Ice-Templating for the Elaboration of Oxygen Permeation Asymmetric Tubular Membrane with Radial Oriented Porosity
Abstract
:1. Introduction
2. Materials and Methods
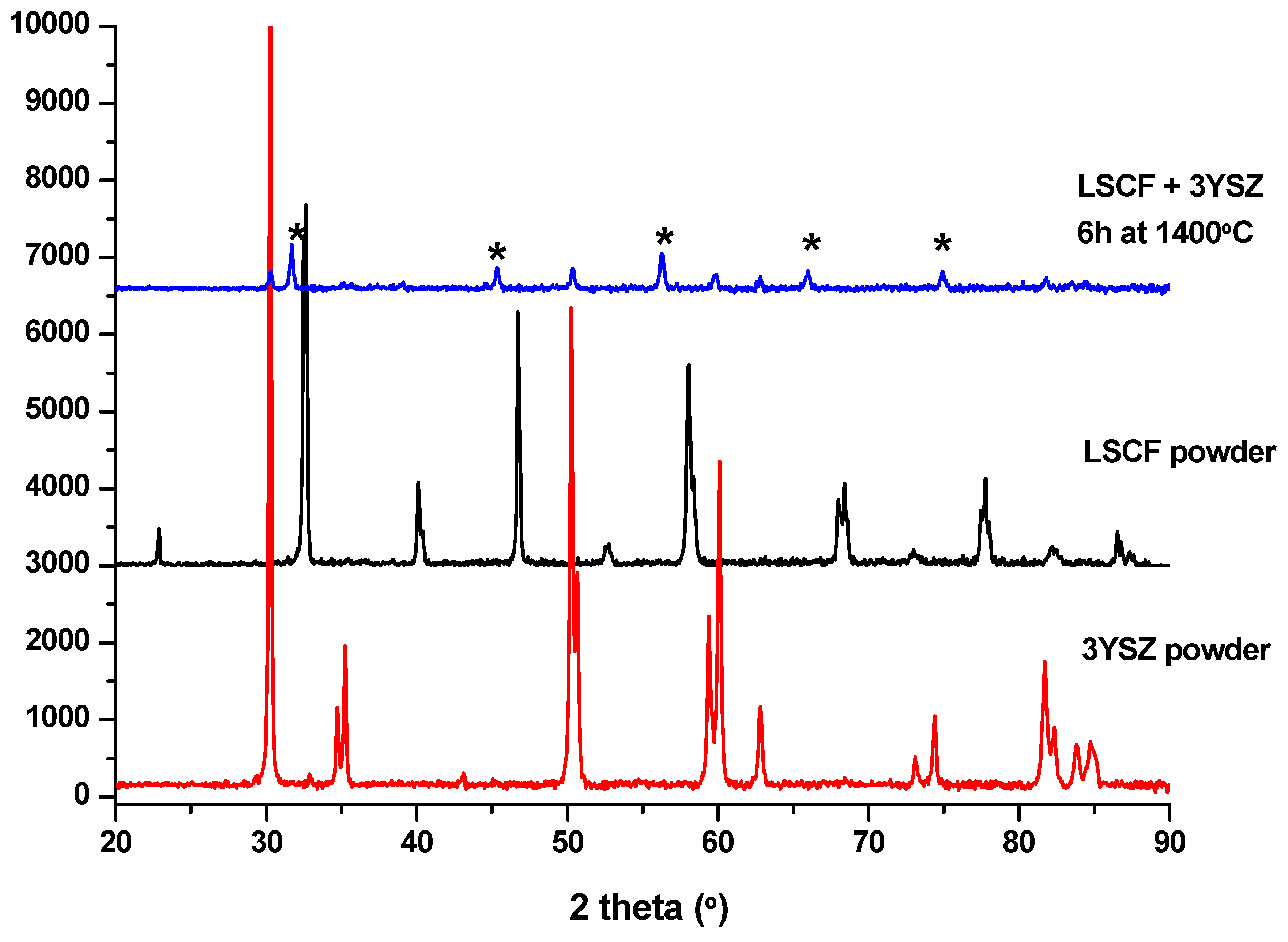
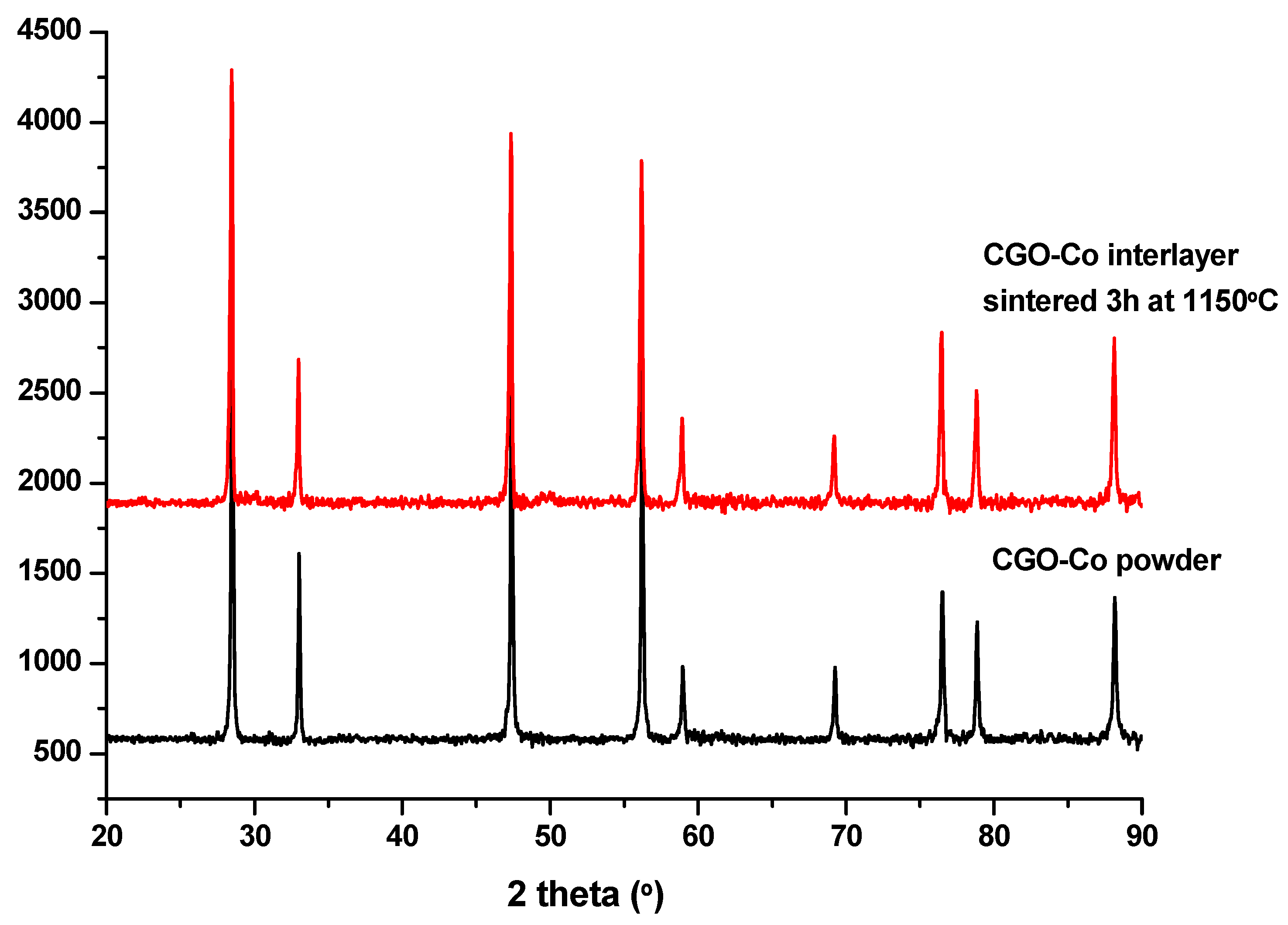
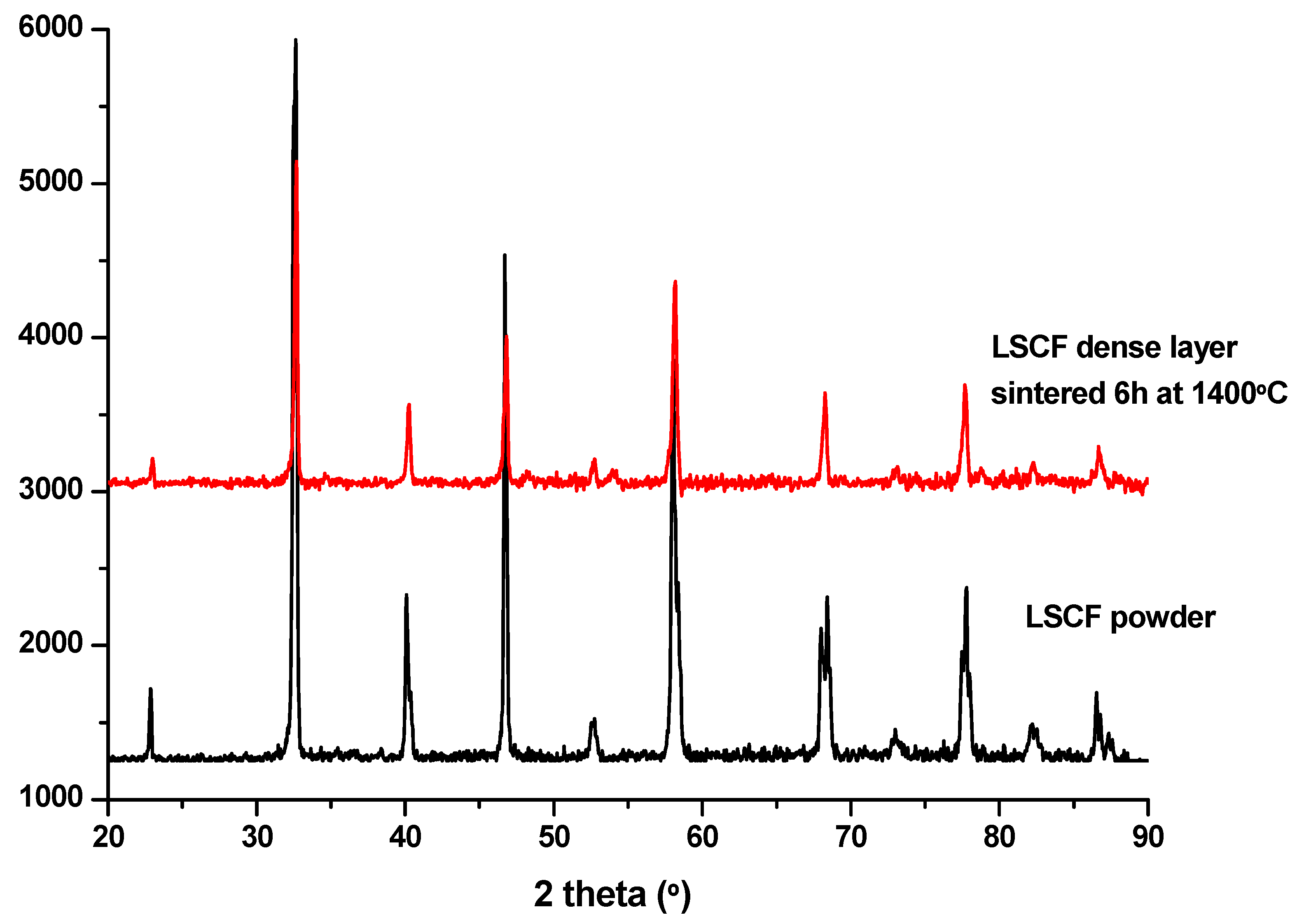
3. Results and Discussion
Preliminary Work
4. Asymmetric Tubular Membrane Characterization
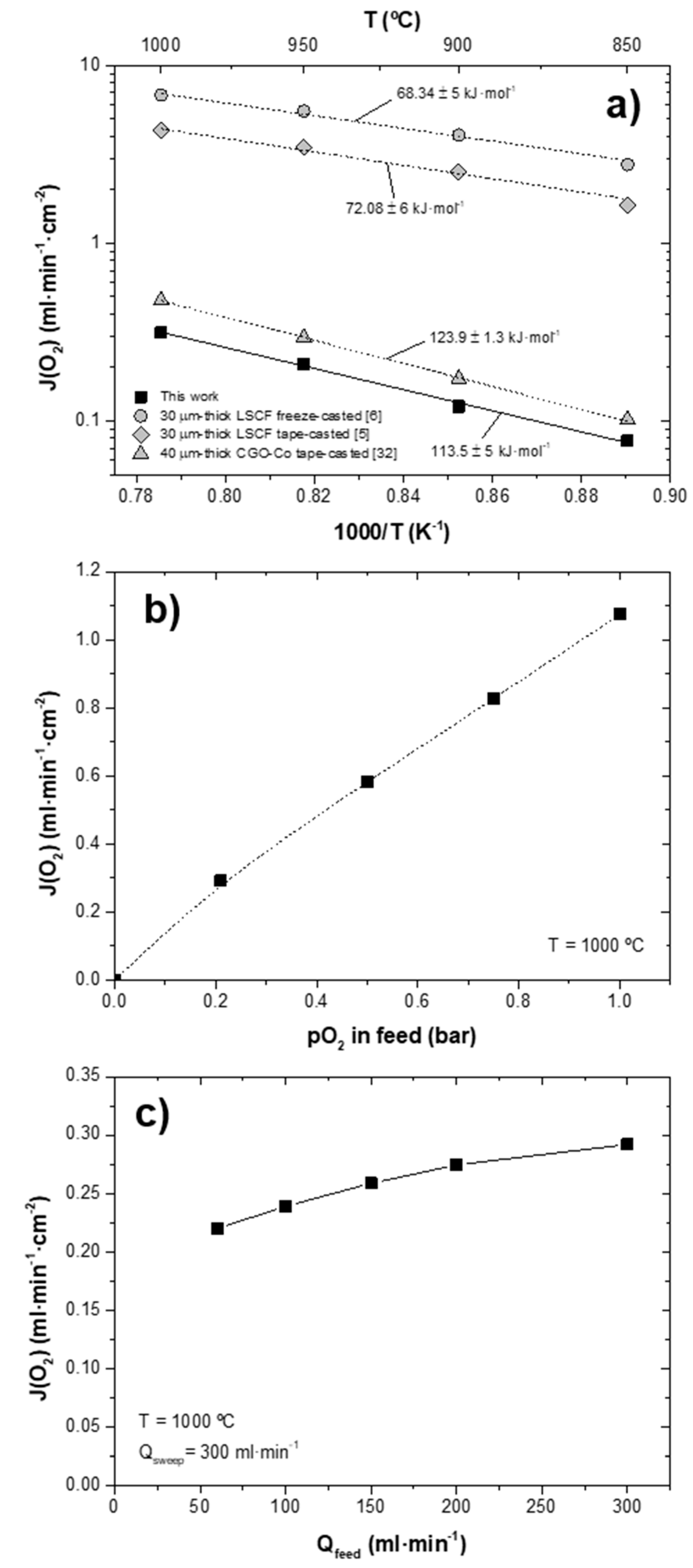
5. Oxygen Permeation Evaluation
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hashim, S.S.; Mohamed, A.R.; Bhatia, S. Oxygen separation from air using ceramic-based membrane technology for sustainable fuel production and power generation. Renew. Sustain. Energy Rev. 2011, 15, 1284–1293. [Google Scholar] [CrossRef]
- Bredesen, R.; Sogge, A. A technical and economic assessment of membrane reactors for hydrogen and syngas production. In Proceedings of the Seminar on the Ecological Applications of Innovative Membrane Technology in the Chemical Industry, Cetraro, Italy, 1 May 1996. [Google Scholar]
- Den Exter, M.; Haije, W.; Vente, J. Viability of ITM Technology for Oxygen Production and Oxidation Processes: Material, System, and Process Aspects. In Inorganic Membranes for Energy and Environmental Applications; Bose, A., Ed.; Springer: New York, NY, USA, 2009; pp. 27–51. [Google Scholar]
