatures correlated with lower fungal CFUs m−3 (Figure 2). Other relationships among CFUs, PM, and meteorological measures were not significant. There were also no discernable patterns of CFU fluctuation in relation to the time of day.
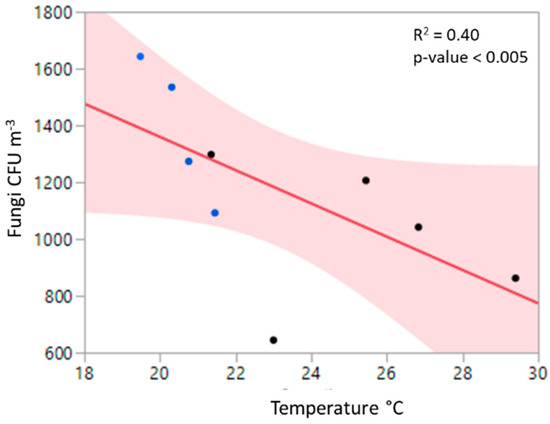
Figure 2.
Negative correlation between air temperature at sampling location and fungal CFUs per cubic meter of air. Shaded area shows 95% confidence limits of the mean. Blue dots indicate background air and black dots represent smoke.
4. Discussion
4.1. Sampling Bioaerosols from Aircraft Platforms
This study demonstrates the utility of using a UAS to sample living microbes in smoke plumes above wildland fires. The benefits of using this UAS sampling method to determine wildland fire bioaerosols emissions include: positioning of the UAS directly above the flaming front, increasing the precision of sampling by reducing the influence of entrained background air; high maneuverability to respond in real time to dynamic smoke plume behavior; remote launch capability; ground-based piloting; low technological complexity; and low cost. In comparison to fixed-wing aircraft, there also exist sampling effectiveness benefits. Because large, fixed-wing aircraft must travel at certain speeds to maintain lift, the organisms sampled commonly impact the sampling surface (be it liquid, growth medium, or filter) at high speeds, which influences the size distribution and survival of the sampled organisms and may cause reductions or inconsistencies in the organisms captured through bounce or inconsistent inertial impaction [29,32]. Desiccation on filters, resulting from high and prolonged air flow, can lead to a significant reduction in cell viability and therefore limit inferences about organism viability and vitality [34,35]. Most impaction devices achieve known sampling efficiencies for targeted microbe size ranges by controlling the speed at which particles enter the device and limiting sampling time to decrease the desiccation of organisms [36].
An alternative option is to use the speed of a small, remotely operated fixed-wing aircraft in lieu of an incorporated vacuum pump. In these cases, the volume sampled is estimated using the area of an exposed sampling plane (e.g., a remotely-opened petri-dish), the speed of the aircraft and the overall sampling duration to estimate the volume of air sampled [37,38]. While this may be a lightweight solution for some research objectives, challenges arise when sampling an air mass that frequently changes location, dimension, density, and windspeed, such as a smoke plume produced by wildland fire.
4.2. Species Composition and Abundance
That seven unique species were found in smoke compared to three in background samples suggests that combustion or heating of vegetative and soil material is aerosolizing particles that may otherwise not be common in the background air of the location sampled. However, no two samples of either background or smoke had the same composition of microbes, reflective of the high variability among samples evidenced in other bioaerosol studies [10]. The number of CFUs reflects not only the presence of an organism but its vitality. The slightly higher number of CFUs in the background air evidenced in this study may, therefore, reflect one of two things: either smoke reduces the abundance while influencing the composition of aerosols, or organisms in smoke have been impacted by combustion (physio-chemical effects) and viability is reduced. Due to the small sample size, outliers also had an impact. The one background sample with the highest CFU count was 81% composed of a single bacterial genus, Bacillus. In either case, it is well known that the vast majority of microbes are either unknown, unculturable, or both [39]. Therefore, these measures are likely low estimates of the total viable organisms aerosolized in wildland fire smoke or present in background air.
