The Microvertebrates of Shanidar Cave: Preliminary Taphonomic Findings
Abstract
:1. Introduction
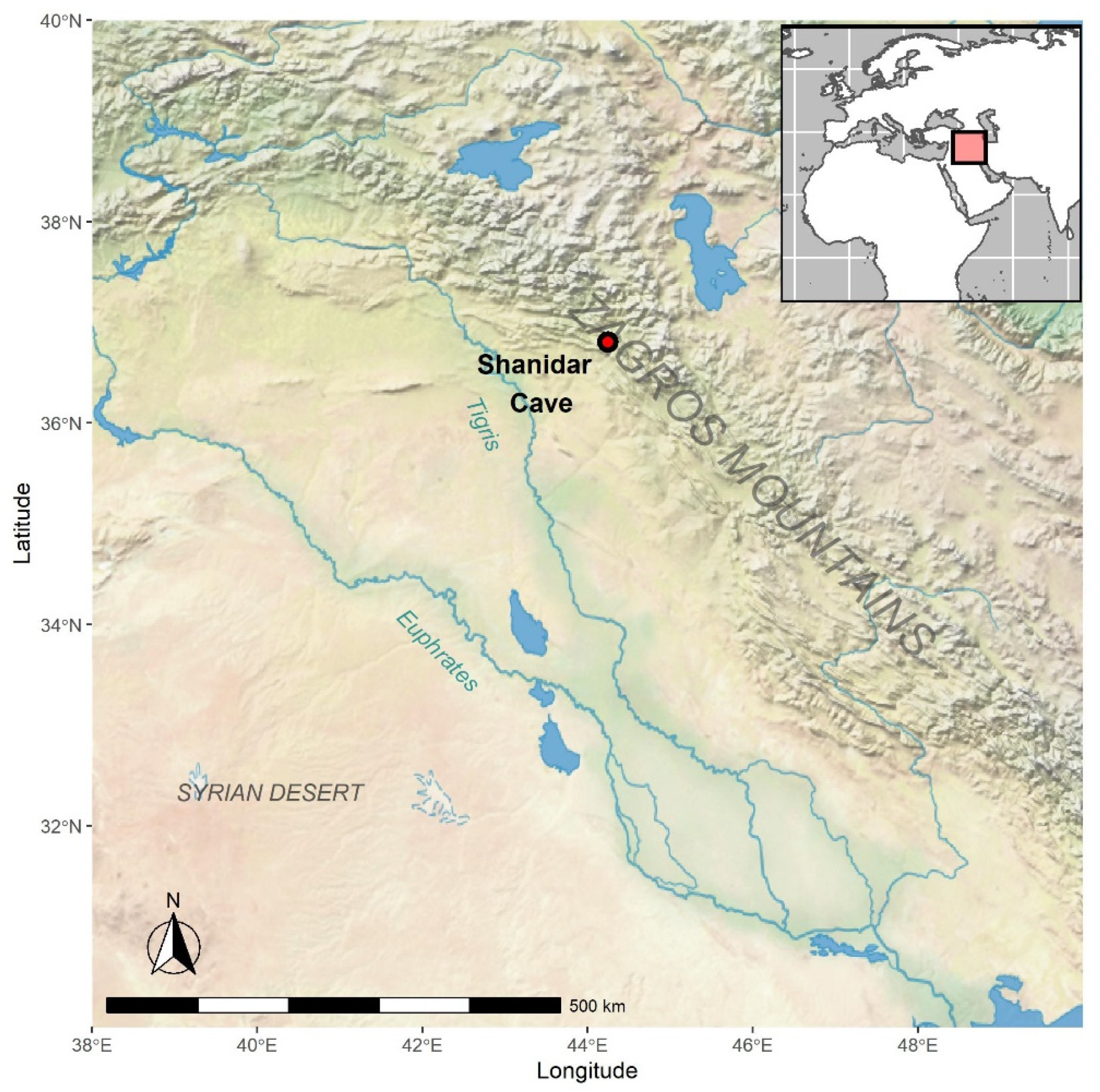
1.1. The Site
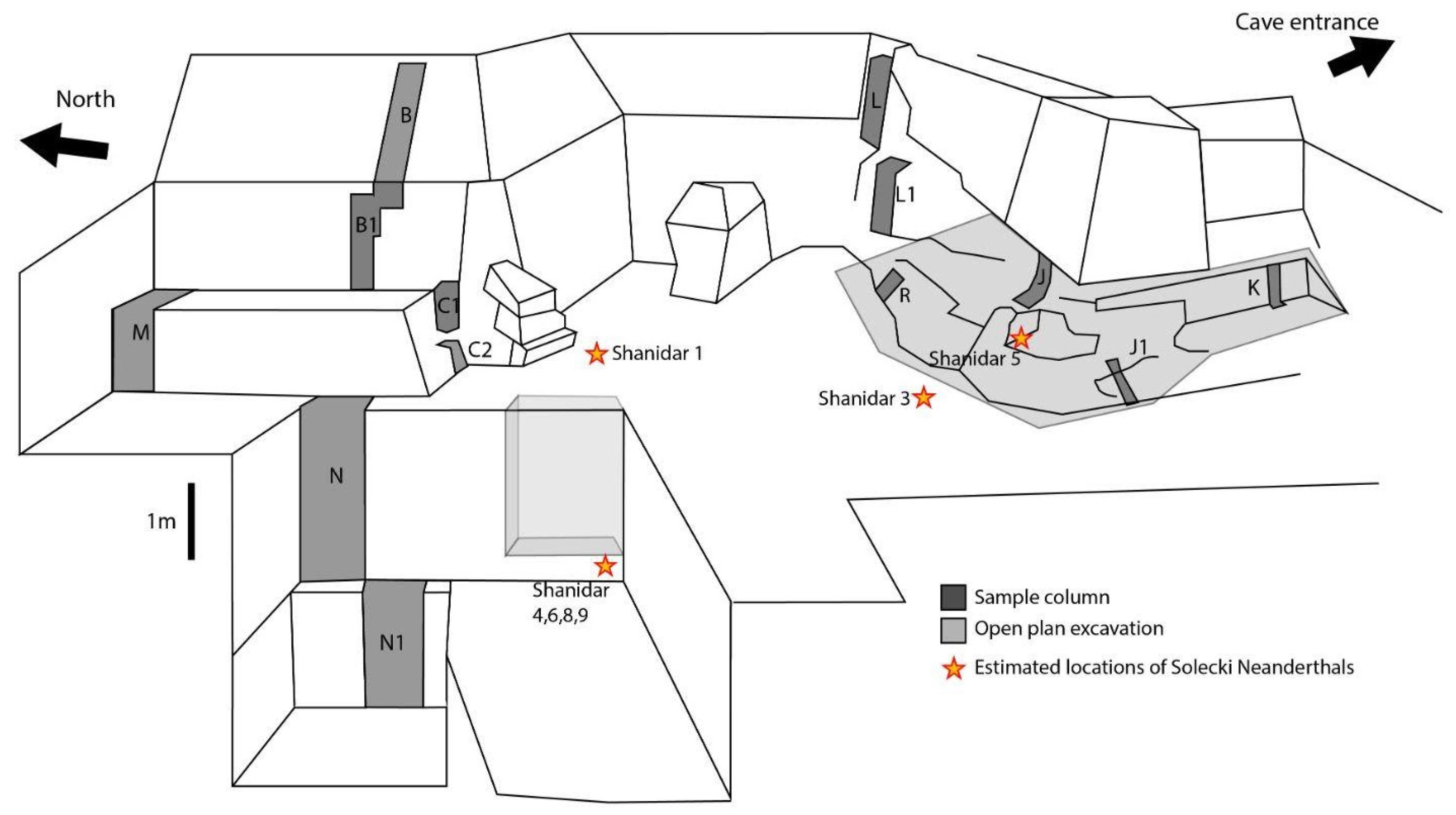
1.1.1. Excavations 1951–1960
1.1.2. Recent Excavations
1.1.3. Stratigraphy and Dating of the Cave
1.2. Taphonomy and Microvertebrate Studies
2. Materials and Methods
2.1. Sampling and Processing of Material
2.2. Taphonomic Methods
3. Results
3.1. Breakage
3.1.1. Humeri and Femora Results
3.1.2. Teeth Results
3.1.3. Comparison between the Postcranial and the Dental Breakage Patterns
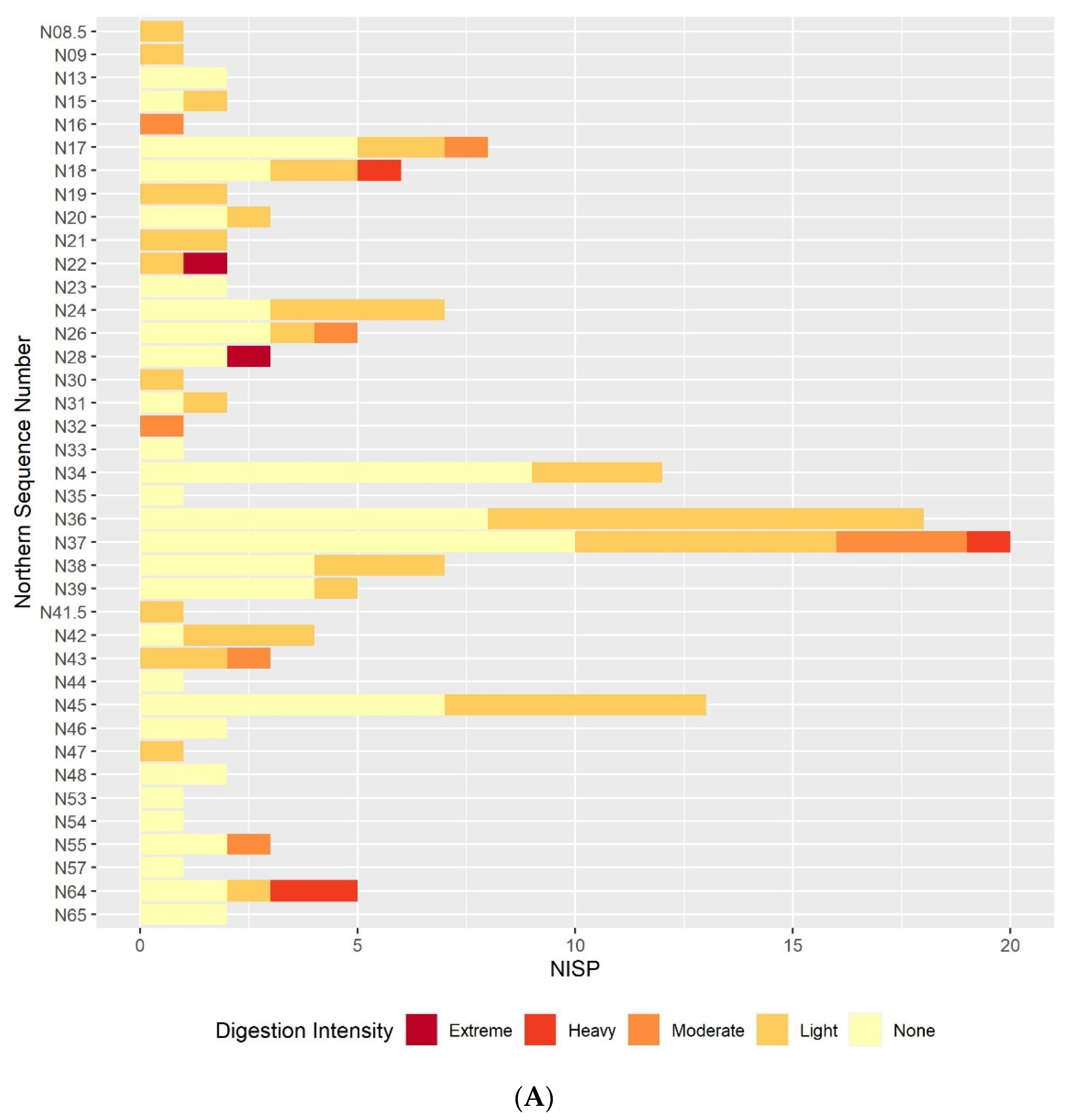
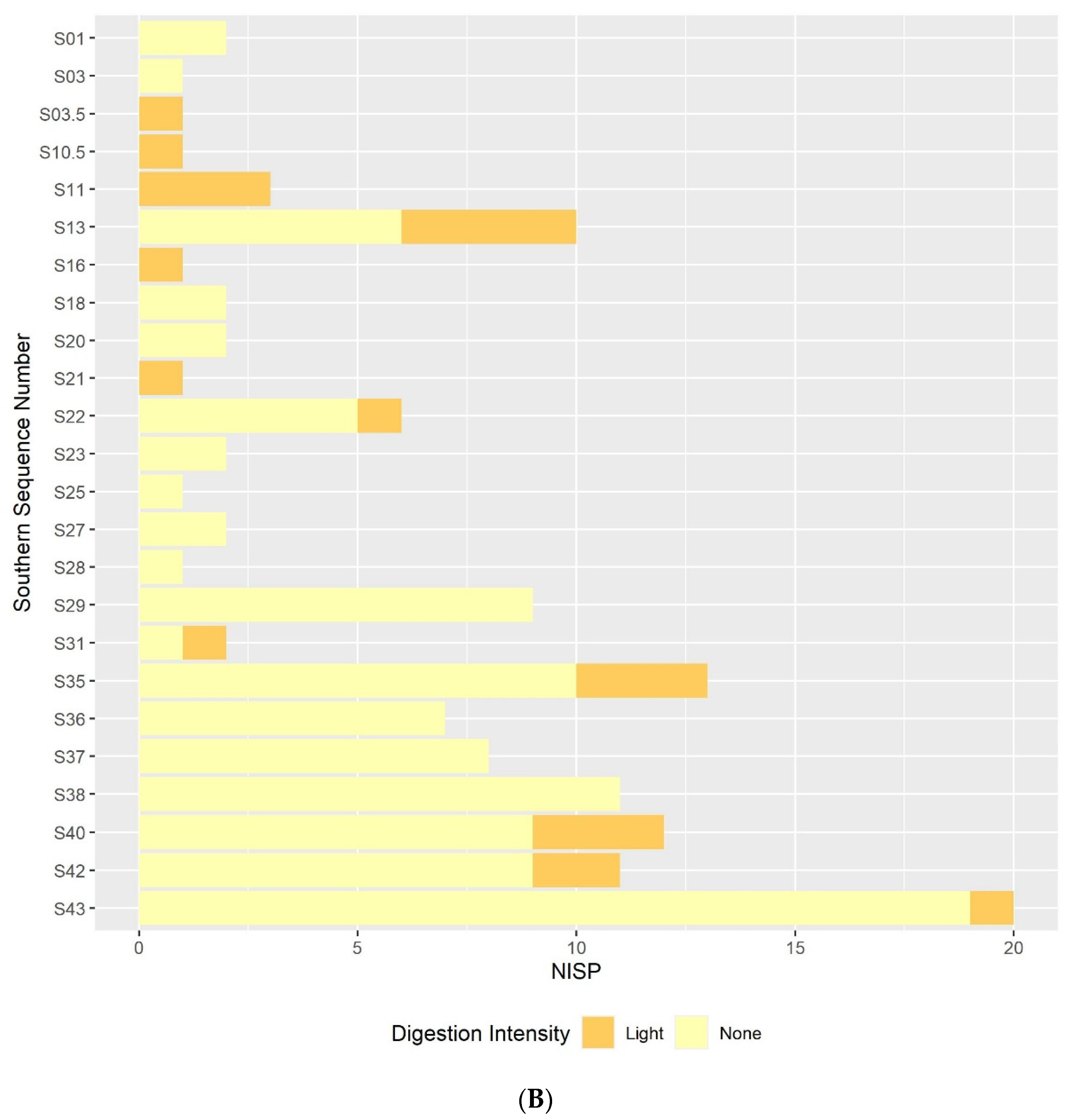
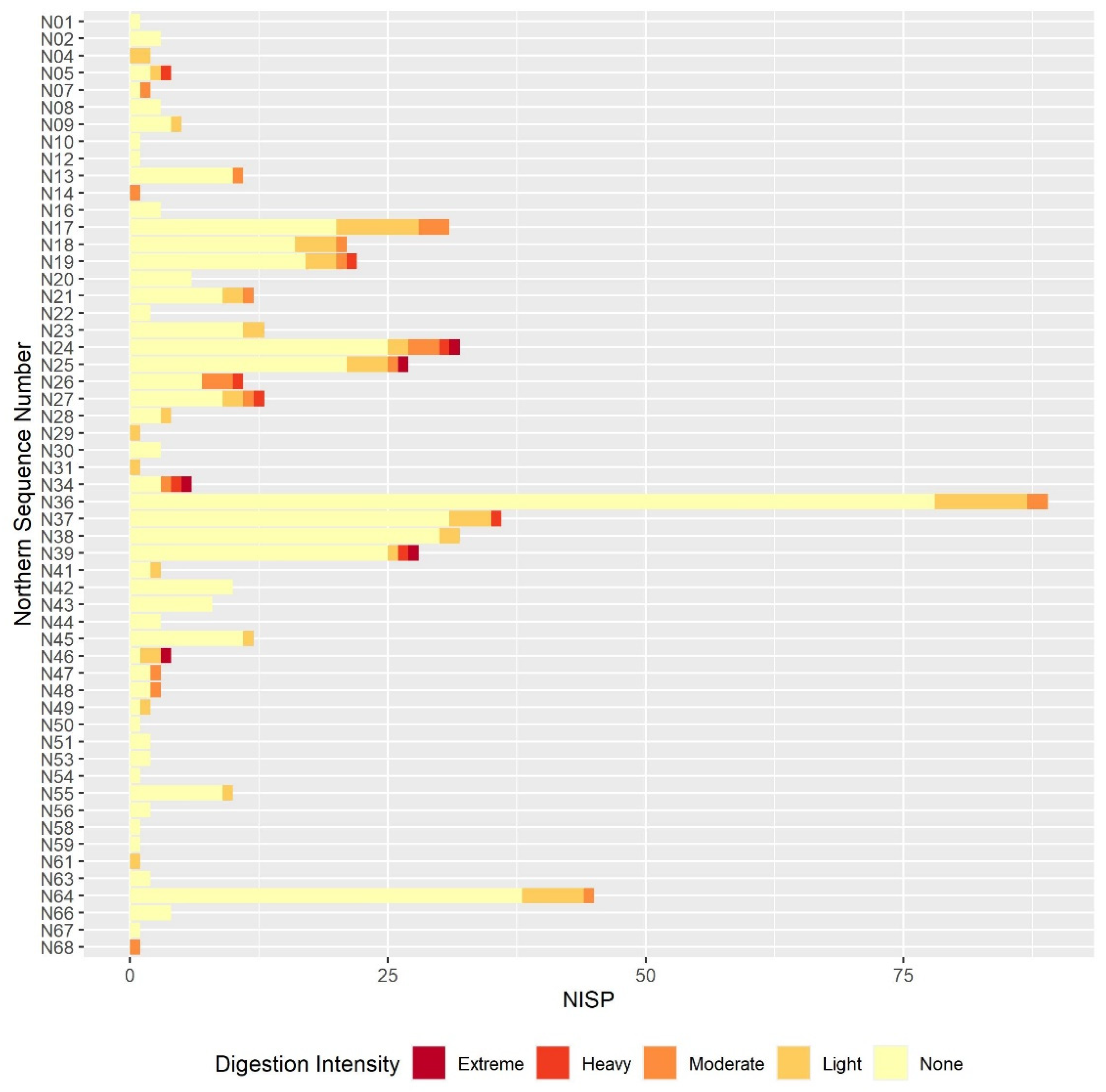
3.2. Digestion
3.2.1. Humeri and Femora Results
3.2.2. Molar Results
3.2.3. Comparison between the Postcranial and the Molar Digestion Patterns
3.3. Other Taphonomic Marks
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stahl, P.W. The recovery and interpretation of microvertebrate bone assemblages from archaeological contexts. J. Archaeol. Method Theory 1996, 3, 31–75. [Google Scholar] [CrossRef]
- Lyman, R.L. Vertebrate Taphonomy; Cambridge Manuals in Archaeology; Cambridge University Press: Cambridge, UK, 1994; ISBN 978-0-521-45840-5. [Google Scholar]
- Fiacconi, M.; Hunt, C.O. Pollen taphonomy at Shanidar Cave (Kurdish Iraq): An initial evaluation. Rev. Palaeobot. Palynol. 2015, 223, 87–93. [Google Scholar] [CrossRef]
- Solecki, R.S. Prehistory in Shanidar Valley, Northern Iraq. Science 1963, 139, 179–193. [Google Scholar] [PubMed]
- Becerra-Valdivia, L.; Douka, K.; Comeskey, D.; Bazgir, B.; Conard, N.J.; Marean, C.W.; Ollé, A.; Otte, M.; Tumung, L.; Zeidi, M.; et al. Chronometric investigations of the Middle to Upper Paleolithic transition in the Zagros Mountains using AMS radiocarbon dating and Bayesian age modelling. J. Hum. Evol. 2017, 109, 57–69. [Google Scholar] [CrossRef]
- Solecki, R.S. Notes on a brief archaeological reconnaissance of cave sites in the Rowanduz district of Iraq. Sumer 1952, 8, 37–48. [Google Scholar]
