Understanding the Impact of Trampling on Rodent Bones
Abstract
:1. Introduction
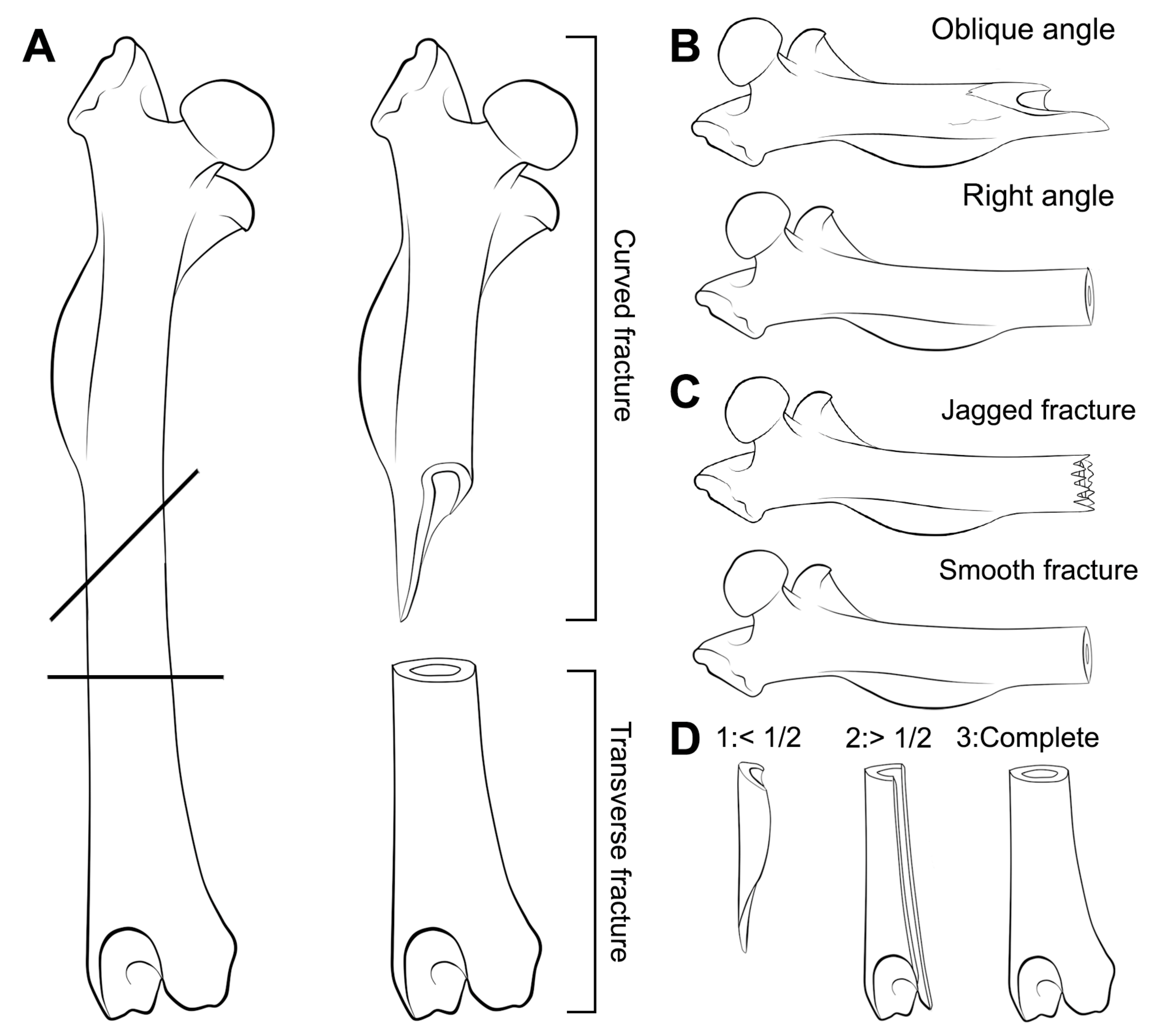
2. Material and Methods
Case Study of Wonderwerk Cave
3. Results
3.1. Compression of Pellets
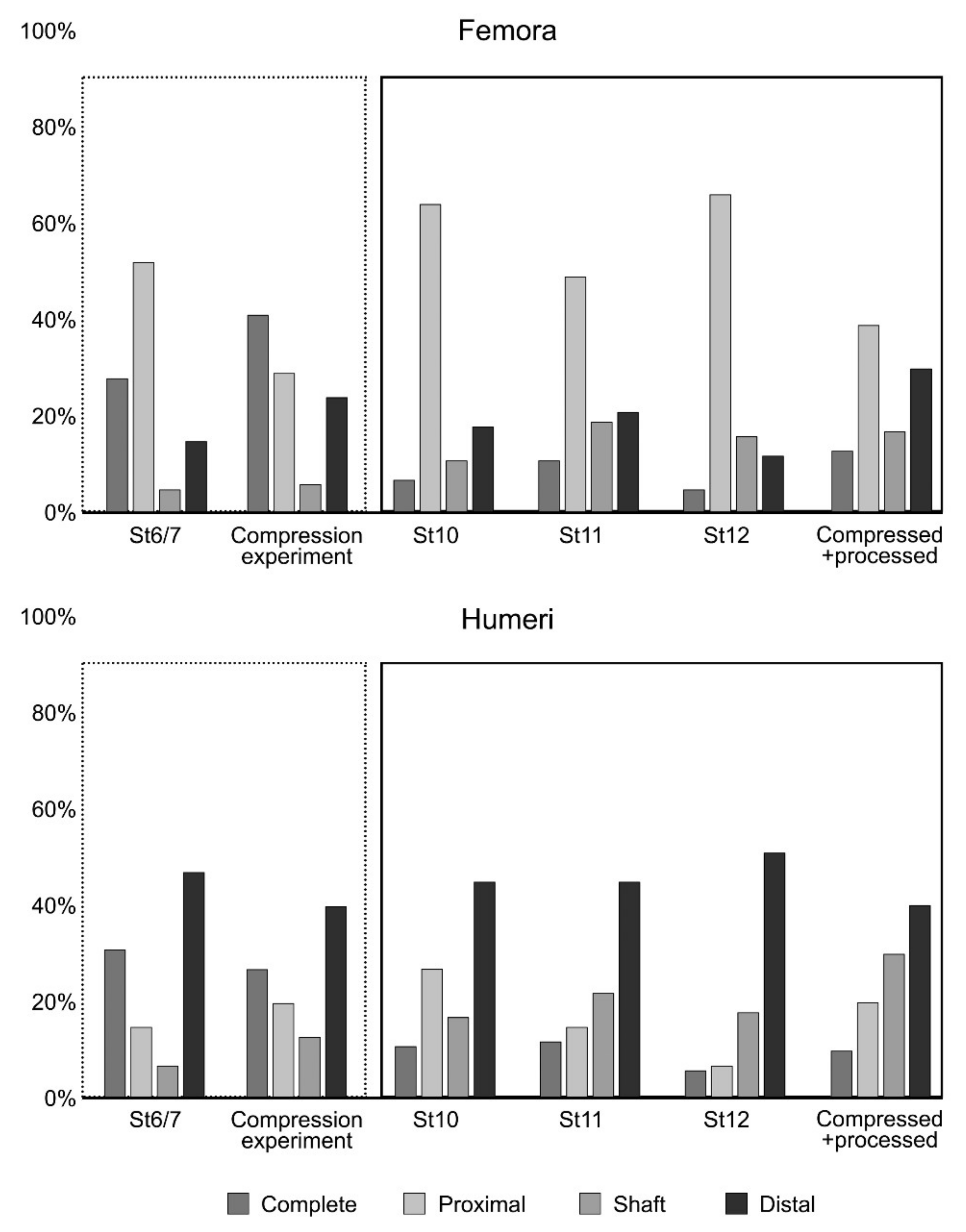
3.2. Compression of Skeletal Elements
4. Discussion
- (A)
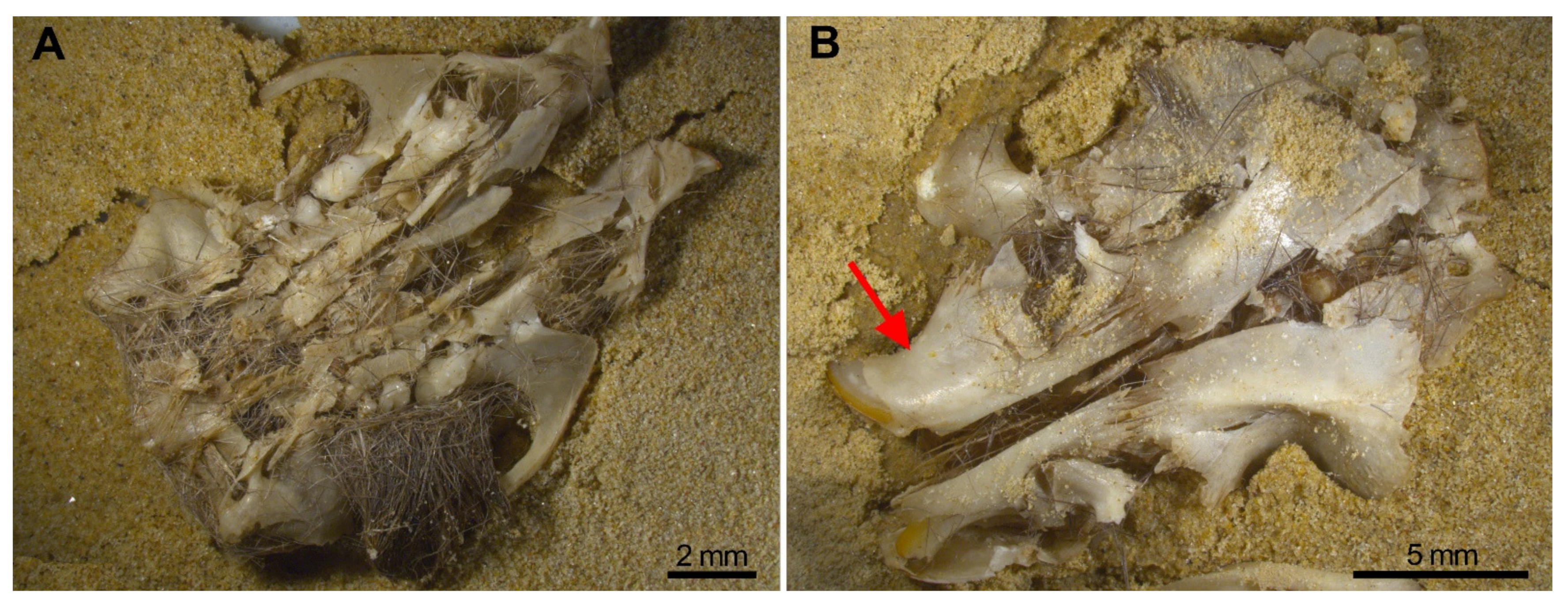
- Breakage of skulls, reduction in numbers of maxillae. In our experiment, we have proved the pneumatic behaviour of the skull which deformed leaving a mass of bones and teeth. The bones of the skull detached into small unidentifiable bone fragments, especially when subjected to water or gravitational movements such as sifting, leaving the zygomatic arches apart from the maxillae which frequently bear the M1 in situ. Incisors were also frequently found in the interior of the alveoli, and both traits have been observed in the Wonderwerk Cave fossil assemblage. Consequently, both observations can be added as taphonomic criteria of trampling in a fossil site.
