An Assessment of Soil Phytolith Analysis as a Palaeoecological Tool for Identifying Pre-Columbian Land Use in Amazonian Rainforests
Abstract
1. Introduction
2. Aims and Approach
- The ring-ditch region of northern Bolivia, for which we present new data.
- The geoglyph region of eastern Acre state, Brazil, where we examine previously published phytolith data [20].
3. Methods
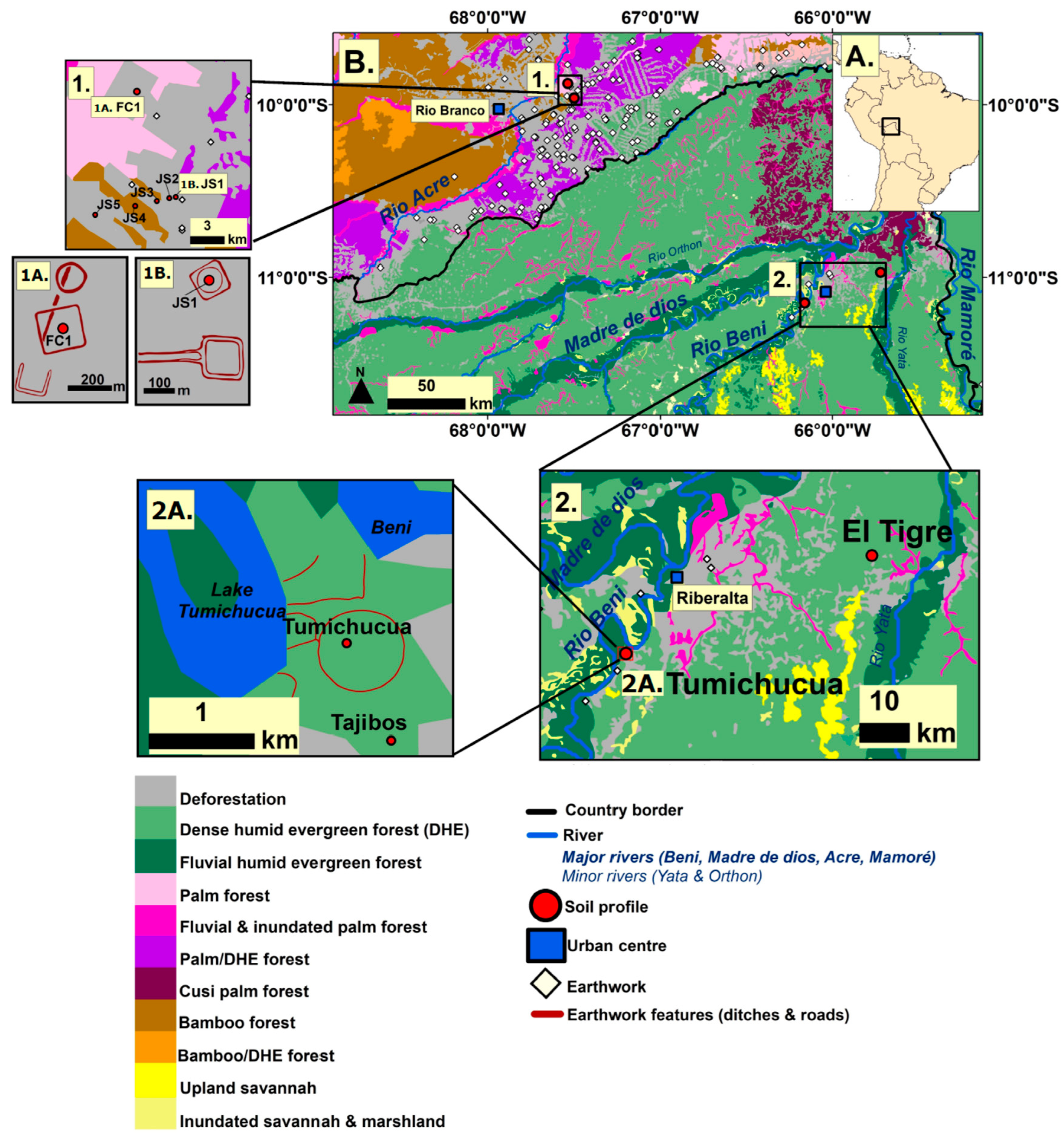
3.1. Study Areas
3.1.1. Ring Ditches of Northern Bolivia
3.1.2. Geoglyphs of Eastern Acre, Brazil
3.2. Site Selection Strategy and Sampling Design
3.3. Field and Laboratory Methods
4. Results
4.1. Study Area 1—Ring Ditches of Riberalta Region, Northern Bolivia (Figure 2, Figure 3 and Figure 4)
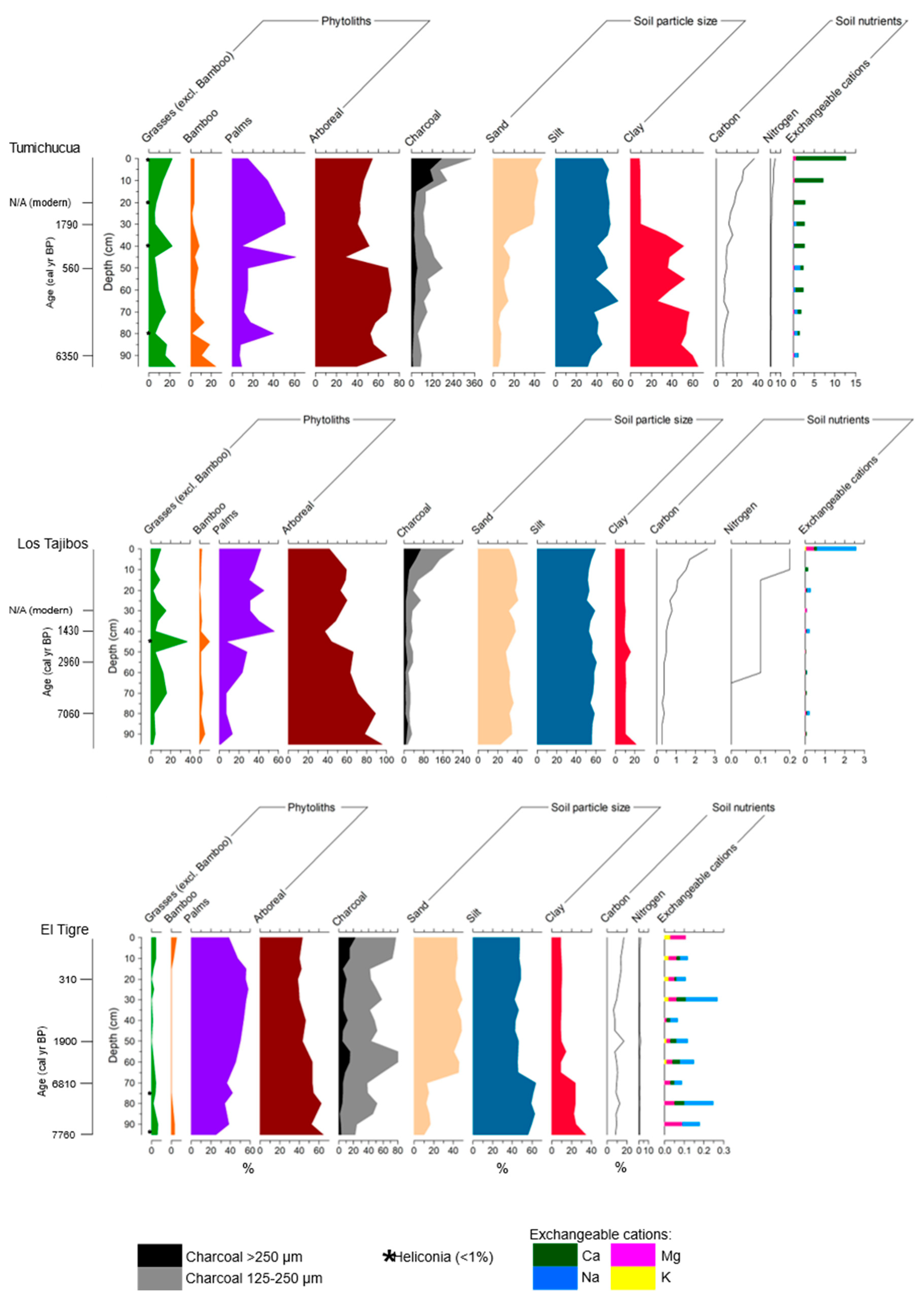

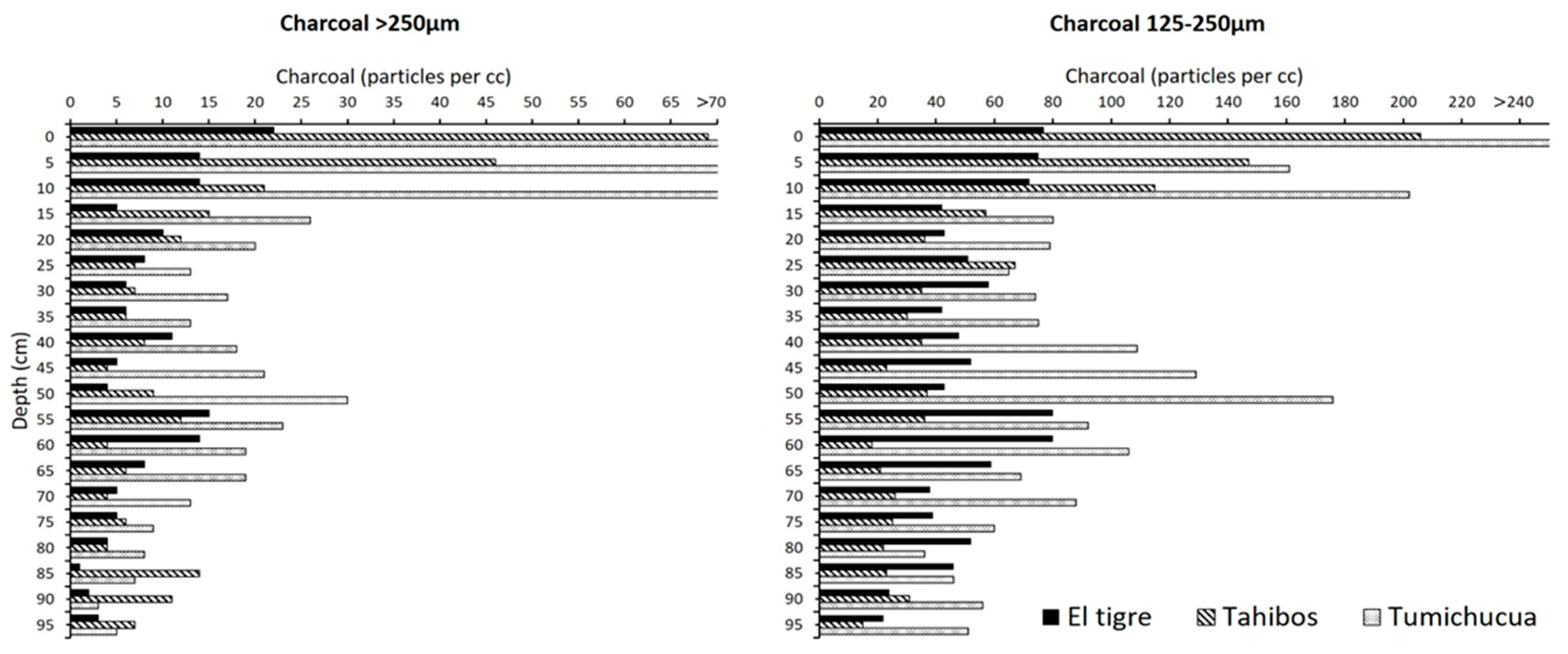
4.1.1. Tumichucua Ring-Ditch
4.1.2. Los Tajibos (1 km from Ring Ditch)
4.1.3. El Tigre (50 km from Tumichucua)
4.2. Study Area 2—Geoglyphs of Eastern Acre, Brazil (Figure 5)

5. Discussion
5.1. Scale of Deforestation
5.1.1. Riberalta Study Area
5.1.2. Acre Study Area
5.2. Forest Management—Palm Enrichment
5.3. Crop Cultivation and Burning
5.4. Soil Properties, Temporal Resolution, and Phytolith Preservation and Translocation
5.5. Recommendations for Future Work
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gross, D.R. Protein Capture and Cultural Development in the Amazon Basin. Am. Anthr. 1975, 77, 526–549. [Google Scholar] [CrossRef]
- Meggers, B.J. Environmental Limitation on the Development of Culture. Am. Anthr. 1954, 56, 801–824. [Google Scholar] [CrossRef]
- Denevan, W.M. Cultivated Landscapes of Native Amazonia and the Andes; Oxford University Press: New York, NY, USA, 2001. [Google Scholar]
- Erickson, C.L. The Transformation of Environment into Landscape: The Historical Ecology of Monumental Earthwork Construction in the Bolivian Amazon. Diversity 2010, 2, 618–652. [Google Scholar] [CrossRef]
- Erickson, C.L.; Balée, W. (Eds.) The historical ecology of a complex landscape in Bolivia. In Time and Complexity in Historical Ecology: Studies in the Neotropical Lowlands; Columbia University Press: New York, NY, USA, 2006; pp. 187–233. [Google Scholar]
- Mayle, F.E.; Iriarte, J. Integrated palaeoecology and archaeology—A powerful approach for understanding pre-Columbian Amazonia. J. Archaeol. Sci. 2014, 51, 54–64. [Google Scholar] [CrossRef]
- Iriarte, J.; Elliott, S.; Maezumi, S.Y.; Alves, D.; Gonda, R.; Robinson, M.; de Souza, J.G.; Watling, J.; Handley, J. The origins of Amazonian landscapes: Plant cultivation, domestication and the spread of food production in tropical South America. Quat. Sci. Rev. 2020, 248, 106582. [Google Scholar] [CrossRef]
- Maezumi, S.Y.; Alves, D.; Robinson, M.; de Souza, J.G.; Levis, C.; Barnett, R.L.; de Oliveira, E.A.; Urrego, D.; Schaan, D.; Iriarte, J. The legacy of 4,500 years of polyculture agroforestry in the eastern Amazon. Nat. Plants 2018, 4, 540–547. [Google Scholar] [CrossRef]
- Iriarte, J.; Power, M.J.; Rostain, S.; Mayle, F.E.; Jones, H.; Watling, J.; Whitney, B.S.; McKey, D.B. Fire-free land use in pre-1492 Amazonian savannas. Proc. Natl. Acad. Sci. USA 2012, 109, 6473–6478. [Google Scholar] [CrossRef]
- Whitney, B.S.; Dickau, R.; E Mayle, F.; Walker, J.H.; Soto, J.D.; Iriarte, J. Pre-Columbian raised-field agriculture and land use in the Bolivian Amazon. Holocene 2014, 24, 231–241. [Google Scholar] [CrossRef]
- Carson, J.F.; Whitney, B.S.; Mayle, F.E.; Iriarte, J.; Prümers, H.; Soto, J.D.; Watling, J. Environmental impact of geometric earthwork construction in pre-Columbian Amazonia. Proc. Natl. Acad. Sci. USA 2014, 111, 10497–10502. [Google Scholar] [CrossRef]
- Whitney, B.S.; Dickau, R.; Mayle, F.E.; Soto, J.D.; Iriarte, J. Pre-Columbian landscape impact and agriculture in the Monumental Mound region of the Llanos de Moxos, lowland Bolivia. Quat. Res. 2013, 80, 207–217. [Google Scholar] [CrossRef]