- Solecki, R.S. A palaeolithic site in the Zagros Mountains of Northern Iraq, report on a sounding at Shanidar Cave: Part I. Sumer 1952, 8, 127–192. [Google Scholar]
- Solecki, R.S. A palaeolithic site in the Zagros Mountains of Northern Iraq, report on a sounding at Shanidar Cave: Part II. Sumer 1953, 9, 60–93. [Google Scholar]
- Solecki, R.S. Shanidar Cave, a Paleolithic site in Northern Iraq. In Annual Report of the Smithsonian Institution, 1954; Annual Report of the Smithsonian Institution; Smithsonian Institution: Washington, DC, USA, 1955; pp. 389–425. [Google Scholar]
- Solecki, R.S. Three adult Neanderthal skeletons from Shanidar Cave, Northern Iraq. In Annual Report of the Smithsonian Institution, 1959; Annual Report of the Smithsonian Institution; Smithsonian Institution: Washington, DC, USA, 1960; pp. 603–635. [Google Scholar]
- Solecki, R.S. New anthropological discoveries at Shanidar, Northern Iraq. Trans. N. Y. Acad. Sci. 1961, 23, 690–699. [Google Scholar] [CrossRef]
- Solecki, R.S.; Leroi-Gourhan, A. Palaeoclimatology and archaeology in the Near East. Ann. N. Y. Acad. Sci. 1961, 95, 729–739. [Google Scholar] [CrossRef]
- Cowgill, L.W.; Trinkaus, E.; Zeder, M.A. Shanidar 10: A Middle Paleolithic immature distal lower limb from Shanidar Cave, Iraqi Kurdistan. J. Hum. Evol. 2007, 53, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Trinkaus, E. The Shanidar Neandertals; Academic Press: New York, NY, USA, 1983. [Google Scholar]
- Dettwyler, K.A. Can paleopathology provide evidence for “compassion”? Am. J. Phys. Anthropol. 1991, 84, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Solecki, R.S. Shanidar: The Humanity of Neanderthal Man; Allen Lane The Penguin Press: London, UK, 1971; ISBN 978-0-7139-0306-5. [Google Scholar]
- Trinkaus, E.; Zimmerman, M.R. Trauma among the Shanidar Neandertals. Am. J. Phys. Anthropol. 1982, 57, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Leroi-Gourhan, A. The flowers found with Shanidar IV, a Neanderthal burial in Iraq. Science 1975, 190, 562–564. [Google Scholar] [CrossRef]