- (B)
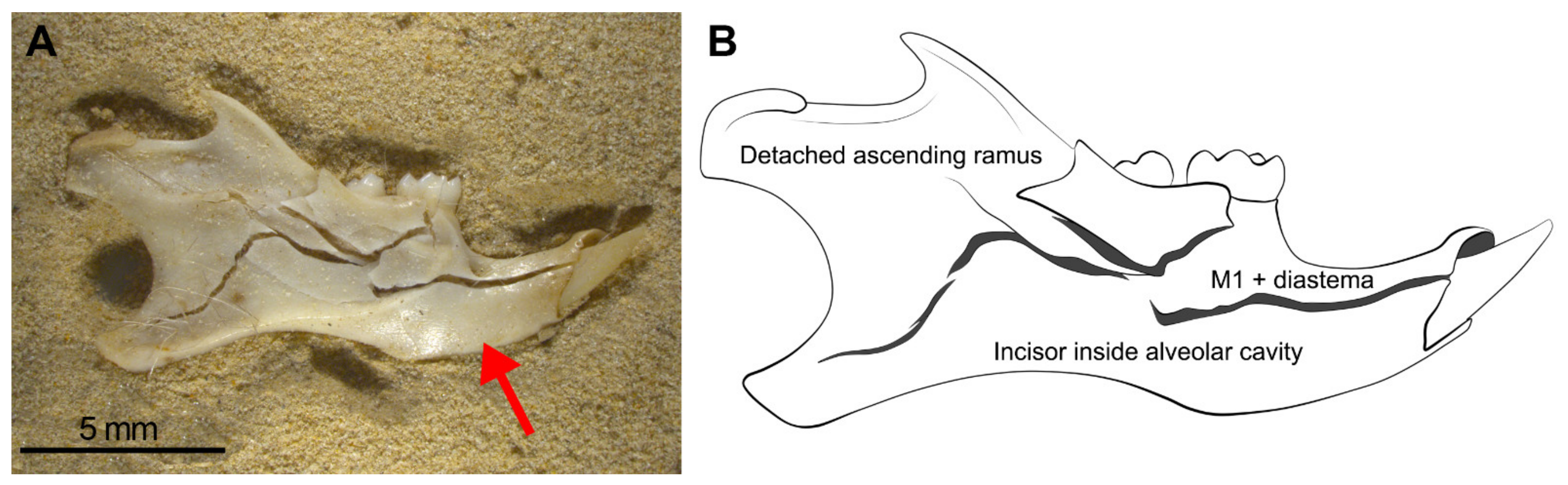
- Considerable loss of teeth from the jaws leading to large numbers of isolated teeth. As seen in Wonderwerk Cave and in the experiment, both mandibles and maxillae showed detachment of molars although in mandibles the M1 remained in situ retained in the diastema portion. The incisors, as observed in maxillae, are also retained in situ (in the alveolar socket of the mandible) and in this type of assemblage it is also common to see the ascending ramus detached from the dental row and the rest of the mandible.
- (C)
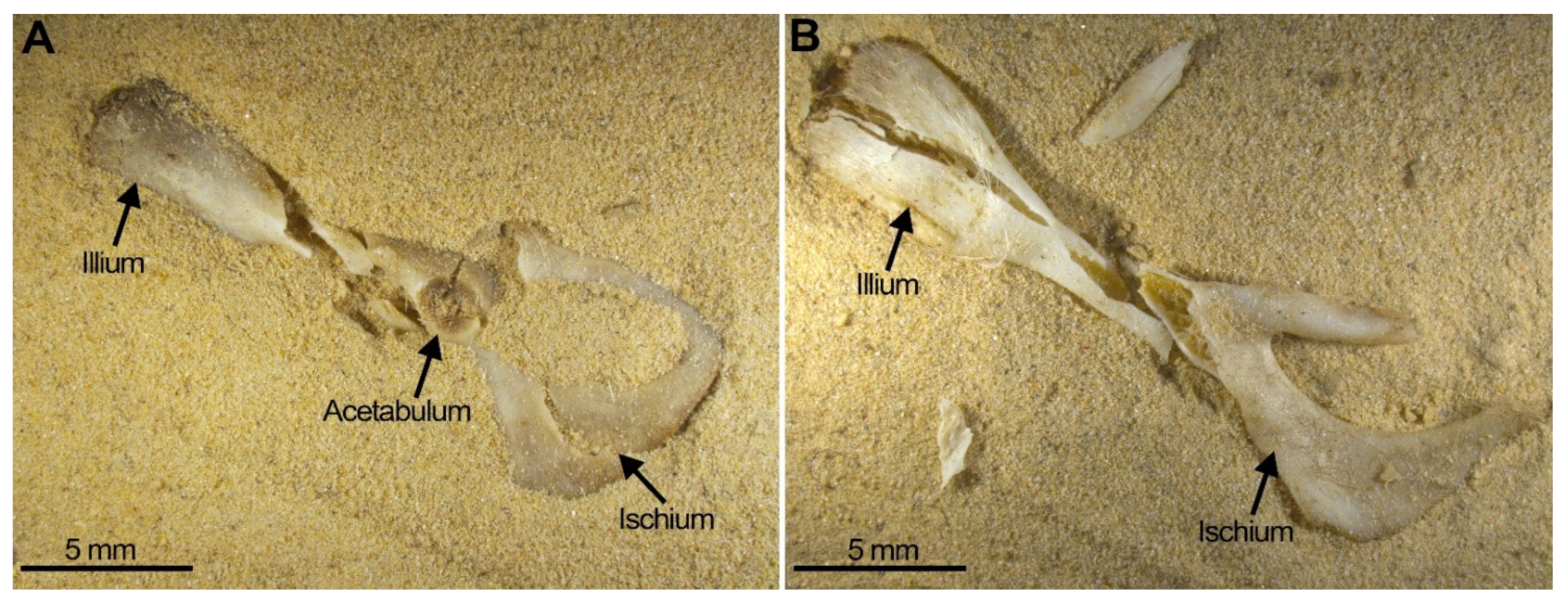