- Bush, M.B.; Silman, M.R.; De Toledo, M.B.; Listopad, C.; Gosling, W.D.; Williams, C.; De Oliveira, P.E.; Krisel, C. Holocene fire and occupation in Amazonia: Records from two lake districts. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 209–218. [Google Scholar] [CrossRef]
- Bush, M.B.; Nascimento, M.N.; Åkesson, C.M.; Cárdenes-Sandí, G.M.; Maezumi, S.Y.; Behling, H.; Correa-Metrio, A.; Church, W.; Huisman, S.N.; Kelly, T.; et al. Widespread reforestation before European influence on Amazonia. Science 2021, 372, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Burn, M.J.; Mayle, F.E.; Killeen, T.J. Pollen-based differentiation of Amazonian rainforest communities and implications for lowland palaeoecology in tropical South America. Palaeogeogr. Palaeoclim. Palaeoecol. 2010, 295, 1–18. [Google Scholar] [CrossRef]
- Gosling, W.D.; Mayle, F.E.; Tate, N.J.; Killeen, T.J. Differentiation between Neotropical rainforest, dry forest, and savanna ecosystems by their modern pollen spectra and implications for the fossil pollen record. Rev. Palaeobot. Palynol. 2009, 153, 70–85. [Google Scholar] [CrossRef]
- Jacobson, G.L.; Bradshaw, R. The Selection of Sites for Paleovegetational Studies. Quat. Res. 1981, 16, 80–96. [Google Scholar] [CrossRef]
- Lane, C.S.; Cummings, K.E.; Clark, J.J. Maize pollen deposition in modern lake sediments: A case study from Northeastern Wisconsin. Rev. Palaeobot. Palynol. 2010, 159, 177–187. [Google Scholar] [CrossRef]
- Pärssinen, M.; Schaan, D.; Ranzi, A. Pre-Columbian geometric earthworks in the upper Purús: A complex society in western Amazonia. Antiquity 2009, 83, 1084–1095. [Google Scholar] [CrossRef]
- Watling, J.; Iriarte, J.; Mayle, F.E.; Schaan, D.; Pessenda, L.C.R.; Loader, N.J.; Street-Perrott, F.A.; Dickau, R.E.; Damasceno, A.; Ranzi, A. Impact of pre-Columbian “geoglyph” builders on Amazonian forests. Proc. Natl. Acad. Sci. USA 2017, 114, 1868–1873. [Google Scholar] [CrossRef]
- Lathrap, D.W. Aboriginal Occupation and Changes in River Channel on the Central Ucayali, Peru. Am. Antiq. 1968, 33, 62–79. [Google Scholar] [CrossRef]
- Lathrap, D.W. The Upper Amazon; Thames and Hudson: Southampton, UK, 1970. [Google Scholar]
- Meggers, B.J. Cultural evolution in Amazonia. In Profiles in Cultural Evolution; Rambo, A.T., Gillogly, K., Eds.; Anthropological Papers, 85; Museum of Anthropology, University of Michigan: Ann Arbor, MI, USA, 1991; pp. 191–216. [Google Scholar]
- Pearsall, D.M. Paleoethnobotany: A Handbook of Procedures; Academic Press: San Diego, CA, USA, 1989. [Google Scholar]
- Hilbert, L.; Neves, E.G.; Pugliese, F.; Whitney, B.S.; Shock, M.; Veasey, E.; Zimpel, C.A.; Iriarte, J. Evidence for mid-Holocene rice domestication in the Americas. Nat. Ecol. Evol. 2017, 1, 1693–1698. [Google Scholar] [CrossRef]
- Watling, J.; Shock, M.P.; Mongeló, G.Z.; Almeida, F.O.; Kater, T.; De Oliveira, P.E.; Neves, E.G. Direct archaeological evidence for Southwestern Amazonia as an early plant domestication and food production centre. PLoS ONE 2018, 13, e0199868. [Google Scholar] [CrossRef]
- McMichael, C.H.; Piperno, D.R.; Bush, M.B.; Silman, M.R.; Zimmerman, A.R.; Raczka, M.F.; Lobato, L.C. Sparse Pre-Columbian Human Habitation in Western Amazonia. Science 2012, 336, 1429–1431. [Google Scholar] [CrossRef] [PubMed]
- McMichael, C.H.; Bush, M.; Piperno, D.R.; Silman, M.R.; Zimmerman, A.; Anderson, C. Spatial and temporal scales of pre-Columbian disturbance associated with western Amazonian lakes. Holocene 2011, 22, 131–141. [Google Scholar] [CrossRef]
- McMichael, C.H.; Piperno, D.R.; Neves, E.G.; Bush, M.B.; Almeida, F.O.; Mongeló, G.; Eyjolfsdottir, M.B. Phytolith Assemblages Along a Gradient of Ancient Human Disturbance in Western Amazonia. Front. Ecol. Evol. 2015, 3, 141. [Google Scholar] [CrossRef]
- Alexandre, A.; Meunier, J.-D.; Colin, F.; Koud, J.-M. Plant impact on the biogeochemical cycle of silicon and related weathering processes. Geochim. Cosmochim. Acta 1997, 61, 677–682. [Google Scholar] [CrossRef]