- Solecki, R.S. The implications of the Shanidar cave Neanderthal flower burial. Ann. N. Y. Acad. Sci. 1977, 293, 114–124. [Google Scholar] [CrossRef]
- Sommer, J.D. The Shanidar IV ‘flower burial’: A re-evaluation of Neanderthal burial ritual. Camb. Archaeol. J. 1999, 9, 127–129. [Google Scholar] [CrossRef]
- Perkins, D. Prehistoric fauna from Shanidar, Iraq. Science 1964, 144, 1565–1566. [Google Scholar] [CrossRef]
- Evins, M. A Study of the Fauna from the Mousterian Deposits at Shanidar Cave, Northeastern Iraq. Master’s Thesis, University of Chicago, Chicago, IL, USA, 1981. [Google Scholar]
- Evins, M. The fauna from Shanidar Cave: Mousterian wild goat exploitation in Northeastern Iraq. Paléorient 1982, 8, 37–58. [Google Scholar] [CrossRef]
- Reynolds, T.; Boismier, W.; Farr, L.; Abdulmutalb, D.; Barker, G. New investigations at Shanidar Cave, Iraqi Kurdistan. In The Archaeology of the Kurdistan Region of Iraq and Adjacent Regions; Kopanias, K., MacGinnis, J., Eds.; Archaeopress: Oxford, UK, 2016; pp. 369–372. ISBN 978-1-78491-394-6. [Google Scholar]
- Hunt, C.O.; Hill, E.A.; Reynolds, T.; Abdulmutalb, D.; Farr, L.; Lane, R.; Szabó, K.; Barker, G. An incised shell object from Baradostian (Early Upper Palaeolithic) layers in Shanidar Cave, Iraqi Kurdistan. J. Archaeol. Sci. Rep. 2017, 14, 318–322. [Google Scholar] [CrossRef]
- Pomeroy, E.; Mirazón Lahr, M.; Crivellaro, F.; Farr, L.; Reynolds, T.; Hunt, C.O.; Barker, G. Newly discovered Neanderthal remains from Shanidar Cave, Iraqi Kurdistan, and their attribution to Shanidar 5. J. Hum. Evol. 2017, 111, 102–118. [Google Scholar] [CrossRef]
- Pomeroy, E.; Bennett, P.; Hunt, C.O.; Reynolds, T.; Farr, L.; Frouin, M.; Holman, J.; Lane, R.; French, C.; Barker, G. New Neanderthal remains associated with the ‘flower burial’ at Shanidar Cave. Antiquity 2020, 94, 11–26. [Google Scholar] [CrossRef] [Green Version]