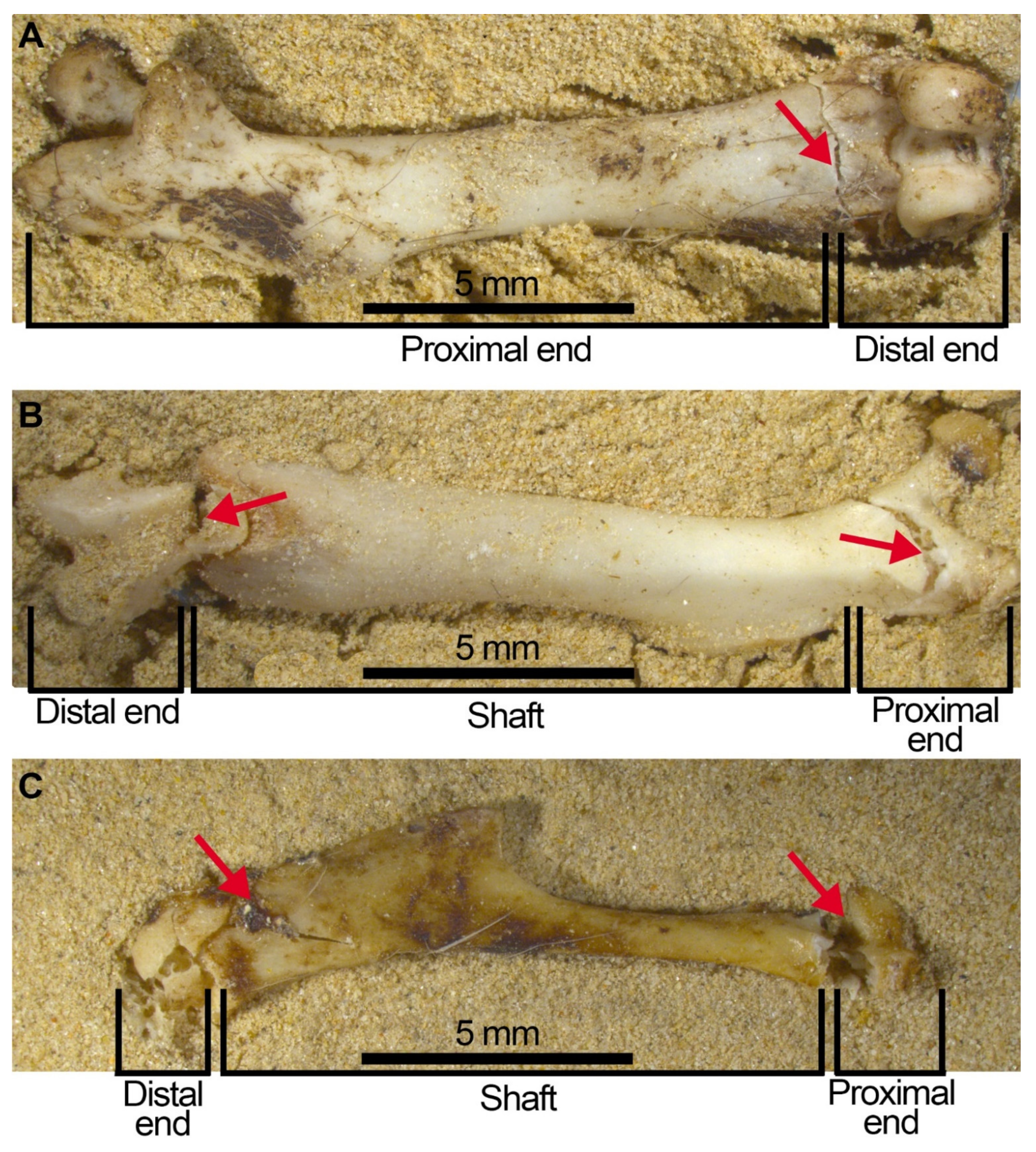
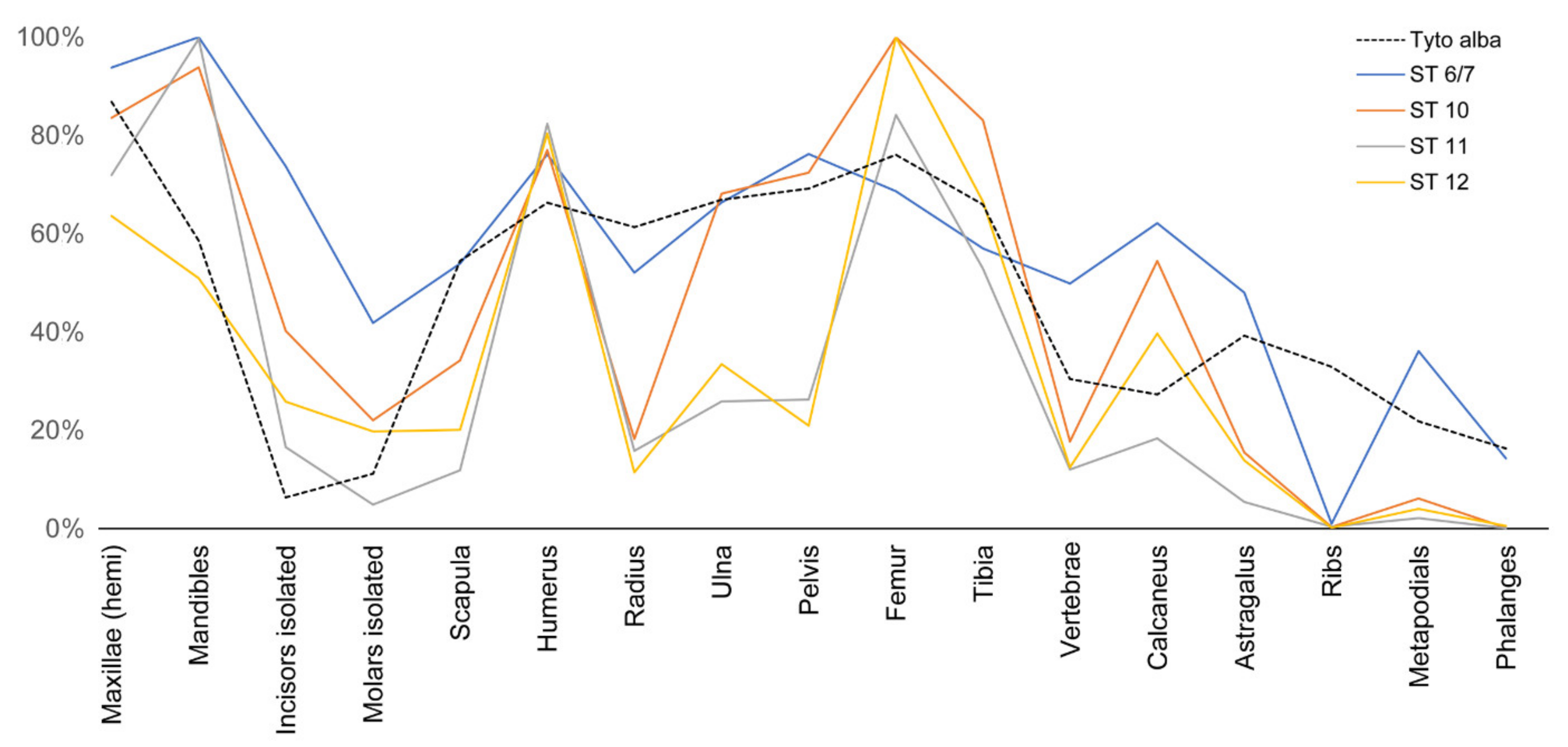
- Considerable breakage of larger postcranial elements and some degree of loss, but no loss or breakage of smaller elements (calcanei, talli). High frequency of calcanei and astragali is one of the most representative features of microfaunal assemblages of Wonderwerk Cave site, as well as modern assemblages exposed to trampling. In addition, a high frequency of complete bones was observed when compression was undertaken on wet bones together with jagged edges, fissures and cracks, which were frequently transversal to the length of the bone.