- Rajesh, S.; Pereira, J.R.S.; Figueiredo, F.M.L.; Marques, F.M.B. Performance of Carbonate—LaCoO3 and La0.8Sr0.2Co0.2Fe0.8O3-δ Composite Cathodes under Carbon Dioxide. Electrochim. Acta 2014, 125, 435–442. [Google Scholar] [CrossRef]
- Serra, J.M.; Garcia-Fayos, J.; Baumann, S.; Schulze-Küppers, F.; Meulenberg, W.A. Oxygen permeation through tape-cast asymmetric all-La0.6Sr0.4Co0.2Fe0.8O3−δ membranes. J. Membr. Sci. 2013, 447, 297–305. [Google Scholar] [CrossRef]
- Gaudillere, C.; Garcia-Fayos, J.; Serra, J. Enhancing oxygen permeation through hierarchically-structured perovskite membranes elaborated by freeze-casting. J. Mater. Chem. A 2013. [Google Scholar] [CrossRef]
- Deville, S. Freeze-casting of porous ceramics: A review of current achievements and issues. Adv. Eng. Mater. 2008, 10, 155–169. [Google Scholar] [CrossRef]
- Gaudillere, C.; Garcia-Fayos, J.; Balaguer, M.; Serra, J.M. Enhanced Oxygen Separation through Robust Freeze-Cast Bilayered Dual-Phase Membranes. ChemSusChem 2014, 7, 2554–2561. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Chua, W. Fabrication of a dense La0.2Sr0.8CoO3−δ/CoO composite membrane by utilizing the electroless cobalt plating technique. J. Membr. Sci. 2002, 198, 95–108. [Google Scholar] [CrossRef]
- Middleton, H.; Diethelm, S.; Ihringer, R.; Larrain, D.; Sfeir, J.; Van Herle, J. Co-casting and co-sintering of porous MgO support plates with thin dense perovskite layers of LaSrFeCoO3. J. Eur. Ceram. Soc. 2004, 24, 1083–1086. [Google Scholar] [CrossRef]