- Cabanes, D.; Weiner, S.; Shahack-Gross, R. Stability of phytoliths in the archaeological record: A dissolution study of modern and fossil phytoliths. J. Archaeol. Sci. 2011, 38, 2480–2490. [Google Scholar] [CrossRef]
- Bartoli, F.; Wilding, L.P. Dissolution of Biogenic Opal as a Function of its Physical and Chemical Properties. Soil Sci. Soc. Am. J. 1980, 44, 873–878. [Google Scholar] [CrossRef]
- Osterrieth, M.; Madella, M.; Zurro, D.; Alvarez, M.F. Taphonomical aspects of silica phytoliths in the loess sediments of the Argentinean Pampas. Quat. Int. 2007, 193, 70–79. [Google Scholar] [CrossRef]
- Fishkis, O.; Ingwersen, J.; Lamers, M.; Denysenko, D.; Streck, T. Phytolith transport in soil: A field study using fluorescent labelling. Geoderma 2010, 157, 27–36. [Google Scholar] [CrossRef]
- Kaczorek, D.; Puppe, D.; Busse, J.; Sommer, M. Effects of phytolith distribution and characteristics on extractable silicon fractions in soils under different vegetation—An exploratory study on loess. Geoderma 2019, 356, 113917. [Google Scholar] [CrossRef]
- Hart, D.M. The influence of soil fauna on phytolith distribution in a soil. In Phytoliths and Starch Research in the Australian-Pacific-Asian Regions: The State of the Art, Terra Australis 19; Hart, D.M., Wallis, L.A., Eds.; Pandanus Books: Canberra, Australia, 2003; pp. 83–91. [Google Scholar]
- Madella, M.; Lancelotti, C. Taphonomy and phytoliths: A user manual. Quat. Int. 2012, 275, 76–83. [Google Scholar] [CrossRef]
- Piperno, D.R. Phytoliths: A Comprehensive Guide for Archaeologists and Paleoecologists; AltaMira Press: Oxford, UK, 2006. [Google Scholar]
- Piperno, D.R. Phytolith Analysis: An Archaeological and Geological Perspective; Academic Press: San Diego, CA, USA, 1984. [Google Scholar]
- Piperno, D.R.; Pearsall, D.M. The Origins of Agriculture in the Lowland Neotropics; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Fredlund, G.G.; Tieszen, L.T. Modern Phytolith Assemblages from the North American Great Plains. J. Biogeogr. 1994, 21, 321. [Google Scholar] [CrossRef]
- Iriarte, J. Assessing the feasibility of identifying maize through the analysis of cross-shaped size and three-dimensional morphology of phytoliths in the grasslands of southeastern South America. J. Archaeol. Sci. 2003, 30, 1085–1094. [Google Scholar] [CrossRef]
- Ollendorf, A.L. Toward a classification scheme of sedge (Cyperaceae) phytoliths. In Phytolith Systematics: Emerging Issues; Rapp, G., Mulholand, S.C., Eds.; Plenum Press: New York, NY, USA, 1992; pp. 91–111. [Google Scholar]
- Bozarth, S.R. Diagnostic Opal Phytoliths from Rinds of Selected Cucurbita Species. Am. Antiq. 1987, 52, 607–615. [Google Scholar] [CrossRef]
- Piperno, D.R.; Ranere, A.J.; Holst, I.; Iriarte, J.; Dickau, R. Starch grain and phytolith evidence for early ninth millennium B.P. maize from the Central Balsas River Valley, Mexico. Proc. Natl. Acad. Sci. USA 2009, 106, 5019–5024. [Google Scholar] [CrossRef] [PubMed]
- Bozarth, S.R.; Price, K.; Woods, W.I.; Neves, E.G.; Rebellato, R. Phytoliths and terra preta: The Hatahara site example. In Amazonian Dark Earths: Wim Sombroek’s Vision; Woods, W.I., Teixeira, W.G., Lehmann, J., Steiner, C., Winkler Prins, A., Rebellato, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 85–98. [Google Scholar]
- Lombardo, U.; Iriarte, J.; Hilbert, L.; Ruiz-Pérez, J.; Capriles, J.M.; Veit, H. Early Holocene crop cultivation and landscape modification in Amazonia. Nature 2020, 581, 190–193. [Google Scholar] [CrossRef]
- Godwin, H. Pollen Analysis. An outline of the problems and potentialities of the method. New Phytol. 1934, 33, 278–304. [Google Scholar] [CrossRef]
- Larson, B.M.H.; Barrett, S.C.H. A comparative analysis of pollen limitation in flowering plants. Biol. J. Linn. Soc. 2000, 69, 503–520. [Google Scholar] [CrossRef]
- Macdonald, G.M. Methodological falsification and the interpretation of palaeoecological records: The cause of the early Holocene birch decline in western Canada. Rev. Palaeobot. Palynol. 1993, 79, 83–97. [Google Scholar] [CrossRef]