5. Conclusions
- -
- There are distinct patterns of bone breakage and anatomical element survival that can be used to recognise the involvement of trampling in small mammal assemblages.
- -
- Several patterns can be proposed based on the results obtained in this and previous experiments. The primary indications of trampling are: (1) Presence of edentulous jaws, (2) high postcranial breakage and (3) high frequency of complete calcanei and astragali.
- -
- Taphonomic categories, especially with reference to cranial elements, reflect other types of breakage patterns due to compression, especially with reference to mandibles and maxillae. Long bones under compression produce characteristic fissures and cracks (transversal to the metaphysis) that may be recorded on complete or almost complete bones which indicate trampling, although these observations need further study and experiments.
- -
- Results from the experiments we have undertaken were compared to those obtained in the actualistic experiment of human trampling (validated here) and compared to the taphonomic results of the Oldowan and Earlier Stone Age small mammal assemblages from Wonderwerk Cave (South Africa) (i.e., Strata 12, 11, 10 and 6/7). Comparison showed that trampling was an important factor responsible for the high degree of breakage observed in these assemblages. Trampling could have been caused by the predators (barn owl) themselves, as well as by other terrestrial animals or humans that visited the cave. The effects of trampling have been augmented by sifting and processing of the samples.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lyman, R.L. Vertebrate Taphonomy; Cambridge University Press: Cambridge, UK, 1994; p. 524. [Google Scholar]
- Andrews, P. Experiments in taphonomy. J. Archaeol. Sci. 1995, 22, 147–153. [Google Scholar] [CrossRef]
- Denys, C. Taphonomy and experimentation. Archaeometry 2002, 44, 469–484. [Google Scholar] [CrossRef]
- Fernández, F.J.; Montalvo, C.I. Actualistic Taphonomy of Small Mammals from Owl Pellets in South America and Its Archaeological Implication. Glob. J. Arch. Anthropol. 2017, 2, 1–4. [Google Scholar]
- Andrews, P.J.; Cook, J. Natural modifications to bones in a temperate setting. Man 1985, 20, 675–691. [Google Scholar] [CrossRef]
- Behrensmeyer, A.K.; Gordon, K.D.; Yanagi, G.T. Trampling as a cause of bone surface damage and pseudo-cutmarks. Nature 1986, 319, 768–771. [Google Scholar] [CrossRef]
- Olsen, S.L.; Shipman, P. Surface modification on bone: Trampling versus butchery. J. Archaeol. Sci. 1988, 15, 535–553. [Google Scholar] [CrossRef]
- Domínguez-Rodrigo, M.; De Juan, S.; Galan, A.B. A new protocol to differentiate trampling marks from butchery cut marks. J. Archaeol. Sci. 2009, 36, 2643–2654. [Google Scholar] [CrossRef]
- Denys, C.; Patou-Mathis, M. Manuel de Taphonomie (Collection Archéologiques); Errance: Paris, France, 2014; p. 248. [Google Scholar]
- Courtenay, L.A.; Huguet, R.; Yravedra, J. Scratches and grazes: A detailed microscopic analysis of trampling phenomena. J. Microsc. 2020, 277, 107–117. [Google Scholar] [CrossRef]
- Andrews, P. Owls, Caves and Fossils; The Natural History Museum: London, UK, 1990; p. 222. [Google Scholar]
- Madgwick, R. What makes bones shiny? Investigating trampling as a cause of bone abrasion. Archaeol. Anthropol. Sci. 2014, 6, 163–173. [Google Scholar] [CrossRef]
- Blasco, R.; Rosell, J.; Fernández-Peris, J.; Cáceres, I.; Verges, J.M. A new element of trampling: An experimental application on the Level XII faunal record of Bolomor Cave (Valencia, Spain). J. Archaeol. Sci. 2008, 35, 1605–1618. [Google Scholar] [CrossRef]
- Domínguez-Rodrigo, M.; Saladié, P.; Cáceres, I.; Hughet, R.; Yravedra, J.; Rodríguez-Hidalgo, A.; Martín, P.; Pinera, A.; Marín, J.; Gené, C.; et al. Use and abuse of cut mark analyses: The Rorschach effect. J. Archaeol. Sci. 2017, 86, 14–23. [Google Scholar] [CrossRef]
- Courtenay, L.A.; Huguet, R.; González-Aguilera, D.; Yravedra, J. A hybrid Geometric Morphometric deep learning approach for cut and trampling Mark classification. Appl. Sci. 2019, 10, 150. [Google Scholar] [CrossRef] [Green Version]
