Synthesis and Characterization of Ceria- and Samaria-Based Powders and Solid Electrolytes as Promising Components of Solid Oxide Fuel Cells
Abstract
1. Introduction
2. Experimental
2.1. Synthesis of Xerogels, Powders and Ceramic Materials of the Composition (CeO2)1-x(Sm2O3)x (x = 0.05, 0.10, 0.20)
2.2. Material and Methods
3. Results and Discussion
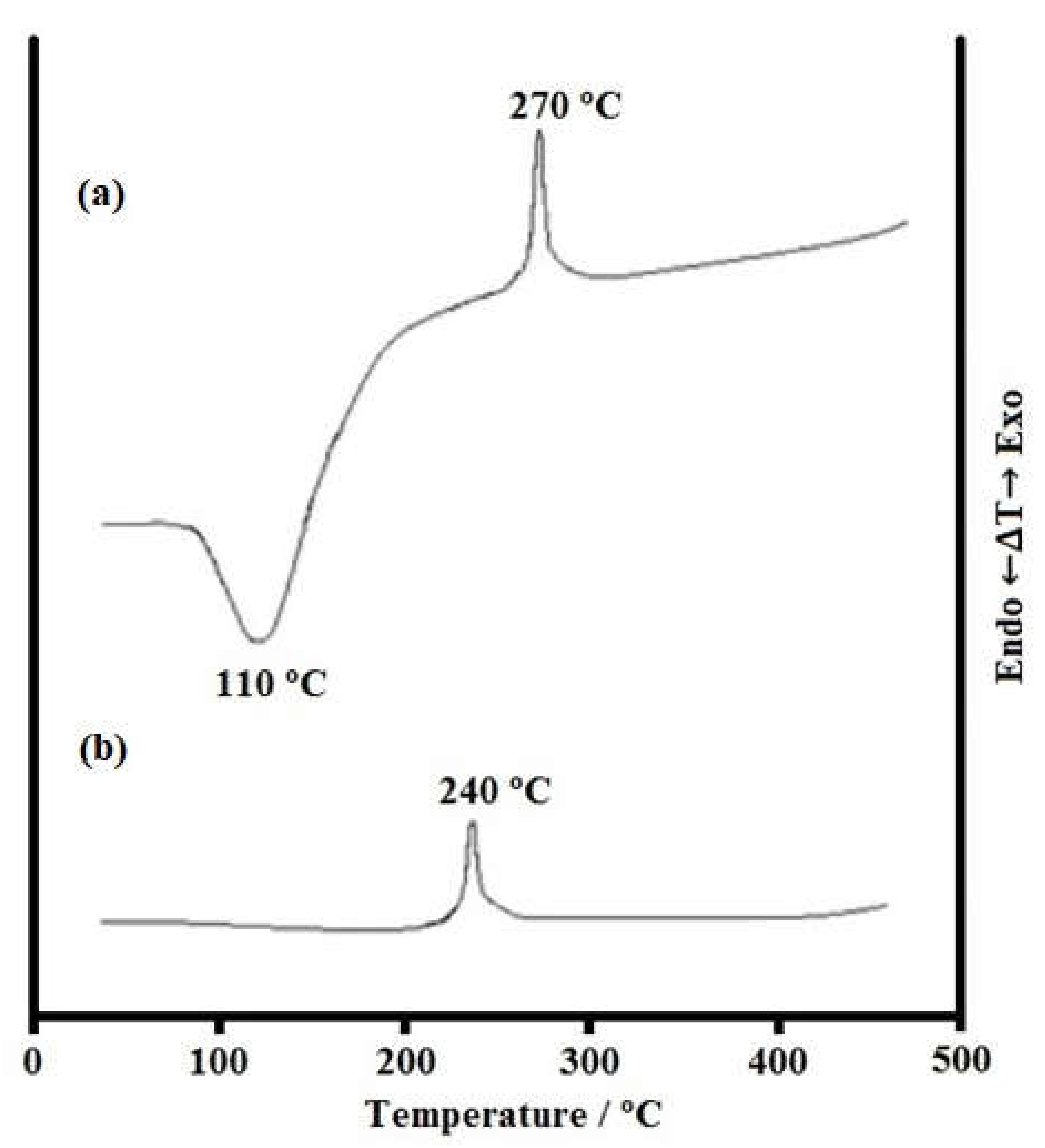
3.1. Study of the Synthesized Xerogels Thermolysis
3.2. Low-Temperature Nitrogen Adsorption Characterization of the Microstructure of Powders Synthesized by Co-Precipitation of Hydroxides
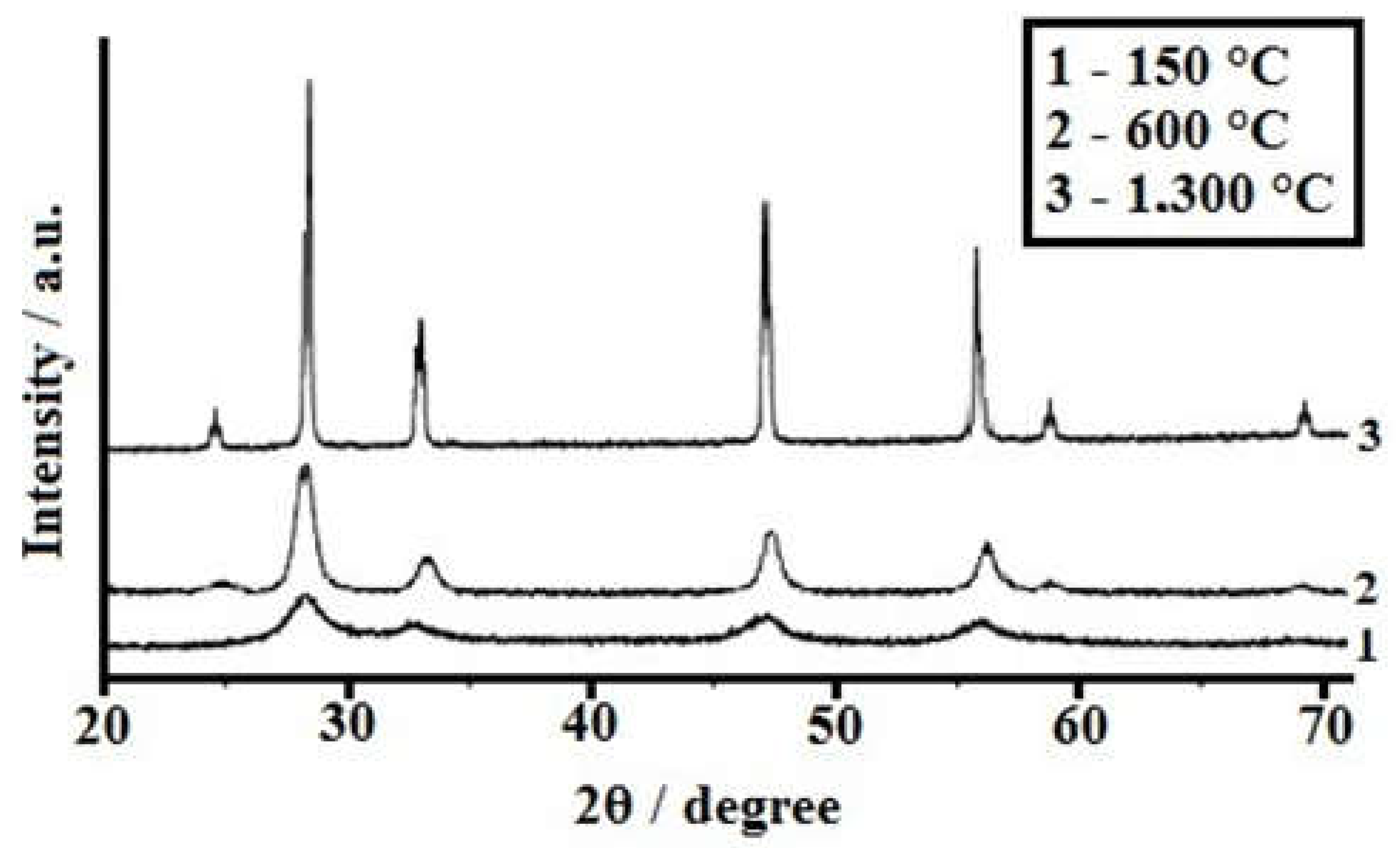
3.3. Study of the Crystal Structure of the Obtained Powders
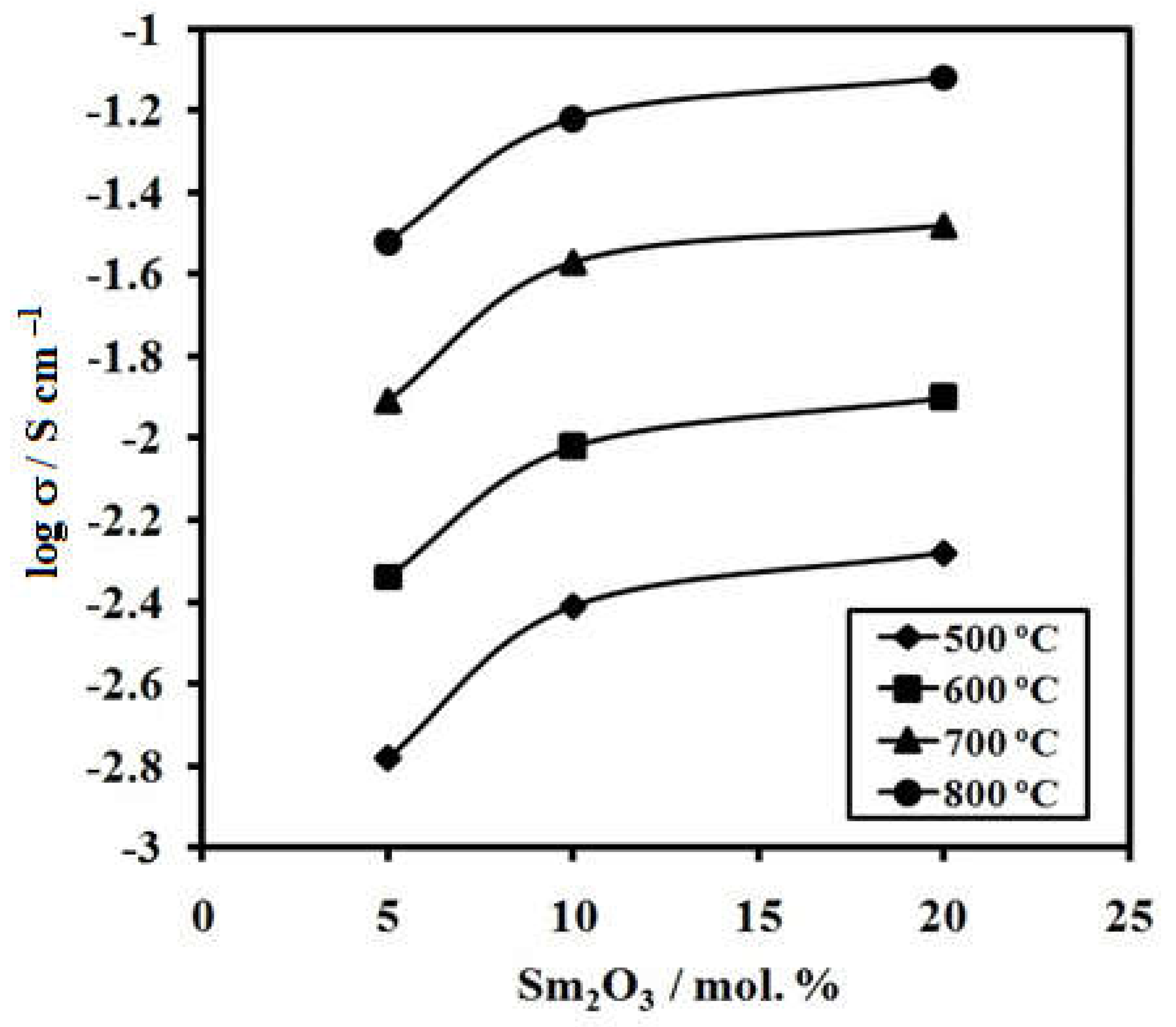
3.4. Electrical Properties of Ceramic Samples