Two of the three species unique to the background samples were found only in the ground-based samples, and as both Lysinbacillus sp. and Sporolactobacillus sp. are soil-dwelling bacteria, it is unlikely that they would be aerosolized above tree canopy heights unless significant soil disturbance and wind turbulence occur. Altenaria alternata, one of the species found in smoke in this study and in [20] is a common aerosol and is a widely recognized plant pathogen and human allergen [40]. Cladosporium is a common indoor and outdoor mold genus with over 500 species, while Epicoccum nigrum, found in both smoke and background air in this study, is a common fungus that can be both a plant pathogen and endophyte [41,42]. Nocardia spp. are relatively common aerobic actinomycetes found in soil and vegetation and are known to cause a broad range of human health impacts collectively referred to as “Nocardiosis” [11]. Inhaled Nocardia sp. cells can lead to cardio-pulmonary, respiratory, and central nervous system illnesses [11]. Also only found in smoke, the Paenibacillus genus includes facultative anaerobic endospore-forming bacteria. Some species serve as beneficial nitrogen-fixers in soil, while others are plant growth-promoting rhizobacteria and serve as biopesticides that can protect roots from fungal pathogens [43]. During a wildland fire and in the days, weeks, and months following, the transport of bioaerosols has unknown consequences for post-fire restoration and recovery of fire-affected, adjacent, and distant ecosystems [21].
Cell viability and vitality are key considerations for determining the ecological and atmospheric impacts of aerosolized microbes [44,45]. Living microbes in the troposphere continue metabolic respiration, producing respiratory products such as endotoxins and even reproduce in their aerosolized state [45]. Some living microbes (e.g., numerous bacteria) are also ice nucleators and are effective at higher temperatures than other nucleation particles [44]. The density of CFUs evidenced here from even low-intensity fire suggests that the unique microbes aerosolized through combustion processes may be plentiful enough to contribute to the role of living biological aerosols in rain or ice nucleation [46].
4.3. Relationships of Bioaerosols and Environmental Factors, Including Particulate Matter
Contrary to our expectations, there was no significant correlation between PM mass concentrations and total, fungal, or bacterial CFUs in our study. Although this may be the product of our limited sample size, [19] and [47] also found no correlation between PM and even xerophilic fungal spore levels. However, [20] reported a strong correlation between coarse carbon particles (presumably from biomass burning) and fungal spore counts on microscope slides, although the particles were not differentiated by size and the viability of the bioaerosols was not examined. The lack of consistency in the literature concerning the relationship between PM and fungal spores or colonies reflects the influence of different source communities and/or environmental conditions, as well as the potential that fungal spores are being entrained in convection columns from outside the combustion zone. Gusty winds during the first day of sampling may have also played a role in mixing background and smoke air masses in this study, decoupling PM from the bioaerosols.
Another potential influence is combustion efficiency: higher efficiency combustion produces less PM and more water vapor per unit mass of fuel consumed. It may be that increased combustion efficiency promotes some wet discharge of ascospores and/or basidiospores near the combustion zone. The mechanisms for spore aerosolization in the combustion zone have yet to be examined, although simple laboratory experiments have indicated that dry discharge of spores during direct combustion of fungal colonies does occur (Kobziar, unpublished data). Aerosolization of bacteria and fungi may be occurring during all stages of combustion: heating of fuels, pyrolysis, and during both flaming and smoldering combustion. It is likely that over the course of the sampling period, each of these processes contributes microbes to the smoke plumes to varying extents. Unlike most fungal spore results, PM levels have been shown to be positively correlated with bacterial cell counts in air pollution studies [19,47]. Results from [22] suggest that overall colony-forming units, driven mostly by bacterial colonies, are more abundant closer to combustion sources, where both convective wind turbulence and PM levels are likely higher.
Investigation into the relationships between meteorological factors and bioaerosols typically include larger-scale and seasonal patterns. For example, when assessed day-to-day, RH and precipitation were negatively correlated with background Alternaria and Cladosporium spores in a two-year study in Portugal [43]. The authors attributed this result to increased wet deposition rates. In contrast, temperature was positively correlated with both Alternaria and Cladosporium spore levels measured over the course of two years in the atmosphere in the same study [48]. The day-to-day patterns are related to the phenology of the source communities, while diurnal fluctuations may better reflect physiological characteristics of the organisms and their tolerance to environmental stressors. As our sampling scope only covered two days, our results are more likely attributable to physiological characteristics of the organisms and their survival through the unique circumstances characterizing combustion and related aerosolization processes.
Cladosporium spp., ascospore, and basidiospore concentrations are known to exhibit significant diurnal fluctuations, peaking in the morning and evening, when osmotic stress and temperatures are typically lower, and with the lowest concentrations observed between 1200 and 1600 h [49]. Real-time fluorescence techniques also indicate increases in bioparticles when temperatures are lowest and RH is highest [49]. Although we found some correlation with RH and temperature, our results showed no pattern in relation to time sampled, which may be due to the discontinuous temporal scope of our sampling. To account for known diurnal patterns of bioaerosol presence, future smoke bioaerosol campaigns should attempt to sample throughout the day to help control for any non-combustion related patterns.