- Pomeroy, E.; Hunt, C.O.; Reynolds, T.; Abdulmutalb, D.; Asouti, E.; Bennett, P.; Bosch, M.; Burke, A.; Farr, L.; Foley, R.; et al. Issues of theory and method in the analysis of Paleolithic mortuary behavior: A view from Shanidar Cave. Evol. Anthropol. 2020, 29, 263–279. [Google Scholar] [CrossRef]
- Friedman, I.; Smith, R.L. Part I, the development of the method. Am. Antiq. 1960, 25, 476–493. [Google Scholar] [CrossRef]
- Brothwell, D.R.; Jones, R. The relevance of small mammal studies to archaeology. In Research Problems in Zooarchaeology; Routledge: London, UK; Institute of Archaeology: London, UK, 1978; pp. 47–57. [Google Scholar]
- Cucchi, T.; Papayianni, K.; Cersoy, S.; Aznar-Cormano, L.; Zazzo, A.; Debruyne, R.; Berthon, R.; Bălășescu, A.; Simmons, A.; Valla, F.; et al. Tracking the Near Eastern origins and European dispersal of the Western house mouse. Sci. Rep. 2020, 10, 8276. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, T. Animals as Neighbors: The Past and Present of Commensal Species; Michigan State University Press: East Lansing, MI, USA, 2013. [Google Scholar]
- Avery, D.M. Micromammals and paleoenvironmental interpretation in Southern Africa. Geoarchaeology 1988, 3, 41–52. [Google Scholar] [CrossRef]
- Belmaker, M.; Bar-Yosef, O.; Belfer-Cohen, A.; Meshveliani, T.; Jakeli, N. The environment in the Caucasus in the Upper Paleolithic (Late Pleistocene): Evidence from the small mammals from Dzudzuana cave, Georgia. Quat. Int. 2016, 425, 4–15. [Google Scholar] [CrossRef]
- Belmaker, M.; Hovers, E. Ecological change and the extinction of the Levantine Neanderthals: Implications from a diachronic study of micromammals from Amud Cave, Israel. Quat. Sci. Rev. 2011, 30, 3196–3209. [Google Scholar] [CrossRef]
- Maul, L.C.; Smith, K.T.; Barkai, R.; Barash, A.; Karkanas, P.; Shahack-Gross, R.; Gopher, A. Microfaunal remains at Middle Pleistocene Qesem Cave, Israel: Preliminary results on small vertebrates, environment and biostratigraphy. J. Hum. Evol. 2011, 60, 464–480. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.T.; Maul, L.C.; Flemming, F.; Barkai, R.; Gopher, A. The microvertebrates of Qesem Cave: A comparison of the two concentrations. Quat. Int. 2016, 398, 233–245. [Google Scholar] [CrossRef]