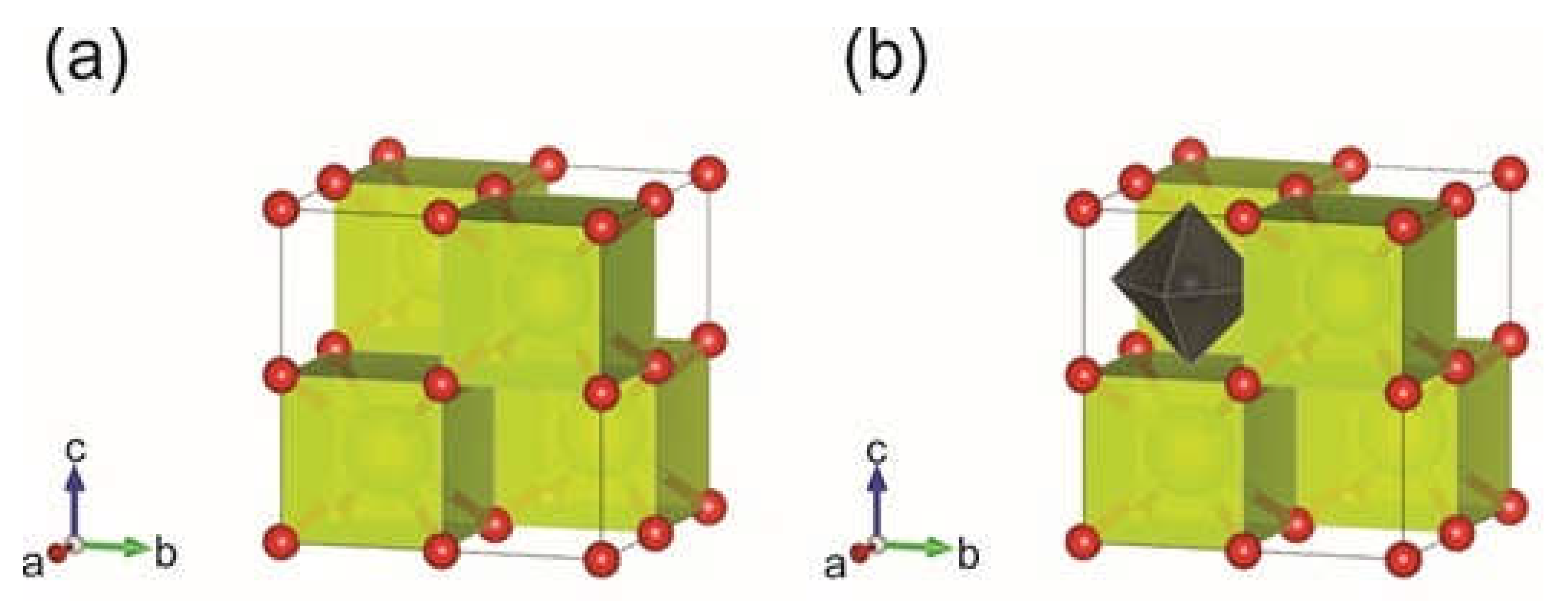
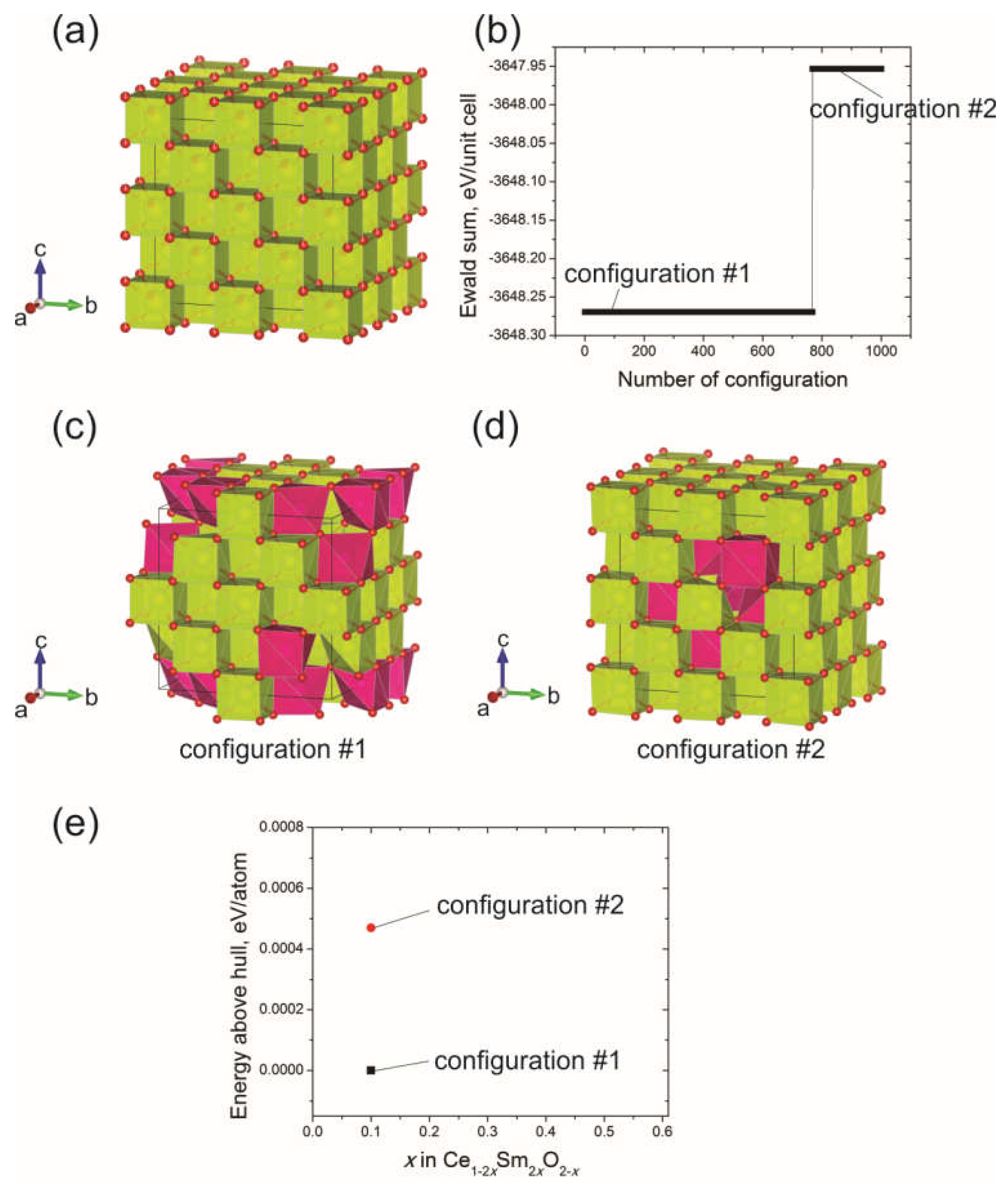
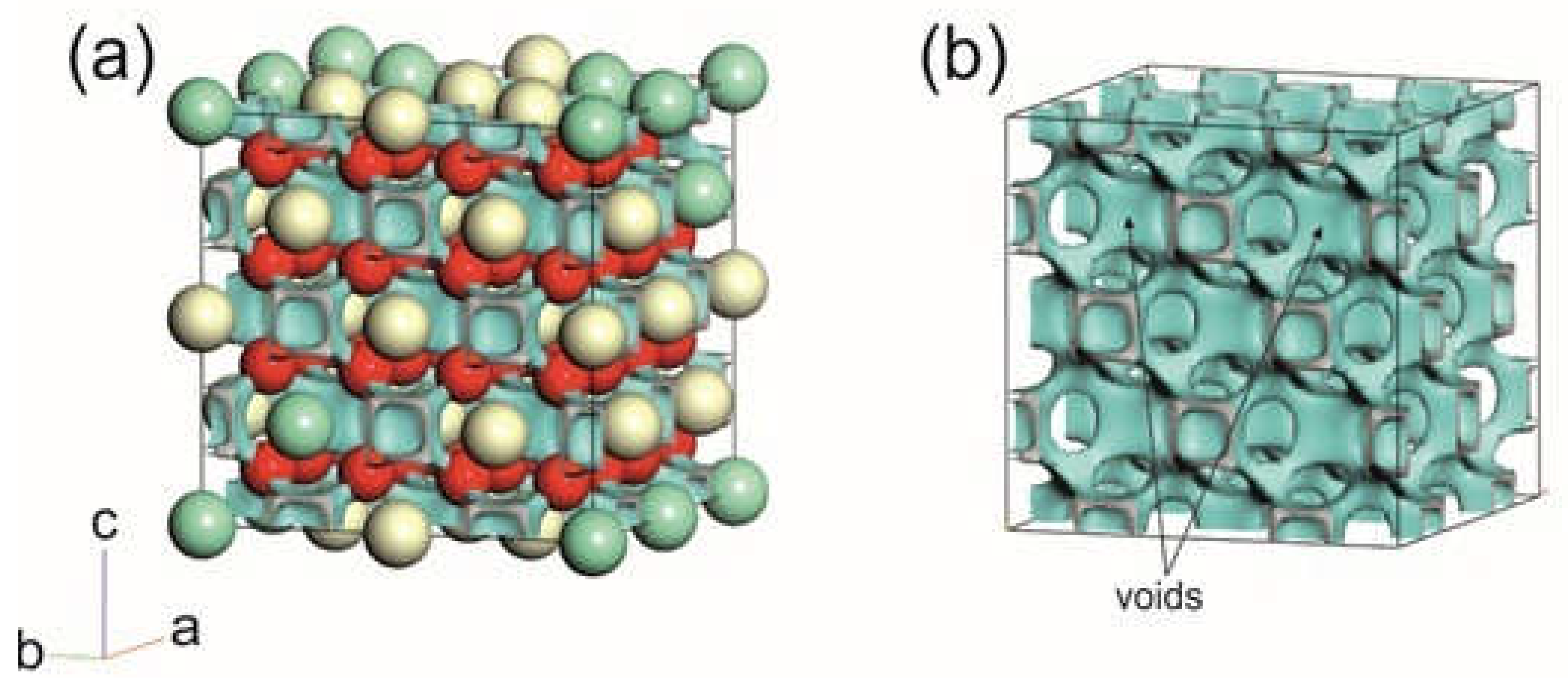
3.5. Ab Initio Studies of Samarium-Doped Ceria
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maric, R.; Mirshekari, G. Solid Oxide Fuel Cells: From Fundamental Principles to Complete Systems, 1st ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar] [CrossRef]
- Pachauri, R.K.; Chauhan, Y.K. A study, analysis and power management schemes for fuel cells. Renew. Sustain. Energy Rev. 2015, 43, 1301–1319. [Google Scholar] [CrossRef]
- Tarancón, A. Strategies for Lowering Solid Oxide Fuel Cells Operating Temperature. Energies 2009, 2, 1130–1150. [Google Scholar] [CrossRef]
- Sunandana, C.S. Introduction to Solid State Ionics: Phenomenology and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar] [CrossRef]