- Cifuentes-Alcobendas, G.; Domínguez-Rodrigo, M. Deep learning and taphonomy: High accuracy in the classification of cut marks made on fleshed and defleshed bones using convolutional neural networks. Sci. Rep. 2019, 9, 18933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villa, P.; Mahieu, E. Breakage patterns of human long bones. J. Hum. Evol. 1991, 21, 27–48. [Google Scholar] [CrossRef]
- Nicholson, R.A. A morphological investigation of burnt animal bone and an evaluation of its utility in archaeology. J. Archaeol. Sci. 1993, 20, 411–428. [Google Scholar] [CrossRef]
- Marin-Monfort, M.D.; Pesquero, M.D.; Fernández-Jalvo, Y. Compressive marks from gravel substrate on vertebrate remains: A preliminary experimental study. Quat. Int. 2014, 330, 118–125. [Google Scholar] [CrossRef] [Green Version]
- Frontini, R.; Morales-Muñiz, A.; Fernández-Jalvo, Y.; Roselló-Izquierdo, E.; Pesquero-Fernández, M.D.; Hernández, A.B.; García, L.A. Evaluation of size-related Salmonid fish vertebrae deformation due to compression. An experimental approach. Archaeol. Anthropol. Sci. 2021, Submitted. [Google Scholar]
- Fernández-Jalvo, Y.; Andrews, P. Experimental effects of water abrasion on bone fragments. J. Taphon. 2003, 1, 147–163. [Google Scholar]
- Gaudzinski-Windheuser, S.; Kindler, L.; Rabinovich, R.; Gofen-Inbar, N. Testing heterogeneity in faunal assemblages from archaeological sites. Tumbling and trampling experiments at the early-Middle Pleistocene site of Gesher Benot Ya’aqov (Israel). J. Archaeol. Sci. 2010, 37, 3170–3190. [Google Scholar] [CrossRef]
- Sahle, Y.; El Zaatari, S.; White, T.D. Hominid butchers and biting crocodiles in the African Plio–Pleistocene. Proc. Natl. Acad. Sci. USA 2017, 114, 13165–13169. [Google Scholar] [CrossRef] [Green Version]
- Frontini, R.; Roselló-Izquierdo, E.; Morales-Muñiz, A.; Denys, C.; Guillaud, E.; Fernández-Jalvo, Y.; Pesquero-Fernández, M.D. Compression and digestion as agents of vertebral deformation in fish remains: An experimental study to interpret archaeological assemblages. J. Archaeol. Method Theory 2021, 8, 1–8. [Google Scholar]
- Van Valkenburgh, B. Incidence of tooth breakage among large, predatory mammals. Am. Nat. 1988, 131, 291–302. [Google Scholar] [CrossRef]
- Tanner, J.B.; Zelditch, M.L.; Lundrigan, B.L.; Holekamp, K.E. Ontogenetic change in skull morphology and mechanical advantage in the spotted hyena (Crocuta crocuta). Int. J. Morphol. 2010, 271, 353–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palmqvist, P.; Martínez-Navarro, B.; Pérez-Claros, J.A.; Torregrosa, V.; Figueirido, B.; Jiménez-Arenas, J.M.; Espigares, M.P.; Ros-Montoya, S.; De Renzi, M. The giant hyena Pachycrocuta brevirostris: Modelling the bone-cracking behavior of an extinct carnivore. Quat. Int. 2011, 243, 61–79. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Andrews, P. Atlas of Taphonomic Identifications: 1001+ Images of Fossil and Recent Mammal Bone Modification; Springer: Berlin/Heidelberg, Germany, 2016; p. 355. [Google Scholar]
- Frontini, R.; Fernández-Jalvo, Y.; Pesquero, M.D.; Vechi, R.; Bayón, C. Abrasion in archaeological fish bones from sand dunes. An experimental approach. Archaeol. Anthropol. Sci. 2019, 11, 4891–4907. [Google Scholar] [CrossRef]
- Marin-Monfort, M.D.; García-Morato, S.; Andrews, P.; Avery, M.D.; Chazan, M.; Horwitz, L.K.; Fernández-Jalvo, Y. The owl that never left!, Taphonomy of Earlier Stone Age small mammal assemblages from Wonderwerk Cave (South Africa). Quat. Int. 2022, 614, 111–125. [Google Scholar] [CrossRef]
- Williams, J. Small Mammal Deposits in Archaeology: A Taphonomic Investigation of Tyto Alba (Barn Owl) Nesting and Roosting Sites. Ph.D. Thesis, University of Sheffield, Sheffield, UK, 2001, unpublished. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 27 January 2022).
- Mangiafico, S.S.; Summary and Analysis of Extension Program Evaluation in R, Version 1.18.8. Monte Carlo Simulation. 2016. Available online: https://rcompanion.org/handbook/ (accessed on 27 January 2022).