- Seppä, H.; Bennett, K. Quaternary pollen analysis: Recent progress in palaeoecology and palaeoclimatology. Prog. Phys. Geogr. Earth Environ. 2003, 27, 548–579. [Google Scholar] [CrossRef]
- Beekma, J.; Zonta, A.; Keijzer, B. Base Ambiental para el Dessarrollo; Departamento de Pando y la Provincia Vaca Diez, Servicio Holandes de Cooperación al Desarrollo SNV: Pichincha, Ecuador, 1996; p. 91. [Google Scholar]
- Myers, G.P.; Newton, A.C.; Melgarejo, O. The influence of canopy gap size on natural regeneration of Brazil nut (Bertholletia excelsa) in Bolivia. For. Ecol. Manag. 2000, 127, 119–128. [Google Scholar] [CrossRef]
- Wasson, J.G.; Barrera, S.; Barrère, B.; Binet, D.; Collomb, D.; Gonzales, I.; Gourdin, F.; Guyot, J.L.; Rocabado, G. Hydro-ecoregions of the Bolivian Amazon: A geographical framework for the functioning of river ecosystems. In The Ecohydrology of South American Rivers and Wetlands; McClain, M.E., Ed.; International Association of Hydrological Sciences: Wallingford, UK, 2002; pp. 69–91. [Google Scholar]
- Ibisch, P.L.; Beck, S.G.; Gerkmann, B.; Carretero, A. Ecoregions and ecosystems. In Biodiversity: The Richness of Bolivia; Ibisch, P.L., Mérida, G., Eds.; Editorial FAN: Santa Cruz, CA, USA, 2004; pp. 47–88. [Google Scholar]
- Quesada, C.A.; Lloyd, J.; Anderson, L.O.; Fyllas, N.M.; Schwarz, M.; Czimczik, C.I. Soils of Amazonia with particular reference to the RAINFOR sites. Biogeosciences 2011, 8, 1415–1440. [Google Scholar] [CrossRef]
- Saunaluoma, S. Pre-Columbian Earthworks in the Riberalta Region of the Bolivian Amazon. Amaz.-Rev. Antropol. 2010, 2, 104–138. [Google Scholar] [CrossRef]
- Arnold, D.E.; Pendery, S.R.; Prettol, K.A.; Webster, G.S.; Greenfield, H.J. News and Short Contributions. J. Field Archaeol. 1988, 15, 457. [Google Scholar] [CrossRef]
- Watling, J.; Saunaluoma, S.; Pärssinen, M.; Schaan, D. Subsistence practices among earthwork builders: Phytolith evidence from archaeological sites in the southwest Amazonian interfluves. J. Archaeol. Sci. Rep. 2015, 4, 541–551. [Google Scholar] [CrossRef]
- Prümers, H.; Betancourt, C.J. 100 años de investigación arqueológica en los Llanos de Mojos. Arqueoantropol. Año 2014, 4, 11–54. [Google Scholar]
- de Souza, J.G.; Schaan, D.P.; Robinson, M.; Barbosa, A.D.; Aragão, L.E.; Marimon, B.H., Jr.; Marimon, B.S.; da Silva, I.B.; Khan, S.S.; Nakahara, F.R.; et al. Pre-Columbian earth-builders settled along the entire southern rim of the Amazon. Nat. Commun. 2018, 9, 1125. [Google Scholar] [CrossRef]
- Schaan, D.P. Sacred Geographies of Ancient Amazonia: Historical Ecology of Social Complexity; Left Coast Press: Walnut Creek, CA, USA, 2012. [Google Scholar]
- Saunaluoma, S.; Schaan, D. Monumentality in Western Amazonian formative societies: Geometric ditched enclosures in the Brazilian state of Acre. Antiqua 2012, 2, e1. [Google Scholar] [CrossRef]
- McMichael, C.H.; Palace, M.W.; Golightly, M. Bamboo-dominated forests and pre-Columbian earthwork formations in south-western Amazonia. J. Biogeogr. 2014, 41, 1733–1745. [Google Scholar] [CrossRef]
- Lopez-Gonzalez, G.; Lewis, S.L.; Burkitt, M.; Phillips, O. ForestPlots.net Database. 2009. Available online: www.forestplots.net (accessed on 12 September 2017).
- ForestPlots.net.; Blundo, C.; Carilla, J.; Grau, R.; Malizia, A.; Malizia, L.; Osinaga-Acosta, O.; Bird, M.; Bradford, M.; Catchpole, D.; et al. Taking the pulse of Earth’s tropical forests using networks of highly distributed plots. Biol. Conserv. 2021, 260, 108849. [Google Scholar] [CrossRef]
- Dickau, R.; Whitney, B.S.; Iriarte, J.; Mayle, F.E.; Soto, J.D.; Metcalfe, P.; Street-Perrott, F.A.; Loader, N.J.; Ficken, K.J.; Killeen, T.J. Differentiation of neotropical ecosystems by modern soil phytolith assemblages and its implications for palaeoenvironmental and archaeological reconstructions. Rev. Palaeobot. Palynol. 2013, 193, 15–37. [Google Scholar] [CrossRef]
- Iriarte, J.; Paz, E.A. Phytolith analysis of selected native plants and modern soils from southeastern Uruguay and its implications for paleoenvironmental and archeological reconstruction. Quat. Int. 2007, 193, 99–123. [Google Scholar] [CrossRef]