- Liang, J. Low temperature fabrication of dense gadolinia-doped ceria electrolyte with enhanced electrical conductivity. Electrochim. Acta 2015, 178, 321–328. [Google Scholar] [CrossRef]
- Rekas, M. Electrolytes for Intermediate Temperature Solid Oxide Fuel Cells. Arch. Met. Mater. 2015, 60, 891–896. [Google Scholar] [CrossRef]
- Mahato, N.; Banerjee, A.; Gupta, A.; Omar, S.; Balani, K. Progress in material selection for solid oxide fuel cell technology: A review. Prog. Mater. Sci. 2015, 72, 141–337. [Google Scholar] [CrossRef]
- Sal’Nikov, V.V.; Pikalova, E.Y. Raman and impedance spectroscopic studies of the specific features of the transport properties of electrolytes based on CeO. Phys. Solid State 2015, 57, 1944–1952. [Google Scholar] [CrossRef]
- Egorova, T.L.; Kalinina, M.V.; Simonenko, T.; Shilova, O.A.; Sevastyanov, V.G.; Kuznetsov, N.T. Liquid-phase synthesis and physicochemical properties of xerogels, nanopowders and thin films of the CeO2–Y2O3 system. Russ. J. Inorg. Chem. 2016, 61, 1061–1069. [Google Scholar] [CrossRef]
- Ramos-Alvarez, P.; Villafuerte-Castrejón, M.E.; González, G.; Cassir, M.; Flores-Morales, C.; Chávez-Carvayar, J.A. Ceria-based electrolytes with high surface area and improved conductivity for intermediate temperature solid oxide fuel cells. J. Mater. Sci. 2016, 52, 519–532. [Google Scholar] [CrossRef]
- Kalinina, M.V.; Morozova, L.; Egorova, T.L.; Arsent’Ev, M.Y.; Drozdova, I.; Shilova, O.A. Synthesis and physicochemical properties of a solid oxide nanocomposite based on a ZrO2–Y2O3–Gd2O3–MgO system. Glas. Phys. Chem. 2016, 42, 505–511. [Google Scholar] [CrossRef]