- López-González, F.; Grandal-d’Anglade, A.; Vidal-Romaní, J.R. Deciphering bone depositional sequences in caves through the study of manganese coatings. J. Archaeol. Sci. 2006, 33, 707–717. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Andrews, P.; Sevilla, P.; Requejo, V. Digestion versus abrasion features in rodent bones. Lethaia 2014, 47, 323–336. [Google Scholar] [CrossRef]
- Andrews, P. Owls, Caves and Fossils; The University of Chicago Press: Chicago, IL, USA, 1990; ISBN 978-0-565-01118-5. [Google Scholar]
- Fernández-Jalvo, Y.; Scott, L.; Andrews, P. Taphonomy in palaeoecological interpretations. Quat. Sci. Rev. 2011, 30, 1296–1302. [Google Scholar] [CrossRef]
- Mayhew, D.F. Avian predators as accumulators of fossil mammal material. Boreas 1977, 6, 25–31. [Google Scholar] [CrossRef]
- Denys, C.; Stoetzel, E.; Andrews, P.; Bailon, S.; Rihane, A.; Huchet, J.B.; Fernández-Jalvo, Y.; Laroulandie, V. Taphonomy of small predators multi-taxa accumulations: Palaeoecological implications. Hist. Biol. 2018, 30, 868–881. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Andrews, P.; Denys, C.; Sesé, C.; Stoetzel, E.; Marin-Monfort, D.; Pesquero, D. Taphonomy for taxonomists: Implications of predation in small mammal studies. Quat. Sci. Rev. 2016, 139, 138–157. [Google Scholar] [CrossRef]
- Fernández-Jalvo, Y.; Andrews, P. Atlas of Taphonomic Identifications: 1001+ Images of Fossil and Recent Mammal Bone Modification; Springer: New York, NY, USA, 2016. [Google Scholar]
- Jenkins, E.L. Unwanted Inhabitants? The Microfauna from Çatalhöyük and Pınarbası; VDM: Saarbrücken, Germany, 2009; ISBN 978-3-639-11213-9. [Google Scholar]
- Marín Arroyo, A.B.; Landete Ruiz, M.D.; Vidal Bernabeu, G.; Seva Román, R.; González Morales, M.R.; Straus, L.G. Archaeological implications of human-derived manganese coatings: A study of blackened bones in El Mirón Cave, Cantabrian Spain. J. Archaeol. Sci. 2008, 35, 801–813. [Google Scholar] [CrossRef]