5. Conclusions
This work provides new evidence for UAS capacity to sample wildland fire aerosolization of living microbes directly from smoke plumes above a forest canopy. Biogenic materials emitted during wildland fire combustion represent a wide variety of sources and a range of sizes, including pollen, plant fragments, seeds, and microbial organisms. The concentration of colony-forming units we collected fall within the range of those reported at the site of large African dust storms, which are considered significant vectors for aerobiological distribution globally [50]. Only a handful of studies have examined the relationship between smoke-impacted air either by measuring bioaerosols directly [20,21,22] or indirectly using biochemical indicators of organism presence and combustion products [23,24]. Only one published study has sampled smoke directly from combustion sources to determine whether viable fungi or bacteria were aerosolized [22]. This study builds on that work by using a UAS to enable the mobile positioning of an active microbial sampling device coupled with environmental monitors directly in the dynamic smoke plume of a wildland fire. The potential impact of the world’s wildland fires on microbial biological diversity and human health warrants further examination using this and other technological advancements.
Our results suggest that this UAS platform has significant potential for future studies of bioaerosols, including using multiple UAS systems to sample fresh and aged smoke to parameterize bioaerosol transport models. Comparisons among the various sampling devices available for bioaerosol capture (e.g., impingers, cyclones, impactors, filters) need further exploration for use on the mobile UAS platform to reduce the potential impacts of rotor wash [31] and desiccation, and to promote both physical and biological sampling efficiencies [35]. Current estimates of global bioaerosol emission rates vary greatly, ranging from ~10 to 1000 Tg annually [45], while estimates of the overall biological component of total aerosols by mass range up to 25% [2]. As a significant component of total aerosol content, emphasis on the contribution of wildland fire to aerosol emissions can help improve our limited understanding of the role of bioaerosols in climate forcing, biological diversity, and terrestrial-atmosphere interactions.
Author Contributions
Conceptualization, L.N.K., M.R.A.P. and T.J.D.; Data curation, M.R.A.P.; Formal analysis, L.N.K., M.R.A.P.; Funding acquisition, L.N.K. and A.C.W.; Investigation, L.N.K., M.R.A.P., A.C.W. and K.N.N.; Methodology, L.N.K., M.R.A.P., A.C.W., K.N.N., T.J.D. and M.R.; Project administration, L.N.K.; Resources, L.N.K. and M.R.; Supervision, L.N.K.; Visualization, L.N.K.; Writing—original draft, L.N.K., M.R.A.P., and K.N.N.; Writing—review & editing, L.N.K., M.R.A.P., A.C.W., K.N.N., T.J.D. and M.R.
Funding
This work was supported in part by grant no. IDAZ-MS-0115/project accession no. 1009933 from the USDA National Institute of Food and Agriculture.
Acknowledgments
We are grateful for the Tall Timbers Research Station and staff in providing logistical support and conducting the prescribed burns studied here, including Kevin Hiers, Morgan Varner, Casey Teske, and Dave Grimm among others. Christian Trucco assisted in payload design. The University of Idaho Forest Pathology and Plant Symbiosis Lab under George Newcombe assisted in species identification. The Desert Research Institute provided in-kind support for UAS piloting and sampling operations. Thanks to Jennifer Andrew for initial contributions.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Andreae, M.O.; Merlet, P. Emission of trace gases and aerosols from biomass burning. Glob. Biogeochem. Cycles 2001, 15, 955–966. [Google Scholar] [CrossRef]