- Shilova, O.A.; Antipov, V.N.; Tikhonov, P.A.; Kruchinina, I.Y.; Arsent’Ev, M.Y.; Panova, T.I.; Morozova, L.V.; Moskovskaya, V.V.; Kalinina, M.V.; Tsvetkova, I.N. Ceramic nanocomposites based on oxides of transition metals for ionistors. Glas. Phys. Chem. 2013, 39, 570–578. [Google Scholar] [CrossRef]
- Rempel, A.A.; Gusev, A.I. Nanocrystalline Materials; Cambridge International Science Publishing: Cambridge, UK, 2004; p. 351. ISBN 978-1-898326-26-7. [Google Scholar]
- Tian, C. Ionic conductivities, sintering temperatures and microstructures of bulk ceramic CeO2 doped with Y2O. Solid State Ionics 2000, 134, 89–102. [Google Scholar] [CrossRef]
- Durán, P.; Villegas, M.; Capel, F.; Recio, P.; Moure, C. Low-temperature sintering and microstructural development of nanocrystalline Y-TZP powders. J. Eur. Ceram. Soc. 1996, 16, 945–952. [Google Scholar] [CrossRef]
- Klabunde, K.; Sergeev, G. Nanochemistry, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2013; ISBN 9780444594099. [Google Scholar]
- Kroger, F.A. The Chemistry of Imperfect Crystals; North-Holland Publishing Company-Amsterdam: Amsterdam, The Netherlands, 1964; p. 1039. [Google Scholar] [CrossRef]
- Russian Standard GOST 473.4-81, 1981. (In Russian), Products Chemically Resistant and Heat-Resistant Ceramic. Method for Determining Apparent Density and Apparent Porosity GOST 473.4 - 81. 1981. Available online: https://docs.cntd.ru/document/1200013968 (accessed on 23 November 2005).