- Lee, T.H.; Yang, Y.L.; Jacobson, A.J.; Abeles, B.; Zhou, M. Oxygen permeation in dense SrCo0.8Fe0.2O3—δ membranes: Surface exchange kinetics versus bulk diffusion. Solid State Ionics 1997, 100, 77–85. [Google Scholar] [CrossRef]
- Balachandran, U.; Dusek, J.T.; Maiya, P.S.; Ma, B.; Mieville, R.L.; Kleefisch, M.S.; Udovich, C.A. Ceramic membrane reactor for converting methane to syngas. Catal. Today 1997, 36, 265–272. [Google Scholar] [CrossRef]
- Balachandran, U.; Dusek, J.T.; Mieville, R.L.; Poeppel, R.B.; Kleefisch, M.S.; Pei, S.; Kobylinski, T.P.; Udovich, C.A.; Bose, A.C. Dense ceramic membranes for partial oxidation of methane to syngas. Appl. Catal. A Gener. 1995, 133, 19–29. [Google Scholar] [CrossRef]
- Li, S.; Jin, W.; Huang, P.; Xu, N.; Shi, J.; Lin, Y.S. Tubular lanthanum cobaltite perovskite type membrane for oxygen permeation. J. Membr. Sci. 2000, 166, 51–61. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, G.; Dong, X.; Jiang, W.; Jin, W.; Xu, N. Fabrication of asymmetric tubular mixed-conducting dense membranes by a combined spin-spraying and co-sintering process. J. Membr. Sci. 2012, 415–416, 313–319. [Google Scholar] [CrossRef]