- Jaenicke, R. Abundance of Cellular Material and Proteins in the Atmosphere. Science 2005, 308, 73. [Google Scholar] [CrossRef] [PubMed]
- Charland, A.M.; Clements, C.B. Kinematic structure of a wildland fire plume observed by Doppler lidar. J. Geophys. Res. Atmos. 2013, 118, 3200–3212. [Google Scholar] [CrossRef]
- O’Brien, J.J.; Loudermilk, E.L.; Hornsby, B.; Hudak, A.T.; Bright, B.C.; Dickinson, M.B.; Hiers, J.K.; Teske, C.; Ottmar, R.D. High-resolution infrared thermography for capturing wildland fire behaviour: RxCADRE 2012. Int. J. Wildland Fire 2016, 25, 62–75. [Google Scholar] [CrossRef]
- O’Brien, J.J.; Hiers, J.K.; Varner, J.M.; Hoffman, C.M.; Dickinson, M.B.; Michaletz, S.T.; Loudermilk, E.L.; Butler, B.W. Advances in Mechanistic Approaches to Quantifying Biophysical Fire Effects. Current. Forest. Report. 2018, 4, 161–177. [Google Scholar] [CrossRef]
- Kobziar, L.; Moghaddas, J.; Stephens, S.L. Tree mortality patterns following prescribed fires in a mixed conifer forest. Can. J. For. Res. 2006, 36, 3222–3238. [Google Scholar] [CrossRef][Green Version]
- Pingree, M.R.A.; Kobziar, L.N. The myth of the biological threshold: A review of biological responses to soil heating associated with wildland fire. For. Ecol. Manag. 2019, 432, 1022–1029. [Google Scholar] [CrossRef]
- Glassman, S.I.; Levine, C.R.; DiRocco, A.M.; Battles, J.J.; Bruns, T.D. Ectomycorrhizal fungal spore bank recovery after a severe forest fire: Some like it hot. ISME J. 2016, 10, 1228–1239. [Google Scholar] [CrossRef]
- Polymenakou, P.N.; Polymenakou, P.N. Atmosphere: A Source of Pathogenic or Beneficial Microbes? Atmosphere 2012, 3, 87–102. [Google Scholar] [CrossRef]
- Fröhlich-Nowoisky, J.; Burrows, S.M.; Xie, Z.; Engling, G.; Solomon, P.A.; Fraser, M.P.; Mayol-Bracero, O.L.; Artaxo, P.; Begerow, D.; Conrad, R.; et al. Biogeography in the air: Fungal diversity over land and oceans. Biogeosciences 2012, 9, 1125–1136. [Google Scholar] [CrossRef]
- Lerner, P.I. Nocardiosis. Clin. Infect. Dis. 1996, 22, 891–903. [Google Scholar] [CrossRef]
- Brown, G.D.; Denning, D.W.; Gow, N.A.R.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden Killers: Human Fungal Infections. Sci. Transl. Med. 2012, 4, 165rv13. [Google Scholar] [CrossRef]
- Sharma, D.; Dutta, B.K.; Singh, A.B.; Shome, B.R. Aerobiological, biochemical and immunological studies on some of the dominant Aspergillus species of South Assam (India). Aerobiologia 2007, 23, 201. [Google Scholar] [CrossRef]
- Schmale, D.G.; Ross, S.D. Highways in the sky: Scales of atmospheric transport of plant pathogens. Annu. Rev. Phytopathol. 2015, 53, 591–611. [Google Scholar] [CrossRef]
- Hoose, C.; Kristjánsson, J.E.; Burrows, S.M. How important is biological ice nucleation in clouds on a global scale? Environ. Res. Lett. 2010, 5, 024009. [Google Scholar] [CrossRef]
- Hara, K.; Zhang, D. Bacterial abundance and viability in long-range transported dust. Atmos. Environ. 2012, 47, 20–25. [Google Scholar] [CrossRef]
- Smith, D.J.; Jaffe, D.A.; Birmele, M.N.; Griffin, D.W.; Schuerger, A.C.; Hee, J.; Roberts, M.S. Free Tropospheric Transport of Microorganisms from Asia to North America. Microb. Ecol. 2012, 64, 973–985. [Google Scholar] [CrossRef]
- Franzetti, A.; Gandolfi, I.; Gaspari, E.; Ambrosini, R.; Bestetti, G. Seasonal variability of bacteria in fine and coarse urban air particulate matter. Appl. Microbiol. Biotechnol. 2011, 90, 745–753. [Google Scholar] [CrossRef]