- Morcote-Ríos, G.; Bernal, R.; Raz, L. Phytoliths as a tool for archaeobotanical, palaeobotanical and palaeoecological studies in Amazonian palms. Bot. J. Linn. Soc. 2016, 182, 348–360. [Google Scholar] [CrossRef]
- Watling, J.; Iriarte, J.; Whitney, B.; Consuelo, E.; Mayle, F.; Castro, W.; Schaan, D.; Feldpausch, T. Differentiation of neotropical ecosystems by modern soil phytolith assemblages and its implications for palaeoenvironmental and archaeological reconstructions II: Southwestern Amazonian forests. Rev. Palaeobot. Palynol. 2016, 226, 30–43. [Google Scholar] [CrossRef]
- Clark, J.S. Particle Motion and the Theory of Charcoal Analysis: Source Area, Transport, Deposition, and Sampling. Quat. Res. 1988, 30, 67–80. [Google Scholar] [CrossRef]
- Whitlock, C.; Larsen, C. Charcoal as a fire proxy. In Tracking Environmental Change Using Lake Sediments, Volume 3: Terrestrial, Algal and Siliceous Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 75–98. [Google Scholar]
- McCormac, F.G.; Hogg, A.G.; Blackwell, P.G.; E Buck, C.; Higham, T.F.G.; Reimer, P.J. Shcal04 Southern Hemisphere Calibration, 0–11.0 Cal Kyr BP. Radiocarbon 2004, 46, 1087–1092. [Google Scholar] [CrossRef]
- Reimer, P.J.; Bard, E.; Bayliss, A.; Beck, J.W.; Blackwell, P.G.; Ramsey, C.B.; Buck, C.E.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. IntCal13 and Marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 2013, 55, 1869–1887. [Google Scholar] [CrossRef]
- Fraysse, F.; Pokrovsky, O.S.; Schott, J.; Meunier, J.-D. Surface properties, solubility and dissolution kinetics of bamboo phytoliths. Geochim. Cosmochim. Acta 2006, 70, 1939–1951. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Q.; Li, X.; Jia, X.; Wei, X.; Shao, M. Determination of Soil Texture by Laser Diffraction Method. Soil Sci. Soc. Am. J. 2015, 79, 1556–1566. [Google Scholar] [CrossRef]
- Campbell, J.R. Limitations in the laser particle sizing the soils. In Advances in Regolith; Roach, I.C., Ed.; Cooperative Research Centre for Landscape Environments and Mineral Exploration (CRC LEME): Canberra, Australia, 2003; pp. 38–42. [Google Scholar]
- Rodwell, D.L. Soil Science: Methods and Applications; Longman Group UK Ltd.: London, UK, 1994; pp. 139–141. [Google Scholar]
- Gouveia, S.; Pessenda, L.; Aravena, R.; Boulet, R.; Scheel-Ybert, R.; Bendassoli, J.; Ribeiro, A.; Freitas, H. Carbon isotopes in charcoal and soils in studies of paleovegetation and climate changes during the late Pleistocene and the Holocene in the southeast and centerwest regions of Brazil. Glob. Planet. Chang. 2002, 33, 95–106. [Google Scholar] [CrossRef]
- Pessenda, L.C.; Gomes, B.M.; Aravena, R.; Ribeiro, A.S.; Boulet, R.; Gouveia, S.E. The carbon isotope record in soils along a forest-cerrado ecosystem transect: Implications for vegetation changes in the Rondonia state, southwestern Brazilian Amazon region. Holocene 1998, 8, 599–603. [Google Scholar] [CrossRef]
- Pessenda, L.C.R.; Ribeiro, A.D.S.; Gouveia, S.E.M.; Aravena, R.; Boulet, R.; Bendassolli, J.A. Vegetation dynamics during the late Pleistocene in the Barreirinhas region, Maranhão State, northeastern Brazil, based on carbon Isotopes in soil organic matter. Quat. Res. 2004, 62, 183–193. [Google Scholar] [CrossRef]
- Pessenda, L.C.; Ledru, M.-P.; Gouveia, S.E.; Aravena, R.; Ribeiro, A.S.; Bendashsollil, J.A.; Boulet, R. Holocene palaeoenvironmental reconstruction in northeastern Brazil inferred from pollen, charcoal and carbon isotope records. Holocene 2004, 15, 812–820. [Google Scholar] [CrossRef]
- Watling, J.; Castro, M.T.; Simon, M.F.; Rodrigues, F.O.; de Medeiros, M.B.; De Oliveira, P.E.; Neves, E.G. Phytoliths from native plants and surface soils from the Upper Madeira river, SW Amazonia, and their potential for paleoecological reconstruction. Quat. Int. 2020, 550, 85–110. [Google Scholar] [CrossRef]
- Parr, J.F. Effect of fire on phytolith coloration. Geoarchaeology 2006, 21, 171–185. [Google Scholar] [CrossRef]