- Ito, W.; Nagai, T.; Sakon, T. Oxygen separation from compressed air using a mixed conducting perovskite-type oxide membrane. Solid State Ionics 2007, 178, 809–816. [Google Scholar] [CrossRef]
- Zhu, X.; Sun, S.; Cong, Y.; Yang, W. Operation of perovskite membrane under vacuum and elevated pressures for high-purity oxygen production. J. Membr. Sci. 2009, 345, 47–52. [Google Scholar] [CrossRef]
- Moon, J.W.; Hwang, H.J.; Awano, M.; Maeda, K. Preparation of NiO-YSZ tubular support with radially aligned pore channels. Mater. Lett. 2003, 57, 1428–1434. [Google Scholar] [CrossRef]
- Moon, Y.W.; Shin, K.H.; Koh, Y.H.; Yook, S.W.; Han, C.M.; Kim, H.E. Novel ceramic/camphene-based co-extrusion for highly aligned porous alumina ceramic tubes. J. Am. Ceram. Soc. 2012, 95, 1803–1806. [Google Scholar] [CrossRef]
- Liu, R.; Yuan, J.; Wang, C.A. A novel way to fabricate tubular porous mullite membrane supports by TBA-based freezing casting method. J. Eur. Ceram. Soc. 2013, 33, 3249–3256. [Google Scholar] [CrossRef]
- Seub, J.; Leloup, J.; Richaud, S.; Deville, S.; Guizard, C.; Stevenson, A.J. Fabrication of ice-templated tubes by rotational freezing: Microstructure, strength, and permeability. J. Eur. Ceram. Soc. 2017, 37, 2423–2429. [Google Scholar] [CrossRef]
- Knibbe, R.; Hjelm, J.; Menon, M.; Pryds, N.; Søgaard, M.; Wang, H.J.; Neufeld, K. Cathode–Electrolyte Interfaces with CGO Barrier Layers in SOFC. J. Am. Ceram. Soc. 2010, 93, 2877–2883. [Google Scholar] [CrossRef]