- Kim, H.Y. Statistical notes for clinical researchers: Chi-squared test and Fisher’s exact test. Restor. Dent. Endod. 2017, 42, 152–155. [Google Scholar] [CrossRef]
- Malan, B.D.; Cooke, H.B.S. A preliminary account of the Wonderwerk Cave, Kuruman. S. Afr. J. 1941, 37, 300–312. [Google Scholar]
- Malan, B.D.; Wells, L.H. A further report on the Wonderwerk Cave, Kuruman. S. Afr. J. 1943, 40, 258–270. [Google Scholar]
- Humphreys, A.J.B.; Thackeray, A.I. Ghaap and Gariep: Later Stone Age Studies in the Northern Cape; South African Archaeological Society: Cape Town, South Africa, 1983; p. 328. [Google Scholar]
- Beaumont, P.B.; Vogel, J.C. On a timescale for the past million years of human history in central South Africa. S. Afr. J. Sci. 2006, 102, 217–228. [Google Scholar]
- Beaumont, P.B. The edge: More on fire-making by about 1.7 million years ago at Wonderwerk Cave in South Africa. Curr. Anthropol. 2011, 52, 585–595. [Google Scholar] [CrossRef]
- Chazan, M.; Ron, H.; Matmon, A.; Porat, N.; Goldberg, P.; Yates, R.; Avery, M.; Sumner, A.; Horwitz, L.K. First radiometric dates for the Earlier Stone Age sequence in Wonderwerk Cave, South Africa. J. Hum. Evol. 2008, 55, 1–11. [Google Scholar] [CrossRef]
- Horwitz, L.K.; Chazan, M. Past and present at Wonderwerk cave (Northern Cape Province, South Africa). Afr. Archaeol. Rev. 2015, 32, 595–612. [Google Scholar] [CrossRef] [Green Version]
- Ecker, M.; Brink, J.; Chazan, M.; Horwitz, L.K.; Lee Thorp, J.A. Radiocarbon dates constrain the timing of environmental and cultural shifts in the Holocene strata of Wonderwerk Cave, South Africa. Radiocarbon 2017, 59, 1067–1086. [Google Scholar] [CrossRef] [Green Version]
- Shaar, R.; Matmon, A.; Horwitz, L.K.; Ebert, Y.; Chazan, M. Magnetostratigraphy and cosmogenic dating of Wonderwerk Cave: New constraints for the chronology of the South African Earlier Stone Age. Quat. Sci. Rev. 2021, 259, 106907. [Google Scholar] [CrossRef]
- Avery, D.M. Micromammals as paleoenvironmental indicators of the southern African Quaternary. Trans. R. Soc. S. Afr. 2007, 62, 17–23. [Google Scholar] [CrossRef]
- Avery, D.M. Rodents and other micromammals from the Pleistocene strata in Excavation 1 at Wonderwerk Cave, South Africa: A work in progress. Quat. Int. 2022, 614, 23–36. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Avery, D.M. Pleistocene micromammals and their predators at Wonderwerk Cave, South Africa. Afr. Archaeol. Rev. 2015, 32, 751–791. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Tormo, L.; Andrews, P.; Marin-Monfort, M.D. Taphonomy of burnt bones from Wonderwerk Cave (South Africa). Quat. Int. 2018, 495, 19–29. [Google Scholar] [CrossRef]
- Fernández-López, S.R. Taphonomic concepts of or a theoretical biochronology. Revista Española de Paleontología 1991, 6, 37–49. [Google Scholar]
- Goldberg, P.; Berna, F.; Chazan, M. Deposition and diagenesis in the Earlier Stone Age of Wonderwerk Cave, Excavation 1, South Africa. Afr. Archaeol. Rev. 2015, 32, 613–643. [Google Scholar] [CrossRef]
- Currey, J.D. The mechanical properties of bone. Clin. Orthop. Relat. Res. 1970, 73, 210–231. [Google Scholar] [CrossRef] [Green Version]
- Burstein, A.H.; Zika, J.M.; Heiple, K.G.; Klein, L. Contribution of collagen and mineral to the elastic-plastic properties of bone. J. Bone. Joint. Surg. Br. 1975, 57, 956–961. [Google Scholar] [CrossRef]
- Reilly, D.T.; Burstein, A.H. The mechanical properties of cortical bone. J. Bone. Joint. Surg. Br. 1974, 56, 1001–1022. [Google Scholar] [CrossRef]
- Turner, C.H. Bone strength: Current concepts. Ann. N. Y. Acad. Sci. 2006, 1068, 429–446. [Google Scholar] [CrossRef]
- Nyman, J.S.; Roy, A.; Shen, X.; Acuna, R.L.; Tyler, J.H.; Wang, X. The influence of water removal on the strength and toughness of cortical bone. J. Biomech. 2006, 39, 931–938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agnew, A.M.; Bolte, J.H. Bone fracture. Biomechanics and risk. In Bone Histology: An Anthropological Perspective; Crowder, C., Stout, S., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 237–256. [Google Scholar]
- Terry, R.C. Owl pellet taphonomy: A preliminary study of the post-regurgitation taphonomic history of pellets in a temperate forest. Palaios 2004, 19, 497–506. [Google Scholar] [CrossRef]