- Porter, R.; Salim, M.; Ararat, K. A provisional checklist of the birds of Iraq. Marsh Bull. 2010, 5, 56–95. [Google Scholar]
- Bazgir, B.; Ollé, A.; Tumung, L.; Becerra-Valdivia, L.; Douka, K.; Higham, T.; Van Der Made, J.; Picin, A.; Saladié, P.; López-Garciá, J.M.; et al. Understanding the emergence of modern humans and the disappearance of Neanderthals: Insights from Kaldar Cave (Khorramabad Valley, Western Iran). Sci. Rep. 2017, 7, 43460. [Google Scholar] [CrossRef]
- Rey-Rodríguez, I.; López-García, J.-M.; Blain, H.-A.; Stoetzel, E.; Denys, C.; Fernández-García, M.; Tumung, L.; Ollé, A.; Bazgir, B. Exploring the landscape and climatic conditions of Neanderthals and Anatomically Modern Humans in the Middle East: The rodent assemblage from the Late Pleistocene of Kaldar Cave (Khorramabad Valley, Iran). Quat. Sci. Rev. 2020, 236, 106278. [Google Scholar] [CrossRef]
- Rey-Rodríguez, I.; Stoetzel, E.; López-García, J.M.; Denys, C. Implications of modern barn owl pellets analysis for archaeological studies in the Middle East. J. Archaeol. Sci. 2019, 111, 105029. [Google Scholar] [CrossRef]
- Charter, M.; Izhaki, I.; Meyrom, K.; Motro, Y.; Leshem, Y. Diets of barn owls differ in the same agricultural region. Wilson J. Ornithol. 2009, 121, 378–383. [Google Scholar] [CrossRef]
- Williams, J.P. Small Mammal Deposits in Archaeology: A Taphonomic Investigation of Tyto Alba (Barn Owl) Nesting and Roosting Sites. Ph.D. Thesis, University of Sheffield, Sheffield, UK, 2001. [Google Scholar]





| Order | Family | Subfamily | Species |
|---|---|---|---|
| Rodentia | |||
| Cricetidae | |||
| Arvicolinae | |||
| Microtus socialis s.l. | |||
| Ellobius sp. | |||
| Arvicola cf. amphibius | |||
| Cricetinae | |||
| Cricetulus sp. | |||
| Mesocricetus sp. | |||
| Muridae | |||
| Murinae | |||
| Apodemus (Sylvaemus) sp. | |||
| Mus cf. musculus | |||
| Micromys sp. | |||
| Gerbillinae | Meriones cf. persicus | ||
| Calomyscidae | |||
| Calomyscus sp. | |||
| Eulipotyphla | |||