- Qiu, L.; Ichikawa, T.; Hirano, A.; Imanishi, N.; Takeda, Y. Ln1−xSrxCo1−yFeyO3−δ (Ln = Pr, Nd, Gd; x = 0.2, 0.3) for the electrodes of solid oxide fuel cells. Solid State Ionics 2003, 158, 55–65. [Google Scholar] [CrossRef]
- Zhou, X.D.; Scarfino, B.; Anderson, H.U. Electrical conductivity and stability of Gd-doped ceria/Y-doped zirconia ceramics and thin films. Solid State Ionics 2004, 175, 19–22. [Google Scholar] [CrossRef]
- Mori, M.; Hiei, Y.; Itoh, H.; Tompsett, G.A.; Sammes, N.M. Evaluation of Ni and Ti-doped Y2O3 stabilized ZrO2 cermet as an anode in high-temperature solid oxide fuel cells. Solid State Ionics 2003, 160, 1–14. [Google Scholar] [CrossRef]
- Antunes, B.C. Production of Volumetric Ceramic Porous Burners by Direct Foaming Process. Ph.D. Thesis, University of Bremen, Advanced Ceramics Institute, Bremen, Germany, 2010. [Google Scholar]
- Schulze-Küppers, F.; Baumann, S.; Tietz, F.; Bouwmeester, H.J.M.; Meulenberg, W.A. Towards the fabrication of La0.98−xSrxCo0.2Fe0.8O3−δ perovskite-type oxygen transport membranes. J. Eur. Ceram. Soc. 2014, 34, 3741–3748. [Google Scholar] [CrossRef]
- Pérez-Coll, D.; Núñez, P.; Abrantes, J.C.C.; Fagg, D.P.; Kharton, V.V.; Frade, J.R. Effects of firing conditions and addition of Co on bulk and grain boundary properties of CGO. Solid State Ionics 2005, 176, 2799–2805. [Google Scholar] [CrossRef]