- Pivovarova, A.P.; Strakhov, V.I.; Popov, V.P. On the mechanism of electron conductivity in lanthanum metaniobate. Tech. Phys. Lett. 2002, 28, 815–817. [Google Scholar] [CrossRef]
- Dell’Agli, G.; Spiridigliozzi, L.; Marocco, A.; Accardo, G.; Frattini, D.; Kwon, Y.; Yoon, S.P. Morphological and crystalline evolution of Sm-(20 mol%)–doped ceria nanopowders prepared by a combined co-precipitation/hydrothermal synthesis for solid oxide fuel cell applications. Ceram. Int. 2017, 43, 12799–12808. [Google Scholar] [CrossRef]
- Andersson, D.A.; Simak, S.I.; Skorodumova, N.V.; Abrikosov, I.A.; Johansson, B. Optimization of ionic conductivity in doped ceria. Proc. Natl. Acad. Sci. USA 2006, 103, 3518–3521. [Google Scholar] [CrossRef] [PubMed]
- Hart, G.L.W.; Forcade, R.W. Algorithm for generating derivative structures. Phys. Rev. B 2008, 77, 224115. [Google Scholar] [CrossRef]
- Antipov, E.V.; Khasanova, N.R.; Fedotov, S. Perspectives on Li and transition metal fluoride phosphates as cathode materials for a new generation of Li-ion batteries. IUCrJ 2015, 2, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Barpanda, P.; Oyama, G.; Nishimura, S.-I.; Chung, S.-C.; Yamada, A. A 3.8-V earth-abundant sodium battery electrode. Nat. Commun. 2014, 5, 4358. [Google Scholar] [CrossRef] [PubMed]
- Kalinina, M.V.; Duskina, D.A.; Kovalko, N.Y.; Myakin, S.V.; Arsent’Ev, M.Y.; Khristyuk, N.A.; Shilova, O.A. Liquid-Phase Synthesis and Physical and Chemical Properties of Ceramic Electrolyte Nanomaterials in the CeO2–Sm2O3 System for Solid Oxide Fuel Cells. Inorg. Mater. Appl. Res. 2020, 11, 1229–1235. [Google Scholar] [CrossRef]
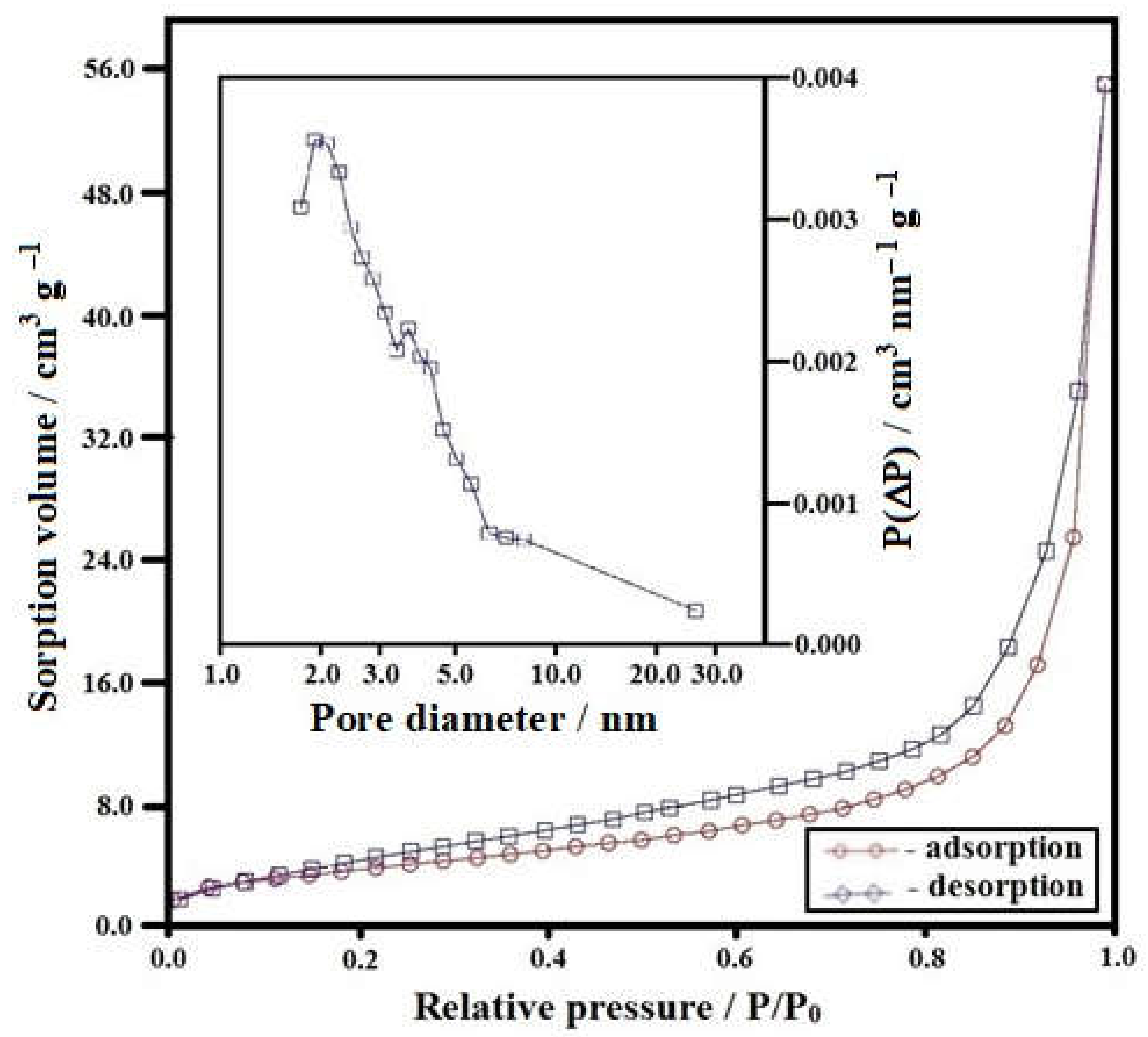



| Composition | Specific Surface Area Ss, m2/g | Average Pore Size Dpor, Nm | Specific Pore Volume Vpor, cm3/g |
|---|---|---|---|
| (CeO2)0.95(Sm2O3)0.05 | 50 | 3.6 | 0.080 |
| (CeO2)0.90(Sm2O3)0.10 | 78 | 2.5 | 0.086 |