- Haas, D.; Galler, H.; Luxner, J.; Zarfel, G.; Buzina, W.; Friedl, H.; Marth, E.; Habib, J.; Reinthaler, F.F. The concentrations of culturable microorganisms in relation to particulate matter in urban air. Atmos. Environ. 2013, 65, 215–222. [Google Scholar] [CrossRef]
- Mims, S.A.; Mims, F.M. Fungal spores are transported long distances in smoke from biomass fires. Atmos. Environ. 2004, 38, 651–655. [Google Scholar] [CrossRef]
- Camacho, I.; Góis, A.; Camacho, R.; Nóbrega, V. Fernandez The impact of urban and forest fires on the airborne fungal spore aerobiology. Aerobiologia 2018, 34, 585–592. [Google Scholar] [CrossRef]
- Kobziar, L.N.; Pingree, M.R.A.; Larson, H.; Dreaden, T.J.; Green, S.; Smith, J.A. Pyroaerobiology: The aerosolization and transport of viable microbial life by wildland fire. Ecosphere 2018, 9, e02507. [Google Scholar] [CrossRef]
- Rajput, P.; Anjum, M.H.; Gupta, T. One year record of bioaerosols and particles concentration in Indo-Gangetic Plain: Implications of biomass burning emissions to high-level of endotoxin exposure. Environ. Pollut. 2017, 224, 98–106. [Google Scholar] [CrossRef]
- Yang, Y.; Chan, C.; Tao, J.; Lin, M.; Engling, G.; Zhang, Z.; Zhang, T.; Su, L. Observation of elevated fungal tracers due to biomass burning in the Sichuan Basin at Chengdu City, China. Sci. Total Environ. 2012, 431, 68–77. [Google Scholar] [CrossRef]
- Hirst, J.M. An Automatic Volumetric Spore Trap. Ann. Appl. Biol. 1952, 39, 257–265. [Google Scholar] [CrossRef]
- Lin, B.; Bozorgmagham, A.; Ross, S.D.; Schmale, D.G., III. Small fluctuations in the recovery of fusaria across consecutive sampling intervals with unmanned aircraft 100 m above ground level. Aerobiologia 2013, 29, 45–54. [Google Scholar] [CrossRef]
- West, J.; Kimber, R. Innovations in air sampling to detect plant pathogens. Ann. Appl. Biol. 2015, 166, 4–17. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, S.; Sadyś, M.; Smith, M.; Tormo-Molina, R.; Skjøth, C.A.; Maya-Manzano, J.M.; Silva-Palacios, I.; Gonzalo-Garijo, Á. Potential sources of airborne Alternaria spp. spores in South-west Spain. Sci. Total Environ. 2015, 533, 165–176. [Google Scholar] [CrossRef]
- Haig, C.W.; Mackay, W.G.; Walker, J.T.; Williams, C. Bioaerosol sampling: Sampling mechanisms, bioefficiency and field studies. J. Hosp. Infect. 2016, 93, 242–255. [Google Scholar] [CrossRef]
- Villa, T.F.; Gonzalez, F.; Miljievic, B.; Ristovski, Z.D.; Morawska, L. An Overview of Small Unmanned Aerial Vehicles for Air Quality Measurements: Present Applications and Future Prospectives. Sensors 2016, 16, 1072. [Google Scholar] [CrossRef]
- Nelson, K.; Boehmler, J.; Khlystov, A.; Moosmüller, H.; Samburova, V.; Bhattarai, C.; Wilcox, E.; Watts, A. A Multipollutant Smoke Emissions Sensing and Sampling Instrument Package for Unmanned Aircraft Systems: Development and Testing. Fire 2019, 2, 32. [Google Scholar] [CrossRef]
- Haddrell, A.E.; Thomas, R.J. Aerobiology: Experimental Considerations, Observations, and Future Tools. Appl. Environ. Microbiol. 2017, 83, e00809–e00817. [Google Scholar] [CrossRef]
- Macher, J.M. Positive-hole correction of multiple-jet impactors for collecting viable microorganisms. Am. Ind. Hyg. Assoc. J. 1989, 50, 561–568. [Google Scholar] [CrossRef]
- Grinshpun, S.A.; Willeke, K.; Ulevicius, V.; Juozaitis, A.; Terzieva, S.; Donnelly, J.; Stelma, G.N.; Brenner, K.P. Effect of Impaction, Bounce and Reaerosolization on the Collection Efficiency of Impingers. Aerosol Sci. Technol. 1997, 26, 326–342. [Google Scholar] [CrossRef]
- Dybwad, M.; Skogan, G.; Blatny, J.M. Comparative Testing and Evaluation of Nine Different Air Samplers: End-to-End Sampling Efficiencies as Specific Performance Measurements for Bioaerosol Applications. Aerosol Sci. Technol. 2014, 48, 282–295. [Google Scholar] [CrossRef]