- Guillermo, A.A.; Montalvo, C.I.; Fernández, F.J. Palimpsest of micromammal deposits in an archaeological rock shelter (Álvarez 4, late Holocene) from northwestern Patagonia, Argentina. Archaeol. Anthropol. Sci. 2021, 13, 178. [Google Scholar] [CrossRef]




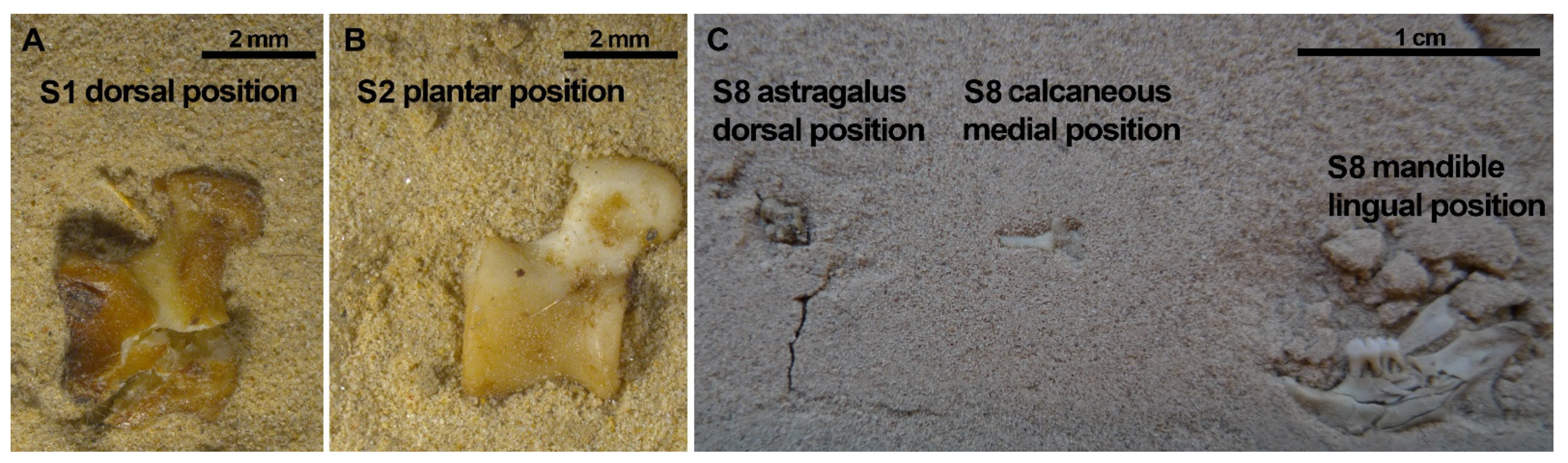
| SETS | State | Size | Skulls | Mandibles | Femora | Humeri | Pelves | Astragali | Calcanei |
|---|---|---|---|---|---|---|---|---|---|
| Set 1 | Dry | Large | Dorsal | Lingual | Anterior | Anterior | Ventral | Dorsal Plantar | Absent |
| Set 2 | Dry | Large | Ventral | Buccal | Anterior Posterior | Posterior | Dorsal | Dorsal | Medial |
| Set 3 | Dry | Small | Dorsal | Lingual | Anterior | Anterior | Ventral | Plantar | Lateral |
| Set 4 | Dry | Small | Ventral | Buccal | Anterior Posterior | Anterior | Dorsal | Dorsal | Medial |
| Set 5 | Wet | Large | Dorsal | Buccal | Posterior | Posterior | Ventral | Plantar | Lateral |
| Set 6 | Wet | Large | Ventral | Lingual | Anterior Posterior | Anterior | Dorsal | Dorsal | Medial |
| Set 7 | Wet | Small | Dorsal | Buccal | Posterior Anterior | Posterior | Ventral | Plantar | Lateral |
| Set 8 | Wet | Small | Ventral | Lingual | Posterior | Lateral | Dorsal | Dorsal | Medial |
| STRATA | St 6/7 | St10 | St11 | St12 | ||||
|---|---|---|---|---|---|---|---|---|
| FEMORA (Total N) | N = 1255 | % | N = 3072 | % | N = 393 | % | N = 1451 | % |
| Complete | 353 | 28% | 216 | 7% | 43 | 11% | 72 | 5% |
| Proximal (+Prox + 1/2) | 650 | 52% | 1953 | 64% | 191 | 49% | 962 | 66% |
| Shaft | 61 | 5% | 351 | 11% | 76 | 19% | 236 | 16% |
| Distal (+Dist + 1/2) | 191 | 15% | 552 | 18% | 83 | 21% | 181 | 12% |
| HUMERI (Total N) | N = 1425 | % | N = 2975 | % | N = 372 | % | N = 1185 | % |
| Complete | 446 | 31% | 325 | 11% | 49 | 12% | 85 | 6% |
| Proximal (+Prox + 1/2) | 212 | 15% | 803 | 27% | 60 | 15% | 104 | 7% |
| Shaft | 100 | 7% | 505 | 17% | 85 | 22% | 258 | 18% |
| Distal (+Dist + 1/2) | 667 | 47% | 1342 | 45% | 178 | 45% | 738 | 51% |
| Skulls (n = 8) | N | % |
|---|---|---|
| M1 + zygomatic arch | 7 | 88 |
| Incisor in socket | 8 | 100 |
| Isolated molars | 8 | 100 |
| Mandibles (n = 8) | ||
| Complete or longitudinal fissures | 4 | 33 |
| M1 + diastema | 7 | 58 |
| Detached molars | 4 | 33 |
| Incisor in socket | 7 | 58 |
| Detached ascending ramus | 4 | 33 |
| Pelvis (n = 8) | N | |
|---|---|---|
| Almost Complete | 1 | 13 |
| Articular Cavity (Damaged) | 2 | 25 |
| Cavity + Illium | 0 | 0 |
| Cavity + Ischium | 3 | 38 |
| Cavity + Pubis Branches | 2 | 25 |
| Experiment Sample | ||
|---|---|---|
| Femora (n = 12) | Compression experiment (n) % | Compressed + processed (n) % |
| complete | (7) 41 | (3) 13 |
| proximal | (5) 29 | (9) 39 |
| shaft | (1) 6 | (4) 17 |
| distal | (4) 24 | (7) 30 |
| Humeri (n = 10 *) | ||
| complete | (4) 27 | (2) 10 |
| proximal | (3) 20 | (4) 20 |
| shaft | (2) 13 | (6) 30 |
| distal | (6) 40 | (8) 40 |
| Femur | Humeri | |||
|---|---|---|---|---|
| Stratum | Compressed | Compressed + Processed | Compressed | Compressed + Processed |
| St 6/7 | 0.190 | 0.006 | 0.085 | 0.004 |
| St 10 | 0.001 | 0.052 | 0.046 | 0.518 |
| St 11 | 0.005 | 0.656 | 0.049 | 0.753 |
| St 12 | 0.001 | 0.010 | 0.002 | 0.099 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Jalvo, Y.; Rueda, L.; Fernández, F.J.; García-Morato, S.; Marin-Monfort, M.D.; Montalvo, C.I.; Tomassini, R.; Chazan, M.; Horwitz, L.K.; Andrews, P. Understanding the Impact of Trampling on Rodent Bones. Quaternary 2022, 5, 11. https://doi.org/10.3390/quat5010011
Fernández-Jalvo Y, Rueda L, Fernández FJ, García-Morato S, Marin-Monfort MD, Montalvo CI, Tomassini R, Chazan M, Horwitz LK, Andrews P. Understanding the Impact of Trampling on Rodent Bones. Quaternary. 2022; 5(1):11. https://doi.org/10.3390/quat5010011
Chicago/Turabian StyleFernández-Jalvo, Yolanda, Lucía Rueda, Fernando Julian Fernández, Sara García-Morato, María Dolores Marin-Monfort, Claudia Ines Montalvo, Rodrigo Tomassini, Michael Chazan, Liora K. Horwitz, and Peter Andrews. 2022. "Understanding the Impact of Trampling on Rodent Bones" Quaternary 5, no. 1: 11. https://doi.org/10.3390/quat5010011
APA StyleFernández-Jalvo, Y., Rueda, L., Fernández, F. J., García-Morato, S., Marin-Monfort, M. D., Montalvo, C. I., Tomassini, R., Chazan, M., Horwitz, L. K., & Andrews, P. (2022). Understanding the Impact of Trampling on Rodent Bones. Quaternary, 5(1), 11. https://doi.org/10.3390/quat5010011