- Toledo, M.; Poorter, L.; Peña-Claros, M.; Alarcón, A.; Balcázar, J.; Chuviña, J.; Leaño, C.; Licona, J.C.; Ter Steege, H.; Bongers, F. Patterns and Determinants of Floristic Variation across Lowland Forests of Bolivia. Biotropica 2010, 43, 405–413. [Google Scholar] [CrossRef]
- Alves, D.T. Dark Earth Plant Management in the Lower Tapajos. Ph.D. Thesis, University of Exeter, Exeter, UK, 2017. [Google Scholar]
- Blecker, S.W.; McCulley, R.L.; Chadwick, O.A.; Kelly, E.F. Biologic cycling of silica across a grassland bioclimosequence. Glob. Biogeochem. Cycles 2006, 20, GB3023. [Google Scholar] [CrossRef]
- Fishkis, O.; Ingwersen, J.; Streck, T. Phytolith transport in sandy sediment: Experiments and modeling. Geoderma 2009, 151, 168–178. [Google Scholar] [CrossRef]
- Piperno, D.R.; McMichael, C.; Bush, M.B. Further evidence for localized, short-term anthropogenic forest alterations across pre-Columbian Amazonia. Proc. Natl. Acad. Sci. USA 2017, 114, E4118–E4119. [Google Scholar] [CrossRef]
- Watling, J.; Iriarte, J.; Mayle, F.E.; Schaan, D.; Pessenda, L.C.R.; Loader, N.J.; Street-Perrott, F.A.; Dickau, R.E.; Damasceno, A.; Ranzi, A. Reply to Piperno et al.: It is too soon to argue for localized, short-term human impacts in interfluvial Amazonia. Proc. Natl. Acad. Sci. USA 2017, 114, E4120–E4121. [Google Scholar] [CrossRef]
- Saunaluoma, S. Geometric Earthworks in the State of Acre, Brazil: Excavations at the Fazenda Atlântica and Quinauá Sites. Lat. Am. Antiq. 2012, 23, 565–583. [Google Scholar] [CrossRef]
- Saunaluoma, S.; Pärssinen, M.; Schaan, D. Diversity of Pre-colonial Earthworks in the Brazilian State of Acre, Southwestern Amazonia. J. Field Archaeol. 2018, 43, 362–379. [Google Scholar] [CrossRef]
- Balée, W. Contingent Diversity on Anthropic Landscapes. Diversity 2010, 2, 163–181. [Google Scholar] [CrossRef]
- Levis, C.; Costa, F.R.C.; Bongers, F.; Peña-Claros, M.; Clement, C.R.; Junqueira, A.B.; Neves, E.G.; Tamanaha, E.K.; Figueiredo, F.O.G.; Salomão, R.P.; et al. Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 2017, 355, 925–931. [Google Scholar] [CrossRef]
- Clement, C.R.; Denevan, W.M.; Heckenberger, M.J.; Junqueira, A.B.; Neves, E.G.; Teixeira, W.G.; Woods, W.I. The domestication of Amazonia before European conquest. Proc. R. Soc. B Boil. Sci. 2015, 282, 20150813. [Google Scholar] [CrossRef]
- Levis, C.; Flores, B.M.; Moreira, P.A.; Luize, B.G.; Alves, R.P.; Franco-Moraes, J.; Lins, J.; Konings, E.; Peña-Claros, M.; Bongers, F.; et al. How People Domesticated Amazonian Forests. Front. Ecol. Evol. 2018, 5, 171. [Google Scholar] [CrossRef]
- Levis, C.; de Souza, P.F.; Schietti, J.; Emilio, T.; Pinto, J.L.P.D.V.; Clement, C.R.; Costa, F.R.C. Historical Human Footprint on Modern Tree Species Composition in the Purus-Madeira Interfluve, Central Amazonia. PLoS ONE 2012, 7, e48559. [Google Scholar] [CrossRef]
- de Oliveira, E.A.; Marimon-Junior, B.H.; Marimon, B.S.; Iriarte, J.; Morandi, P.S.; Maezumi, S.Y.; Nogueira, D.S.; Aragao, L.E.; da Silva, I.B.; Feldpausch, T.R. Legacy of Amazonian Dark Earth soils on forest structure and species composition. Glob. Ecol. Biogeogr. 2020, 29, 1458–1473. [Google Scholar] [CrossRef]
- Gehring, C.; Zelarayán, M.C.; Luz, R.L.; Almeida, R.B.; Boddey, R.M.; Leite, M.F.A. Babassu palm (Attalea speciosa Mart.) super-dominance shapes its surroundings via multiple biotic, soil chemical, and physical interactions and accumulates soil carbon: A case study in eastern Amazonia. Plant Soil 2020, 454, 447–460. [Google Scholar] [CrossRef]
- Rocha, G.P.; Vieira, D.L.; Simon, M.F. Fast natural regeneration in abandoned pastures in southern Amazonia. For. Ecol. Manag. 2016, 370, 93–101. [Google Scholar] [CrossRef]
- Aleman, J.; Leys, B.; Apema, R.; Bentaleb, I.; Dubois, M.A.; Lamba, B.; Lebamba, J.; Martin, C.; Ngomanda, A.; Truc, L.; et al. Reconstructing savanna tree cover from pollen, phytoliths and stable carbon isotopes. J. Veg. Sci. 2011, 23, 187–197. [Google Scholar] [CrossRef]