| Soricidae | |||
| Crocidurinae | |||
| Crocidura cf. suaveolens |
| Element | Portion Present | Number of Specimens Examined | Proportion (%) 1 |
|---|---|---|---|
| Humerus | Proximal epiphysis | 20 | 6.0 |
| Proximal epiphysis and shaft | 10 | 3.0 | |
| Shaft | 39 | 11.6 | |
| Distal epiphysis and shaft | 67 | 20.0 | |
| Distal epiphysis | 174 | 51.8 | |
| Complete | 26 | 7.7 | |
| Femur | Proximal epiphysis | 176 | 51.3 |
| Proximal epiphysis and shaft | 95 | 27.7 | |
| Shaft | 24 | 7.0 | |
| Distal epiphysis and shaft | 7 | 2.0 | |
| Distal epiphysis | 12 | 3.5 | |
| Complete | 29 | 8.5 | |
| Unidentified | Shaft | 3 | 100 |
| Sample Column or Area | Femora | Humeri | ||
|---|---|---|---|---|
| NISP | % Broken 1 | NISP | % Broken 1 | |
| Area SH18GS | 165 | 90.9 | 163 | 94.5 |
| Area SH19GS | 69 | 92.8 | 70 | 88.6 |
| Sample Column J | 4 | 100.0 | 0 | - |
| Sample Column J1 | 2 | 100.0 | 4 | 100.0 |
| Sample Column L | 1 | 100.0 | 2 | 100.0 |
| Sample Column L1 | 3 | 100.0 | 3 | 100.0 |
| Sample Column B | 1 | 100.0 | 0 | - |
| Sample Column B1 | 42 | 97.6 | 6 | 100.0 |
| Sample Column M | 42 | 92.9 | 38 | 94.7 |
| Sample Column N | 14 | 85.7 | 30 | 93.3 |
| Sample Column N1 | 113 | 91.2 | 20 | 85.0 |
| Total | 317 | 92.4 | 336 | 92.9 |
| Sample Column or Area | In Situ | Isolated Teeth | ||
|---|---|---|---|---|
| NISP | % Broken | NISP | % Broken | |
| Sample Column B | 5 | 20.0 | 30 | 53.3 |
| Sample Column B1 | 1 | 0.0 | 20 | 35 |
| Sample Column J | 4 | 0.0 | 31 | 51.6 |
| Sample Column J1 | 0 | - | 8 | 50 |
| Sample Column L | 0 | - | 8 | 50 |
| Sample Column L1 | 2 | 0.0 | 52 | 55.8 |
| Sample Column M | 15 | 0.0 | 283 | 44.2 |
| Sample Column N | 7 | 14.3 | 127 | 31.5 |
| Sample Column N1 | 17 | 5.9 | 122 | 19.7 |
| Area SH18GS | 58 | 14.7 | 728 | 36.0 |
| Area SH19GS | 56 | 14.3 | 267 | 21.0 |
| Total | 175 | 12.0 | 1676 | 34.8 |
| Element | Observed Digestion | NISP | Proportion (%) |
|---|---|---|---|
| Femur | None | 183 | 53.4 |
| Light | 125 | 36.4 | |
| Moderate | 26 | 7.6 | |