- Baque, L.; Padmasree, K.P.; Reyes, M.A.C.; Troiani, H.; Arce, M.D.; Serquis, A.; Soldati, A. Effect of Cobalt-Doped Electrolyte on the Electrochemical Performance of LSCFO/CGO Interfaces. ECS Trans. 2016, 72, 117–121. [Google Scholar] [CrossRef]
- Balaguer, M.; Solis, C.; Roitsch, S.; Serra, J.M. Engineering microstructure and redox properties in the mixed conductor Ce0.9Pr0.1O2-[small delta] + Co 2 mol%. Dalton Trans. 2014, 43, 4305–4312. [Google Scholar] [CrossRef]
- Gaudillere, C.; Garcia-Fayos, J.; Serra, J.M. Oxygen Permeation Improvement under CO2-Rich Environments through Catalytic Activation of Hierarchically Structured Perovskite Membranes. ChemPlusChem 2014, 79, 1720–1725. [Google Scholar] [CrossRef]
- Garcia-Fayos, J.; Søgaard, M.; Kaiser, A.; Serra, J.M. Oxygen permeation studies in surface Pd-activated asymmetric Ce0.9Gd0.1O1.95 membranes for application in CO2 and CH4 environments. Sep. Purif. Technol. 2019, 216, 58–64. [Google Scholar] [CrossRef]
- Bouwmeester, H.; Burggraaf, A. Dense Ceramic Membranes for Oxygen Separation. In Handbook of Solid State Electrochemistry; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar] [CrossRef]
- Escribano, J.A.; García-Fayos, J.; Serra, J.M. Shaping of 3YSZ porous substrates for oxygen separation membranes. J. Eur. Ceram. Soc. 2017, 37, 5223–5231. [Google Scholar] [CrossRef]