- Buttner, M.P.; Stetzenbach, L.D. Evaluation of Four Aerobiological Sampling Methods for the Retrieval of Aerosolized Pseudomonas syringae. Appl. Environ. Microbiol. 1991, 57, 1268–1270. [Google Scholar]
- Jimenez-Sanchez, C.; Hanlon, R.; Aho, K.A.; Powers, C.; Morris, C.E.; Schmale, D.G. Diversity and Ice Nucleation Activity of Microorganisms Collected With a Small Unmanned Aircraft System (sUAS) in France and the United States. Front. Microbiol. 2018, 9, 1667. [Google Scholar] [CrossRef]
- Powers, C.W.; Hanlon, R.; Grothe, H.; Prussin, A.J.; Marr, L.C.; Schmale, D.G. Coordinated Sampling of Microorganisms Over Freshwater and Saltwater Environments Using an Unmanned Surface Vehicle (USV) and a Small Unmanned Aircraft System (sUAS). Front. Microbiol. 2018, 9, 1668. [Google Scholar] [CrossRef]
- Vitorino, L.; Bessa, L. Microbial Diversity: The Gap between the Estimated and the Known. Diversity 2018, 10, 46. [Google Scholar] [CrossRef]
- Bush, R.K.; Prochnau, J.J. Alternaria-induced asthma. J. Allergy Clin. Immunol. 2004, 113, 227–234. [Google Scholar] [CrossRef]
- Mahadevakumar, S.; Jayaramaiah, K.M.; Janardhana, G.R. First Report of Leaf Spot Disease Caused by Epicoccum nigrum on Lablab purpureus in India. Plant Dis. 2014, 98, 284. [Google Scholar] [CrossRef] [PubMed]
- de Fávaro, L.C.L.; de Sebastianes, F.L.S.; Araújo, W.L. Epicoccum nigrum P16, a Sugarcane Endophyte, Produces Antifungal Compounds and Induces Root Growth. PLoS ONE 2012, 7, e36826. [Google Scholar] [CrossRef] [PubMed]
- Kloepper, J.W.; Leong, J.; Teintze, M.; Schroth, M.N. Pseudomonas siderophores: A mechanism explaining disease-suppressive soils. Curr. Microbiol. 1980, 4, 317–320. [Google Scholar] [CrossRef]
- Conen, F.; Morris, C.E.; Leifeld, J.; Yakutin, M.V.; Alewell, C. Biological residues define the ice nucleation properties of soil dust. Atmos. Chem. Phys. 2011, 11, 9643–9648. [Google Scholar] [CrossRef]
- Fröhlich-Nowoisky, J.; Kampf, C.J.; Weber, B.; Huffman, J.A.; Pöhlker, C.; Andreae, M.O.; Lang-Yona, N.; Burrows, S.M.; Gunthe, S.S.; Elbert, W.; et al. Bioaerosols in the Earth system: Climate, health, and ecosystem interactions. Atmos. Res. 2016, 182, 346–376. [Google Scholar] [CrossRef]
- Petters, M.D.; Parsons, M.T.; Prenni, A.J.; DeMott, P.J.; Kreidenweis, S.M.; Carrico, C.M.; Sullivan, A.P.; McMeeking, G.R.; Levin, E.; Wold, C.E.; et al. Ice nuclei emissions from biomass burning. J. Geophys. Res. Atmospheres 2009, 114, D07209. [Google Scholar] [CrossRef]
- Fan, X.-Y.; Gao, J.-F.; Pan, K.-L.; Li, D.-C.; Dai, H.-H.; Li, X. More obvious air pollution impacts on variations in bacteria than fungi and their co-occurrences with ammonia-oxidizing microorganisms in PM2.5. Environ. Pollut. 2019, 251, 668–680. [Google Scholar] [CrossRef]
- Almeida, E.; Caeiro, E.; Todo-Bom, A.; Ferro, R.; Dionísio, A.; Duarte, A.; Gazarini, L. The influence of meteorological parameters on Alternaria and Cladosporium fungal spore concentrations in Beja (Southern Portugal): Preliminary results. Aerobiologia 2018, 34, 219–226. [Google Scholar] [CrossRef]
- Healy, D.A.; Huffman, J.A.; O’Connor, D.J.; Pöhlker, C.; Pöschl, U.; Sodeau, J.R. Ambient measurements of biological aerosol particles near Killarney, Ireland: A comparison between real-time fluorescence and microscopy techniques. Atmos. Chem. Phys. 2014, 14, 8055–8069. [Google Scholar] [CrossRef]
- Kellogg, C.A.; Griffin, D.W. Aerobiology and the global transport of desert dust. Trends Ecol. Evolut. 2006, 21, 638–644. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).