| Heavy | 7 | 2 | |
| Extreme | 2 | 0.6 | |
| Humerus | None | 276 | 82.1 |
| Light | 56 | 16.7 | |
| Moderate | 4 | 1.2 |
| Observed Digestion | Isolated Molars | In Situ Molars | ||
|---|---|---|---|---|
| NISP | Proportion (%) | NISP | Proportion (%) | |
| None | 968 | 80.0 | 25 | 89.3 |
| Light | 162 | 13.2 | 2 | 7.1 |
| Moderate | 62 | 5.1 | 1 | 3.6 |
| Heavy | 24 | 2.0 | 0 | 0 |
| Extreme | 10 | 0.8 | 0 | 0 |
| Total | 1226 | - | 28 | - |
| Sample Column | Observed Digestion (%) | Total NISP | ||||
|---|---|---|---|---|---|---|
| None | Light | Moderate | Heavy | Extreme | ||
| Sample Column B | 70.0 | 20.0 | 5.0 | 5.0 | 0.0 | 20 |
| Sample Column B1 | 85.7 | 0.0 | 14.3 | 0.0 | 0.0 | 14 |
| Sample Column J | 90.5 | 4.8 | 0.0 | 0.0 | 4.8 | 21 |
| Sample Column J1 | 83.3 | 16.7 | 0.0 | 0.0 | 0.0 | 6 |
| Sample Column L | 25.0 | 50.0 | 0.0 | 25.0 | 0.0 | 4 |
| Sample Column L1 | 80.0 | 20.0 | 0.0 | 0.0 | 0.0 | 20 |
| Sample Column M | 75.3 | 14.4 | 7.2 | 2.1 | 1.0 | 194 |
| Sample Column N | 81.7 | 8.5 | 1.4 | 4.2 | 4.2 | 71 |
| Sample Column N1 | 86.1 | 11.1 | 2.8 | 0.0 | 0.0 | 72 |
| Area SH18GS | 76.6 | 14.4 | 5.9 | 2.5 | 0.7 | 611 |
| Area SH19GS | 86.5 | 10.4 | 3.1 | 0.0 | 0.0 | 193 |
| Total | 79.0 | 13.2 | 5.1 | 2.0 | 0.8 | 1226 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tilby, E.; Miracle, P.; Barker, G. The Microvertebrates of Shanidar Cave: Preliminary Taphonomic Findings. Quaternary 2022, 5, 4. https://doi.org/10.3390/quat5010004
Tilby E, Miracle P, Barker G. The Microvertebrates of Shanidar Cave: Preliminary Taphonomic Findings. Quaternary. 2022; 5(1):4. https://doi.org/10.3390/quat5010004
Chicago/Turabian StyleTilby, Emily, Preston Miracle, and Graeme Barker. 2022. "The Microvertebrates of Shanidar Cave: Preliminary Taphonomic Findings" Quaternary 5, no. 1: 4. https://doi.org/10.3390/quat5010004
APA StyleTilby, E., Miracle, P., & Barker, G. (2022). The Microvertebrates of Shanidar Cave: Preliminary Taphonomic Findings. Quaternary, 5(1), 4. https://doi.org/10.3390/quat5010004