| (CeO2)0.80(Sm2O3)0.20 | 83 | 1.5 | 0.092 |
| Composition | b ρteor g/cm3 | a Apparent density ρexp g/cm3 | c ρrel, % | CSR, nm (1300 °C) | f P, % | d σi·10−2 S/cm (700 °C) | e Ea, eV | Structure, Parameter a |
|---|---|---|---|---|---|---|---|---|
| (CeO2)0.95(Sm2O3)0.05 | 7.23 | 6.55 | 91 | 69 | 2.0 | 1.2 | 1.35 | F a = 5.4292 |
| (CeO2)0.90(Sm2O3)0.10 | 6.98 | 6.33 | 91 | 68 | 3.8 | 2.7 | 1.31 | F a = 5.4391 |
| (CeO2)0.80(Sm2O3)0.20 | 6.90 | 6.25 | 91 | 65 | 6.2 | 3.3 | 1.29 | F a = 5.4471 |
| T, °C | ti | te |
|---|---|---|
| 300 | 0.85 | 0.15 |
| 400 | 0.80 | 0.20 |
| 500 | 0.78 | 0.22 |
| 600 | 0.75 | 0.25 |
| 700 | 0.73 | 0.27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalinina, M.V.; Dyuskina, D.A.; Arsent’ev, M.Y.; Mjakin, S.V.; Shilova, O.A. Synthesis and Characterization of Ceria- and Samaria-Based Powders and Solid Electrolytes as Promising Components of Solid Oxide Fuel Cells. Ceramics 2022, 5, 1102-1114. https://doi.org/10.3390/ceramics5040078
Kalinina MV, Dyuskina DA, Arsent’ev MY, Mjakin SV, Shilova OA. Synthesis and Characterization of Ceria- and Samaria-Based Powders and Solid Electrolytes as Promising Components of Solid Oxide Fuel Cells. Ceramics. 2022; 5(4):1102-1114. https://doi.org/10.3390/ceramics5040078
Chicago/Turabian StyleKalinina, Marina V., Daria A. Dyuskina, Maxim Y. Arsent’ev, Sergey V. Mjakin, and Olga A. Shilova. 2022. "Synthesis and Characterization of Ceria- and Samaria-Based Powders and Solid Electrolytes as Promising Components of Solid Oxide Fuel Cells" Ceramics 5, no. 4: 1102-1114. https://doi.org/10.3390/ceramics5040078
APA StyleKalinina, M. V., Dyuskina, D. A., Arsent’ev, M. Y., Mjakin, S. V., & Shilova, O. A. (2022). Synthesis and Characterization of Ceria- and Samaria-Based Powders and Solid Electrolytes as Promising Components of Solid Oxide Fuel Cells. Ceramics, 5(4), 1102-1114. https://doi.org/10.3390/ceramics5040078