- Lobera, M.P.; Balaguer, M.; García-Fayos, J.; Serra, J.M. Catalytic Oxide-Ion Conducting Materials for Surface Activation of Ba0.5Sr0.5Co0.8Fe0.2O3-δ Membranes. ChemistrySelect 2017, 2, 2949–2955. [Google Scholar] [CrossRef]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaudillere, C.; Garcia-Fayos, J.; Plaza, J.; Serra, J.M. Ice-Templating for the Elaboration of Oxygen Permeation Asymmetric Tubular Membrane with Radial Oriented Porosity. Ceramics 2019, 2, 246-259. https://doi.org/10.3390/ceramics2020020
Gaudillere C, Garcia-Fayos J, Plaza J, Serra JM. Ice-Templating for the Elaboration of Oxygen Permeation Asymmetric Tubular Membrane with Radial Oriented Porosity. Ceramics. 2019; 2(2):246-259. https://doi.org/10.3390/ceramics2020020
Chicago/Turabian StyleGaudillere, Cyril, Julio Garcia-Fayos, Jorge Plaza, and José M. Serra. 2019. "Ice-Templating for the Elaboration of Oxygen Permeation Asymmetric Tubular Membrane with Radial Oriented Porosity" Ceramics 2, no. 2: 246-259. https://doi.org/10.3390/ceramics2020020
APA StyleGaudillere, C., Garcia-Fayos, J., Plaza, J., & Serra, J. M. (2019). Ice-Templating for the Elaboration of Oxygen Permeation Asymmetric Tubular Membrane with Radial Oriented Porosity. Ceramics, 2(2), 246-259. https://doi.org/10.3390/ceramics2020020