- Maezumi, S.Y.; Robinson, M.; De Souza, J.; Urrego, D.H.; Schaan, D.; Alves, D.; Iriarte, J. New Insights from Pre-Columbian Land Use and Fire Management in Amazonian Dark Earth Forests. Front. Ecol. Evol. 2018, 6, 111. [Google Scholar] [CrossRef]
- Maezumi, S.Y.; Whitney, B.S.; Mayle, F.E.; de Souza, J.G.; Iriarte, J. Reassessing climate and pre-Columbian drivers of paleofire activity in the Bolivian Amazon. Quat. Int. 2018, 488, 81–94. [Google Scholar] [CrossRef]
- Denevan, W.M. Stone versus metal axes: The ambiguity of shifting cultivation in prehistoric Amazonia. J. Steward Anthrop. Soc. 1992, 20, 153–165. [Google Scholar]
- Glaser, B.; Birk, J.J. State of the scientific knowledge on properties and genesis of Anthropogenic Dark Earths in Central Amazonia (terra preta de Índio). Geochim. Cosmochim. Acta 2012, 82, 39–51. [Google Scholar] [CrossRef]
- Robinson, M.; Jaimes-Betancourt, C.; Elliott, S.; Maezumi, S.Y.; Hilbert, L.; Alves, D.; de Souza, J.G.; Iriarte, J. Anthropogenic soil and settlement organisation in the Bolivian Amazon. Geoarchaeology 2020, 36, 388–403. [Google Scholar] [CrossRef]
- Pessenda, L.C.R.; Gouveia, S.E.M.; Aravena, R. Radiocarbon Dating of Total Soil Organic Matter and Humin Fraction and Its Comparison with 14C Ages of Fossil Charcoal. Radiocarbon 2001, 43, 595–601. [Google Scholar] [CrossRef]
- Balesdent, J.; Basile-Doelsch, I.; Chadoeuf, J.; Cornu, S.; Derrien, D.; Fekiacova, Z.; Hatté, C. Atmosphere–soil carbon transfer as a function of soil depth. Nature 2018, 559, 599–602. [Google Scholar] [CrossRef]
- Song, Z.; McGrouther, K.; Wang, H. Occurrence, turnover and carbon sequestration potential of phytoliths in terrestrial ecosystems. Earth-Sci. Rev. 2016, 158, 19–30. [Google Scholar] [CrossRef]
- Strömberg, C.A.; Dunn, R.E.; Crifò, C.; Harris, E.B. Phytoliths in paleoecology: Analytical considerations, current use, and future directions. In Methods in Paleoecology: Reconstructing Cenozoic Terrestrial Environments and Ecological Communities, Vertebrate Paleobiology and Paleoanthropology; Croft, D.A., Su, D.F., Simpson, S.W., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 235–287. [Google Scholar]
- Grave, P.; Kealhofer, L. Assessing Bioturbation in Archaeological Sediments using Soil Morphology and Phytolith Analysis. J. Archaeol. Sci. 1999, 26, 1239–1248. [Google Scholar] [CrossRef]
- Kerns, B.K.; Moore, M.M.; Hart, S.C. Estimating forest-grassland dynamics using soil phytolith assemblages and d13C of soil organic matter. Écoscience 2001, 8, 478–488. [Google Scholar] [CrossRef]
- McClaran, M.P.; Umlauf, M. Desert grassland dynamics estimated from carbon isotopes in grass phytoliths and soil organic matter. J. Veg. Sci. 2000, 11, 71–76. [Google Scholar] [CrossRef]
- Morris, L.R.; West, N.E.; Ryel, R.J. Testing soil phytolith analysis as a tool to understand vegetation change in the sagebrush steppe and pinyon-juniper woodlands of the Great Basin Desert, USA. Holocene 2010, 20, 697–709. [Google Scholar] [CrossRef]
- Piperno, D.R.; Becker, P. Vegetational History of a Site in the Central Amazon Basin Derived from Phytolith and Charcoal Records from Natural Soils. Quat. Res. 1996, 45, 202–209. [Google Scholar] [CrossRef]
- Iler, R. The Chemistry of Silica. Solubility, Polymerization, Colloid and Surface Properties, and Biochemistry; John Wiley and Sons: Chichester, UK, 1979. [Google Scholar]
- Cabanes, D.; Shahack-Gross, R. Understanding Fossil Phytolith Preservation: The Role of Partial Dissolution in Paleoecology and Archaeology. PLoS ONE 2015, 10, e0125532. [Google Scholar] [CrossRef] [PubMed]
- Shoji, S.; Nanzyo, M.; Dahlgren, R. Volcanic Ash Soils: Genesis, Properties, and Utilization; Elsevier Science Publishers B.V.: Amsterdam, The Netherlands, 1994; Volume 21. [Google Scholar]
- Watling, J.; Almeida, F.; Kater, T.; Zuse, S.; Shock, M.P.; Mongeló, G.; Bespalez, E.; Santi, J.R.; Neves, E.G. Arqueobotânica de ocupações ceramistas na Cachoeira do Teotônio. Boletim do Museu Paraense Emílio Goeldi. Ciênc. Hum. 2020, 15, 2178–2547. [Google Scholar] [CrossRef]
- Piperno, D.R.; McMichael, C. Phytoliths in modern plants from amazonia and the neotropics at large: Implications for vegetation history reconstruction. Quat. Int. 2020, 565, 54–74. [Google Scholar] [CrossRef]

| Site | Publication Code | Sample Depth (cm) | Sample Type | Conventional Radiocarbon Age (a BP ± 2 σ) | Calibrated Age (cal. a BP ± 2 σ) | Average (cal. a BP ± 2 σ) |
|---|---|---|---|---|---|---|
| Tumichucua | UCIAMS-223844 | 20 | charcoal | n/a | n/a | n/a |
| SUERC-88899 | 30 | charcoal | 1846 ± 37 | 1876–1702 | 1790 | |
| SUERC-88906 | 50 | charcoal | 490 ± 37 | 623–493 | 560 | |
| SUERC-88907 | 90 | charcoal | 5546 ± 37 | 6403–6289 | 6350 | |
| Los Tajibos | SUERC-88908 | 30 | charcoal | n/a | n/a | n/a |
| SUERC-88909 | 40 | charcoal | 1513 ± 35 | 1523–1327 | 1430 | |
| SUERC-88910 | 55 | charcoal | 2847 ± 37 | 3068–2861 | 2960 | |
| SUERC-88911 | 80 | charcoal | 6160 ± 38 | 7165–6951 | 7060 | |
| El Tigre | SUERC-88887 | 20 | charcoal | 209 ± 37 | 311 | 310 |
| SUERC-88888 | 50 | charcoal | 1942 ± 37 | 1988–1820 | 1900 | |
| SUERC-88889 | 70 | charcoal | 5975 ± 38 | 6925–6693 | 6810 | |
| SUERC-88890 | 100 | charcoal | 6920 ± 39 | 7838–7675 | 7760 |
| Taxa Present in Both Phytolith Surface Sample and Inventory | % of Phytoliths in Surface Sample | % of Stems in Plot (dbh > 10 cm) | Taxa with Known/Potentially Diagnostic Phytoliths Present in Inventory but Not Present in the Phytolith Surface Sample (dbh > 10 cm) | % of Stems in Plot (dbh > 10 cm) |
|---|---|---|---|---|
| Arecaceae | 38 | 40 | Arecaceae: | 10 |
| 9 | |||
| 0.1 | |||
| 0.1 | |||
| Annonaceae (5 spp.) | 2 | |||
| Phenakospermum guyannense | 8 | |||
| Total: | 20 |
| Species | Economic spp. Stems% | Total Stems% |
|---|---|---|
| Pseudolmedia laevis (Moraceae) | 25 | 7 |
| Socratea exorrhiza (Arecaceae) | 3 | 1 |
| Micropholis guyanensis (Sapotaceae) | 1 | 0.3 |
| Helicostylis tomentosa (Moraceae) | 1 | 0.1 |
| Bertholletia excelsa (Lecythidaceae) | 1 | 0.1 |
| Pourouma minor (Urticaceae) | 1 | 0.1 |
| Brosimum lactescens (Moraceae) | 13 | 3 |
| Brosimum guianense (Moraceae) | 2 | 0.5 |
| Leonia glycycarpa (Violaceae) | 8 | 2 |
| Virola calophylla (Myristicaceae) | 2 | 0.5 |
| Conceveiba guianensis (Euphorbiaceae) | 1 | 0.2 |
| Aspidosperma excelsum (Apocynaceae) | 1 | 0.2 |
| Iryanthera laevis (Myristicaceae) | 3 | 1 |
| Laetia procera (Salicaceae) | 0.5 | 0.1 |
| Total: | 62.5 | 16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hill, J.; Black, S.; Araujo-Murakami, A.; Boot, R.; Brienen, R.; Feldpausch, T.; Leigue, J.; Murakami, S.; Monteagudo, A.; Pardo, G.; et al. An Assessment of Soil Phytolith Analysis as a Palaeoecological Tool for Identifying Pre-Columbian Land Use in Amazonian Rainforests. Quaternary 2023, 6, 33. https://doi.org/10.3390/quat6020033
Hill J, Black S, Araujo-Murakami A, Boot R, Brienen R, Feldpausch T, Leigue J, Murakami S, Monteagudo A, Pardo G, et al. An Assessment of Soil Phytolith Analysis as a Palaeoecological Tool for Identifying Pre-Columbian Land Use in Amazonian Rainforests. Quaternary. 2023; 6(2):33. https://doi.org/10.3390/quat6020033
Chicago/Turabian StyleHill, James, Stuart Black, Alejandro Araujo-Murakami, Rene Boot, Roel Brienen, Ted Feldpausch, John Leigue, Samaria Murakami, Abel Monteagudo, Guido Pardo, and et al. 2023. "An Assessment of Soil Phytolith Analysis as a Palaeoecological Tool for Identifying Pre-Columbian Land Use in Amazonian Rainforests" Quaternary 6, no. 2: 33. https://doi.org/10.3390/quat6020033
APA StyleHill, J., Black, S., Araujo-Murakami, A., Boot, R., Brienen, R., Feldpausch, T., Leigue, J., Murakami, S., Monteagudo, A., Pardo, G., Peña-Claros, M., Phillips, O. L., Toledo, M., Vos, V., Zuidema, P., & Mayle, F. E. (2023). An Assessment of Soil Phytolith Analysis as a Palaeoecological Tool for Identifying Pre-Columbian Land Use in Amazonian Rainforests. Quaternary, 6(2), 33. https://doi.org/10.3390/quat6020033






